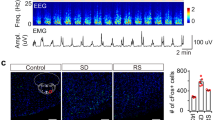
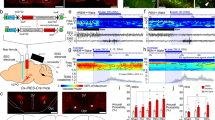
Abstract
Sleep cycles consist of rapid alterations between arousal states, including transient perturbation of sleep rhythms, microarousals, and full-blown awake states. Here we demonstrate that the calretinin (CR)-containing neurons in the dorsal medial thalamus (DMT) constitute a key diencephalic node that mediates distinct levels of forebrain arousal. Cell-type-specific activation of DMT/CR+ cells elicited active locomotion lasting for minutes, stereotyped microarousals, or transient disruption of sleep rhythms, depending on the parameters of the stimulation. State transitions could be induced in both slow-wave and rapid eye-movement sleep. The DMT/CR+ cells displayed elevated activity before arousal, received selective subcortical inputs, and innervated several forebrain sites via highly branched axons. Together, these features enable DMT/CR+ cells to summate subcortical arousal information and effectively transfer it as a rapid, synchronous signal to several forebrain regions to modulate the level of arousal.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Saper, C. B., Fuller, P. M., Pedersen, N. P., Lu, J. & Scammell, T. E. Sleep state switching. Neuron 68, 1023–1042 (2010).
Brown, R. E., Basheer, R., McKenna, J. T., Strecker, R. E. & McCarley, R. W. Control of sleep and wakefulness. Physiol. Rev. 92, 1087–1187 (2012).
Halász, P. & Bódizs, R. Dynamic Structure of NREM Sleep. (Springer-Verlag, London, UK, 2013).
Terzano, M. G. & Parrino, L. Origin and significance of the cyclic alternating pattern (CAP). Sleep. Med. Rev. 4, 101–123 (2000).
Schieber, J. P., Muzet, A. & Ferriere, P. J. Les phases d’activation transitoire spontanées au cours du sommeil normal chez l’homme. [Phases of spontaneous transitory activation during normal sleep in humans]. Arch. Sci. Physiol. (Paris) 25, 443–465 (1971).
Sakurai, T. The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nat. Rev. Neurosci. 8, 171–181 (2007).
Van der Werf, Y. D., Witter, M. P. & Groenewegen, H. J. The intralaminar and midline nuclei of the thalamus. Anatomical and functional evidence for participation in processes of arousal and awareness. Brain Res. Brain Res. Rev. 39, 107–140 (2002).
Kirouac, G. J. Placing the paraventricular nucleus of the thalamus within the brain circuits that control behavior. Neurosci. Biobehav. Rev. 56, 315–329 (2015).
Colavito, V., Tesoriero, C., Wirtu, A. T., Grassi-Zucconi, G. & Bentivoglio, M. Limbic thalamus and state-dependent behavior: The paraventricular nucleus of the thalamic midline as a node in circadian timing and sleep/wake-regulatory networks. Neurosci. Biobehav. Rev. 54, 3–17 (2015).
Unzai, T., Kuramoto, E., Kaneko, T. & Fujiyama, F. Quantitative analyses of the projection of individual neurons from the midline thalamic nuclei to the striosome and matrix compartments of the rat striatum. Cereb. Cortex 27, 1164–1181 (2017).
Lövblad, K. O., Bassetti, C., Mathis, J. & Schroth, G. MRI of paramedian thalamic stroke with sleep disturbance. Neuroradiology 39, 693–698 (1997).
Hermann, D. M. et al. Evolution of neurological, neuropsychological and sleep-wake disturbances after paramedian thalamic stroke. Stroke 39, 62–68 (2008).
Jones, E. G. The thalamic matrix and thalamocortical synchrony. Trends Neurosci. 24, 595–601 (2001).
Fuller, P. M., Sherman, D., Pedersen, N. P., Saper, C. B. & Lu, J. Reassessment of the structural basis of the ascending arousal system. J. Comp. Neurol. 519, 933–956 (2011).
Anaclet, C. et al. Basal forebrain control of wakefulness and cortical rhythms. Nat. Commun. 6, 8744 (2015).
Winsky, L., Montpied, P., Arai, R., Martin, B. M. & Jacobowitz, D. M. Calretinin distribution in the thalamus of the rat: immunohistochemical and in situ hybridization histochemical analyses. Neuroscience 50, 181–196 (1992).
Fortin, M., Asselin, M. C., Gould, P. V. & Parent, A. Calretinin-immunoreactive neurons in the human thalamus. Neuroscience 84, 537–548 (1998).
Peng, Z. C., Grassi-Zucconi, G. & Bentivoglio, M. Fos-related protein expression in the midline paraventricular nucleus of the rat thalamus: basal oscillation and relationship with limbic efferents. Exp. Brain Res. 104, 21–29 (1995).
Otake, K., Kin, K. & Nakamura, Y. Fos expression in afferents to the rat midline thalamus following immobilization stress. Neurosci. Res. 43, 269–282 (2002).
Zhu, L., Wu, L., Yu, B. & Liu, X. The participation of a neurocircuit from the paraventricular thalamus to amygdala in the depressive like behavior. Neurosci. Lett. 488, 81–86 (2011).
Bubser, M., Scruggs, J. L., Young, C. D. & Deutch, A. Y. The distribution and origin of the calretinin-containing innervation of the nucleus accumbens of the rat. Eur. J. Neurosci. 12, 1591–1598 (2000).
Do-Monte, F. H., Quiñones-Laracuente, K. & Quirk, G. J. A temporal shift in the circuits mediating retrieval of fear memory. Nature 519, 460–463 (2015).
Penzo, M. A. et al. The paraventricular thalamus controls a central amygdala fear circuit. Nature 519, 455–459 (2015).
Steriade, M., Oakson, G. & Ropert, N. Firing rates and patterns of midbrain reticular neurons during steady and transitional states of the sleep-waking cycle. Exp. Brain Res. 46, 37–51 (1982).
Beas, B. S. et al. The locus coeruleus drives disinhibition in the midline thalamus via a dopaminergic mechanism. Nat. Neurosci. 21, 963–973 (2018).
Berndt, A., Lee, S. Y., Ramakrishnan, C. & Deisseroth, K. Structure-guided transformation of channelrhodopsin into a light-activated chloride channel. Science 344, 420–424 (2014).
Watson, B. O., Levenstein, D., Greene, J. P., Gelinas, J. N. & Buzsáki, G. Network homeostasis and state dynamics of neocortical sleep. Neuron 90, 839–852 (2016).
Lecci, S. et al. Coordinated infraslow neural and cardiac oscillations mark fragility and offline periods in mammalian sleep. Sci. Adv. 3, e1602026 (2017).
Otake, K. & Nakamura, Y. Single midline thalamic neurons projecting to both the ventral striatum and the prefrontal cortex in the rat. Neuroscience 86, 635–649 (1998).
Dong, X., Li, S. & Kirouac, G. J. Collateralization of projections from the paraventricular nucleus of the thalamus to the nucleus accumbens, bed nucleus of the stria terminalis, and central nucleus of the amygdala. Brain. Struct. Funct. 222, 3927–3943 (2017).
Paré, D. & Smith, Y. Thalamic collaterals of corticostriatal axons: their termination field and synaptic targets in cats. J. Comp. Neurol. 372, 551–567 (1996).
Zhang, L., Kolaj, M. & Renaud, L. P. Suprachiasmatic nucleus communicates with anterior thalamic paraventricular nucleus neurons via rapid glutamatergic and gabaergic neurotransmission: state-dependent response patterns observed in vitro. Neuroscience 141, 2059–2066 (2006).
Pedersen, N. P. et al. Supramammillary glutamate neurons are a key node of the arousal system. Nat. Commun. 8, 1405 (2017).
Fremeau, R. T. Jr et al. The expression of vesicular glutamate transporters defines two classes of excitatory synapse. Neuron 31, 247–260 (2001).
Cicchetti, F., Lacroix, S., Beach, T. G. & Parent, A. Calretinin gene expression in the human thalamus. Brain. Res. Mol. Brain. Res. 54, 1–12 (1998).
Gent, T. C., Bandarabadi, M., Herrera, C. G. & Adamantidis, A. R. Thalamic dual control of sleep and wakefulness. Nat. Neurosci. 21, 974–984 (2018).
Matyas, F. et al. Motor control by sensory cortex. Science 330, 1240–1243 (2010).
Hunter, J. & Jasper, H. H. Effects of thalamic stimulation in unanaesthetised animals; the arrest reaction and petit mal-like seizures, activation patterns and generalized convulsions. Electroencephalogr. Clin. Neurophysiol. 1, 305–324 (1949).
Moruzzi, G. & Magoun, H. W. Brain stem reticular formation and activation of the EEG. Electroencephalogr. Clin. Neurophysiol. 1, 455–473 (1949).
Schlag, J. D., Chaillet, F. & Herzet, J. P. Thalamic reticular system and cortical arousal. Science 134, 1691–1692 (1961).
Vanderwolf, C. H. & Stewart, D. J. Thalamic control of neocortical activation: a critical re-evaluation. Brain Res. Bull. 20, 529–538 (1988).
Padilla-Coreano, N., Do-Monte, F. H. & Quirk, G. J. A time-dependent role of midline thalamic nuclei in the retrieval of fear memory. Neuropharmacology 62, 457–463 (2012).
Zhu, Y., Wienecke, C. F. R., Nachtrab, G. & Chen, X. A thalamic input to the nucleus accumbens mediates opiate dependence. Nature 530, 219–222 (2016).
Choi, E. A. & McNally, G. P. Paraventricular thalamus balances danger and reward. J. Neurosci. 37, 3018–3029 (2017).
Do-Monte, F. H., Minier-Toribio, A., Quiñones-Laracuente, K., Medina-Colón, E. M. & Quirk, G. J. Thalamic regulation of sucrose seeking during unexpected reward omission. Neuron 94, 388–400.e4 (2017).
Betley, J. N. et al. Neurons for hunger and thirst transmit a negative-valence teaching signal. Nature 521, 180–185 (2015).
Labouèbe, G., Boutrel, B., Tarussio, D. & Thorens, B. Glucose-responsive neurons of the paraventricular thalamus control sucrose-seeking behavior. Nat. Neurosci. 19, 999–1002 (2016).
Christoffel, D. J. et al. Excitatory transmission at thalamo-striatal synapses mediates susceptibility to social stress. Nat. Neurosci. 18, 962–964 (2015).
Yerkes, R. M. & Dodson, J. D. The relation of strength of stimulus to rapidity of habit-formation. J. Comp. Neurol. Psychol. 18, 459–482 (1908).
Umbriaco, D., Watkins, K. C., Descarries, L., Cozzari, C. & Hartman, B. K. Ultrastructural and morphometric features of the acetylcholine innervation in adult rat parietal cortex: an electron microscopic study in serial sections. J. Comp. Neurol. 348, 351–373 (1994).
Pinault, D. A novel single-cell staining procedure performed in vivo under electrophysiological control: morpho-functional features of juxtacellularly labeled thalamic cells and other central neurons with biocytin or Neurobiotin. J. Neurosci. Methods 65, 113–136 (1996).
Lopes, G. et al. Bonsai: an event-based framework for processing and controlling data streams. Front. Neuroinform. 9, 7 (2015).
Rossant, C. et al. Spike sorting for large, dense electrode arrays. Nat. Neurosci. 19, 634–641 (2016).
Furuta, T. et al. In vivo transduction of central neurons using recombinant Sindbis virus: Golgi-like labeling of dendrites and axons with membrane-targeted fluorescent proteins. J. Histochem. Cytochem. 49, 1497–1508 (2001).
Porrero, C. et al. A simple and efficient in vivo non-viral RNA transfection method for labeling the whole axonal tree of individual adult long-range projection neurons. Front. Neuroanat. 10, 27 (2016).
Harsan, L.-A. et al. Mapping remodeling of thalamocortical projections in the living reeler mouse brain by diffusion tractography. Proc. Natl. Acad. Sci. USA 110, E1797–E1806 (2013).
Giber, K. et al. A subcortical inhibitory signal for behavioral arrest in the thalamus. Nat. Neurosci. 18, 562–568 (2015).
Barthó, P. et al. Ongoing network state controls the length of sleep spindles via inhibitory activity. Neuron 82, 1367–1379 (2014).
Acknowledgements
We thank Z.J. Huang (CSHL, NY, USA) for providing us with the Calb2-Cre mice and C. Smerdou and C. Ballesteros (CIMA, University of Navarre, Spain) for synthesizing the Sindbis-Pal-eGFP RNA construct. The technical help of K. Faddi, K. Varga, A. Jász and E. Szabo-Egyud is acknowledged. The authors thank the Nikon Microscopy Center at IEM, Nikon Austria GmbH, and Auro-Science Consulting Ltd for kindly providing microscopy support and thank the Human Brain Research Laboratory (IEM/HAS) for the preparation of human material. The authors thank J. Poulet, B. Hangya, and H. Bokor for comments and discussions on the manuscript. This work was supported by the National Office for Research and Technology (NKTH-ANR-09-BLAN-0401, Neurogen to L.A; K119650 to P.B.; FK124434 to F.M.; PD124034 to B.B.), “Lendület” Program of the Hungarian Academy of Sciences (LP2012-23; B.B.), Hungarian Korean Joint Laboratory Program, Hungarian Brain Research Program (grants no. KTIA_NAP_13-2-2015-0010 to F.M., KTIA_NAP_13-2-2014-0016 to P.B. and KTIA_13_NAP-A-I/1 to L.A.), ERC (FRONTHAL, 742595 to L.A.), and HBP-FLAG-ERA (118886 to L.A.).
Author information
Authors and Affiliations
Contributions
F.M. and L.A. designed the experiments; F.M. and Á.B. performed the anatomical experiments; F.M. and G.K performed the freely moving EEG recordings; G.K. performed the freely moving data analysis with support from P.B.; K.K. and A.M. performed the freely moving unit recording and data analysis; F.M., V.K., and B.B. performed the behavioral experiments and data analysis; C.D. performed the axon analysis in PrL and the human histology; C.P. performed the electroporation with support from F.C.; I.S. provided the human thalamic samples; F.M., G.K., and L.A. wrote the paper, which was edited by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Activity of DMT cells before and after spontaneous arousals in freely sleeping conditions.
(a-c), Low power confocal images of a representative injection site at three coronal levels following a single injection of AAV-DIO-ChR2(H134)-eYFP into DMT of CR-Cre mice (n = 4 mice). (d), High power confocal images of the co-localization of CR immunostaining and eYFP in DMT 6 weeks after the viral injection. (e), An example of an optically tagged DMT/CR+ cell which increased firing activity together with (but not before) the sleep/wake transition in freely moving conditions. From left-to-right, waveforms (WF, top) and autocorrelogram (ACG, bottom), peri-event time histogram upon optogenetic tagging and change of firing rate of the sample DMT/CR+ cell. (f), Population data for the activity of those DMT/CR+ units which increased their firing only at the onset of sleep/wake transition (0/+; n = 8/31) but not before. (g), Population data for the activity of those DMT/CR+ units which displayed anticipatatory firing before the onset of the sleep/wake transition (+/all; n = 20/31). 1 s bins indicate the averages of z-scores, while green line shows its variance (SD). Red dashed lines represent the significance levels for P < 0.05 Z-score values (1.96). (h), Pie chart shows the distribution of firing rate changes for all 31 DMT/CR+ neurons before and after the sleep/wake transition. (i), Pie chart shows the distribution of firing rate changes for all 34 DMT/CR- before and after sleep/wake transition. Activation before EMG ON vs. others, DMT/CR+ vs. DMT/CR- Fisher’s exact test, two-tailed, P = 0.0012.
Supplementary Figure 2 Distribution of c-Fos-activated cells in the DMT/CR+ region.
a1-a3, Overlap between foot-shock activated c-Fos-positive neurons and CR expression in DMT (n = 4 mice).
Supplementary Figure 3 Response of DMT/CR+ cell activity to tail-pinch.
(a), Experimental design to monitor the effects of tail pinch on the DMT/CR+ cells firing along with the prelimbic cortical (PrL) LFP. (b-c), Individual responses of single DMT/CR+ cells which increased (b) or decreased (c) their firing to tail pinch. Black and red traces indicate the corresponding raw and filtered (1–3 Hz) PrL LFP, respectively. (d), Top, population data for responses to tail-pinch of DMT/CR+ (n = 13 cells). 6/13 DMT/CR+ neurons responded with elevated activity (PRE 1.26 ± 0.62 Hz; TAIL 4.96 ± 3.43 Hz;) which persisted long after the termination of the stimulus (3.39 ± 2.73Hz in the 40 s post stimulus period). One-way ANOVA, F(2, 15) = 5.3735, P = 0.0174; Fisher’s LSD, Pre vs. Tail, P = 0.0053; Pre vs Post, P = 0.0748; Tail vs. Post, P = 0.1978. The remaining neurons (7/13) decreased their activity during tail pinch (Pre 0.91 ± 0.31 Hz; Tail 0.17 ± 0.26 Hz; Post 0.12 ± 0.2 Hz,). One-way ANOVA, F(2, 18) = 50.533, P < 0.0001; Fisher’s LSD, Pre vs. Tail, P < 0.0001; Pre vs Post, P < 0.0001; Tail vs. Post P = 0.7262. Bottom, changes of PrL delta power underlying tail pinch. Black and grey dots indicate individual cells which increase or decrease their firing, respectively, during tail pinch [group (increase vs. decrease)-wise comparison: Repeated measures of ANOVA, F(2, 16) = 0.187, P = 0.8314; Effect (Pre-Tail-Post)-wise comparison: one-way ANOVA, F(2, 27) = 61.372, P < 0.0001; Fisher’s LSD, Pre vs. Tail, P < 0.0001; Pre vs Post, P < 0.0001; Tail vs. Post P = 0.8692; while light and dark green colored bars represent their mean ± SD, respectively. #P < 0.1; *P < 0.05; **P < 0.01, ***P < 0.001; n.s., non-significant.
Supplementary Figure 4 SwichR-mediated silencing of DMT/CR+ cells.
a, Experimental design to validate SwichR-mediated silencing of DMT/CR+ cells. b-d, Juxtacellular recording of a DMT cell using 0.5 s (a), 1 s (b) and 2 s (c) long blue laser light under urethane anesthesia.
Supplementary Figure 5 Quantification of arousal events and features of the EMG ON states evoked by stimulations with different durations.
(a), Definitions of EMG ON and OFF (active/inactive) states for quantitative purposes (onset, duration, probability) based on the raw EMG signal. Black, 300–600 Hz filtered EMG; purple, standard deviation of EMG; green, threshold for EMG active state. Three criteria were used. 1: standard deviation (+2.1–5 SD of baseline) above threshold; 2: EMG ON states shorter than 0.5 s are discarded since they represent muscle twitches rather than arousals or microarousals. 3: Gaps shorter than 2 s between EMG ON states were filled with ON state, since it is unlikely that mice go back to sleep for 2 s. (b), Cumulative distribution of the latency of spontaneously occurring (black) or optogenetically evoked arousals (1s; red, 10s blue) in a 60 s window after stimulation onset. Shaded area represents ± s.e.m. (c), Average durations of spontaneous and evoked arousals, using different stimulus durations (n = 225 trials in 5 mice, spontaneous (sp), 2.67 ± 0.77; 0.5 s, 2.08 ± 0.78 s; 1 s, 3.22 ± 0.8 s; 2 s, 9.59 ± 4.02 s; 10 s, 56.41 ± 2 s; Repeated measures of ANOVA, F(3,12) = 836.88, P < 0.0001. Pairwise comparison with Bonferroni correction shows significant difference only for 0.5 s vs 10 s, P < 0.0001).The horizontal lines in the box plots indicate medians, the box limits indicate first and third quantiles, and the vertical whisker lines indicate minimum and maximum values.
Supplementary Figure 6 Alteration of EEG, not EMG activity, is the primary response after DMT/CR+ stimulation.
(a), Average peri-stimulus distribution of the probability of EMG ON states (black), normalized delta power (red) and raw EMG activity (gray) during microarousals following 1 s DMT/CR+ stimulations in NREM sleep (n = 5 mice). Microarousals with longer than 2 s latencies are included. Note the instantaneous drop in delta activity followed by the onset of EMG activity after several seconds. (b), In this figure in the top the evoked EMG ON states are grouped according to onset latencies (0–1 s, 1–2 s, etc, n = 521 trials in 5 mice) and labeled with different colors and in the bottom the corresponding average peristimulus EEG delta powers data are shown with the same colors. Note, that the drop in the delta power is tightly linked to the stimulation regardless of the onset of EMG ON state demonstrating the primacy of EEG response. (c), Distribution of time differences between the drop in delta activity and the onset of EMG ON states in case of individual evoked arousal events. Bin size is 0.25 s. In the vast majority of the cases (89%) the EEG response occurs first.
Supplementary Figure 7 State dependency of DMT/CR+ responses and characterization of evoked VB responses.
(a), Spike triggered EEG averages of the cortical evoked responses during DMT/CR+ stimulations resulting in microarousals (red) or sleep throughs (blue) in NREM sleep (n = 5 mice). The traces are triggered by the first stimulation of the 1 s long 10 Hz train. No difference in cortical response can be observed in these two conditions. (b-c), Normalized, average delta (b) and sigma (c) powers preceding DMT/CR+ stimulations resulting in microarousals (red) or sleep throughs (blue) in NREM sleep (n = 5 mice). No systematic difference is present between the two conditions. Light blue and red lines represents ± s.e.m. (d-f), Average peristimulus distribution of EMG ON states (d), delta (e) and sigma (f) power during microarousals (MA, red) and sleep-through (ST, blue) in case of 1 s VB stimulations n = 274 trials in 4 unilateral stimulations. Note prolonged disturbance of sigma activity even in case of sleep-throughs. Light blue and red lines represents ± s.e.m.
Supplementary Figure 8 Stimulation intensity vs. arousal (EMG ON) probability curves for individual DMT/CR+ and VB mice.
(a), DMT/CR+ mice (n = 5) (b), VB mice (n = 4). Black curves, moving averages of 30 points; red curves, sigmoid fitted on data; cyan horizontal lines, probability of spontaneous arousal (EMG ON) within 10s (using the same criteria as for evoked). Blue dots indicate trials of laser intensities at which the stimulation evoked microarousal (1) or sleep-through (0). Each panel contains the laser intensity range used for the given animal.
Supplementary Figure 9 Widespread projection of DMT/CR+ neurons in different forebrain regions.
(a–k), AAV-DIO-ChR2-eYFP labeled axonal processes (green) in a CR-Cre mouse arising from DMT. DMT/CR+ fibers can be found in all layers (L1-L6) of the prelimbic (PrL, a) and insular cortex (IC; b), in L6 of the primary somatosensory cortex (S1; c), deep layers of the temporal association cortex (TeA; d), nucleus accumbens (NAc; e), lateral septum (LS; f), dorsomedial part of the caudate putamen (dmCPu) and bed nucleus of the stria terminalis (BNST; g); interstitial nucleus of the posterior limb of the anterior commissure (IPAC), olfactory tubercle (Tu), substantia innominata (SI) and ventral pallidum (VP; h); amygdalostriatal transition area (Astr), centrolateral amygdala (CeL), basolateral amygdala (BLA) but not lateral amygdala (LA; i); lateral hypothalamus (LH; j) and thalamic reticularis nucleus (TRN; k). Note the varying density of DMT/CR+ axonal arbor around the cholinergic (Chat; red in h) and orexinergic cells (Orx, red in j). The territory of TRN is labeled by parvalbumin (PV, red) in k. LV, lateral ventricle. (l–n), Schematic drawings of the experiments (top) and representative low power confocal image of a CR-immunostained (red) sections of DMT (bottom). Yellow dots indicate the position of DMT neurons retrogradely labeled from the prelimbic cortex (l; PrL; 891/922 cell, 96.64 %; n = 5 mice), amygdala (m; AMY; 416/438 cells, 94.98 %, n = 4 mice) and nucleus accumbens (n; NAc; 2081/2114 cell, 98.44 %, n = 5 mice). Graphs in the left corner of the images show the proportion of CR+/CTB-labeled cells in the DMT and represent means ± SD.
Supplementary Figure 10 Functional connectivity of DMT/CR+ neurons in PrL, NAc, and BLA.
(a), The magnitude of evoked multi-unit activity (eMUA) in PrL, NAc and BLA by DMT/CR+ stimulation at 10 Hz depends on laser intensity. (b), Quantification of the peak amplitude for the 1st, 2nd, 5th and 10th pulses of an 1 s long 10 Hz optical stimulation eMUA display depression in PrL (top; n = 6 animals; One-way ANOVA, F(3, 20)=14.788, P < 0.0001; Newman-Keuls test, 1st vs. 2nd, P = 0.4790; 1st vs. 5th, P = 0.0236; 1st vs. 10th, P = 0.0002) and NAc (middle; n = 3; One-way ANOVA, F(3, 16)=15.254, P < 0.0001; Newman-Keuls test, 1st vs. 2nd, P = 0.1322; 1st vs. 5th, P = 0.0008; 1st vs. 10th, P = 0.0003) and less prominently in BLA (bottom; n = 3, One-way ANOVA, F(3, 8)=1.7067, p = 0.2424). (c), Representative evoked LFP signal in PrL by 1 (left) and 10 Hz (right) optical stimulation of DMT/CR+ neurons (dashed lines). Note the lack of augmenting responses. (d), Population data (n = 6) show no changes in evoked LFP amplitude at 1 Hz (One-way ANOVA, F(3, 20) = 0.30281, p = 0.823) but significant depression at 10 Hz (F(3, 20)=38.227, P < 0.0001; Newman-Keuls test, 1st vs. 2nd, P = 0.2326; 1st vs. 5th, P = 0.0002; 1st vs. 10th, P = 0.0002). Data are means ± SD; *P < 0.05, ***P < 0.001.
Supplementary Figure 11 Multiple forebrain targets of DMT/CR+ cells.
(a-l), Distribution of single (red and green dots) and double labeled DMT cells (yellow triangles) from PrL and NAc (top; a-d), BLA and PrL (middle; e-h), and BLA and NAc (bottom; i-l) at two AP levels. (m-o), A single cell labelling of a DMT/CR+ neuron with Pal-eGFP-Sindbis. Confocal images in m show the colocalization of GFP (green, top) with CR (red, middle). Note the neighboring CR+ (*) and a CR- (#) cell. n, A low magnification confocal image indicates the location of the same labeled cell in DMT/CR+ region. o, A partial reconstruction of the same DMT/CR+ cell (green) shows its axon branching (black) in many forebrain sites including cortical [insular (IC) and piriform cortices (Pir) as well as basolateral amygdala (BLA)] and subcortical [nucleus reticular thalami (nRT), nucleus basalis (NB), nucleus accumbens (NAc), anterior cortical amygdaloid nucleus (ACo), central amygdala (CeA), interstitial nucleus of the posterior limb of the anterior commissure (IPAC) and medial amygdala (MeA)] structures. (p-r), Following double retrograde tracings using Fluorogold (FG) and choleratoxin B (CTB) injected into any combination of the three main DMT/CR+ targets (PrL, NAc or AMY) revealed largely non-overlapping population of projecting neurons in any other brain region examined beside DMT (see above). This indicates that only DMT/CR+ neurons provide significant amount of branching collaterals linking these regions. In this example, FG is injected into PrL (a, green) and CTB into into NAc (b, red). In the two brain regions which contained neurons projecting to both sites, (the supramammilary nucleus, SUM, p and the ventral tegmental area, VTA, q) to ratio of dual projecting cells were extremely low (9/700 neurons, 1.3%; n = 4 mice) (r).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11
Supplementary Video 1
Ten seconds long stimulation of DMT/CR+ cells evokes full blown arousal. 10 s activation of DMT/CR+ cells during natural sleep. Green lamp on the left (red circle) indicates the start of the laser stimulus. Note normal awakening behavior, including stretching.
Supplementary Video 2
One second long stimulation of DMT/CR+ cells evokes microarousal. Similar conditions but 1 s optogenetic activation of DMT/CR+ neurons as in Supplementary Video 1. Note only short head and neck movements.
Rights and permissions
About this article
Cite this article
Mátyás, F., Komlósi, G., Babiczky, Á. et al. A highly collateralized thalamic cell type with arousal-predicting activity serves as a key hub for graded state transitions in the forebrain. Nat Neurosci 21, 1551–1562 (2018). https://doi.org/10.1038/s41593-018-0251-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-018-0251-9
This article is cited by
-
Region-selective control of the thalamic reticular nucleus via cortical layer 5 pyramidal cells
Nature Neuroscience (2023)
-
Neuro-orchestration of sleep and wakefulness
Nature Neuroscience (2023)
-
Parasubthalamic calretinin neurons modulate wakefulness associated with exploration in male mice
Nature Communications (2023)
-
Variable cardiac responses in rhesus macaque monkeys after discrete mediodorsal thalamus manipulations
Scientific Reports (2023)
-
AgRP neurons control structure and function of the medial prefrontal cortex
Molecular Psychiatry (2022)