Abstract
HIVACAT T-cell immunogen (HTI) is a novel human immunodeficiency virus (HIV) vaccine immunogen designed to elicit cellular immune responses to HIV targets associated with viral control in humans. The AELIX-002 trial was a randomized, placebo-controlled trial to evaluate as a primary objective the safety of a combination of DNA.HTI (D), MVA.HTI (M) and ChAdOx1.HTI (C) vaccines in 45 early-antiretroviral (ART)-treated individuals (44 men, 1 woman; NCT03204617). Secondary objectives included T-cell immunogenicity, the effect on viral rebound and the safety of an antiretroviral treatment interruption (ATI). Adverse events were mostly mild and transient. No related serious adverse events were observed. We show here that HTI vaccines were able to induce strong, polyfunctional and broad CD4 and CD8 T-cell responses. All participants experienced detectable viral rebound during ATI, and resumed ART when plasma HIV-1 viral load reached either >100,000 copies ml−1, >10,000 copies ml−1 for eight consecutive weeks, or after 24 weeks of ATI. In post-hoc analyses, HTI vaccines were associated with a prolonged time off ART in vaccinees without beneficial HLA (human leukocyte antigen) class I alleles. Plasma viral load at the end of ATI and time off ART positively correlated with vaccine-induced HTI-specific T-cell responses at ART cessation. Despite limited efficacy of the vaccines in preventing viral rebound, their ability to elicit robust T-cell responses towards HTI may be beneficial in combination cure strategies, which are currently being tested in clinical trials.
Similar content being viewed by others
Main
Therapeutic vaccines designed to enhance human immunodeficiency virus (HIV)-specific T-cell immunity have been postulated to be a key component of any HIV cure strategy1. Different therapeutic vaccine candidates have been shown to be safe, immunogenic and able to induce broad and functional T- and B-cell immune responses2,3,4,5. However, no reduction in HIV-1 viral reservoirs, prevention of viral rebound or suppressed viremia off ART have been reported in randomized, placebo-controlled trials of vaccines, given alone or in combination with latency-reversing agents5,6,7.
One potential reason for these suboptimal trial outcomes may have been T-cell immunogen designs and the induction of virus-specific T-cell responses with ineffective or insufficient antiviral activity. To overcome this, HTI (HIVACAT T-cell immunogen)-based vaccines were designed to induce functional HIV-1-specific T-cell responses that were associated with better viral control in more than 1,000 HIV-1 clade B and C infected individuals within a broad HLA (human leukocyte antigen) class I and class II allele coverage8 targeting the most vulnerable sites of HIV-1. The HTI immunogen includes 16 HIV-1 regions from Gag, Pol, Nef and Vif that induce T-cell responses of high functional avidity and cross-reactivity and target regions of overall low diversity/entropy, even though these regions were not predicted by stringent conservation algorithms, but were based on human trial data9,10. Importantly, in independent cohorts of viremic controllers and individuals with breakthrough infection after being vaccinated with full-length proteins, recognition of viral protein segments covered by HTI were found to be generally subdominant, but, when detected, were associated with better viral control and viral inhibition of clade-matched HIV isolates11. The 16 identified HIV-1 regions were assembled in a 529aa immunogen sequence (HTI) and expressed both in a plasmid DNA (DNA.HTI, D)12 and two viral-vectored vaccines based on a modified Vaccinia virus Ankara (MVA.HTI, M)13 and a chimpanzee adenovirus (ChAdOx1.HTI, C)14.
AELIX-002 was a phase I, first-in-human, randomized, double-blind, placebo-controlled study to evaluate the safety, immunogenicity and effect on viral rebound of DNA.HTI, MVA.HTI and ChAdOx1.HTI HIV-1 vaccines administered in a heterologous prime-boost regimen to 45 virally suppressed, early-treated individuals with HIV-1 infection.
Results
A total of 45 participants (44 men and 1 woman), virologically suppressed for at least one year, were recruited from an existing Early-ART cohort15. Acute/recent infection at ART initiation was confirmed based on any of the following criteria: (1) positive plasma HIV-1 RNA with negative serology, (2) positive Gag p24 antigen; (3) indeterminate western blot; (4) absence of the p31 band in a positive western blot in the context of a known exposure/reported acute retroviral syndrome; and/or (5) negative HIV antibody test <24 weeks from the first positive test and before starting ART. Participants were randomized 2:1 to receive vaccines or placebo. DNA.HTI or placebo were given at weeks 0, 4 and 8 and MVA.HTI or placebo were given at weeks 12 and 20. All participants completed the first vaccination regimen (DDDMM (n = 30) or placebo (n = 15)). Of them, 42 reconsented to start a second vaccination regimen after a favorable report from the safety monitoring committee (SMC) once the last participant had reached week 32 of the follow-up. The second vaccination regimen started after a minimum of 24 weeks from last MVA.HTI or placebo vaccination. Participants received ChadOx.HTI or placebo at weeks 0, 12 and MVA.HTI or placebo at week 24. Finally, 41 participants (CCM (n = 26) or placebo (n = 15)) entered an analytical treatment interruption (ATI) eight weeks after completing the last series of vaccination (CCM or placebo; Fig. 1).
Demographics
Table 1 presents the baseline characteristics. ART was initiated after a median (range) of 55 (12–125) and 64 (6–140) days after the estimated date of HIV-1 acquisition in placebo and vaccine recipients, respectively. All participants were receiving an integrase strand transfer inhibitor (INSTI)-based ART regimen at inclusion. Median (range) time with undetectable viral load at enrollment was 18 (13–56) and 27 (12–55) months, and median CD4+ T-cell counts (range) were 826 (549–2,156) and 727 (553–1,336) cells per mm3 in the placebo and in the vaccine group, respectively (not significant for all parameters). Three placebo (20%) and seven (23%) vaccine recipients expressed any HLA class I allele associated with spontaneous control of HIV replication, respectively (that is HLA-B*27:05, -B*57:01, -B*15:17 and/or -B*15:03). In addition, six (40%) placebo recipients and nine (30%) vaccinees expressed HLA class I alleles associated with HIV disease progression (that is, HLA-B*07:02, -B*08:01, -B*35:01/02/03, -B*53:01 and/or -B*54/55/56)16.
Pre-ART HIV-1 viral sequencing
Full-genome deep sequencing was performed on HIV-1 viral sequences isolated within the first four weeks of ART initiation from 41 participants. Of the 41 participants, 32 (78%) had subtype B viruses. Phylogenetic distance to a reference sequence (HXB2) and the coverage by the HTI immunogen were comparable between placebo and vaccine recipients for any of the HIV-1 proteins included in the HTI immunogen (Extended Data Fig. 1a–c). The median (range) number of pre-ART CTL (cytotoxic T lymphocyte) escape mutants within sequences included in the HTI immunogen was 7 (2–11) and 5 (2–8) in the placebo and vaccine recipients, respectively (Mann–Whitney, P = 0.0364; Extended Data Fig. 1d). The degree of pre-ART CTL escape in HTI-covered regions was not associated with replication fitness of the participants’ autologous virus (Extended Data Fig. 1e).
Safety
The severity and intensity of adverse events (AEs) were assessed by the investigator according to the Division of DAIDS table for grading the severity of adult and pediatric adverse events, version 2.1 (March 2017). Overall, vaccines were safe and well-tolerated (Extended Data Table 1). All participants reported solicited AEs related to vaccinations, which were mostly mild (grade 1–2) and transient, except one participant who reported grade 3 asthenia lasting <72 h after the third MVA.HTI vaccination. A total of 440 related AEs were recorded during the entire vaccination phase (111 in placebo and 329 in vaccine recipients), of which 76 and 229 occurred after placebo or DDDMM administrations and 35 and 100 after placebo or CCM (Supplementary Tables 1–4). The most frequent AEs related to vaccinations were pain at the injection site and a flu-like syndrome. There were only two serious adverse events (SAEs) during the study—an episode of acute infectious gastroenteritis due to Campylobacter jejuni and an acute appendicitis that required hospitalization, both in vaccine recipients (Extended Data Table 2). No laboratory abnormalities related to vaccinations were reported.
Immunogenicity
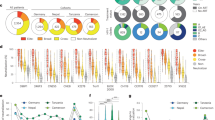
Total HIV-1 and HTI-specific T cells were assessed by an ex vivo interferon (IFN)-γ-detecting enzyme-linked immunosorbent spot (ELISpot) assay. Both vaccination regimens (DDDMM and CCM) were immunogenic. The median (range) increase in the total frequencies of HTI-specific T cells from baseline to the peak immunogenicity timepoint after the overall vaccination regimen was 100 (0–498) spot-forming cells (SFCs) per million peripheral blood mononuclear cells (PBMCs) in the placebo group and 1,499 (120–3,150) in the vaccine group (Mann–Whitney t-test, P < 0.0001; Fig. 2a and Extended Data Table 3). This corresponded to an increase in HTI magnitude of more than twofold in ten (67%) and more than threefold in one (7%) of the placebo recipients compared to 29 (97%) and 24 (80%) of vaccine recipients (Fisher’s exact test, P = 0.0117 and P < 0.0001, respectively; Extended Data Table 3). To determine the breadth of vaccine-induced T-cell responses, PBMCs obtained at study entry and after DDDMM and CCM or placebo were expanded in vitro and tested against individual 15-mer overlapping peptides (OLPs) covering the HTI immunogen (n = 147). A cumulative breadth over the entire vaccination period of a median (range) of 5 (1–13) IFN-γ-producing responses to individual HTI-covered OLPs was detected in vaccinees without any specific pattern of immunodominance across the HIV subproteins covered by the HTI immunogen, in contrast to 3 (1–8) and predominantly gag-specific responses in placebo recipients (Mann–Whitney t-test, P = 0.0125; Fig. 2b,c). Responses to HTI were already present in 31 participants (20 vaccine and 11 placebo recipients) before ART was initiated. The maximal magnitude of HTI-specific responses achieved during the intervention phase positively correlated with the magnitude of pre-ART HTI-specific T-cell responses (Spearman’s ρ = 0.5343, P = 0.0024 and ρ = 0.4632, P = 0.0147 for vaccine recipients at their peak immunogenicity timepoints after DDDMM or CCM, respectively; Extended Data Fig. 2a). Although the HTI magnitude at the peak immunogenicity timepoint was higher after DDDMM in vaccinees with pre-ART HTI-specific responses compared to those without any HTI-detectable responses before ART initiation (median (range) of 2,203 (460–3,200) versus 808 (60–1,595) SFCs per million PBMCs, Mann–Whitney t-test, P = 0.0380), these differences were no longer statistically significant at ATI initiation (median (range) of 795 (165–2,705) versus 595 (50–980) SFCs per million PBMCs, Mann–Whitney t-test, P = 0.1012; Extended Data Fig. 2b). To determine whether HTI vaccination was able to shift the focus of the virus-specific T cells, the percentage of HTI-specific T-cell frequencies divided by the total HIV-1 proteome-specific T-cell frequencies was calculated at each timepoint. At the time of ATI start, the median (range) of 14% (0–50) versus 67% (0–100) of the total anti-HIV-1 T-cell response was HTI-specific in placebo and vaccine recipients, respectively (Mann–Whitney t-test P < 0.001; Fig. 2d).
a, Magnitude (sum of SFCs per 106 PBMCs for HTI pools P1–P10) over the AELIX-002 study in placebo (blue) and vaccine (red) recipients over the two vaccination regimens (DDDMM/PPPPP and CCM/PPP) up to the start of the ATI period. b, Breadth of vaccine-elicited responses towards individual OLPs spanning the entire HTI sequence in the 15 placebo and 30 vaccine recipients. Boxplots represent the median and IQR, and the P values correspond to comparisons between the indicated timepoints using the Wilcoxon signed-rank test. c, Distribution of HTI-specific responses within the different HIV-1 subproteins included in the HTI immunogen of cumulative breadth at AELIX-002 study entry (top) and after completion of the last series of vaccinations (bottom) for each placebo (P1 to P15) and vaccine (V1 to V26) recipient. d, Average distribution of total HIV-1 T cells according to their specificity at the indicated timepoints. HTI-specific responses are shown for placebo (blue) and vaccine (red) recipients. The other non-HTI HIV-1 specific responses are shown in gray. P values correspond to a comparison between the proportion of HTI-specific responses at each timepoint. Fisher’s exact test is used for comparisons between groups. e, Proportion of HTI-specific CD4+ and CD8+ T cells secreting IFN-γ (left) or both IFN-γ and GzmB (right) after completion of the last series of HTI vaccinations (DDDMM-CCM/PPPP-PPP). Data are presented as median and IQR for the sum of IFN-γ+ and IFN-γ+/GzmB+ for each of the four HTI peptide pool stimulations. A Wilcoxon–Mann–Whitney test is used for comparison between placebo (n = 12) and vaccine (n = 20) groups. f, Polyfunctionality of HTI-specific CD4+ and CD8+ T cells analyzed by Boolean gating. Pie charts and boxplots per treatment group (placebo n = 15, vaccine n = 26) illustrate the relative and absolute proportion of each of the different subsets (cells producing two, three or four cytokines), respectively. On each boxplot, the central line indicates the median, and the bottom and top edges of the box indicate the 25th and 75th percentiles, respectively. The whiskers extend to 1.5 times the IQR. P-values correspond to the Mann–Whitney test per row, adjusted for multiple comparisons. g, Changes in viral inhibition capacity to laboratory-adapted HIV-1 strains (placebo n = 15, vaccine n = 26) and autologous HIV-1 (placebo n = 14, vaccine n = 23) at study entry, after DDDMM/PPPPP and CCM/PPP regimens for placebo (blue) and vaccine (red) recipients. Boxplots represent median and IQR, and the P values correspond to comparisons between the indicated timepoints using the Wilcoxon signed-rank test. SCR, screening; BSL, baseline; D, DNA.HTI; M, MVA.HTI; C, ChAdOx1.HTI; P, placebo.
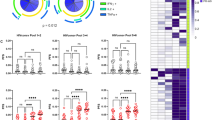
To further characterize the vaccine-induced T cells, intracellular cytokine staining for IFN-γ, GranzymeB (GzmB), interleukin-2 (IL-2) and tumor necrosis factor-α (TNF-α) was performed in samples obtained four weeks after the last CCM or placebo vaccination (week 28) with or without in vitro stimulation with four different peptide pools covering the HTI immunogen. T-cell lineage, phenotype, activation and exhaustion surface markers were included in the panel. The results showed that HTI-specific responses, defined as the sum of the HTI-IFN-γ+ populations for each of the four HTI peptide pool stimulations, were both CD4 and CD8 T-cell-mediated (Fig. 2e). Polyfunctionality analyses showed that, compared to placebo recipients, vaccinees had a higher frequency of bi and three-function CD8 T cells expressing IFN-γ/GzmB or IFN-γ/GzmB/TNF-α, whereas CD4 T cells predominantly expressed combinations of IL-2, IFN-γ and TNF-α (Fig. 2f). Importantly, and despite the intense vaccination regimen used in the study (DDDMM-CCM), T-cell exhaustion markers were not increased in HTI-specific T cells in vaccinees compared to placebo recipients after completing the last series of vaccination (Supplementary Table 5).
Finally, we measured the in vitro antiviral capacity of CD8+ T cells by a standard viral inhibition assay (VIA)17 using autologous CD4+ T cells infected with two laboratory-adapted HIV-1 strains (BaL (R5 tropic virus) and IIIB (X4 tropic virus)) as well as with the autologous HIV virus. Median (interquartile range (IQR)) percentages of inhibition of the BaL-isolate increased in the vaccine group from 46(17; 75)% at baseline to 75(9; 88)% at the end of the intervention (Wilcoxon t-test, P = 0.0805), but it remained unchanged in the placebo group (34 (17; 60)% at baseline and 37 (14; 63)% at the end of the intervention (Wilcoxon t-test, P = 0.9153)). When using IIIB viruses and a participant’s autologous viruses, significant changes in VIA were detected as well (Wilcoxon t-test, P = 0.0014 and 0.0176) in vaccinees in contrast to placebo recipients. However, absolute increases in viral inhibition capacity were of minor magnitude, probably due to the high inhibition capacity against the autologous virus already present at study entry, and consistent with early treatment initiation (Fig. 2g).
Effect on viral rebound during an ATI
Of the participants, 41 (15 placebo and 26 vaccine recipients) interrupted ART and were monitored weekly for a maximum of 24 weeks. Criteria for ART resumption included a single HIV-1 plasma viral load (pVL) of >100,000 copies ml−1, eight consecutive determinations of >10,000 copies ml−1, two repeated CD4+ cell counts of <350 cells mm−3 and/or development of grade 3 or higher-severity clinical symptoms suggestive of an acute retroviral syndrome (ARS)—whichever appeared first. The ATI period partially overlapped with the first COVID-19 outbreak in Spain, with a State of Alarm declared from 16 March 2020 to 20 June 2020. Risk mitigation strategies were quickly implemented during the pandemic to reduce premature withdrawals while ensuring participants’ safety. ATI was tolerated well overall (Supplementary Table 6). The frequency of sexually transmitted infections (STIs) in the study population was similar to those previously reported in MSM (men who have sex with other men)18, but importantly was relatively lower during the ATI period than during the intervention phase of the study (7 versus 17 cases of STI per 100 persons per year, respectively). Viral suppression to undetectable levels was achieved by the 12th week after ART resumption in all 35 participants assessed at the end-of-study visit.
As shown in Fig. 3a,b, pVL rebound (defined as pVL > 50 copies ml−1) was detected in all 41 participants after ART discontinuation at a median (range) time of 2 (1–6) and 3 (1–9) weeks in placebo and vaccine recipients, respectively (Mann–Whitney t-test, P = 0.1942). Time to pVL rebound, peak viremia, time to peak viremia, slope of increasing pVL or area under the curve (AUC) pVL during the ATI were comparable between placebo and vaccine recipients (Extended Data Table 4). Of the participants, 25 (61%) resumed ART after one determination of pVL > 100,000 copies ml−1, and one (2%) participant after eight consecutive determinations of >10,000 copies ml−1. Three participants (one in the placebo and two in the vaccine group) showed symptoms compatible with ARS, but they were grade 1–2 and did not proceed to ART resumption. Four (9%) participants resumed ART at weeks 9, 12, 22 and 23 of ATI without reaching any pre-specified ART resumption criteria in the context of the COVID-19 pandemic (details are provided in Supplementary Table 7). Eleven (27%) participants completed 24 weeks of ATI, seven of them with sustained pVL < 2,000 copies ml−1. Five participants resumed ART at week 24, and the remaining six participants (two placebo and four vaccine recipients) opted to remain off ART and entered an ATI-extension protocol with monthly monitoring for up to a total of 72 weeks of ATI (NCT04385875). Four participants (one placebo and three vaccine recipients) completed the ATI extension with sustained pVL < 2,000 copies ml−1 after 72 weeks off ART (Extended Data Fig. 3), and then resumed ART. Reasons for starting ART included worries about HIV transmission, previous good tolerability to ART and the burden of additional HIV prevention tools required for viremic individuals. In a post-hoc survival analysis for time off ART during the ATI, participants without any beneficial HLA class I alleles (32 of the 41 participants that entered the ATI period), one (8%) of the placebo and eight (40%) of the vaccine recipients were able to remain off ART for 22 weeks (∆ 32%, 80% confidence interval (CI) (7.6; 55.7) and 95% CI (−1.6; 64.9); log-rank test P = 0.1834 for all ATI), with pVL < 2,000 copies ml−1 being observed in one placebo and five vaccine recipients, respectively (Fig. 3c)
a,b, Individual HIV-1 pVL during the 24 weeks of ATI, shown for all placebo (blue) or vaccine (red) recipients (a) and in those without any beneficial HLA associated with spontaneous viral control (b). Lines are interrupted at the week of ART resumption. Dotted lines represent the detection limit and the two different virologic thresholds for ART resumption (10,000 and 100,000 HIV-1 RNA copies per ml, respectively). c, Proportion of participants without any beneficial HLA allele associated with spontaneous viral control in the placebo and vaccine arms remaining off ART following treatment interruption. The log-rank test is used for comparison between groups over the entire ATI period. The proportion of participants, delta and 80% CI are shown for week 22 of ATI, before the last two vaccine recipients resumed ART for COVID-19-related reasons without fulfilling any per-protocol virological criteria. pVL, plasma viral load; ART, antiretroviral treatment.
Exploratory objectives
Reservoir
Amplicon signal issues occurred for six (14%) participants (three placebo and three vaccine recipients) for whom intact proviral DNA assay (IPDA) determinations were not available. Intact HIV-1 DNA represented a median (IQR) of 23% (9; 42) of the total HIV-1 DNA. Total and intact proviral HIV-1 DNA were highly correlated (Spearman’s ρ = 0.6673, P < 0.0001 at study entry and ρ = 0.8716, P < 0.0001 at ATI start). No differences in reservoir decay were found between groups, either measured by total proviral HIV-1 DNA (21% versus 16% decay in the placebo and vaccine groups, respectively, Wilcoxon t-test, P = 0.4291) or by IPDA (68% versus 66% decay in the placebo and vaccine groups, respectively, Wilcoxon t-test, P = 0.7892) (Extended Data Fig. 4).
Correlate analyses
Potential immune and viral correlates associated with longer time off ART (that is, less risk to reach the ART resumption criteria of HIV-1 pVL > 100,000 or consecutive HIV-1 pVL > 10,000 for more than eight weeks) were assessed in the subgroup of individuals that did not harbor any HLA class I allele associated with spontaneous HIV control. The magnitude of the HTI-specific T-cell response at ATI start was significantly associated with both prolonged time off ART and with lower pVL at the end of ATI in vaccinees (Spearman’s ρ = 0.6469, P = 0.0021 and ρ = −0.6837, P = 0.0009, respectively; Fig. 4a,b) but not in placebo recipients. Similarly (albeit not statistically significant), the cumulative breadth of HTI-specific responses at ATI start was associated with longer time off ART (Spearman’s ρ = 0.4235, P = 0.0628; Supplementary Fig. 1). In terms of specificities within HTI, for those vaccinees remaining off ART for longer than 12 weeks (n = 8), we did not observe differences in the pattern of responses induced across the different HIV protein segments covered by HTI (Supplementary Fig. 1).
a–f, Correlation between time off ART (left) and HIV-1 pVL at the end of ATI at the ART resumption timepoint (right) for HTI magnitude at ATI start (a,b) and proportion of CD8+ (c,d) and CD4+ (e,f) GzmB-secreting T cells in placebo (blue) and vaccine (red) recipients. Spearman’s correlation is used. ART, antiretroviral treatment; pVL, plasma viral load; ATI, analytical treatment interruption.
As for T-cell functionality, the frequency of CD8+—and to a lesser extent CD4+—T cells expressing GzmB+ was positively correlated with time off ART and with lower HIV-1 pVL at the end of ATI in vaccine, but not in placebo recipients (Fig. 4c–f). Although vaccinees showed an increase in in vitro viral inhibition capacity, this was not associated with any of the ATI outcomes. As for viral factors, we ruled out the possibility that pre-existing CTL escape in sequences covered by HTI immunogen and/or replication fitness of the participants’ autologous virus could have influenced the ability of vaccine-induced responses to control virus replication during ATI. Vaccine recipients that remained off ART for longer periods of time did not show any significant correlation with the number of HLA-adapted footprints in pre-ART sequences (Spearman’s ρ = −0.0160, P = 0.9467; Extended Data Fig. 5a) and were able to control viruses not only with low but also with medium and high replicative capacity (Extended Data Fig. 5b). Levels of total or intact proviral HIV-1 DNA at ATI start were not associated with time to viral rebound or with longer time off ART (Extended Data Fig. 5c,d). However, the majority of participants that remained off ART for >12 weeks were among those with lower reservoir levels.
Finally, as the distribution of time off ART was quite binary rather than continuous (≤12 or >12 weeks), univariate logistic regression models were used to identify factors that could influence length of time to ART resumption. In addition to the pre-ART pVL, most of the immune parameters measured at ATI start increased the odds of time off ART > 12 weeks (for example, HTI magnitude \({\widehat {\rm{OR}}}\) (odds ratio) 1.46, 95% CI (1.16; 1.99), P = 0.0052; frequency of HTI-specific CD8+ GzmB+ T cells at ATI start \(\widehat {\rm{OR}}\) 1.07, 95% CI (1.01; 1.14), P = 0.0240; Fig. 5). Conversely, reservoir levels were not associated with higher chances of remaining off ART in the regression model. Importantly, in a multivariate logistic regression model including most critical demographic covariates, such as pre-ART pVL and CD4/CD8 ratio at AELIX-002 entry, there was an increased probability for being off ART after 12 weeks of ATI for the vaccinees compared to placebo recipients (\(\widehat {\rm{OR}}\) 8.25, 95% CI (1.05; 140.36); Extended Data Table 5).
Discussion
The double-blind, placebo-controlled, randomized AELIX-002 study has demonstrated that HTI vaccines were safe, well-tolerated and able to induce strong, polyfunctional and broad CD4 and CD8 T-cell responses focused on the HTI immunogen sequence. In agreement with preclinical data in NHP (non-human primates)19 and clinical trials in similar populations using other T-cell vaccines only5,6, all participants showed detectable viral rebound during the ATI. However, in exploratory analyses we observed a positive efficacy signal on the ability to remain off ART during a 24-week ATI (that is, to avoid reaching an HIV-1 pVL of >100,000 copies ml−1 or >10,000 copies ml−1 for eight consecutive weeks as per the protocol-defined ART resumption criteria) in vaccinees without beneficial HLA genetics compared to placebo recipients. The AELIX-002 trial is a randomized, placebo-controlled trial testing therapeutic T-cell vaccines in an early ART-treated population that shows a correlation between vaccine-induced immune responses and both lower post-rebound viremia and extended time off ART, providing an opportunity to identify correlates of improved viral control.
The AELIX-002 trial results support the idea that the induction of HIV-specific T cells is a key factor in improving post-rebound viral suppression during an ATI, while validating the design of the HTI immunogen to induce functional T-cell responses to vulnerable sites of the virus. Indeed, the HTI vaccines used in AELIX-002 showed good coverage of the autologous viral sequences, despite some evidence of pre-existing CTL escape20. Importantly, HTI vaccination induced strong, long-lasting GzmB-secreting CD8+ T cells along with an improved ability to inhibit replication of CCR5-tropic, CXCR4-tropic and, importantly, autologous HIV virus with a broad range of viral replicative fitness. Additionally, vaccine-induced responses targeted different HTI subunits, confirming that the HTI immunogen design does contain multiple T-cell targets that can mediate effective HIV control ex vivo.
Studies testing a combination of TLR7 agonists and bNAbs in NHP have observed a correlation between lower pre-ART pVL in acute infection and time to viral rebound during an ATI21. In contrast, in AELIX-002, lower pre-ART pVL was not associated with longer time to first detectable pVL during the ATI, but it was positively correlated with time off ART. Importantly, in exploratory multivariate models, the association of vaccination with extended time off ART remained statistically significant, even after accounting for participants’ levels of pre-ART viremia and CD4/CD8 ratio.
Different approaches have been developed to establish high-throughput assays to quantify the replication-competent viral reservoir relevant for cure-related trials, including the IPDA assay, which allows measurement of genetically intact proviruses and excludes the majority of defective proviruses22,23. In AELIX-002, although the intact proviral HIV-1 DNA declined preferentially over time relative to total proviruses, we did not detect differences in the reservoir decay from baseline to ATI associated with therapeutic vaccination, suggesting that such a reduction reflected natural decay curves due to early treatment15. In contrast to others who have reported an association between a delay in viral rebound and lower intact proviral DNA levels after vesatolimod treatment in viremic controllers24, we did not detect any correlation between levels of intact proviral DNA and time to viral rebound in our early-treated population. Of note, seven (17%) participants that entered the ATI period had no detectable levels of intact HIV-1 proviruses at the time of ART cessation and yet experienced viral rebound during the ATI.
Despite the extended vaccination regimen used in AELIX-002, vaccinations were safe and well-tolerated, and safety profiles were comparable to other HIV vaccines using the same vector platforms both in HIV-negative25 or HIV-positive individuals2. No serious related AEs or laboratory abnormalities were observed after either DDDMM or CCM vaccinations, including any suspected vaccine-induced immune thrombotic thrombocytopenia (VITT), as described for ChAdOx1-vectored COVID-19 vaccines26; although our sample size was rather limited to detect such rare events. Notewothy, T-cell exhaustion markers were not increased in vaccinees compared to placebo recipients.
Similar to the ATI viral kinetics in the AELIX-002 trial, in which all participants experienced a fast viral rebound, Okoye et al. have recently shown in the NHP model that CD8+ T cells contribute to reduce the viral set point, although they were not able to prevent viral recrudescence19. These data suggest that HIV antigenic stimulation might be necessary to trigger an effective immune response during the ATI. This, in turn, has important implications for the design of ATI trials where ART resumption criteria may need to be permissive enough to allow for such a transient viremia27,28,29. Initial peak viremia may, however, also be associated with risks for onward virus transmission, mutational T-cell escape, reseeding of the viral reservoir and/or excessive inflammatory responses giving rise to ARS. Therefore, it is critical to balance research objectives and the well-being of participants while considering, in collaboration with community advisory boards, effective transmission risk-reduction strategies30. In AELIX-002, ART resumption criteria during the ATI were well-accepted among participants, as well as all transmission-risk reduction strategies implemented, which included PrEP provision to sexual partners, psychological support and active surveillance for asymptomatic STI. Of note, the AELIX-002 study, and the ATI phase in particular, was ongoing when the first COVID-19 outbreak occurred in Spain. This severely impacted many clinical trial sites, as most non-COVID-related hospital activities, including clinical research, had to be paused. Rapid establishment of a risk-mitigation plan overseen by an external SMC during the emergency outbreak was critical to minimize the impact of the COVID-19 pandemic on the conduct of AELIX-002, as some investigators have recommended recently31,32.
The main limitations of our trial include the sample size, which did not allow for a powered subgroup analysis in individuals without beneficial HLA genetics, as well as the selected study population, which limited extrapolation of our results to HIV populations other than those treated early during acute/recent HIV infection and in which both cis-gender and transgender women are usually underrepresented. In addition, the regimen used in AELIX-002 consisted of two different vaccination regimens of DDDMM, further boosted by CCM vaccines; overall, this does not represent a clinically feasible vaccination regimen, but it did serve to set up an efficacy proof of concept of the HTI immunogen design. In fact, we acknowledge that the efficacy endpoint of time off ART in our study is a function of the ART resumption criteria used in the protocol and, importantly, is not yet translatable into clinical practice.
Our findings strongly support the further use of HTI vaccines in simpler regimens, given alone or in combination with other immunomodulatory agents to improve their efficacy, to achieve more clinically relevant virological outcomes and to be better aligned with the most current target product profile for an HIV cure indication33. For example, to avoid viral rebound, or partially curtail fast and severe viral recrudesence, and to improve the level of virus control, we and others have proposed strategies combining therapeutic vaccines with bNAbs, which at the same time may enhance suppressive capacity of vaccine-induced responses through a vaccinal effect34,35,36. In this sense, the BCN03 and AELIX-003 clinical trials (NCT05208125 and NCT04364035, respectively) are currently exploring the safety and immunogenicity of a ChAdOx1.HTI/MVA.HTI vaccine regimen with a recombinant HIV-1 envelope SOSIP protein (ConM SOSIP.v7 gp140) or with a TLR7 agonist (vesatolimod) including an ATI with the same ART resumption criteria as in AELIX-002.
In conclusion, this first administration of a heterologous prime-boost regimen of HTI vaccines in early ART-treated individuals with HIV infection was safe and immunogenic. In exploratory analyses, AELIX-002 showed a potential signal for improved post-rebound viral control after ART discontinuation in a subset of individuals who did not already possess a beneficial HLA genotype; this requires validation in future studies. These data provide support for the use of HTI vaccines as a T-cell-stimulating backbone for future combination cure strategies, with the addition of immunomodulators, bNAbs or alternative vaccine vectors to boost their efficacy.
Methods
Study design
AELIX-002 (clinicaltrials.gov NCT03204617) enrolled 45 HIV-positive early-treated individuals at the Infectious Diseases Department of the Hospital Germans Trias i Pujol (HUGTIP), Badalona, Spain. The first and last participants were recruited on 20 July 2017 and 5 June 2018, respectively. The last study visit was conducted on 10 March 2021. AELIX-002 was a phase I, proof-of-concept, first-in-human, randomized, double-blind, placebo-controlled study to evaluate the safety, immunogenicity and effect on viral rebound during an ATI of three novel HIV-1 vaccines (DNA.HTI (D), MVA.HTI (M) and ChAdOx1.HTI (C)) administered in a heterologous prime-boost regimen consisting of DDDMM and CCM versus placebo.
Participants had to be aged 18–65 years and have a history of triple-drug ART initiated within six months after estimated HIV-1 acquisition with an HIV-1 viral load of <50 HIV-1 RNA copies ml−1 and CD4+ T cells >400 cells mm−3 for at least 12 and 6 months before inclusion, respectively. An in-house algorithm based on the Fiebig classification of HIV infection15,37 and each participant’s available HIV-1 diagnostic tests were used to calculate the estimated date of HIV-1 acquisition for each individual.
Before inclusion, all participants signed an informed consent previously reviewed by a local Community Advisory Board. The study was approved by the institutional ethical review board of HUGTIP (ref. no. AC-15-108-R) and by the Spanish Regulatory Authorities, and was conducted in accordance with the principles of the Helsinki Declaration and local personal data protection law (LOPD 15/1999).
For safety purposes, participants were randomized (2:1) into three sequential recruitment blocks after blinded safety reports were approved by an external SMC. A sentinel group of three participants (two vaccine recipients and one placebo recipient) was first enrolled, and one participant was randomized per day and monitored 24 h after each vaccination (group 1) to allow for the next sentinel participant to be vaccinated. The rest of the participants were part of the non-sentinel groups: group 2 (n = 12) and group 3 (n = 30). After completion of the first vaccination regimen (DDDMM/placebo), all 45 participants were offered to participate in a second phase of the study, which included a booster vaccination regimen with CCM or placebo (while maintaining the same treatment allocation from the initial regimen) and an ATI period of 24 weeks. Between DDDMM/placebo and CCM/placebo phases of the study, participants were kept on suppressive ART and performed clinical follow-ups every 12 weeks (‘roll-over’ period).
Criteria to proceed to ATI and resume ART
Eight weeks after the last vaccination (DDDMM-CCM or placebo) participants underwent an ATI of up to 24 weeks of duration if they had (1) received all vaccinations, (2) maintained a pVL of <50 copies ml−1 and CD4+ T cells of >400 cells mm−3 and (3) there was no evidence of active syphilis, hepatitis B or hepatitis C infections. Before ATI start, HIV-seronegative participants’ sexual partners were offered PrEP through a trial-specific PrEP-provision program. During the ATI, weekly visits were performed at HUGTIP, Badalona or at BCN-Checkpoint, Barcelona at participants’ convenience. During the COVID-19 pandemic, remote visits and home-based blood draws were carried out. Criteria to resume ART included a single pVL of >100,000 copies ml−1, pVL of >10,000 and ≤100,000 copies ml−1 for eight consecutive weeks, CD4+ T cells <350 cells mm−3 in two consecutive determinations, development of a ≥grade 3 ARS, at the participant’s request or investigator criteria. As part of investigator criteria, active surveillance for STIs was performed during the ATI and, if suggestive of unprotected sex with partners with unknown HIV status and/or HIV-negative partners not taking PrEP, ART was recommended to prevent HIV transmission. All participants off ART after 24 weeks of ATI were offered to resume ART except when pVL < 2,000 copies ml−1. These participants were invited to participate in an ATI-extension protocol (NCT04385875). Criteria for ART resumption during the ATI-extension phase included one determination of pVL > 100,000 copies ml−1 or pVL > 2,000 copies ml−1 for eight consecutive weeks. Psychological assessments of the impact of the ATI on the emotional and sexual sphere were evaluated using trial-specific questionnaires by clinical psychologists at the HIV unit before entering the ATI, 12 weeks after the ATI, four weeks after ART was resumed and at the participants’ request. Participants were followed 4 and 12 weeks after ART was resumed. The protocol and a list of amendments to the protocol are available as Supplementary files 1 and 2.
Study vaccines
The HTI immunogen is a chimeric protein sequence (total length of 529 amino acids (aa)) designed based on human immune reactivity8 that includes 26 regions in HIV-1 Gag (45%), Pol (44%), Vif (8%) and Nef (3%) proteins identified in these analyses that (1) were preferentially targeted by participants with low viral loads and largely independent of beneficial HLA class I genotypes, (2) turned out to be more conserved than the rest of the proteome and (3) elicited responses of higher functional avidity and broader variant cross-reactivity than responses to other regions9.
The DNA.HTI vaccine (D) is a circular and double-stranded DNA plasmid vector of 5,676 base pairs derived from the pCMVkan expression vector backbone expressing the codon-optimized HTI gene, preceded by the human granulocyte-macrophage colony-stimulating factor (GM-CSF) signal peptide for better secretion12. The DNA.HTI drug substance is manufactured, quality-control-tested and released in accordance with the requirements of good manufacturing practice (cGMP) by the Clinical Biotechnology Centre (CBC), Bristol Institute for Transfusion Sciences, University of Bristol, UK.
The MVA.HTI vaccine (M, modified Vaccinia virus Ankara) is a live, attenuated recombinant vaccinia (pox) virus attenuated by serial passages in cultured chicken-embryo fibroblasts that contains six large deletions from the parental virus genome13. The size of MVA.HTI after insertion of a transgene coding for the HTI insert is estimated to be ~179.6 kbp. Production was carried out by the German company IDT Biologika, and preparation, verification of the genetic stability and MSV and WSV storage were carried out at IDT under cGMP conditions and according to EU regulations.
The ChAdOx1.HTI vaccine (C)- is a replication-defective recombinant chimpanzee adenovirus (ChAd) vector based on a chimpanzee adenoviral isolate Y2514 that encodes the HTI sequence. ChAdOx1.HTI was derived by subcloning the HTI antigen sequence into the generic ChAdOx1 BAC. The plasmid resulting from this subcloning (pC255; 40,483 bp) was linearized and transfected into commercial HEX293A T-REx cells to produce the vectored vaccine ChAdOx1.HTI. The ChAdOx1.HTI batch for non-clinical use was produced at the University of Oxford (UK), and large-scale amplification and purification of ChAdOx1.HTI were performed at ReiThera/Advent (Italy) according to cGMP.
Objectives
The primary objective of the study was to evaluate the safety and tolerability of HIV-1 vaccines DNA.HTI, MVA.HTI and ChAdOx1.HTI, administered intramuscularly as part of heterologous prime-boost regimen (DDDMM-CCM) in early-treated HIV-1-positive individuals. Secondary objectives included (1) evaluating the immunogenicity of DDDMM and CCM, (2) evaluating whether vaccination was able to prevent or delay viral rebound, induce post-rebound viral control and/or prevent or delay the need for resumption of ARV therapy during an ATI and (3) assessing the safety of the ATI period. Further immune (flow cytometry and viral inhibition assay) and viral evaluations (viral reservoir, autologous HIV-1 sequence and replicative fitness) were conducted as exploratory analyses. Post-hoc univariate and multivariate regression models were performed to explore potential correlates of virus control during ATI.
Safety
Safety was assessed by an analysis of local and systemic reactogenicity and laboratory data. All solicited local and systemic AEs were recorded during seven days after administration of each investigational medicinal product using a ‘participant reactogenicity diary card’. Unsolicited AEs and SAEs were recorded at any point during the study. AEs were graded according to the Division of DAIDS table for grading the severity of adult and pediatric adverse events, version 2.1 (March 2017). Throughout the study, AEs were analyzed by period: from screening to ATI start and by DDDMM/CCM or placebo; during ATI and after ART resumption. The primary safety endpoint of the study was the proportion of participants who develop grade ≥3 AEs (including SAE) related to the investigational medicinal product (IMP) administration. AEs were specified as related or unrelated to the IMPs by the investigator. Per the Manual for Expedited Reporting of Adverse Events to DAIDS (version 2.0, January 2010), AEs were reported as related if there was reasonable possibility that the AE may be related to the study agent(s), as suggested by a plausible, reasonable time sequence existing in relation to administration of the drug, the observed manifestation coincided with the known adverse reactions profile of the implicated drug, and the event could not be or was unlikely to be explained by a concurrent disease or by other drugs or chemical substances. If there was not a reasonable possibility that the AE was related to the study agent(s), the AE was reported as unrelated.
SMC and risk-mitigation plan during the COVID-19 pandemic
An SMC formed by three external experts in pharmacovigilance and HIV vaccine trials plus four non-voting sponsor representatives reviewed all blinded safety data from the study at pre-specified timepoints (that is, before progressing recruitment groups and every three months thereafter). The SMC also reviewed and approved a risk-mitigation plan established to minimize the impact of the COVID-19 pandemic on the conduct of the trial. This plan included weekly ATI assessments with home-based blood draws by personnel protected with personal protective equipment and remote visits via phone; a taxi service for on-site visits; 24-h/7-d phone availability for reporting any COVID-19 symptoms; SARS-CoV-2 polymerase chain reaction (PCR) testing before any IMP dosing; and provision of ART by courier. The SMC met virtually every week from 16 March 2020 to 28 May 2020 to review all blinded safety and laboratory data, and decisions on whether to continue with the trial were based on the evolving situation of the local epidemic, site capacity and a case-by-case discussion. New ICF versions with emerging information on COVID-19 were also developed and reviewed by the institutional ethical review board of HUGTIP.
High-resolution HLA-A, -B and -C typing
The QIAsymphony DNA kit (Qiagen) was used for genomic DNA extraction. Genomic DNA was genotyped by screening for HLA class I molecules (HLA-A, HLA-B and HLA-C genes) at high resolution at the Histocompatibility and Immunogenetics Laboratory (www.bancsang.net). Briefly, three loci were genotyped simultaneously by an in-house multiplex long-range PCR (LRPCR). The library was prepared (enzymatic fragmentation, adapter ligation and barcoding) from the PCR pools using the NGSgo kit (GenDx) according to the manufacturer’s instructions. The final denatured library was sequenced using a NextSeq or MiSeq sequencer (Illumina). HLA class I genotype determination was performed with NGSengine 2.9.1 software (GenDx) using the IMGT database as reference.
CCR5-Δ32 genotyping
DNA was extracted from cryopreserved PBMCs stored from roll-over phase timepoints from participants entering the ATI (n = 41). DNA samples were amplified using fluorescent PCR in a 9700 Gene Amp PCR System or 2720 Thermal Cycler (Applied Biosystems) as described in ref. 38. The forward (TTCATTACACCTGCAGCTCTC) and reverse (FAM-CCTGTTAGAGCTACTGCAATTAT) primers produced a 270-bp product for the CCR5-Δ32 allele and a 302-bp PCR product for the CCR5-WT allele. After amplification, 0.5 μl of PCR products was mixed in a 1:10 dilution with 24 μl of Hi-Di formamide (Applied Biosystems) and 0.7 μl of Gene Scan-500 ROX Size Standard (Applied Biosystems) and denatured at 94 °C for 5 min. The capillary electrophoresis was carried out in a 3130xlGenetic Analyzer (Applied Biosystems) and samples were analyzed with GeneMapper software (Applied Biosystems).
Sequencing
Whole-genome deep sequencing of the HIV-1 genome, including gag, pol, vif and nef genes, was performed using the Illumina NexteraXT protocol and a MiSeq platform with 300-bp paired-end sequencing length. Raw sequencing data were analyzed wih PASeq v 1.14 (www.paseq.org39). In brief, quality filter and adapter trimming was performed using trimmomatic40. High-quality sequences were aligned against the HXB2R reference using Bowtie241. The consensus sequence at 20% frequency threshold was called using samtools42 and a multiple alignment including all sequences was generated using MAFFT43. For each sample-specific consensus nucleotide sequence, subtyping was performed using the COMET online tool44, and the Tamura–Nei nucleotide and Jones–Taylor–Thornton (JTT) amino-acid distances versus HXB2R and HTI sequences, respectively, were calculated using the R::phangorn package45. The number of mismatches (hamming) versus the HTI sequence was also calculated for all segments and aggregated at the protein level. The percentage difference (%AA.mm versus HTI) was calculated over the total length of the segment, correcting for the uncovered position in each sample. Group comparisons were performed using the Mann–Whitney t-test.
IFN-γ-ELISpot assay
Total HTI and HIV-1-specific T cells were assessed ex vivo using freshly isolated PBMCs with an IFN-γ-detecting enzyme-linked immunoabsorbent spot assay (ELISpot IFN-γ Mabtech kit) as previously described2. 15-mer peptides overlapping by 11 amino acids were combined into ten pools spanning different HIV-1 proteins/subproteins of 7–22 peptides per pool corresponding to the HTI vaccine insert (P1–P10, total n = 111 peptides, Thermo Fisher) and eight pools of 62–105 peptides per pool spanning the rest of the HIV-1 viral protein sequences (OUT P1–P8, total n = 637 peptides, obtained through the NIH AIDS Reagent Program). All peptide pools used in fresh ELISpots were tested in duplicate with a final concentration of individual peptide of 1.55 μg ml−1. Medium only was used as no-peptide negative control in quadruplicate wells. Positive controls included two peptide pools covering lytic (n = 16) and latent (n = 36) Epstein–Barr viral proteins (1.55 μg ml−1, Thermo Fisher), phytohaemagglutinin (PHA; 50 μg ml−1, Sigma) and a chicken-embryo-fibroblast peptide pool (2 μg ml−1) consisting of 32 previously defined human CD8+ T-cell epitopes from cytomegalovirus, Epstein–Barr virus and influenza virus (Pantec). Spots were counted using an automated Cellular Technology Limited (C.T.L.) ELISpot reader unit. The threshold for positive responses was set at ≥50 SFCs per 106 PBMCs (five spots per well), greater than the mean number of SFCs in negative control wells plus three standard deviations of the negative control wells, or more than three times the mean of negative control wells, whichever was higher.
Mapping of HTI-specific responses
IFN-γ ELISpot assays using 147 individual overlapping peptides covering the entire HTI sequence were performed in in vitro expanded T cells. Participants’ cryopreserved PBMCs obtained at baseline (week 0) and after DDDMM (week 24) and CCM or placebo vaccinations (week 28) were expanded using an anti-CD3 mAb (12F6) and kept in culture until sufficient cell numbers were reached for each timepoint46. Two consecutive overlapping peptides were considered one individual HTI response, and the highest magnitude of the sequential responses was taken as the magnitude for each identified response. The results were expressed as the number of positive responses to individual peptides as well as the distribution among the different HIV subprotein regions covered by HTI: Vif-Nef, Pol-Int, Pol-RT, Pol-Prot, Gag-p2p7p1p7, Gag-p24 and Gag p17.
Intracellular cytokine staining assay
Cryopreserved PBMCs from week 28 (four weeks after completion of the last series of vaccinations, DDDMM-CCM) were used for stimulation with four pools of 9–43 peptides per pool spanning p17, p24/p15, Pol and Vif/Nef regions included in the HTI vaccine insert. Peptides were added at a final concentration of 5 μg ml−1 of each peptide in the presence of both 1.4 µg ml−1 of anti-CD28 (BD Bioscience) and 1.4 µg ml−1 anti-CD49d (BD Bioscience). As positive controls for the assay, cells were cultured alone in the presence of (1) anti-CD3/28 Dynabeads (Thermo Fisher Scientific) according to the manufacturer’s instructions or (2) 10 ng ml−1 phorbol 12-myristate 13-acetate (PMA,Sigma) and 1 µM ionomycin (Sigma). Cells stimulated with only anti-CD28 and anti-CD49d antibodies or with DMSO were used as negative controls. Stimulated cells were incubated for 6 h at 37 °C in 5% CO2, in the presence of 4 µl of monensin (GolgiStop, BD Bioscience). After 6 h of stimulation, cells were incubated with a Live/Dead fixable Violet Dead cell stain kit (Invitrogen), for exclusion of dead cells, along with the exclusion of monocytes and B cells by including in the dump channel anti-CD14 and anti-CD19 antibodies. Surface markers of T-cell lineage (CD3, CD4 and CD8), follicular T cells (CXCR5 and PD1), T-cell phenotype (CD45RA and CCR7), T-cell activation (CD69 and HLADR) and T-cell exhaustion (TIGIT, PD1) were included as well. Cells were fixed and permeabilized using the Cell Fixation and Cell Permeabilization Kit (Invitrogen) and intracellularly stained for INF-γ, GrazymeB, IL-2 and TNF-α. Details on the used antibodies can be found in the Reporting summary. Cells were resuspended in phosphate buffered saline supplemented with 1% FBS and acquired on an LSR Fortessa flow cytometer (BD, Unidad de Citometria, IGTP) and analyzed using FlowJo. The gating strategy is shown in Supplementary Fig. 2. When needed for variably expressed antigens, fluorescence minus one was included to define the boundaries between positive and negative populations. At least 100,000 total events were recorded. The frequencies of cells that produce all possible combinations of intracellular cytokines were calculated using the Boolean gating function of the FlowJo software. Data were reported after background subtraction (from the unstimulated negative control), and HTI-specific responses were defined as the sum of the specific population for each of the four HTI peptide pool stimulations.
In vitro viral suppressive capacity (VIA assay)
CD8+ T-cell-mediated viral inhibition capacity was measured at 1:1 and 1:10 CD8-effector to CD4-target ratios, as previously described47,48. Autologous CD4+ cells were obtained as targets from samples before vaccination where CD8+ cells were depleted by magnetic bead separation (MACS Milteny Biotec). CD8+-depleted cells (CD4+-enriched fraction) were stimulated with PHA for three days and then infected by spinoculation with HIV-1 BAL and IIIB laboratory-adapted strains and autologous HIV-1 viruses at a multiplicity of infection of 0.001. HIV-infected cells were cultured in triplicates in R10 medium with 20 U ml−1 of IL-2 in 96-well round-bottomed plates, alone or together with effector CD8+ T cells obtained by positive magnetic bead separation the same day from an additional vial of cryopreserved PBMCs from baseline and after DDDMM (week 24) and CCM or placebo (week 28) vaccinations. Viral replication was measured as the production of HIV-1 antigen p24 in culture supernatants (pg p24 per ml) at day 5 of co-culture using an Innogenetics p24 Elisa kit, and inhibition was expressed as a percentage with respect to the positive control of each virus (that is, infection in the absence of CD8+ T cells).
Total and intact proviral HIV-1 DNA
To distinguish deleted and/or hypermutated proviruses from intact proviruses, total and intact proviral (IPDA) HIV-1 DNA copies in CD4+ T cells were measured at screening and ATI start in extracts of lyzed CD4+ T cells by digital droplet PCR (ddPCR), as previously described49. Samples from 41 participants that entered into the ATI period were processed at Accelevir Diagnostics. The DNA shearing index was calculated, and values for intact and defective proviruses were normalized to copies per 106 input cells (determined by RPP30, the gene encoding Ribonuclease P protein subunit p30) and adjusted for shearing using the DNA shearing index. Results were expressed as HIV-1 DNA copies (counts) per 106 CD4+ T cells.
Viral fitness of participants’ autologous HIV-1 viruses
The viral replication capacity of autologous HIV-1 viruses was measured for 38 of the 41 participants that entered into the ATI period. For isolation of autologous HIV-1 viruses, the CD4-enriched fraction of cryopreserved PBMCs stored at HIV-1 diagnosis pre/or within the first weeks of ART initiation were thawed and co-cultured with CD8-depleted PBMCs previously activated from three different healthy donors until HIV-1 was collected from supernatants. To determine the viral replication kinetics, a pool of PBMCs from three healthy donors, previously stimulated with 20 U ml−1 of IL-2 and PHA for three days, were infected by spinoculation at a multiplicity of infection of 0.001. HIV-1 antigen p24 was measured in culture supernatants (pg p24 per ml) using a commercial ELISA kit from Innogenetics at days 0, 3, 4, 5, 6 and 7 post-infection, and replication capacity was calculated by fitting a linear model to the log-transformed p24 data during the exponential growth phase. Uninfected cells and those infected with laboratory-adapted CCR5- and CXCR4-tropic viruses (HIV-1NL43, HIV-1BaL and HIV-1IIIB isolates isolates) in the presence and absence of the antiretroviral AZT were used as reference values or controls.
Statistics
There was no power calculation for this study. The sample size was proposed to provide preliminary safety information on the vaccine regimen (primary objective). As a means to characterize the statistical properties of this study for the safety primary endpoint, in terms of the chances of observing an AE, 30 participants in the active group provided a high probability (78.5%) that this study would observe at least one event if the event occurred in the population with a true rate of 5%.
Time to viral load detection was calculated from the ATI start date to the date of first occurrence of pVL of ≥50 copies ml−1 and time off ART was calculated from the ATI start date to the date of ART resumption. Participants who prematurely resumed ART for COVID-19-related reasons were not censored for the survival analysis. The time to event was derived using the number of days between the ATI start date and the date of event expressed in weeks (number of days/7). The Kaplan–Meier estimator was used to describe the time to ART resumption, and survival functions were compared using the log-rank test. Differences in medians between groups were compared using the Mann–Whitney test and Fisher test, when corresponding. Spearman’s ρ was used for correlations. All tests were two-sided, unadjusted for multiple comparisons, with 5% error rate. Post-hoc univariate logistic regression models (the list of considered covariates is provided in Extended Data Table 5) were considered to select the covariates with P < 0.25 to be included in the multivariate models. All selected covariates were analyzed for possible multicollinearity. Considering the final selected covariates, multivariate logistic regression models were adjusted for the binary outcome of time off ART ≥ 12 weeks versus <12 weeks. Analyses were performed using R project 3.6.2 (https://www.r-project.org/) and GraphPad Prism version 9.1.2 for Windows (GraphPad Software, https://www.graphpad.com). Flow cytometry data were preprocessed using FlowJo software version 10.6 and imported into Pestle2/SPICE software v5.35 (Vaccine Research Center, NIAID/NIH) for graphical representation. Polyfunctional bar plots per treatment group were compared using the Mann–Whitney test per row, with individual ranks computed for each comparison. The two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli was used to control for false discovery rate. All performed analyses matched the pre-specified statistical analysis plan (AELIX002-SAP, version 2, from 10 July 2020).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this Article.
Data availability
Deep sequencing raw data obtained from sequencing have been deposited in GenBank (accession no. PRJNA751460). Requests for access to the study data can be submitted through the Yale Open Data Access (YODA) Project site at http://yoda.yale.edu.
References
Shan, L. et al. Stimulation of HIV-1-specific cytolytic T lymphocytes facilitates elimination of latent viral reservoir after virus reactivation. Immunity 36, 491–501 (2012).
Mothe, B. et al. Therapeutic vaccination refocuses T-cell responses towards conserved regions of HIV-1 in early treated individuals (BCN 01 study). EClinicalMedicine 11, 65–80 (2019).
Mothe, B. et al. HIVconsv vaccines and romidepsin in early-treated HIV-1-infected individuals: safety, immunogenicity and effect on the viral reservoir (Study BCN02). Front. Immunol. 11, 823 (2020).
Fidler, S. et al. Antiretroviral therapy alone versus antiretroviral therapy with a kick and kill approach, on measures of the HIV reservoir in participants with recent HIV infection (the RIVER trial): a phase 2, randomised trial. Lancet 395, 888–898 (2020).
Colby, D. J. et al. Safety and immunogenicity of Ad26 and MVA vaccines in acutely treated HIV and effect on viral rebound after antiretroviral therapy interruption. Nat. Med. 26, 498–501 (2020).
Sneller, M. C. et al. A randomized controlled safety/efficacy trial of therapeutic vaccination in HIV-infected individuals who initiated antiretroviral therapy early in infection. Sci. Transl. Med. 9, eaan8848 (2017).
Søgaard, O. S. et al. The depsipeptide romidepsin reverses HIV-1 latency in vivo. PLoS Pathog. 11, e1005142 (2015).
Mothe, B. et al. Definition of the viral targets of protective HIV-1-specific T cell responses. J. Transl. Med. 9, 208 (2011).
Mothe, B. et al. A human immune data-informed vaccine concept elicits strong and broad T-cell specificities associated with HIV-1 control in mice and macaques. J. Transl. Med. 13, 60 (2015).
Mothe, B. et al. CTL responses of high functional avidity and broad variant cross-reactivity are associated with HIV control. PLoS ONE 7, e29717 (2012).
Hancock, G. et al. Identification of effective subdominant anti-HIV-1 CD8+ T cells within entire post-infection and post-vaccination immune responses. PLoS Pathog. 11, e1004658 (2015).
Kulkarni, V. et al. Comparison of immune responses generated by optimized DNA vaccination against SIV antigens in mice and macaques. Vaccine 29, 6742–6754 (2011).
Létourneau, S. et al. Design and pre-clinical evaluation of a universal HIV-1 vaccine. PLoS ONE 2, e984 (2007).
Dicks, M. D. J. et al. A novel chimpanzee adenovirus vector with low human seroprevalence: improved systems for vector derivation and comparative immunogenicity. PLoS ONE 7, e40385 (2012).
Bayón-Gil, Á. et al. HIV-1 DNA decay dynamics in early treated individuals: practical considerations for clinical trial design. J. Antimicrob. Chemother. 75, 2258–2263 (2020).
Goulder, P. J. R. & Walker, B. D. HIV and HLA Class I: an evolving relationship. Immunity https://doi.org/10.1016/j.immuni.2012.09.005 (2012).
Yang, H. et al. Antiviral inhibitory capacity of CD8+ T cells predicts the rate of CD4+ T-cell decline in HIV-1 infection. J. Infect. Dis. 206, 552–561 (2012).
Werner, R. N., Gaskins, M., Nast, A. & Dressler, C. Incidence of sexually transmitted infections in men who have sex with men and who are at substantial risk of HIV infection—a meta-analysis of data from trials and observational studies of HIV pre-exposure prophylaxis. PLoS ONE 13, e0208107 (2018).
Okoye, A. A. et al. CD8+ T cells fail to limit SIV reactivation following ART withdrawal until after viral amplification. J. Clin. Invest. 131, e141677 (2021).
Deng, K. et al. Broad CTL response is required to clear latent HIV-1 due to dominance of escape mutations. Nature 517, 381–385 (2015).
Borducchi, E. N. et al. Antibody and TLR7 agonist delay viral rebound in SHIV-infected monkeys. Nature 563, 360–364 (2018).
Gaebler, C. et al. Sequence evaluation and comparative analysis of novel assays for intact proviral HIV-1 DNA. J. Virol. 95, e01986920 (2021).
M, A.-M. et al. Recommendations for measuring HIV reservoir size in cure-directed clinical trials. Nat. Med. 26, 1339–1350 (2020).
SenGupta, D. et al. The TLR7 agonist vesatolimod induced a modest delay in viral rebound in HIV controllers after cessation of antiretroviral therapy. Sci. Transl. Med. 13, eabg3071 (2021).
Hayton, E.-J. et al. Safety and tolerability of conserved region vaccines vectored by plasmid DNA, simian adenovirus and modified vaccinia virus ankara administered to human immunodeficiency virus type 1-uninfected adults in a randomized, single-blind phase I trial. PLoS ONE 9, e101591 (2014).
Greinacher, A. et al. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N. Engl. J. Med. 384, 2092–2101 (2021).
Namazi, G. et al. The Control of HIV after Antiretroviral Medication Pause (CHAMP) study: post-treatment controllers identified from 14 clinical studies. J. Infect. Dis. https://doi.org/10.1093/infdis/jiy479 (2018).
Julg, B. et al. Recommendations for analytical antiretroviral treatment interruptions in HIV research trials—report of a consensus meeting. Lancet HIV 6, e259–e268 (2019).
Fajnzylber, J. M. et al. Frequency of post treatment control varies by ART restart and viral load criteria. AIDS 35, 2225–2227 (2021).
Dubé, K. et al. Ethical and practical considerations for mitigating risks to sexual partners during analytical treatment interruptions in HIV cure-related research. HIV Res. Clin. Pract. 22, 14–30 (2021).
Peluso, M. J. et al. Operationalizing HIV cure-related trials with analytic treatment interruptions during the SARS-CoV-2 pandemic: a collaborative approach. Clin. Infect. Dis. Publ. Infect. Dis. Soc. Am. 72, 1843–1849 (2021).
Fidler, S. et al. HIV cure research in the time of COVID-19—antiretroviral therapy treatment interruption trials: a discussion paper. J. Virus Erad. 7, 100025 (2021).
Lewin, S. R. et al. Multi-stakeholder consensus on a target product profile for an HIV cure. Lancet HIV 8, e42–e50 (2021).
Nishimura, Y. et al. Early antibody therapy can induce long-lasting immunity to SHIV. Nature 543, 559–563 (2017).
Mendoza, P. et al. Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature 561, 479–484 (2018).
Caskey, M. Broadly neutralizing antibodies for the treatment and prevention of HIV infection. Curr. Opin. HIV AIDS 15, 49–55 (2020).
Fiebig, E. W. et al. Dynamics of HIV viremia and antibody seroconversion in plasma donors: implications for diagnosis and staging of primary HIV infection. AIDS 17, 1871–1879 (2003).
Enrich, E. et al. Analysis of the Spanish CCR5-Δ32 inventory of cord blood units: lower cell counts in homozygous donors. Bone Marrow Transpl. 53, 741–748 (2018).
Noguera-Julian, M. et al. Next-generation human immunodeficiency virus sequencing for patient management and drug resistance surveillance. J. Infect. Dis. 216, S829–S833 (2017).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Pineda-Peña, A. C. et al. Automated subtyping of HIV-1 genetic sequences for clinical and surveillance purposes: performance evaluation of the new REGA version 3 and seven other tools. Infect. Genet. Evol. 19, 337–348 (2013).
Schliep, K. P. phangorn: phylogenetic analysis in R. Bioinformatics 27, 592–593 (2011).
Kawana-Tachikawa, A. et al. Effect of maraviroc intensification on HIV-1-specific T cell immunity in recently HIV-1-infected individuals. PLoS ONE 9, e87334 (2014).
Yang, H. et al. Antiviral inhibitory capacity of CD8+ T cells predicts the rate of CD4+ T-cell decline in HIV-1 infection. J. Infect. Dis 206, 552–561 (2012).
Ross-Umbert, M. In vivo effects of romidepsin on T-cell activation, apoptosis and function in the BCN02 HIV-1 Kick&Kill Clinical Trial. Front. Immunol 11, 418 (2020).
Bruner, K. M. et al. A quantitative approach for measuring the reservoir of latent HIV-1 proviruses. Nature 566, 120–125 (2019).
Acknowledgements
Special thanks go to all the volunteers participating in this study for their perseverance and dedication. We especially thank the SMC members E. Negredo, M. Farré and F. Garcia for their commitment to oversee the risk-mitigation plan during the challenging COVID-19 pandemic. We thank R. Ayen, L. Gómez, C. Ramírez and E. Grau from the sample processing laboratory at IrsiCaixa, Badalona, Spain for their technical assistance throughout the study. We thank B. K. Felber and G. N. Pavlakis from the NCI-Frederick, MD, USA and A. Crook from the Jenner Institute, The Nuffield Department of Medicine, University of Oxford, UK for their assistance in the construction and provision of the pre-GMP starting materials of DNA.HTI, MVA.HTI and ChAdOx1.HTI under MTA contracts. This work was supported by AELIX Therapeutics SL, and partly by grants from the Spanish Research Agency and the European Regional Development Fund under contract no. RTC-2017-6473-1 ((AEI/FEDER, UE)/(MINECO/FEDER, UE)), the European Union’s Horizon 2020 research and innovation program under grant agreement 681137-EAVI2020, NIH grant P01-AI131568 and by ISCIII PI20/01039. L.B. held a PhD grant from ISCIII (RH CM20/00097) during the conduct of the study. This study was funded by AELIX Therapeutics SL, who participated in the design of the study, provided the IMP of the study, oversaw all safety monitoring activities, data analysis and interpretation, and reviewed the manuscript.
Author information
Authors and Affiliations
Consortia
Contributions
C.B., I.M., J.M. and B.M. conceived and designed the study. L.B. and A.L. additionally contributed to the study design in further study amendments. L.B., J.C., C.L., M.L., J.M., B.M., F.P. and A.R. contributed with clinical development of the study. A.L., M.L., B.O.-T., F.P., F.P.E. and D.S. contributed to data management and overall study coordination. T.H. and E.G.W. helped with IMP production. M.C., S.C., T.E., A.L.L., M.P. and M.R.-U. performed the experiments. Y.A.-S., A.L.L., J.M., B.M., M.R.-R. and M.N.-J. undertook the statistical analysis. L.B., C.B., J.M. and B.M. drafted the manuscript. L.B., A.L., I.M., D.S., B.C., C.B., J.M., B.M. and R.P. critically revised the manuscript for important intellectual content. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.B., B.M. and A.L.L. are co-inventors of the HTI immunogen (patent application PCT/EP2013/051596). C.B., B.M. and I.M. are co-inventors of US patent application no. 62/935,519 and US patent application no. 62/851,546, which have relevance to the vaccine regimen used in this study. B.M. reports consultancy personal fees from AELIX Therapeutics SL, as well as speakers’ fees from Gilead, Janssen and ViiV Healthcare, outside the submitted work. C.B. is co-founder, CSO and shareholder of AELIX Therapeutics SL and serves as an advisor for Tendel Therapies and OmniScope, outside the submitted work. M.N.-J. is co-founder and shareholder of Nano1Health SL, outside the scope of the submitted work. I.M. is a shareholder of, and acts as a consultant to, AELIX Therapeutics SL. He is also the CMO of Orion Biotechnology, outside the scope of the submitted work. J.M. has received research funding, consultancy fees and lecture sponsorships from, and has served on advisory boards for, various laboratories (MSD, Abbvie, Boehringer Ingelheim, Gilead Sciences, Viiv Healthcare, Janssen-Cilag and Bristol-Myers-Squibb). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Zaza Ndhlovu, Cecilia Moore and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alison Farrell, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Pre-ART HIV-1 sequences.
Coverage of the participant’s HIV-1 sequences by HTI vaccine. Comparison of distance with HXB2 (Tamura-Nei) in (a) and HTI (JTT) (b) for placebo (n=15, blue) and vaccine (n=26, red) recipients. Mann-Whitney t-test is shown. c, Genetic distance between the placebo (blue) and vaccine (red) recipient’s pre-ART HIV-1 sequences and different HIV-1 proteins included in the HTI immunogen. Mann-Whitney t-test is shown d, Number of total HLA (left) and HLA-adapted (right) polymorphisms on pre-ART HIV-1 sequences from placebo (n=15, blue) and vaccine (n=26, red) recipients. Mann-Whitney t-test is shown. For (a)-(d), median with interquartile range is shown. e, Correlation between the number of pre-ART CTL escape footprints and replicative fitness of autologous pre-ART HIV-1 sequences from placebo (n=14, blue) and vaccine (n=24, red) recipients. Spearman’s correlation is used. Pbo: placebo, Vax: vaccine.
Extended Data Fig. 2 Role of pre-existing responses to HTI.
a, Relation between pre-ART HTI-specific responses and expanded HTI responses at peak immunogenicity timepoints after DDDMM, CCM and ATI start in all vaccine recipients are shown and individuals with any beneficial HLA allele are represented in triangle. Spearman’s correlation is used. b, Peak HTI- magnitude after DDDMM (n=30), CCM (n=27) and at ATI start (n=26) for all the vaccine recipients having or not having any HTI responses before any ART (Pre, defined as HTI magnitude >50 SFC/106 PBMC). Individuals with a beneficial HLA are shown in triangle symbols. Median with interquartile range is shown. Wilcoxon-Mann-Whitney test was used for comparison between treatments. C: ChAdOx1.HTI, D: DNA.HTI, M: MVA.HTI, P: placebo, ART: antiretroviral treatment, ATI: analytical treatment interruption.
Extended Data Fig. 3 ATI-extension beyond 24 weeks.
Extended individual pVL up to 72 weeks is shown for placebo (blue) or vaccine (red) recipients that entered into the ATI-extension protocol (>24 weeks). Lines are interrupted on day of ART resumption, that is at week 72 or before. Dotted lines represent individuals with beneficial HLA class I alleles. ATI: analytical treatment interruption, pVL: plasma viral load.
Extended Data Fig. 4 HIV-1 reservoir decay throughout the study.
Fold-change decay for total (placebo n=15, vaccine n=26) and intact (placebo n=12, vaccine n=23) proviral HIV-1 DNA by treatment group from study entry to ATI start. Median and interquartile range are represented. Mann-Whitney test is used. SCR: screening, ATI: analytical treatment interruption.
Extended Data Fig. 5 Virological correlates with ATI outcomes in participants without any beneficial HLA allele.
a, Correlation between number of HLA-adapted polymorphisms in pre-ART HIV-1 sequences and time off ART in placebo (blue) and vaccine (red) recipients. b, Correlation between replication capacity of participant’s autologous HIV-1 virus (relative to NL43) replication and time off ART in placebo (blue) and vaccine (red) recipients. Correlation between levels of total (c) and intact (d) proviral HIV-1 DNA at ATI and time off ART in placebo (blue) and vaccine (red) recipients. Spearman’s correlation is used. ART: antiretroviral treatment, ATI: analytical treatment interruption.
Supplementary information
Supplementary Information
Supplementary Tables 1–7, Figs. 1 and 2, document 1 (study protocol), document 2 (list of protocol amendments), document 3 (list of members of the AELIX-002 Study Group) and document 4 (consortium checklist).
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bailón, L., Llano, A., Cedeño, S. et al. Safety, immunogenicity and effect on viral rebound of HTI vaccines in early treated HIV-1 infection: a randomized, placebo-controlled phase 1 trial. Nat Med 28, 2611–2621 (2022). https://doi.org/10.1038/s41591-022-02060-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-022-02060-2
This article is cited by
-
Immune targeting of HIV-1 reservoir cells: a path to elimination strategies and cure
Nature Reviews Microbiology (2024)
-
Immunogenic arenavirus vector SIV vaccine reduces setpoint viral load in SIV-challenged rhesus monkeys
npj Vaccines (2023)
-
Prevention, treatment and cure of HIV infection
Nature Reviews Microbiology (2023)
-
Therapeutic vaccination following early antiretroviral therapy elicits highly functional T cell responses against conserved HIV-1 regions
Scientific Reports (2023)
-
Elevated glutamate impedes anti-HIV-1 CD8 + T cell responses in HIV-1-infected individuals on antiretroviral therapy
Communications Biology (2023)