Abstract
Previous studies report an association between maternal diabetes mellitus (MDM) and attention-deficit/hyperactivity disorder (ADHD), often overlooking unmeasured confounders such as shared genetics and environmental factors. We therefore conducted a multinational cohort study with linked mother–child pairs data in Hong Kong, New Zealand, Taiwan, Finland, Iceland, Norway and Sweden to evaluate associations between different MDM (any MDM, gestational diabetes mellitus (GDM) and pregestational diabetes mellitus (PGDM)) and ADHD using Cox proportional hazards regression. We included over 3.6 million mother–child pairs between 2001 and 2014 with follow-up until 2020. Children who were born to mothers with any type of diabetes during pregnancy had a higher risk of ADHD than unexposed children (pooled hazard ratio (HR) = 1.16, 95% confidence interval (CI) = 1.08-1.24). Higher risks of ADHD were also observed for both GDM (pooled HR = 1.10, 95% CI = 1.04-1.17) and PGDM (pooled HR = 1.39, 95% CI = 1.25-1.55). However, siblings with discordant exposure to GDM in pregnancy had similar risks of ADHD (pooled HR = 1.05, 95% CI = 0.94-1.17), suggesting potential confounding by unmeasured, shared familial factors. Our findings indicate that there is a small-to-moderate association between MDM and ADHD, whereas the association between GDM and ADHD is unlikely to be causal. This finding contrast with previous studies, which reported substantially higher risk estimates, and underscores the need to reevaluate the precise roles of hyperglycemia and genetic factors in the relationship between MDM and ADHD.
Similar content being viewed by others
Main
Globally, 16% of pregnant women experience hyperglycemia1,2. The prevalence of maternal diabetes mellitus (MDM) has increased worldwide, which is associated with the growing epidemic of obesity, advancing maternal age and improved diagnostic approaches for MDM3,4. There are calls for greater attention to the risks associated with diabetes in pregnancy given the increasing trend of gestational diabetes and preexisting type 2 diabetes5. Animal studies have demonstrated the adverse effects of hyperglycemia during pregnancy on inflammatory responses, intrauterine oxidative stress and imbalance in epigenetic mechanisms, which may contribute to poor neurodevelopment in the offspring6,7.
Attention-deficit/hyperactivity disorder (ADHD) is a neurodevelopmental disorder characterized by hyperactivity, impulsivity and inattentiveness8. Currently, ADHD is estimated to affect 2% to 7% of children worldwide9,10, making it one of the most common disorders among school-aged children. ADHD not only adversely impacts the affected individuals but also poses a substantial burden on their families and the wider society11,12,13,14,15,16,17. A complex interaction between genetic, environmental and psychosocial risk factors is thought to be responsible for the etiology of ADHD8.
Emerging evidence has suggested that both pregestational diabetes mellitus (PGDM) and gestational diabetes mellitus (GDM) are associated with ADHD. A previous meta-analysis found that the offspring of diabetic mothers were at 40% higher risk of ADHD18. However, some of the included studies used self-reported data19,20,21, had limited statistical power20,22 or had limited adjustment for confounders19,20,23,24, especially familial factors, and were predominantly conducted in White populations7,19,20,21,22,23,25,26. To account for these limitations, we conducted the current cohort study based on population-based data covering over 3.6 million mother–child pairs in Hong Kong, New Zealand, Taiwan, Finland, Iceland, Norway and Sweden, with extensive coverage of relevant covariates, to assess the association between MDM and the risk of ADHD in offspring.
Results
This study consisted of children from all live births within the site-specific study period (Table 1). All mother–child pairs were linked with exact deterministic linkage27. Children without valid mother–child linkage or with incomplete birth information (for example, sex or gestational age) or without at least 6 years of follow-up were excluded to allow sufficient follow-up time to capture ADHD outcomes, as a diagnosis is often deferred until a child is of school age28. The follow-up period for each child started on the date of birth and ended on the date of outcome occurrence, date of death, or end of data source-specific study period, whichever came first. Details of the sample size calculation are given in Extended Data Fig. 1.
We identified the start and end of pregnancy by the date of the last menstrual period (LMP) and the child’s date of birth (Fig. 1). In Hong Kong, New Zealand and the Nordic countries, LMP was determined by subtracting gestational age at birth (determined by ultrasound) from the date of birth; in Taiwan, this was defined as the date of delivery minus 280 days29. As hyperglycemia may affect neurodevelopment differently at different trimesters, we divided the pregnancy period into first trimester (0–90 days after the LMP), second trimester (91–180 days after the LMP) and third trimester (181 days after the LMP to delivery). The primary exposure is MDM (including GDM and PGDM). MDM was further classified as GDM (including those receiving and not receiving medications) and PGDM (including type 1 and type 2 PGDM; Extended Data Fig. 2). PGDM refers to existing diabetes before pregnancy, and GDM refers to diabetes diagnosed only during pregnancy. Data source-specific criteria were applied to ensure the best identification of the exposure status (Supplementary Table 1).
aThe earliest date of mothers’ health data in each data source. bBaseline conditions included: demographics, maternal conditions and medication use. For HK analysis, maternal age at delivery and birth year were assessed at the date of delivery, body mass index (BMI) was assessed from LMP − 365 days to LMP − 1 day and all other covariates were assessed before LMP; for analysis in Nordic countries and New Zealand, medication use was assessed from LMP − 365 days to LMP − 1 day, and diagnoses were assessed from LMP − 365 days to delivery date; for Taiwan analysis, all covariates were assessed within 2 years before the date of delivery. cExposure window: (1) Period before pregnancy, (2) first trimester: LMP to LMP + 90 days, (3) second trimester: LMP + 91 days to LMP + 180 days, and (4) third trimester: LMP + 181 days to delivery date. dEarliest of: date of ADHD diagnosis, date of first ADHD medication prescription, date of death, end of database catchment period.
ADHD was defined by each site using specific diagnosis and medication codes as reported in previous studies and detailed in Supplementary Table 1 (refs. 30,31). In Hong Kong, New Zealand and Taiwan, children with either one ADHD diagnosis or one ADHD medication prescription were regarded as having the outcome. In Nordic countries, the outcome was defined as (1) ≥2 records of ADHD diagnoses or (2) ≥1 record of ADHD diagnosis and ≥2 records of ADHD medication prescription fills. In Hong Kong, New Zealand and Nordic countries, these criteria needed to be met at or after the age of 3 years to exclude invalid ADHD diagnoses (Supplementary Table 1).
We identified 3,619,717 mother–child pairs to be included in the analysis (Fig. 2). Overall, 8.0% (n = 30,396), 4.1% (n = 21,326), 13.7% (n = 107,898) and 6.6% (n = 126,425) of children were born to mothers with diabetes in Hong Kong, New Zealand, Taiwan and the Nordic countries, respectively. Child and maternal characteristics are summarized in Supplementary Table 2. Covariate balances were achieved after propensity score (PS) weighting with standardized differences <10%, except for obesity in New Zealand (Supplementary Table 3).
Primary analyses and sibling-matched comparisons
When comparing children born to mothers with any diabetes during pregnancy (NMDM = 286,045) and unexposed children (Nnon-MDM = 3,333,672), we identified a higher risk of ADHD (pooled hazard ratio (HR): 1.16, 95% confidence interval (CI): 1.08-1.24). The cumulative incidence of ADHD for different data sources is shown in Extended Data Fig. 3. Similarly, we identified a higher risk of ADHD across different types of MDM when comparing children whose mothers had GDM (NGDM = 230,798), PGDM (NPGDM = 54,825), type 1 PGDM (Ntype 1-PGDM = 11,444) and type 2 PGDM (Ntype 2-PGDM = 42,977) to those whose mothers did not have diabetes (GDM pooled HR: 1.10, 95% CI: 1.04 to 1.17; PGDM pooled HR: 1.39, 95% CI: 1.25-1.55, type 1 PGDM pooled HR: 1.46, 95% CI: 1.24-1.71; type 2 PGDM pooled HR: 1.38, 95% CI: 1.24-1.53; Fig. 3 and Table 2). We applied sibling-matched analysis for GDM to control for shared familial confounding including unmeasured lifestyle factors. Siblings who were born to the same mother but with discordant exposure to GDM during the respective pregnancy episodes did not differ in the risks of ADHD (pooled HR: 1.05, 95% CI: 0.94-1.17, NGDM = 72,791, Nnon-MDM = 75,082; Fig. 4 and Table 2). Risks of ADHD for children were similar across those whose mothers had GDM diagnosed at different trimesters in Hong Kong; in Taiwan and New Zealand, the risk of ADHD was highest for children born to mothers with GDM diagnosed in the first trimester (Supplementary Table 4).
Data are presented as HRs and 95% CIs, which were adjusted for demographics, socioeconomic status, birth year, multifetal pregnancies, maternal conditions and use of relevant medications using Cox proportional hazard regression, with a significance level of 5% for a two-sided test. No adjustments were made for multiple comparisons. df, degrees of freedom; IV, inverse variance; s.e., standard error; T1DM, type 1 pregestational diabetes; T2DM, type 2 pregestational diabetes.
Comparisons in different MDM exposures
Children born to mothers with GDM may have a lower risk of ADHD when compared with children born to mothers with PGDM (pooled HR: 0.76, 95% CI: 0.61-0.96, NGDM = 230,798, NPGDM = 54,825). Risks of ADHD did not differ between children born to mothers with type 2 PGDM and type 1 PGDM (pooled HR: 1.04, 95% CI: 0.89-1.21, Ntype 1-PGDM = 11,444, Ntype 2-PGDM = 42,977). Children whose mothers had GDM requiring antidiabetic medication had a similar risk of ADHD when compared with children born to unmedicated mothers with GDM (pooled HR: 1.14, 95% CI: 0.92-1.42, Nmedicated = 25,206, Nunmedicated = 205,592; Table 2 and Extended Data Fig. 4).
Sensitivity analyses
Results from the sensitivity analyses were similar to the primary analyses when the analytic cohorts included children with less than 6 years of follow-up and were stratified by sex (Supplementary Table 5). We computed E-values to facilitate the interpretation of results in the presence of unmeasured confounding. The E-values of the pooled results range from 1.43 to 2.28, suggesting that unmeasured confounding with an association magnitude equal or greater to both the exposure and outcome could explain away the observed associations respectively (Supplementary Table 6). In other words, any residual confounding is likely to lead to an even smaller estimate. A post hoc analysis also yielded similar results when the analytic cohorts included only children with 9 or more years of follow-up (Supplementary Table 7). Finally, results from the Poisson and negative binomial regression were similar to the Cox regression in the main analysis (Supplementary Table 8).
Discussion
In this large multinational cohort study, including over 3.6 million mother–child pairs and leveraging a common data model and analytic approach, we found that MDM overall, GDM and PGDM were associated with a small-to-moderate risk of ADHD in offspring. After controlling for shared familial genetic and social factors in the sibling-matched analyses, risks of ADHD did not differ between siblings with discordant exposure to GDM in pregnancy. Due to the discrepancy around the within- and between-family analyses, we speculate that the relationship between GDM exposure and ADHD may be confounded by familial factors.
Compared with a previous meta-analysis which showed a 40% and twofold increased risk of ADHD in children born to mothers with any diabetes and GDM respectively, our study demonstrates a relatively smaller risk estimate of ADHD in children born to mothers with any diabetes after comprehensively controlling for potential confounders. The E-values for the primary analyses ranged from 1.43 to 2.28 and were considered relatively small32. Thus, it is likely that there are unmeasured confounders of this magnitude, such as disease severity or paternal factors, that may explain the identified association in our primary analyses. More importantly, although we observed a higher risk of ADHD in offspring whose mothers had GDM at the population level, the association was null in the sibling-matched analysis. Thus, shared familial or genetic factors are likely to at least partly explain the observed association at least between GDM and ADHD in our primary analyses.
In our study, children born to mothers with medicated GDM had a similar risk of ADHD when compared with those with unmedicated GDM. Existing literature regarding the effects of GDM treatment on the risk of ADHD in offspring is scarce. A study using US private healthcare data found a 38% increased risk in children born to mothers requiring treatment for GDM than those with unmedicated GDM33. Combining our results with currently available evidence, it remains unclear whether maternal antidiabetic medication during pregnancy could increase the risk of ADHD in offspring or if the severity of GDM requiring antidiabetic medication underlies the association. This further poses a question on the role of glycemic control during pregnancy in mitigating the risk of ADHD in children whose mothers have diabetes.
Our study has limitations. First, most data originated from reimbursement or other administrative purposes rather than research purposes, which represent a variety of data sources, healthcare settings, coding practices, diagnostics criteria, and treatment approaches. Nevertheless, data sources included in this study are all based on high-quality territory-wide electronic health records with robust mother–child linkage and comprehensive information on maternal and child medical records that have been used extensively for pregnancy-related studies30,34,35,36. Moreover, we applied data source-specific identification criteria for not only the exposure and outcome but also the covariates to maximize the comparability of variables included in the study. Second, there may be inaccuracy in coding and incompleteness of diagnoses, prescriptions and laboratory test records. However, we used electronic records and the included pregnant women are likely to have received more frequent clinical monitoring throughout pregnancy. Furthermore, we used a comprehensive set of data ranging from diagnosis records, prescription records and laboratory test records for pregnant women. We defined the study outcome as a diagnosis for ADHD or with ADHD medication prescriptions to capture all possible cases to increase the power of our study, but we acknowledge that misclassification could exist, and some children with minor symptoms of ADHD might have been included in the non-ADHD group. Similarly, nondifferential misclassification of ADHD patients may direct our results towards the null, leading to a smaller effect estimate than previous studies. First, ADHD may not be diagnosed until later in life, which may lead to inclusion of undiagnosed ADHD patients in our non-cases. We therefore conducted a post hoc sensitivity analysis (Supplementary Table 7) to assess the effect of insufficient follow-up duration in our main analyses. After only including mother-baby pairs with at least 9 years of follow-up duration, we found a similar result to the main analyses, demonstrating the robustness of our study conclusion. Similarly, the rates of detection and diagnosis of ADHD were expected to vary with age. This finding implies that the hazards for exposed and unexposed children would naturally exhibit deviations from proportionality throughout the observation period (Extended Data Fig. 3)8. We therefore recommend interpreting the HRs we present as a weighted average of the time-varying hazard ratios within our observation period. Nonetheless, sensitivity analyses using Poisson regression and negative binomial regression, which do not rely on the proportional hazards assumption, yielded consistent estimates that support our interpretation and conclusion (Supplementary Table 8). Second, some ADHD medication used in the Nordic countries may be used for other non-ADHD conditions such as narcolepsy. Therefore, ADHD case identification in Nordic countries required at least one ADHD diagnosis and at least two prescription fills for an ADHD medication and thus we were not likely to capture children using ADHD medication for other non-ADHD conditions. Third, maternal lifestyle factors such as physical activity and diet may not be fully captured in electronic health records such that inadequate adjustment for these factors could lead to residual confounding. We thus applied the current study design and analytic approaches to address this concern: 1) the use of sibling-matched analysis for GDM to control for shared familial confounding including lifestyle factors; 2) the computation of E-value to aid the interpretation of the results in presence of unmeasured confounding. For MDM, E-value computation showed that any residual confounding is likely to lead to an even smaller estimate. Therefore, even if these behavioral factors could explain the association, it is unlikely to affect our conclusion that there is a small-to-moderate association between MDM and ADHD, whereas the association between GDM and ADHD is unlikely to be causal. Finally, although the sibling-matched analyses allowed us to control for unmeasured, shared confounding, the design could amplify confounding from factors unique to each sibling37. Therefore, we draw our conclusion based on complementary study designs, including the unrelated cohort analyses, sibling-matched comparisons and computation of E-values. More importantly, our results remain robust in all sensitivity analyses and consistent across all data sources from various populations.
It was hypothesized that hyperglycemia may alter the intrauterine environment with increased inflammation, metabolic stress and lipotoxicity, which may affect the neurodevelopment of offspring6. Our study, however, only found a small-to-moderate effect between MDM and ADHD where the effect is likely to be confounded by shared genetic and familial factors, at least in the case of GDM. Future studies should explore the specific roles of genetic factors and glycemic control during different developmental stages of the human embryonic brain.
Methods
Study design
This is a population-based cohort study with linked mother–child pairs based on healthcare data in Hong Kong, New Zealand, Taiwan and Nordic countries (Finland, Iceland, Norway and Sweden; data nested within the NorPreSS collaboration38). The study results were reported following the STrengthening the Reporting of OBservational studies in Epidemiology statement.
All study sites used pseudonymized patient-level electronic health data derived from the respective territory-wide administrative, clinical, or register databases. The list of data sources is provided in Table 1. We applied a distributed network approach with a common data model to harmonize the data structure and standardize the contents from different data sources. Briefly, the coordinating center at the University of Hong Kong distributed a common analytic package for generating aggregated results based on the common data model38,39. Site investigators conducted the analyses locally in Hong Kong, New Zealand, Taiwan and Nordic countries where data from individual Nordic countries were pooled into one cohort and analyzed centrally38, before sharing aggregated results with the study coordinator. This approach preserved data confidentiality as the individual-level data remained at each site40. Moreover, we were able to maintain the consistency of analyses among sites with common analytics41. The codes used to identify relevant diagnoses and medication prescriptions from each site are presented in Supplementary Table 8.
Covariate assessment
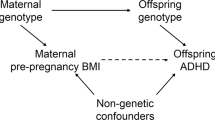
Covariates were selected based on known confounders for the study association and risk factors for the study outcomes30,42,43,44,45,46,47,48,49,50, including demographic factors such as maternal age47,51,52,53, infant sex (as directly recorded by clinicians)54, socioeconomic status55,56, birth year, multifetal pregnancies57 and other maternal factors including smoking44,47,58,59, alcohol consumption47,60,61, psychiatric and neurological conditions47,48,62,63,64, other chronic medical conditions (hypertension51,65, renal disease, inflammatory bowel disease66,67, autoimmune disease46,68,69, thyroid disorders43,47,70,71 and polycystic ovary syndrome45,72,73), BMI44,47,51,74 and use of psychotropic medication42,47,49, antihypertensives, ADHD medication and known or suspected teratogenic medication. Various measures were applied as the proxy of socioeconomic status for each data source according to their respective practice (namely, median household income in Hong Kong, education level in Nordic countries, insurance fees in Taiwan and deprivation quintile in New Zealand). Definitions of the covariates by study site are available in Supplementary Table 8. A schematic directed acyclic graph illustrating the causal relationships between the different covariates and the exposure and/or outcome is shown in Extended Data Fig. 5.
Comparison groups
In our primary analyses, we compared the ADHD status in children born to mothers with any MDM, GDM, any PGDM, type 1 PGDM and type 2 PGDM, with children born to mothers without any diabetes. We also conducted sibling-matched analyses which compared the ADHD status in children born to the same mothers but with discordant GDM exposure. In our secondary analyses, we compared the ADHD status between children born to mothers with different diabetes subtypes, namely GDM and PGDM, type 2 PGDM and type 1 PGDM, and medicated GDM and unmedicated GDM.
Statistical analysis
We estimated HRs of average treatment effect with 95% CIs to study the associations between MDM status and ADHD using Cox proportional hazard regression models. Propensity score (PS) fine-stratification weighting was used to address the differences in baseline covariates. PS, the probability of receiving treatment conditional on the observed characteristics at baseline, can be applied to account for confounding effects efficiently in observational studies50,75. We used PS fine-stratification weighting because of the greater precision, less residual and equivalent bias control compared to traditional PS methods75,76. The PS is first used to create 50 fine strata; weights for both exposure and reference patients in all strata are subsequently calculated based on the total number of patients within each stratum, whereas strata with no exposed or reference patients are dropped out before weight calculation76. We applied robust standard errors to adjust for data clustering. All the covariates listed in Supplementary Table 8 were included in the PS model. Factors with standardized differences greater than 10% were further adjusted in the Cox models77. For missing data, indicator variables for missing maternal characteristics were included in the Nordic (folic acid use, education level, cohabitation, parity, and non-Nordic place of birth) and New Zealand (mother’s socioeconomic status and BMI) models; median imputation was applied for socioeconomic status in Taiwan for missing insurance fees (missing rate = 0.13%).
We conducted sibling-matched analyses to control for shared genetic, familial and environmental confounding factors and used stratified Cox regression with a separate stratum for each family identified by the mother’s unique identification number. Only sibling pairs with discordant exposure and outcome statuses were informative and contributed to the effect estimates.
We pooled the effect estimates from each data source in a meta-analysis using a random-effect model. Meta-analyses were represented in forest plots and the I2 statistic was used to quantify heterogeneity between sites. CIs not overlapping 1.0 were considered statistically significant. Statistical Analysis System (SAS) v9.4 (SAS Institute) and R Foundation for Statistical Computing version 3.6.0 were used for data analysis.
Sensitivity analyses
We conducted sensitivity analyses to test the validity and robustness of the study results. First, we repeated the main analysis in the entire mother-baby cohort, including offspring with less than 6 years of follow-up. Second, we stratified the analyses by offspring’s sex due to a higher prevalence of ADHD in males. Third, to assess the impact of any unmeasured confounders, we computed the E-value, which is defined as the minimum strength of association that an unmeasured confounder would need to have with both exposure and outcome, conditional on the measured covariates, to explain away an observed association78. Fourth, a post hoc sensitivity analysis including only mother-baby pairs with at least 9 years of follow-up was conducted to assess the effect of insufficient follow-up duration in our main analyses. Finally, Poisson and negative binomial regression models were also applied in the sensitivity analyses to test the robustness of our results in the presence of model uncertainty.
Ethics and inclusion statement
The study used healthcare data obtained from Hong Kong, New Zealand, Taiwan and Nordic countries. These data encompassed various sources such as electronic health records, registers, and insurance records. Each participating site followed the relevant local ethics and regulatory frameworks for study approval, namely the Finnish Institute for Health and Welfare (THL/1551/6.02.00/2018, THL/1673/5.05.00/2019) and the Social Insurance Institution of Finland (Kela 148/522/2018 and Kela 117/522/2019) in Finland; the University of Hong Kong/Hospital Authority Hong Kong West Cluster (UW20-051) in Hong Kong; the National Bioethics Committee (VSNb2018060017/03.01) in Iceland; the Norwegian Data Inspectorate (17/02068/Norwegian Data Inspectorate) and the Regional Committee for Medical and Health Research Ethics (2017/2546/REC South-East Norway) in Norway; the New Zealand Health and Disability Ethics Committee (13789) in New Zealand; the Swedish Ethical Review Authority (Dnr 2015/1826-31/2, 2017/2238-32, 2018/1790-32, 2018/2211-32, 2022-04004-02) in Sweden; and the National Cheng Kung University Human Research Ethics Committee (110-453) in Taiwan.
We fully endorse the Nature Portfolio journals’ guidance on authorship and inclusion. All collaborators of this study have fulfilled the criteria for authorship required by Nature Portfolio journals have been included as authors, as their participation was essential for the design and implementation of the study. Roles and responsibilities were agreed among collaborators ahead of the research. This work includes findings that are locally relevant, which have been determined in collaboration with local partners. This research was not severely restricted or prohibited in the setting of the researchers and does not result in stigmatization, incrimination, discrimination or personal risk to participants. Local and regional research relevant to our study was taken into account in citations.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The Hong Kong data used in this study will not be accessible to external parties, as the data custodians have not given permission due to concerns regarding patient privacy protection. Requests can be submitted to hacpaaedr@ha.org.hk (the Central Panel on Administrative Assessment of External Data Requests, the Hospital Authority, Hong Kong). It should be noted that the processing time for such requests may vary as the provided data will be customized for the specific purpose of each project. Individual-level data from the Nordic countries were used under license for the current study and cannot be made publicly available due to data privacy laws. The data are available from the data custodians of the health registers after obtaining the necessary permissions in Finland, Iceland, Norway and Sweden. Due to data privacy laws, individual-level data from the New Zealand cannot be make publicly available. The National Health Research Insurance Database of Taiwan can only be accessed at the Health and Welfare Data Center due to data privacy concerns.
Code availability
R and SAS codes and sample dataset adopted in this study are available on GitHub repository at https://github.com/legao513/DIAMOND-A (ref. 79).
References
Cho, N. H. et al. IDF Diabetes Atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 138, 271–281 (2018).
Yuen, L. et al. Projections of the prevalence of hyperglycaemia in pregnancy in 2019 and beyond: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 157, 107841 (2019).
Djelmis, J. et al. Prevalence of gestational diabetes mellitus according to IADPSG and NICE criteria. Int J. Gynaecol. Obstet. 135, 250–254 (2016).
Lavery, J. A., Friedman, A. M., Keyes, K. M., Wright, J. D. & Ananth, C. V. Gestational diabetes in the United States: temporal changes in prevalence rates between 1979 and 2010. BJOG 124, 804–813 (2017).
Alexopoulos, A. S., Blair, R. & Peters, A. L. Management of preexisting diabetes in pregnancy: a review. JAMA 321, 1811–1819 (2019).
Ornoy, A., Reece, E. A., Pavlinkova, G., Kappen, C. & Miller, R. K. Effect of maternal diabetes on the embryo, fetus, and children: congenital anomalies, genetic and epigenetic changes and developmental outcomes. Birth Defects Res. C. Embryo Today 105, 53–72 (2015).
Instanes, J. T. et al. Attention-deficit/hyperactivity disorder in offspring of mothers with inflammatory and immune system diseases. Biol. Psychiatry 81, 452–459 (2017).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (American Psychiatric Publishing, Inc., Arlington, VA, 2013).
Thomas, R., Sanders, S., Doust, J., Beller, E. & Glasziou, P. Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics 135, e994–e1001 (2015).
Sayal, K., Prasad, V., Daley, D., Ford, T. & Coghill, D. ADHD in children and young people: prevalence, care pathways, and service provision. Lancet Psychiatry 5, 175–186 (2018).
Fleming, M. et al. Educational and health outcomes of children treated for attention-deficit/hyperactivity disorder. JAMA Pediatr. 171, e170691 (2017).
Pastor, P. N. & Reuben, C. A. Identified attention-deficit/hyperactivity disorder and medically attended, nonfatal injuries: US school-age children, 1997-2002. Ambul. Pediatr. 6, 38–44 (2006).
Curry, A. E. et al. Motor vehicle crash risk among adolescents and young adults with attention-deficit/hyperactivity disorder. JAMA Pediatr. 171, 756–763 (2017).
Lee, S. S., Humphreys, K. L., Flory, K., Liu, R. & Glass, K. Prospective association of childhood attention-deficit/hyperactivity disorder (ADHD) and substance use and abuse/dependence: a meta-analytic review. Clin. Psychol. Rev. 31, 328–341 (2011).
Barkley, R. A., Fischer, M., Smallish, L. & Fletcher, K. Young adult follow-up of hyperactive children: antisocial activities and drug use. J. Child Psychol. Psychiatry 45, 195–211 (2004).
Dalsgaard, S., Mortensen, P. B., Frydenberg, M. & Thomsen, P. H. Long-term criminal outcome of children with attention deficit hyperactivity disorder. Crim. Behav. Ment. Health.: CBMH 23, 86–98 (2013).
Leibson, C. L., Katusic, S. K., Barbaresi, W. J., Ransom, J. & O’Brien, P. C. Use and costs of medical care for children and adolescents with and without attention-deficit/hyperactivity disorder. JAMA 285, 60–66 (2001).
Zhao, L. et al. The association of maternal diabetes with attention deficit and hyperactivity disorder in offspring: a meta-analysis. Neuropsychiatr. Dis. Treat. 15, 675–684 (2019).
Bytoft, B. et al. Assessment of attention deficits in adolescent offspring exposed to maternal type 1 diabetes. PLoS One 12, e0169308 (2017).
Nomura, Y. et al. Exposure to gestational diabetes mellitus and low socioeconomic status: effects on neurocognitive development and risk of attention-deficit/hyperactivity disorder in offspring. Arch. Pediatr. Adolesc. Med. 166, 337–343 (2012).
Schmitt, J. & Romanos, M. Prenatal and perinatal risk factors for attention-deficit/hyperactivity disorder. Arch. Pediatr. Adolesc. Med 166, 1074–1075 (2012).
Daraki, V. et al. Effect of parental obesity and gestational diabetes on child neuropsychological and behavioral development at 4 years of age: the Rhea mother–child cohort, Crete, Greece. Eur. Child Adolesc. Psychiatry 26, 703–714 (2017).
Hegvik, T. A., Instanes, J. T., Haavik, J., Klungsoyr, K. & Engeland, A. Associations between attention-deficit/hyperactivity disorder and autoimmune diseases are modified by sex: a population-based cross-sectional study. Eur. Child Adolesc. Psychiatry 27, 663–675 (2018).
Li, J., Zhong, H., Liang, Z. & Xy, L. J. J. C. E. M. Study on correlation between gestational diabetes merged pregnancy-induced hypertension and hyperactivity syndrome in children. J. Clin. Exp. Med. 13, 1583–1585 (2014).
Ji, J., Chen, T., Sundquist, J. & Sundquist, K. Type 1 diabetes in parents and risk of attention deficit/hyperactivity disorder in offspring: a population-based study in Sweden. Diabetes Care 41, 770–774 (2018).
Nielsen, P. R., Benros, M. E. & Dalsgaard, S. Associations between autoimmune diseases and attention-deficit/hyperactivity disorder: a nationwide study. J. Am. Acad. Child Adolesc. Psychiatry 56, 234–240 e231 (2017).
Wang, Z. et al. Advances in epidemiological methods and utilisation of large databases: a methodological review of observational studies on central nervous system drug use in pregnancy and central nervous system outcomes in children. Drug Saf. 42, 499–513 (2019).
Baughman, F. A. Jr. Diagnosis and evaluation of the child with attention-deficit/hyperactivity disorder. Pediatrics 107, 1239 (2001).
Man, K. K. C. et al. Prenatal antidepressant use and risk of attention-deficit/hyperactivity disorder in offspring: population based cohort study. BMJ 357, j2350 (2017).
Chan, A. Y. L. et al. Maternal benzodiazepines and Z-drugs use during pregnancy and adverse birth and neurodevelopmental outcomes in offspring: a population-based cohort study. Psychother. Psychosom. 92, 113–123 (2023).
Halfdanarson, O. et al. Antipsychotic use in pregnancy and risk of attention/deficit-hyperactivity disorder and autism spectrum disorder: a Nordic cohort study. Evid. Based Ment. Health 25, 54–62 (2022).
National Guideline Centre (UK). in Evidence reviews for risk factors for ADHD: attention deficit hyperactivity disorder: diagnosis and management: evidence review A (National Institute for Health and Care Excellence (NICE), London, 2018).
Xiang, A. H. et al. Maternal gestational diabetes mellitus, type 1 diabetes, and type 2 diabetes during pregnancy and risk of ADHD in offspring. Diabetes Care 41, 2502–2508 (2018).
Bjork, M. H. et al. Association of prenatal exposure to antiseizure medication with risk of autism and intellectual disability. JAMA Neurol. 79, 672–681 (2022).
Meng, L. C. et al. Association between maternal benzodiazepine or Z-hypnotic use in early pregnancy and the risk of stillbirth, preterm birth, and small for gestational age: a nationwide, population-based cohort study in Taiwan. Lancet Psychiatry 10, 499–508 (2023).
Wang, Z. et al. Association between prenatal exposure to antipsychotics and attention-deficit/hyperactivity disorder, autism spectrum disorder, preterm birth, and small for gestational age. JAMA Intern. Med. 181, 1332–1340 (2021).
Frisell, T. Invited Commentary: Sibling-comparison designs, are they worth the effort? Am. J. Epidemiol. 190, 738–741 (2021).
Cohen, J. M. et al. A common data model for harmonization in the Nordic Pregnancy Drug Safety Studies (NorPreSS). Norsk Epidemiol. 29, 117–123 (2021).
Lai, E. C. et al. Brief Report: Databases in the Asia-Pacific region: the potential for a distributed network approach. Epidemiology 26, 815–820 (2015).
Lai, E. C. et al. Applying a common data model to Asian databases for multinational pharmacoepidemiologic studies: opportunities and challenges. Clin. Epidemiol. 10, 875–885 (2018).
Lai, E. C. et al. Comparative safety of NSAIDs for gastrointestinal events in Asia-Pacific populations: a multi-database, international cohort study. Pharmacoepidemiol Drug Saf. 27, 1223–1230 (2018).
Christensen, J. et al. Association of prenatal exposure to valproate and other antiepileptic drugs with risk for attention-deficit/hyperactivity disorder in offspring. JAMA Netw. Open 2, e186606 (2019).
Ge, G. M. et al. Maternal thyroid dysfunction during pregnancy and the risk of adverse outcomes in the offspring: a systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 105, 3821–3841 (2020).
Kim, J. H. et al. Environmental risk factors, protective factors, and peripheral biomarkers for ADHD: an umbrella review. Lancet Psychiatry 7, 955–970 (2020).
Kosidou, K. et al. Maternal polycystic ovary syndrome and risk for attention-deficit/hyperactivity disorder in the offspring. Biol. Psychiatry 82, 651–659 (2017).
Nielsen, T. C. et al. Association of maternal autoimmune disease with attention-deficit/hyperactivity disorder in children. JAMA Pediatr. 175, e205487 (2021).
Sciberras, E., Mulraney, M., Silva, D. & Coghill, D. Prenatal risk factors and the etiology of ADHD: review of existing evidence. Curr. Psychiatry Rep. 19, 1 (2017).
Vizzini, L. et al. Maternal anxiety, depression and sleep disorders before and during pregnancy, and preschool ADHD symptoms in the NINFEA birth cohort study. Epidemiol. Psychiatr. Sci. 28, 521–531 (2019).
Wood, M. E. et al. Prenatal triptan exposure and internalising and externalising behaviour problems in 3-year-old children: results from the Norwegian Mother and Child Cohort Study. Paediatr. Perinat. Epidemiol. 30, 190–200 (2016).
Webster-Clark, M. et al. Using propensity scores to estimate effects of treatment initiation decisions: state of the science. Stat. Med. 40, 1718–1735 (2021).
Satman, I. et al. Twelve-year trends in the prevalence and risk factors of diabetes and prediabetes in Turkish adults. Eur. J. Epidemiol. 28, 169–180 (2013).
Li, Y. et al. Maternal age and the risk of gestational diabetes mellitus: a systematic review and meta-analysis of over 120 million participants. Diabetes Res. Clin. Pract. 162, 108044 (2020).
Gao, L., Li, S., Yue, Y. & Long, G. Maternal age at childbirth and the risk of attention-deficit/hyperactivity disorder and learning disability in offspring. Front Public Health 11, 923133 (2023).
Mowlem, F. D. et al. Sex differences in predicting ADHD clinical diagnosis and pharmacological treatment. Eur. Child Adolesc. Psychiatry 28, 481–489 (2019).
Russell, A. E., Ford, T., Williams, R. & Russell, G. The association between socioeconomic disadvantage and attention deficit/hyperactivity disorder (ADHD): a systematic review. Child Psychiatry Hum. Dev. 47, 440–458 (2016).
Roustaei, Z., Anttonen, S., Raisanen, S., Gissler, M. & Heinonen, S. Socioeconomic status, maternal risk factors, and gestational diabetes mellitus across reproductive years: a Finnish register-based study. BMJ Open Diabetes Res. Care 11, e003278 (2023).
Heinonen, K. et al. Behavioural symptoms of attention deficit/hyperactivity disorder in preterm and term children born small and appropriate for gestational age: a longitudinal study. BMC Pediatr. 10, 91 (2010).
Durlach, V. et al. Smoking and diabetes interplay: a comprehensive review and joint statement. Diabetes Metab. 48, 101370 (2022).
Huang, L. et al. Maternal smoking and attention-deficit/hyperactivity disorder in offspring: a meta-analysis. Pediatrics 141, e20172465 (2018).
Li, M. J. et al. Association of alcohol drinking with incident type 2 diabetes and pre-diabetes: the Guangzhou Biobank Cohort Study. Diabetes Metab. Res. Rev. 38, e3548 (2022).
Eilertsen, E. M. et al. Maternal alcohol use during pregnancy and offspring attention-deficit hyperactivity disorder (ADHD): a prospective sibling control study. Int J. Epidemiol. 46, 1633–1640 (2017).
Tao, H. et al. Psychiatric disorders and type 2 diabetes mellitus: a bidirectional Mendelian randomization. Eur. J. Clin. Invest. 53, e13893 (2023).
Wilson, C. A. et al. Systematic review and meta-analysis of risk of gestational diabetes in women with preconception mental disorders. J. Psychiatr. Res. 149, 293–306 (2022).
Yu, M., Zhang, X., Lu, F. & Fang, L. Depression and risk for diabetes: a meta-analysis. Can. J. Diabetes 39, 266–272 (2015).
Wang, H. et al. Maternal hypertensive disorders and neurodevelopmental disorders in offspring: a population-based cohort in two Nordic countries. Eur. J. Epidemiol. 36, 519–530 (2021).
Shero, N. & Pandeya, D. R. Impact of Crohn’s disease during pregnancy on children with attention deficit hyperactivity disorder: a review. Ann. Med Surg. (Lond.) 75, 103369 (2022).
Tarar, Z. I. et al. A national study of pregnancy-related maternal and fetal outcomes in women with inflammatory bowel disease. Int J. Colorectal Dis. 37, 1535–1543 (2022).
Ellul, P. et al. Children with a history of both maternal immune activation and prematurity are not at increased risk of ADHD symptoms. Eur. Child Adolesc. Psychiatry https://doi.org/10.1007/s00787-023-02276-8 (2023).
Zhang, X., Wu, X., Chen, L. & He, L. Autoimmune diseases and risk of gestational diabetes mellitus: a Mendelian randomization study. Acta Diabetol. 61, 161–168 (2024).
Biondi, B., Kahaly, G. J. & Robertson, R. P. Thyroid dysfunction and diabetes mellitus: two closely associated disorders. Endocr. Rev. 40, 789–824 (2019).
Drover, S. S. M. et al. Maternal thyroid function during pregnancy or neonatal thyroid function and attention deficit hyperactivity disorder: a systematic review. Epidemiology 30, 130–144 (2019).
Liu, Q. et al. A genome-wide cross-trait analysis identifies shared loci and causal relationships of type 2 diabetes and glycaemic traits with polycystic ovary syndrome. Diabetologia 65, 1483–1494 (2022).
Maleki, A., Bashirian, S., Soltanian, A. R., Jenabi, E. & Farhadinasab, A. Association between polycystic ovary syndrome and risk of attention-deficit/hyperactivity disorder in offspring: a meta-analysis. Clin. Exp. Pediatr. 65, 85–89 (2022).
Li, L. et al. Maternal pre-pregnancy overweight/obesity and the risk of attention-deficit/hyperactivity disorder in offspring: a systematic review, meta-analysis and quasi-experimental family-based study. Int J. Epidemiol. 49, 857–875 (2020).
Desai, R. J., Rothman, K. J., Bateman, B. T., Hernandez-Diaz, S. & Huybrechts, K. F. A propensity-score-based fine stratification approach for confounding adjustment when exposure is infrequent. Epidemiology 28, 249–257 (2017).
Desai, R. J. & Franklin, J. M. Alternative approaches for confounding adjustment in observational studies using weighting based on the propensity score: a primer for practitioners. BMJ 367, l5657 (2019).
Austin, P. C. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun. Stat. Simul. Comput. 38, 1228–1234 (2009).
VanderWeele, T. J. & Ding, P. Sensitivity analysis in observational research: introducing the E-value. Ann. Intern. Med. 167, 268–274 (2017).
legao513. DIAMOND-A. GitHub https://github.com/legao513/DIAMOND-A (2024).
Acknowledgements
We acknowledge the support from an Australian National Health and Medicine Research Council (NHMRC) – European Union Collaborative Research Grant (NHMRC, grant agreement number APP2007048) and the Taiwan National Science and Technology Council, Taiwan National Health Research Institutes, and Taiwan Health and Welfare Data Science Center. This work was supported by the General Research Fund of Hong Kong Research Grants Council (grant 17112020), NordForsk as part of the Nordic Pregnancy Drug Safety Studies (NorPreSS), project 83539; the Research Council of Norway as part of the International Pregnancy Drug Safety Studies (InPreSS), project 273366; and ADHD Research Network of the Norwegian Centre of Expertise for Neurodevelopmental Disorders and Hypersomnias (NevSom), Oslo University Hospital (project 51379). The study was also partly supported by the Research Council of Norway through its Centres of Excellence funding scheme, project 262700; the Taiwan National Science and Technology Council (ID: 112-2628-B-006-003-) (107-2320-B-006-070-MY3) and the Taiwan National Health Research Institutes (NHRI-11A1-CG-CO-04-2225-1); an Australian National Health and Medical Research Council (NHMRC)-European Union (EU) Collaborative Research Grant (ID: APP2007048). The current work was conducted in collaboration with the TIMESPAN research project, which is funded by the European Union’s Horizon 2020 Research and Innovation Programme (Management of chronic cardiometabolic disease and treatment discontinuity in adult ADHD patients, ID: 965381). H.Z. was supported by a UNSW Scientia Program Award during the conduct of the study. A.Y.L.C. is supported by a grant from the Innovation and Technology Commission of the Hong Kong Special Administrative Region Government. C.E.C. was supported by the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement 844728. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
A.Y.L.C., L.G., I.C.K.W. and K.K.C.M. designed the study. A.Y.L.C. and L.G. did the analysis in Hong Kong, M.H.C.H. and T.C.L. did the analysis in Taiwan, J.H. did the analysis in New Zealand and L.J.K. and C.E.C. did the analysis in Nordic countries. A.Y.L.C. and L.G. wrote the first draft of the paper. All other authors critically reviewed and interpreted the paper. C.E.C., E.C.C.L., H.Z., K.K.C.M. and I.C.K.W. are the principal investigators, providing resources and supervising all steps of this project. All authors contributed to the interpretation, review and editing the paper, and approved the submission of the final version.
Corresponding authors
Ethics declarations
Competing interests
A.H.Y.C. receives research funding from Health Research Council NZ, Oakley Mental Health Foundation, NZ Pharmacy Education and Research Foundation, Ministry of Health, World Health Organisation and is the recipient of fellowships from the Robert Irwin Foundation and Auckland Medical Research Foundation, unrelated to the submitted work. She is also affiliated with the Asthma UK Centre of Applied Research and is on the Board of Asthma NZ and Pharmacy Council of New Zealand. D.C. reports research funding outside the submitted work from the Australian National Health and Medical Research Council, speaker’s fees and honoraria from Novartis, Medice, Servier and Shire/Takeda and royalties from Oxford University Press and Cambridge University Press in the past 3 years. M.G. and M.K.L. report that they received grants from the Innovative Medicines Initiative (Building an ecosystem for better monitoring and communicating the safety of medicines’ use in pregnancy and breastfeeding: validated and regulatory endorsed workflows for fast, optimized evidence generation, IMI ConcePTION, grant agreement number 821520) while conducting the study. J.H. receives research funding from the Health Research Council NZ, Lotteries Health Research (New Zealand), New Zealand Ministry of Health and New Zealand Health Quality Safety Commission unrelated to the submitted work. P.I. reports research funding from the Hong Kong Research Grants Council, Health and Medical Research Fund and Hong Kong Jockey Club Charities Trust. Ø.K. reports participation in regulator-mandated post-authorization safety studies (PASS) of drugs with no relation to the work reported in this paper. The studies are funded by Leo Pharma and Novo Nordisk, with funds paid to the institution where he is employed (no personal fees). W.C.Y.L. reports research grants from Diabetes UK, AIR@InnoHK administered by Innovation and Technology Commission outside the submitted work. J.R. and C.E.C. are employees of the Centre for Pharmacoepidemiology at Karolinska Institutet, which receives funding from pharmaceutical companies and regulatory authorities for drug safety/utilization studies, unrelated to the submitted work. E.S. receives research funding from the UK National Institute of Health Research, United Kingdom Research and Innovation, and the European Innovative Medicines Initiative. E.C.-C.L. reports research funding outside the submitted work from Amgen, Pfizer, Sanofi, Takeda, Roche, IQVIA. H.Z. was supported by a UNSW Scientia Program Award and reports grants from the European Union Horizon 2020, Australian National Health and Medical Research Council (NHMRC), Icelandic Centre for Research, NordForsk Nordic Council of Ministers during the conduct of this study. K.K.C.M. is the recipient of the CW Maplethorpe Fellowship, reports grants from the European Union Horizon 2020, the UK National Institute of Health Research and the Hong Kong Research Grant Council, Hong Kong Innovation and Technology Commission, and reports personal fees from IQVIA unrelated to the submitted work. I.C.K.W. received research grants from Amgen, Janssen, GSK, Novartis, Pfizer, Bayer and Bristol-Myers Squibb and Takeda, Institute for Health Research in England, European Commission, National Health and Medical Research Council in Australia, The European Union’s Seventh Framework Programme for research, technological development, Research Grants Council Hong Kong and Health and Medical Research Fund Hong Kong; consulting fees from IQVIA and World Health Organization; payment for expert testimony for Appeal Court in Hong Kong; serves on advisory committees for Member of Pharmacy and Poisons Board; is a member of the Expert Committee on Clinical Events Assessment Following COVID-19 Immunization; is a member of the Advisory Panel on COVID-19 Vaccines of the Hong Kong Government; is the non-executive director of Jacobson Medical in Hong Kong; and is the founder and director of Therakind Limited (UK), Advance Data Analytics for Medical Science (ADAMS) Limited (HK), Asia Medicine Regulatory Affairs (AMERA) Services Limited and OCUS Innovation Limited (HK, Ireland and UK). A.Y.L.C. is supported by the AIR@innoHK programme of the Hong Kong Innovation and Technology Commission. L.G., M.H.-C.H., L.J.K., R.A., T.B., J.M.C., W.C.L., T.-C.L., S.-C.S., K.C.B.T., K.T. and A.T. declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks David Cochran, Frederick Ho, Eric Ohuma and the other, anonymous, reviewer(s), for their contribution to the peer review of this work. Primary Handling Editor: Sonia Muliyil, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Sample size and power considerations.
Notes: Results are rounded up to the nearest integer. Abbreviations: CC, continuity correction.
Extended Data Fig. 2 General definition of exposure groups.
Abbreviations: GDM, gestational diabetes mellitus; MDM, maternal diabetes mellitus; PGDM, pregestational diabetes mellitus.
Extended Data Fig. 3 Cumulative incidence of ADHD in different comparison groups from the main analyses.
Abbreviations: ADHD, attention-deficit/hyperactivity disorder; MDM, maternal diabetes mellitus.
Extended Data Fig. 4 Results of comparisons between different MDM types.
Notes: Data are presented as hazard ratios and 95% CIs, which were adjusted for demographics, socioeconomic status, birth year, multifoetal pregnancies, maternal conditions and use of relevant medications using Cox proportional hazard regression, with a significance level of 5% for a two-sided test. No adjustments were made for multiple comparisons. Abbreviations: CI, confidence interval; df, degrees of freedom; GDM, gestational diabetes mellitus; IV, inverse variance; MDM, maternal diabetes mellitus; PGDM, pregestational diabetes mellitus; T1DM, type 1 pregestational diabetes mellitus; T2DM, type 2 pregestational diabetes mellitus; SE, standard error.
Extended Data Fig. 5 Directed acyclic graph related to the analyses.
Notes: * including diagnosis or medication prescription; psychiatric and neurological conditions: including ASD, anxiety disorders, bipolar disorders, depression, disorders of psychological development, epilepsy, illicit drug use, intellectual disability, personality disorders, schizophrenia, sleep disorders; other chronic medical conditions: including cluster headache, crohn’s disease and ulcerative colitis, hypertension, migraine or other headaches, polycystic ovary syndrome, renal disease, rheumatoid arthritis and other inflammatory polyarthropathies, thyroid disorders; Medications for psychiatric and neurological conditions: including antihypertensives, antipsychotics, antidepressants, antiepileptics, antiparkinson drugs, anxiolytics, sedatives, opioids, triptans; socioeconomic status: defined by income level, birth institution or maternal education according to each data source.
Supplementary information
Supplementary Information
Supplementary Tables 1–8.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chan, A.Y.L., Gao, L., Hsieh, M.HC. et al. Maternal diabetes and risk of attention-deficit/hyperactivity disorder in offspring in a multinational cohort of 3.6 million mother–child pairs. Nat Med 30, 1416–1423 (2024). https://doi.org/10.1038/s41591-024-02917-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-024-02917-8