Abstract
Characterizing the potential health effects of exposure to risk factors such as red meat consumption is essential to inform health policy and practice. Previous meta-analyses evaluating the effects of red meat intake have generated mixed findings and do not formally assess evidence strength. Here, we conducted a systematic review and implemented a meta-regression—relaxing conventional log-linearity assumptions and incorporating between-study heterogeneity—to evaluate the relationships between unprocessed red meat consumption and six potential health outcomes. We found weak evidence of association between unprocessed red meat consumption and colorectal cancer, breast cancer, type 2 diabetes and ischemic heart disease. Moreover, we found no evidence of an association between unprocessed red meat and ischemic stroke or hemorrhagic stroke. We also found that while risk for the six outcomes in our analysis combined was minimized at 0 g unprocessed red meat intake per day, the 95% uncertainty interval that incorporated between-study heterogeneity was very wide: from 0–200 g d−1. While there is some evidence that eating unprocessed red meat is associated with increased risk of disease incidence and mortality, it is weak and insufficient to make stronger or more conclusive recommendations. More rigorous, well-powered research is needed to better understand and quantify the relationship between consumption of unprocessed red meat and chronic disease.
Similar content being viewed by others

Main
Previous research has broadly shown an association between red meat consumption and increased risks to human health1,2,3,4. The Global Burden of Diseases, Injuries and Risk Factors study (GBD) 2019 estimated that 896,000 (95% uncertainty interval (UI) 536,000–1,250,000) deaths and 23.9 million (15.6–32.0) disability-adjusted life years were attributable to unprocessed red meat consumption globally in 2019 (ref. 5). These and other findings have led institutions such as the World Health Organization, the World Cancer Research Fund (WCRF), the EAT-Lancet Commission and the US Departments of Health and Human Services and Agriculture to recommend limiting red meat intake6,7,8,9. Recommended consumption targets are inconsistent, however, ranging from 14 g d−1 (EAT8), to 50–70 g d−1 (WCRF7), to unrestricted amounts (Nutritional Recommendations Consortium10). Adding to this ambiguity, several studies have found no significant relationship between red meat consumption and risk of death11,12, which has led to further questioning of the strength of evidence in these risk pair associations13,14. To resolve conflicting data and recommendations, a quantitative and objective strategy for assessing the strength of the evidence relating red meat consumption to health outcomes is needed.
Evidence on the health effects of red meat consumption comes primarily from prospective observational cohort studies in which individuals are grouped into categories based on their level of red meat consumption1,15 and estimates of relative risks (RRs) comparing different levels of consumption are reported. Meta-analyses synthesize results from these studies either by pooling effect sizes that compare the extreme categories reported in each study, or by estimating the dose–response hazard ratio per unit of exposure—typically assuming a log-linear relationship—and pooling study-specific results4,16. These methods rely on a number of premises that may limit their utility to capture the effects of risk exposure on health outcomes. One issue involves the assumption of log-linearity, which requires that the hazard ratio for a fixed increment of red meat consumption (for example, 100 g d−1) remains constant across all levels of intake (an increase in consumption from 0 to 100 g d−1 would have the same effect as an increase from 200 to 300 g d−1). Yet evidence indicates that the dose–response relationship for many risk factors attenuates at higher doses17,18 (not log linear). Based on such evidence and in light of limited existing information on the shape of the risk curves for red meat and different health outcomes, it is plausible that the health effects of red meat consumption may not be well characterized by a log-linear function and should be investigated more closely. Another notable issue is that meta-analyses attempting to synthesize findings from cohort studies typically do not account for between-study heterogeneity, which can be a prominent source of bias in epidemiological meta-analyses19.
In this paper we examined the relationship between unprocessed red meat and six health outcomes: breast cancer, colorectal cancer, type 2 diabetes, ischemic heart disease (IHD), ischemic stroke and hemorrhagic stroke. These outcomes were selected using the WCRF criteria for convincing or probable evidence7. To rigorously quantify the dose–response relationship between unprocessed red meat consumption and the selected health outcomes—in addition to the strength of the evidence underlying the results—we systematically evaluated all available prospective epidemiological evidence on the association between unprocessed red meat consumption and each selected health outcome following the burden of proof risk function (BPRF) methodology developed by Zheng et al.20. Specifically, for each risk–outcome pair, we generated a mean risk function by implementing a flexible meta-regression framework that relaxed the conventional assumption of a log-linear dose–response relationship, instead using a data-driven approach to determine the shape of the function using a quadratic spline. The approach accounts for exposure ranges reported in the data and different comparator groups21. We automatically detected outliers using robust statistical trimming methods, tested and corrected for bias related to study design and evaluated publishing and reporting bias. We computed 95% UIs that account for mean effects uncertainty as well as between-study heterogeneity, adjusting for the number of studies. We used the information to calculate uncertainty inclusive of between-study heterogeneity to generate a BPRF20. The BPRF is a conservative risk function (offering an alternative to a mean risk function) that is defined as either the fifth (for harmful risks) or 95th (for protective risks) quantile curve closest to the line of RR equal to one (the null). Thus, the BPRF can be interpreted as the smallest harmful or protective effect at each level of exposure consistent with the available evidence.
We then converted the BPRF for each risk–outcome pair into a risk–outcome score (ROS)20. The ROS summarizes the magnitude and certainty of evidence about a risk or protective factor, with higher positive ROS always corresponding to a stronger relationship and negative ROS corresponding to a failure to reject the null. We further converted the ROS for each risk–outcome pair into a star rating: for both harmful and protective risks, an ROS of <0 received one star, >0–0.14 received two stars, >0.14–0.41 received three stars, >0.41–0.62 received four stars and >0.62 received five stars. Star ratings are designed to offer individuals and policy makers a way to understand the strength of evidence about a risk in a way that is comparable across risk–outcome pairs. The thresholds for each star rating were developed in consultation with collaborators, stakeholders and other audiences and can broadly be interpreted as indicating ‘no evidence of association,’ ‘weak evidence of association,’ ‘moderate evidence of association,’ ‘strong evidence of association’ and ‘very strong evidence of association.’ The main findings and implications for policy of this work are summarized in Table 1.
Results
Overview
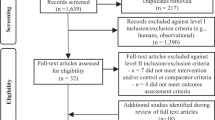
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines22, we systematically searched for literature on the RR of unprocessed red meat consumption, screened identified records and extracted data from reports meeting our inclusion criteria (Methods). Of the 3,286 records identified, 55 reports1,11,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75 met our inclusion criteria, providing information on 37 prospective cohorts and one nested case–control study (Extended Data Fig. 1). These included 18 cohorts and one nested case–control from Europe, 13 cohorts from North America, five cohorts from Asia and one from Australia. In most studies, dietary consumption of unprocessed red meat was assessed using food frequency questionnaires (42 of 55). The number of participants in each study ranged from 639 to 536,969 and the length of follow-up ranged from 4.1 to 32 years. In most studies (45 of 55), RRs were adjusted for major confounders including age, sex and smoking. Supplementary Table 1 (section 1) contains a summary of the main characteristics of all studies included in this analysis, including study design, end point, location, exposure assessment and other characteristics.
We found that removing trimming (including outliers) and removing the imposed model shape constraints did not significantly change the risk curve results. The Supplementary Information provides sensitivity results of these assumptions (section 2.1 and 2.2).
Risk–outcome scores; star ratings; risk curves with all data points, trimmed data points and conventional and conservative UIs; and an interpretation of the findings are available for all risk–outcome pairs at http://vizhub.healthdata.org/burden-of-proof/.
Unprocessed red meat consumption and colorectal cancer
We identified 20 prospective cohorts27,28,35,38,39,42,45,49,50,54,57,58,60,61,63,72,73,75 and one nested case–control study67 to assess the relationship between unprocessed red meat consumption and colorectal cancer among 2,413,032 individuals in total (sample size was calculated as the number of unique data source-location pairs with observations of risk exposure and outcome) over an average of 8.0 years per individual (range of mean/median follow-up per cohort, 4.1–32 years). Ten cohorts were carried out in Europe, eight in North America, two in Asia and one in Australia. Overall, 16,202 colorectal cancer events were recorded across all cohorts, where 14,672 were incident cases and 1,530 were mortality cases. In addition to adjustments for major confounders including age, sex and smoking, the RRs in most cohorts were additionally adjusted for body mass index (BMI) (n = 14) and dietary components such as energy intake and fruit and vegetables (n = 16). Most cohorts reported the RR of colorectal cancer incidence (n = 18); no significant difference between incidence and mortality RRs was detected in covariate selection and no other bias covariates were identified by the covariate selection algorithm of the meta-regression.
We found weak evidence of harmful associations between unprocessed red meat consumption and risk of colorectal cancer; the mean RR at 50 g d−1 relative to no intake was 1.30 (95% UI inclusive of between-study heterogeneity of 1.01–1.64), while the mean RR at 100 g d−1 was 1.37 (1.01–1.78) (Table 2 and Fig. 1), where the UIs account for between-study heterogeneity and other forms of uncertainty. We estimated the exposure-averaged burden of proof RR to be 1.06, indicating that consuming unprocessed red meat in the range of 15th to 85th percentiles of exposure (0 g d−1 to 98 g d−1) was associated with at least a 6% higher risk of colorectal cancer. This corresponds to an ROS of 0.06 and a two-star rating, consistent with weak evidence.
Unprocessed red meat consumption and breast cancer
Six cohorts from Europe and four cohorts from North America were used to evaluate the association between unprocessed red meat intake and incidence of breast cancer26,33,34,37,40,42,51,55,59,65. The total number of participants across all cohorts was 999,428 individuals; the follow-up period ranged from 4.1 to 20.3 years (mean, 8.4 years); and the total number of incident cases was 25,732. In seven cohorts, the RR was adjusted for BMI and in eight cohorts, it was additionally adjusted for dietary components. All studies reported the RR of breast cancer incidence. There were no bias covariates identified by our algorithm as statistically relevant for inclusion in the model.
We found weak evidence of a harmful association between unprocessed red meat intake and risk of breast cancer. The BPRF value (averaged across the 15th to 85th percentiles of red meat consumption, 0–69 g d−1) was 1.03, which was substantially lower than the mean RR of 1.26 (0.98–1.56) and 1.26 (0.98–1.56) at 50 g d−1 and 100 g d−1, respectively. The corresponding ROS is 0.03 (Table 2 and Fig. 2), which translates to a two-star risk and means that unprocessed red meat intake is associated with at least a 3% higher risk of colorectal cancer. When accounting for between-study heterogeneity, the mean RR UI at different exposure levels spanned 1 (Table 2).
Unprocessed red meat consumption and ischemic heart disease
We included 11 prospective cohorts in the analysis of unprocessed red meat consumption and IHD11,23,25,31,36,41,52,56,63,74. The cohorts were from North America (n = 4), Europe (n = 4) and Asia (n = 3). The total number of participants across cohorts was 1,219,288 individuals and the mean duration of follow-up was 11.6 years (range, 5.5–30 years). Overall, 33,490 cases of IHD were recorded across all cohorts. These included 25,222 nonfatal cases, 3,959 fatal cases and 4,309 unspecified nonfatal/fatal cases. In most cohorts, the RRs were adjusted for BMI (n = 8) and dietary components (n = 10). Five studies reported the RR of IHD mortality, four reported for incidence and two reported a combination of incidence and mortality. We evaluated the effect of potential bias covariates, including an indicator that we assessed for differentiating between the RR of incidence and the RR of mortality and found that no bias covariates in our analysis had a significant effect on RR. We therefore did not include any in our model.
We found weak evidence of a harmful association between unprocessed red meat consumption and risk of IHD. The RR was 1.09 (0.99–1.18) at 50 g d−1 and 1.12 (0.99–1.25) at 100 g d−1 (Table 2 and Fig. 3). The corresponding exposure-averaged BPRF was 1.01, which translates to a ROS of 0.01 and a two-star rating at the lower threshold of two-star pairs (at the boundary between weak evidence and no evidence of an association between consumption of unprocessed red meat and increased risk of IHD incidence and mortality).
Unprocessed red meat consumption and type 2 diabetes
We identified 17 prospective cohorts evaluating the relationship between unprocessed red meat consumption and type 2 diabetes among 1,619,574 participants1,29,30,32,43,44,48,53,56,62,64,66,69,70,71. Eight cohorts were from Europe, six from North America and three from Asia. Follow-up time ranged from 4.6 to 28 years (mean 11.6 years). Overall, 58,364 new cases of diabetes and 3,717 deaths from diabetes were recorded. In all cohorts but one, the RR was adjusted for BMI and in 15 cohorts, the RR was adjusted for dietary components. Only one cohort reported the RR of diabetes mortality and the rest reported incidence. No bias covariates were found to be statistically relevant and therefore none were included in the model.
We found evidence of weak harmful effects between unprocessed red meat consumption and risk of type 2 diabetes, with a mean RR of 1.14 (0.97–1.32) at 50 g d−1 relative to no intake and a mean RR of 1.23 (0.96–1.52) at 100 g d−1 relative to no intake (Table 2 and Extended Data Fig. 2). The BPRF value was 1.01 and the corresponding ROS was 0.01, equating to a two-star rating at the lower threshold of two-star pairs (at the boundary between weak evidence and no evidence of an association between consumption of unprocessed red meat and increased risk of type 2 diabetes).
Unprocessed red meat consumption and ischemic stroke
The relationship between consumption of unprocessed red meat and ischemic stroke was assessed in eight prospective cohorts from Europe (n = 4), North America (n = 2) and Asia (n = 2)24,46,47,56,63,68.The number of participants across all cohorts was 1,185,969 individuals; the mean duration of follow-up was 11.4 years (range, 5.5–26 years); and the total number of events was 12,500 (11,996 nonfatal and 504 fatal cases). The RRs were adjusted for BMI (n = 6) in all but two cohorts and for dietary components in all cohorts (n = 8). Most studies reported the RR of ischemic stroke incidence (n = 6). No bias covariates were found to be statistically relevant and therefore none were included in the model.
The exposure-averaged BPRF value for ischemic stroke (averaged between 15th and 85th percentiles of red meat exposure) was 0.98 (Table 2 and Extended Data Fig. 3), which put it opposite null from the mean RR of 1.15 (95% UI inclusive of between-study heterogeneity of 0.93–1.40) at 100 g d−1. The corresponding ROS of –0.02 resulted in a one-star rating, consistent with no evidence of an association between consumption of unprocessed red meat and increased risk of ischemic stroke.
Unprocessed red meat consumption and hemorrhagic stroke
We evaluated the relationship between consumption of unprocessed red meat and hemorrhagic stroke using eight prospective cohorts from Europe (n = 4), North America (n = 2) and Asia (n = 2)24,46,47,56,63,68. The number of participants across all cohorts was 1,185,969 individuals the mean duration of follow-up was 11.4 years (range: 5.5–26 years); and the total number of events was 4,176 (3,646 nonfatal and 530 fatal cases). In addition to adjustments to the RRs for major confounders, all but two cohorts included adjustments for BMI (n = 6) and all were adjusted for dietary components (n = 8). More studies reported the RR of hemorrhagic stroke incidence (n = 6) than mortality. No bias covariates had a significant effect on RR, so none were included in the model.
The exposure-averaged BPRF value for hemorrhagic stroke was 1.14 (Table 2 and Extended Data Fig. 4), which was opposite null from the mean RR of 0.87 (0.56–1.35) at 100 g d−1. The corresponding ROS of −0.13 resulted in a one-star rating, consistent with no evidence of an association between consumption of unprocessed red meat and decreased risk of hemorrhagic stroke.
Minimum risk level of unprocessed red meat intake
The aggregated risk curve for the six outcomes in our analysis combined was minimized at a mean unprocessed red meat consumption level of 0 g d−1 (95% UI 0–200) (Fig. 4). As shown in Fig. 4, the relationship between unprocessed red meat intake and combined-cause incidence and mortality was increasing across the entire exposure domain. Additional details on the results, along with sensitivity tests, are presented in Supplementary Information (section 2.3).
Risk of bias assessment
We assessed potential publication and reporting bias using Egger’s regression and visual inspection of the funnel plots (Figs. 1c–3c and Extended Data Figs. 2c–4c). We found no evidence of publication or reporting bias in five of the six disease outcomes investigated, and mild evidence in ischemic stroke. Supplementary Information (section 3) contains detailed results for our assessment of study quality and risk of bias.
Discussion
We evaluated the relationship between unprocessed red meat consumption and six selected disease outcomes following implementation of a meta-analytic approach20. We found that unprocessed red meat intake had weak evidence of an association with increased risk of colorectal cancer, breast cancer, IHD and type 2 diabetes and no evidence of an association with ischemic stroke and hemorrhagic stroke. In other words, given all the data available on red meat intake and risk of a subsequent outcome, we estimate that consuming unprocessed red meat across an average range of exposure levels increases the risk of subsequent colorectal cancer, breast cancer, IHD and type 2 diabetes at least slightly compared to eating no red meat (by at least 6%, 3%, 1% and 1%, respectively). Furthermore, the conservative interpretation of available data is consistent with no association between consuming unprocessed red meat and the risk of subsequent ischemic stroke or hemorrhagic stroke.
Based upon the star rating categories20, unprocessed red meat and colorectal cancer, breast cancer, IHD and type 2 diabetes are all two-star pairs, whereas the two stroke causes are one-star pairs. These star ratings reflect both the magnitude of the RR and its uncertainty and offer policy makers a simple way to conceptualize and compare the evidence for an association between risk and outcome. However, policy makers should pay attention to all potential risks, even those that only receive one-star or two-star ratings and especially when exposure prevalence is high. Further, the precautionary principle suggests that public policy should pay attention to all potential risks, as it is possible that as evidence accumulates, a stronger association may emerge for some pairs. Conversely, the results for IHD and type 2 diabetes are at the threshold between two-star and one-star ratings and further evidence may show that available data are consistent with no association between elevated unprocessed meat consumption and these outcomes. Because the current evidence for an association between unprocessed red meat and risk of these health outcomes is weak there is a critical need for better data from large-scale, high-quality studies in locations around the world. More evidence either in support of or against an association will allow policy makers to make better-informed decisions on diet recommendations.
A key finding of our analysis is that there is substantial between-study heterogeneity and uncertainty for all six risk–outcome pairs included. This may partly reflect the high degree of heterogeneity often present in data sources used for dietary analysis, which typically comprise observational studies. This heterogeneity limited the sensitivity of our analysis to identify clear—and potentially clinically important—relationships between intake and disease end points. Although visual inspection of the mean risk functions suggests a positive (harmful) relationship between unprocessed red meat intake and colorectal cancer, type 2 diabetes, IHD, ischemic stroke and breast cancer and a negative (protective) relationship with hemorrhagic stroke, the large degree of heterogeneity present, coupled with the moderate mean effects, generated wide UIs for the mean risk functions.
The level of red meat intake to optimize physical health is the subject of much interest and there is a wide range of recommendations in the literature6,7,8,9,10. By aggregating the outcome-specific risk curves computed in the present analysis, we generated a RR curve for the six outcomes combined that minimized risk at 0 g d−1 (95% UI 0–200) of unprocessed red meat consumption. This mean minimum risk is lower than the intake level recommended by the EAT-Lancet Commission (14 g d−1)8. Across the full range of exposures plotted on the combined-cause RR curve, we did not observe a significant relationship between unprocessed red meat consumption and combined-cause incidence and mortality (the 95% uncertainty interval is inclusive of 0). In light of these findings, we contend that consuming no unprocessed red meat likely minimizes the risk of health consequences compared to consuming any, but that the wide uncertainty and low star ratings prevent us from making a strong intake-level recommendation.
Previous meta-analyses have varied in their findings. With respect to colorectal cancer, the WCRF changed the grade of evidence for the relationship between red meat intake and colorectal cancer from possible to probable in 2017, reporting an RR of 1.12 (1.00–1.25)76. This estimate of RR is lower in magnitude than our result but was still found to be significant. For breast cancer, our finding of weak evidence of an association is generally consistent with previous literature, including a previous meta-analyses conducted by Farvid and colleagues (RR, 1.06; 0.99–1.14)77 and Anderson and colleagues (RR, 1.03; 0.99–1.08)78. Previous findings for IHD have been inconsistent, with Bechthold and colleagues finding an association between red meat intake and IHD16, but Abete and colleagues79 and Micha and colleagues80 reporting no association. Several meta-analyses examining the association between red meat consumption and type 2 diabetes identified associations of similar magnitude to our findings, though our UIs are wider; in 2017, Schwingshackl et al. reported an RR of 1.17 (1.08–1.26)15 and in 2011, Pan and colleagues found an RR of 1.19 (1.04–1.37)1. Although previous evidence on red meat and ischemic stroke has generally shown an association, which conflicts with our finding, the uncertainty intervals have been very close to the null. Yang et al.81 found that fresh (unprocessed) red meat consumption was associated with ischemic stroke (RR, 1.15; 1.03–1.29), as did Chen et al.82 (RR, 1.13; 1.01–1.25) and Kim et al.83 (1.24; 1.05–1.46) for total red meat. Any discrepancies between our findings and those in previous meta-analyses is primarily a reflection of our incorporation of between-study heterogeneity, not assuming a log-linear risk function and including newly available data. Our methods that included between-study heterogeneity in the uncertainty estimate revealed substantial uncertainty regarding the relationship between unprocessed red meat consumption and hemorrhagic stroke. Previous evidence regarding this relationship has been inconclusive. A 2019 study based on the EPIC Oxford cohort identified a higher rate of hemorrhagic stroke in vegetarians than in meat eaters, although this finding was not limited to red meat intake83. A meta-analysis by Yang and colleagues reported a pooled RR of 0.88 (0.73–1.06) for each 100 g d−1 increase in intake of unprocessed red meat81. Before that, meta-analyses by Chen and colleagues reported an RR of 0.99 (0.77–1.28)82 and Kaluza and colleagues reported an RR of 1.08 (0.84–1.39)84.
The effects of unprocessed red meat intake we observed vary over consumption levels. In particular, we observed high data inconsistency in evidence at low levels of red meat consumption, illustrating the importance of which foods serve as replacements for red meat. A systematic review of evidence from trials evaluating the effect of red meat on cardiovascular risk factors highlighted this replacement effect, showing that the harmful impact of red meat intake depends on the types of foods consumed in place of red meat85. At a lower level of intake, a wide range of healthy or unhealthy diet components might be substituted for red meat and could increase heterogeneity in the health effects of red meat across studies. We also observed a plateauing of the risk curves at high intake, indicating that above a certain threshold of intake, the risk levels off. Our ability to observe these non-linear relationships suggests that the associations between red meat intake and disease end points are better characterized by a flexible non-log-linear risk curve, allowing the observation of patterns that were potentially obscured in previous research by reliance on log-linear risk curves.
Our analysis has several strengths. We present an approach to dose–response risk curve estimation that uses exposure ranges from cohort studies to infer flexible risk functions without imposing log-linear assumptions. The approach quantifies between-study heterogeneity and uses this information to distinguish risk–outcome pairs with strong evidence from those with weak evidence to inform policies and guidance on unprocessed red meat intake. To explain some of the between-study heterogeneity, the approach uses bias covariates automatically selected from an expert-defined candidate set consistent with GRADE86, Cochrane87 and other evidence-grading criteria. Application of this analysis method to other diet components may yield insights into the shape of risk functions and burden of proof risk functions for a variety of dietary factors. Further, our analysis method and findings are compatible with GBD analyses of prevalence of exposure and background rates of disease outcomes. Collectively, the GBD comparative risk assessment framework allows for the evaluation of the importance of risk–outcome pairs across the full range of risk-attributable burdens and star ratings. For instance, a risk factor with high prevalence of exposure and a two-star relationship with a common and serious health outcome might warrant more policy focus than a risk factor with lower prevalence of exposure and a three-star or four-star relationship with a rare or less severe health outcome. The star rating for a risk–outcome pair is just one component to consider when making policy recommendations, but its compatibility with GBD estimates adds value to our approach.
Although our meta-analysis approach presents an analytic framework to quantify and account for a wide range of source data characteristics that may have obscured aspects of risk–outcome relationships investigated in previous studies, there were a number of limitations related to our source data, our methodological approach and our ability to interpret the data that we were unable to address. First, we did not identify any randomized controlled trials that evaluate the relationship between red meat intake and chronic diseases among adults; therefore, all of the studies included in our analysis were observational and we were unable to definitively assess causality. Second, while many of the sources we included adjusted to some degree for major confounders such as age, sex, smoking, certain other dietary components, cooking method, socioeconomic status and regional dietary patterns, the level and rigor of adjustment varied across studies, leaving the potential for residual confounding. Third, the definition of exposure and unit of exposure were not identical across studies and consumption of red meat was self-reported (and in most studies assessed only at baseline), raising the potential for recall bias and other reporting and/or measurement errors that may have resulted in exposure misclassification. Fourth, we assessed and summarized the risk of bias for each study but included all data points in our model instead of limiting to those scoring ‘low risk of bias’ on standard assessments because of the low availability of data. Fifth, the data came primarily from North America, Europe and Asia, which potentially limits the interpretability and application of our findings to locations outside of these regions. Sixth, although research suggests that red meat intake may be crucially related to infant and child growth and development88,89, this analysis evaluated the effect of red meat consumption on selected chronic disease end points in adults only and did not attempt to address questions of development in children and adolescents. Seventh, our trimming approach requires that the threshold for outliers be user-specified. Our sensitivity analyses demonstrate that fitting 90% of the data was most appropriate, but automation of this process would strengthen the approach. Eighth, types of publication and reporting biases other than those related to the association of reported effect sizes and s.d. are difficult to evaluate within our methodological framework. This is particularly true when studies are more consistent with each other than expected by chance20. Ninth, we used Gaussian priors on the bias covariates to avoid overfitting, which meant that relationships were only identified when there were sufficient studies supporting the estimate of the bias. Using alternative priors might change the biases that were detected. Finally, we chose to calculate an exposure-averaged BPRF and ROS based on a wide range of exposure levels (15th to 85th percentiles). For some pairs, particularly for the one-star pairs, there may be smaller ranges of exposure within which the magnitude of the BPRF was larger and significant, indicating a significant association between risk and outcome at a certain level of exposure. Picking an ‘average’ range of exposure was necessary for assigning each pair a single star rating and we believe a single rating is more useful from a policy perspective because of its simplicity, but this added level of complexity may be worth considering in subsequent analyses.
In conclusion, we applied the BPRF framework of Zheng et al.20 to present a systematic meta-analysis of red meat consumption on six important health outcomes. Our analysis found a high degree of between-study heterogeneity and uncertainty in the existing body of evidence on the health effects of red meat intake. When all forms of uncertainty including between-study heterogeneity were incorporated, we found weak evidence of associations between unprocessed red meat consumption and colorectal cancer, breast cancer, IHD and type 2 diabetes. The available data were consistent with no association between unprocessed red meat consumption and ischemic stroke and hemorrhagic stroke, which received one-star ratings. While there is some evidence that eating red meat increases risk of chronic disease, there is insufficient evidence to make stronger or more conclusive recommendations. More rigorous, well-powered research is needed to better understand and quantify the relationship between red meat consumption and chronic disease.
Methods
Overview
The analytical approach (described previously20) can be summarized in six steps: (1) search and extract data from published studies using a standardized approach; (2) estimate the shape of the exposure versus RR relationship, integrating over exposure ranges in different comparison groups and avoiding the distorting effect of outliers; (3) test and adjust for systematic biases as a function of study attributes; (4) quantify remaining between-study heterogeneity while adjusting for within-study correlation induced by computing RRs for several alternatives with the same reference, as well as the number of studies; (5) evaluate evidence for small-study effects to evaluate potential risks of publication or reporting bias; and (6) estimate the BPRF—quantifying a conservative interpretation of the change in average risk across the range of exposure supported by the evidence—using this estimate to compute the ROS and map it onto a star-rating system, stratifying risk into five levels. Zheng and colleagues21 previously published the technical developments required to implement this approach and disseminated them using open-source Python libraries90,91.
The estimates for our primary indicators from this work—RRs across a range of exposures, BPRFs, ROSs and star ratings for each risk–outcome pair—are not specific to, or disaggregated by, specific populations (we did not estimate by location, sex or age group; though this analysis evaluated the effects of unprocessed red meat consumption on selected chronic disease end points in adults 25 years and older only and breast cancer is only applicable to females). The reports we referenced included information about the self-reported sex of the participants but did not all include sex-specific RR estimates; also, studies were excluded if they did not meet our minimum threshold for confounder adjustment of adjusting for age and sex. These factors precluded us from performing any sex-based analyses. The measures of risk can be considered current until subsequent analyses are made based on newly available data.
We followed the PRISMA guidelines22 through all stages of this study (Extended Data Fig. 1 and Supplementary Tables 3 and 4). This study complies with the Guidelines on Accurate and Transparent Health Estimate Reporting recommendations92 (Supplementary Table 5). This study was approved by the University of Washington Institutional Review Board Committee (study 9060). The systematic review was not registered.
Selecting health outcomes
We selected outcomes on the basis of the availability of epidemiological evidence on their potential relationship with red meat. As detailed by Murray et al.5, risk–outcome pairs were initially selected using the WCRF criteria for convincing or probable evidence. Guidance from WCRF states that probable (strong) evidence is ‘strong enough to support a judgment of a probable causal (or protective) relationship.’7 A probable association generally requires evidence from at least two independent cohort studies, no substantial unexplained between-study heterogeneity, good-quality studies and evidence of biological plausibility.
After evaluating published literature on the relationship between red meat and various disease end points, we found sufficient studies assessing the relationship between red meat consumption and six outcomes: incidence of and mortality due to hemorrhagic stroke, type 2 diabetes, colorectal cancer, IHD and ischemic stroke and incidence of breast cancer. Supplementary Information, section 5, provides more details on the outcomes for which data were sought.
Conducting systematic reviews
We systematically searched PubMed for reports of cohort studies that included meat consumption, selecting reports evaluating the relationship between consumption of red meat and each of the outcomes. These literature searches were last performed on 10 May 2022. To ensure that we were capturing the most recent literature, we also searched Embase and Web of Science for reports published within the past 2 years, as well as the citation lists of recent systematic reviews on health effects of red meat1,15,16,77,78,79,80,81,82,83,93,94,95,96,97,98,99,100,101,102,103 for relevant original investigations. Titles and abstracts of all identified articles were manually screened by one investigator. A second investigator reviewed a random sample of 20% of excluded reports for potential discrepancies; no discrepancies were found. Full texts of potentially relevant articles were manually assessed for eligibility by two investigators. Supplementary Information, section 5.1, contains the full search string.
We defined red meat consumption as total consumption of unprocessed red meat, including beef, lamb and pork, excluding processed meat. Reports involving processed meat were excluded because we aimed to distinguish the health effects of red meat intake per se from the health effects of meat preservatives or preservation byproducts. We also excluded reports that did not report RRs or only reported RR estimates that were unadjusted for key confounders such as age and sex. When duplicate publications from the same study were identified, we only included the report that included the longest duration of follow-up in person-years.
We defined outcomes using the most highly specified diagnosis possible. For stroke outcomes, we excluded data points on total stroke from the hemorrhagic- and ischemic-specific models, as we found that including total stroke data points obscured the unique relationships between red meat and the two types of stroke for which we had sufficient data to model separately. Similarly, for IHD, we excluded RRs from incidence or mortality of unspecified cardiovascular disease outcomes and limited our model to RRs specifically associated with IHD outcomes to most accurately assess the relationship with IHD. Supplementary Information, section 5, contains a full list of inclusion and exclusion criteria. Data from 55 total reports met inclusion criteria for at least one of the six outcomes and were included in the risk–outcome pair-specific analysis1,11,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75.
Data extraction was manually conducted by two investigators. For each study, we extracted data on study name, location, design, population (age, sex, race and sample size), duration of follow-up, exposure definition, exposure assessment method, exposure categories, outcome definition, outcome ascertainment method and specific confounders that were included in the adjusted effect size. For each exposure category, we also collected data on the range of exposure, number of participants, person-years, number of events and risk estimate and associated uncertainty. Supplementary Table 6 contains a full list of extracted variables. We standardized the exposure unit to grams of consumption per day. For reports describing consumption in servings of red meat with no corresponding information about serving size, we assumed a serving size of 85 g d−1 (ref. 104). For reports that gave mean consumption rather than intake range, we calculated the range by using the midpoint between means to provide a cutoff for intake intervals. For undefined lower bounds, we assumed a consumption level of 0 g d−1. For undefined upper bounds when mean and s.d. values were not available, we applied the range from the cohort’s most adjacent quartile or tertile to estimate the upper bound of consumption, specific to each study cohort.
Estimating the shape of the risk–outcome relationship
Following Zheng et al.20, we modeled each non-linear dose–response as a quadratic spline21. For each risk–outcome pair, we first modeled the non-linear RR function without a monotonicity constraint to observe the unconstrained behavior. For outcomes in which the mean curve remained above one across the whole domain and was generally increasing, we then fitted a final model applying a monotonicity constraint to ensure that the mean risk curve was non-decreasing. For outcomes in which the curve decreased then increased and was minimized at a non-zero value, we did not apply a monotonicity constraint but instead implemented a linear-tail constraint (sometimes referred to as ‘natural’ splines in curve-fitting literature) on the left side of the domain to ensure more plausible risk curve behavior at low exposure levels. Linear-tail constraints ensure that the final segment on one side of the domain is linear, rather than a quadratic or cubic binomial and are a common way to regularize spline behavior. We present the sensitivity of the results based on this assumption in the Supplementary Information, section 2.1.
Because knot placement can affect the shape of the risk function when modeling with a spline, we generated a wide range of knot placements and created an ensemble across 50 component models, weighted by their fit to the data and the smoothness of fit to the observations. We also included in the final estimation 10% trimming of the data to avoid sensitivity to outliers; trimming from within the likelihood is an efficient way to identify outliers without manually selecting them21,105. Through this process, we generated a mean RR curve across the range of exposures (a measure of effect size) for each red meat–outcome pair. We obtained uncertainty estimates for the mean risk curves using a parametric bootstrap approach. Details on the fitting procedure, including trimming, ensemble generation and posterior uncertainty estimation, can be found in Zheng et al.20.
Testing for bias across different study designs and characteristics
To capture bias within the studies and data points, we followed the approach of Zheng et al.20, using a Lasso106,107 covariate selection scheme to rank the potential bias covariates. We then converted the dose–response relationship previously estimated by non-linear meta-regression into a new ‘signal’ covariate and used linear meta-regression to systematically test for significant modification of the ‘signal’ by each bias covariate—adding them to the model one at a time based on the Lasso ranking. Significant bias covariates were then included in the spline models; the details of this procedure have been published elsewhere20. Supplementary Information, section 3 provides more information on our risk of bias assessment.
Quantifying between-study heterogeneity, accounting for heterogeneity, uncertainty and small numbers of studies
The variance of between-study random effects is notoriously difficult to estimate. We quantified between-study heterogeneity (the level of disagreement in the inferred relationship between risk and outcome found in each study) by scaling the non-linear RR based on each study using a study-specific random slope with respect to the new bias covariate model. For cases with small numbers of studies, the estimated between-study heterogeneity can often be zero. To safeguard against underestimating the between-study heterogeneity when too few studies were available, we used the Fisher information matrix108 to estimate the variance of the between-study heterogeneity, Zheng et al.20 provides more details. Our main results include the effect of between-study heterogeneity in UIs, although we also present UIs that do not include it (as is standard in conventional meta-regressions) in Supplementary Table 7.
Evaluating potential for publication or reporting bias
To assess potential publication or reporting bias, we used a data-driven approach known as Egger’s regression109 to test for significant correlation between the residuals of the risk function and their s.d. When Egger’s regression failed to detect significant evidence of publication bias, we terminated the process.
When significant bias was detected, we adjusted for it using an appropriate modification of the trim-and-fill algorithm110. The trimming process in extracting the signal covariate is robust both to outliers and to classic cases of publication bias. Given a robust estimate of the mean curve, we applied the ‘fill’ part of the trim-and-fill approach, using the rank statistics and residuals to compute the number of points that need to be ‘filled’. The correct number of the most extreme residuals was then reflected and might affect our resulting estimate of heterogeneity. When fitting for the updated between-study heterogeneity, we used a prior coming from the original fit, with s.d. obtained from the Fisher information.
In addition to this statistical test of publication or reporting bias, we generated funnel plots of the residuals of the risk function and s.d. and inspected them visually. In the presence of significant publication bias, we used the trim-and-fill method to adjust for bias20,110.
Determining minimum risk consumption level
To determine the minimum risk level of red meat consumption, we aggregated the outcome-specific risk curves to generate an aggregated mortality curve for the six causes in our analysis combined, which we used to identify the exposure range that minimizes risk. Specifically, we took a weighted average of the risk curve draws for each outcome, using the GBD 2019 burden of each outcome—with deaths as the metric—as weights5,111. Then we calculated the exposure level that minimized combined-cause mortality for each draw and reported the median and associated 95% UI.
Estimating the burden of proof risk function
Using estimates of the mean risk function and corresponding 95% UIs—inclusive of between-study heterogeneity and reflective of other bias adjustments—as described in the preceding sections, we calculated a BPRF for each risk–outcome pair. Specifically, for γ = between-study heterogeneity, we let γ0.95 represent the 95% quantile according to the asymptotic distribution obtained using Fisher information. We computed the lower envelope of the log-RR curve that included fixed effects uncertainty as well as γ0.95 and found the average value of this curve at specific exposures and along the specified ranges of exposure. The BPRF is defined as the fifth (if harmful) or 95th (if protective) quantile risk curve—inclusive of between-study heterogeneity—closest to the RR equal to one (the null). The BPRF reflects a conservative interpretation of the available evidence and is a measure of the lowest level of excess risk (or risk reduction, for protective risks) that is consistent with the available data; the higher the BPRF, the higher the magnitude and strength of the relationship. This value can be interpreted such that even accounting for between-study heterogeneity and its uncertainty, we are confident that the log RR across the studied unprocessed meat consumption range is at least as high as the BPRF (or at least as low as the BPRF for a protective risk).
We then calculated ROSs as the signed average log RR of the BPRF over the 15th to 85th percentiles of observed exposure20. For example, a mean log BPRF of 0.4 for a harmful risk (where null = 0 for log RR) and a mean log BPRF of –0.4 for a protective risk would both have an ROS of 0.4 because the magnitude of the log RR is the same. In contrast, for risk–outcome pairs with a BPRF opposite the null from the mean risk, ROS would be calculated as negative. A positive risk score indicates that across average levels of exposure, the UI bound that is closest to null is on the same side of null as the mean risk curve. Positive ROSs indicate evidence of a risk–outcome relationship. Overall, a larger positive ROS indicates more consistency in evidence and a higher average effect size across the continuum of the risk. As an estimate of strength of evidence, the ROS is also directly comparable across outcomes, so we can use it to rank disease outcomes by confidence in their relationship to red meat consumption.
Finally, to aid interpretability of our findings for policy makers and research funders, we converted the ROSs to star-rating categories from one to five, with a higher rating indicating that the evidence for and magnitude of a relationship between risk and outcome is stronger. The conservative interpretation suggests that, for one-star pairs, there may be no true association between risk exposure and health outcome. As noted, we further divided the positive ROSs into ranges of at least a 0–15% increase (for harmful risks) or 0–13% decrease (for protective risks) in risk with average exposure (two stars), >15–50% increase or >13–34% decrease (three stars), >50–85% increase or >34–46% decrease (four stars) and greater than 85% increase or greater than 46% decrease (five stars). In ROS terms, the ranges are <0.0, 0.0–0.14, >0.14–0.41, >0.41–0.62 and >0.62 for both harmful and protective risks.
Model validation
We used detailed simulations to validate key components of the meta-regression model. The details of these model validations are available in Zheng et al.20. In brief, we simulated three scenarios: many studies with many data points per study, many studies with few data points per study and few studies with few data points per study. For each simulation, we compared the results from our model with results obtained using existing approaches, including a log-linear meta-analysis implemented in the metafor package112 as well as one-stage113 and two-stage114 competing approaches using the dosresmeta package115. For log-linear relationships, our approach and the metafor package showed similarly superior performance over the one- and two-stage approaches, whereas for non-log-linear relationships, our approach produced uniformly better performance across all scenarios compared to the other approaches.
Statistical analysis
Analyses were carried out using R v.3.6.1, Python v.3.8 and Stata v.17. To validate key aspects of the meta-regression model used in this analysis, the following packages were used, as described by Zheng et al. metafor (R package available for download at https://www.jstatsoft.org/article/view/v036i03) and dosmesreta (R package available for download at https://www.jstatsoft.org/article/view/v072c01).
Statistics and reproducibility
The study was a secondary analysis of existing data involving systematic reviews and meta-analyses. No statistical method was used to predetermine sample size. As the study did not involve primary data collection, randomization, blinding and data exclusions are not relevant to this study; and, as such no data were excluded and we performed no randomization or blinding. We have made our data and code available to foster reproducibility.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The findings from this study were produced using data available in the published literature. Data sources and citations for each risk–outcome pair can be downloaded using the ‘download’ button on each risk curve page available at https://vizhub.healthdata.org/burden-of-proof/. Study characteristics and citations for all input data used in the analyses are also provided in Supplementary Table 1, a template of the data collection form is in Supplementary Table 6 and the results from individual studies are in Supplementary Table 8.
Code availability
All code used for these analyses is publicly available online (https://github.com/ihmeuw-msca/burden-of-proof). This includes code for the meta-regression engine, the model specification interface, both parts of the data processing and risk-specific custom code, as appropriate.
References
Pan, A. et al. Red meat consumption and risk of type 2 diabetes: 3 cohorts of US adults and an updated meta-analysis. Am. J. Clin. Nutr. 94, 1088–1096 (2011).
Demeyer, D., Mertens, B., De Smet, S. & Ulens, M. Mechanisms linking colorectal cancer to the consumption of (processed) red meat: a review. Crit. Rev. Food Sci. Nutr. 56, 2747–2766 (2016).
Bellavia, A., Stilling, F. & Wolk, A. High red meat intake and all-cause cardiovascular and cancer mortality: is the risk modified by fruit and vegetable intake? Am. J. Clin. Nutr. 104, 1137–1143 (2016).
Larsson, S. C. & Orsini, N. Red meat and processed meat consumption and all-cause mortality: a meta-analysis. Am. J. Epidemiol. 179, 282–289 (2014).
Murray, C. J. L. et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1223–1249 (2020).
International Agency for Research on Cancer, World Health Organization. IARC Monographs evaluate consumption of red meat and processed meat. https://www.iarc.who.int/wp-content/uploads/2018/07/pr240_E.pdf (2015).
World Cancer Research Fund/American Institute for Cancer Research. Continuous update project expert report 2018. Recommendations and public health and policy implications. https://www.wcrf.org/diet-and-cancer/ (2018).
Willett, W. et al. Food in the Anthropocene: the EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 393, 447–492 (2019).
US Department of Health and Human Services and US Department of Agriculture. 2015–2020 dietary guidelines for Americans. 8th edn. http://health.gov/dietaryguidelines/2015/guidelines/ (2015).
Johnston, B. C. et al. Unprocessed red meat and processed meat consumption: dietary guideline recommendations from the Nutritional Recommendations (NutriRECS) Consortium. Ann. Intern Med 171, 756–764 (2019).
Nagao, M., Iso, H., Yamagishi, K., Date, C. & Tamakoshi, A. Meat consumption in relation to mortality from cardiovascular disease among Japanese men and women. Eur. J. Clin. Nutr. 66, 687–693 (2012).
Kappeler, R., Eichholzer, M. & Rohrmann, S. Meat consumption and diet quality and mortality in NHANES III. Eur. J. Clin. Nutr. 67, 598–606 (2013).
Stanton, A. V. et al. 36-fold higher estimate of deaths attributable to red meat intake in GBD 2019: is this reliable? Lancet 399, e23–e26 (2022).
Murray, C. J. L. 36-fold higher estimate of deaths attributable to red meat intake in GBD 2019: is this reliable? – Author’s reply. Lancet 399, e27–e28 (2022).
Schwingshackl, L. et al. Food groups and risk of type 2 diabetes mellitus: a systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 32, 363–375 (2017).
Bechthold, A. et al. Food groups and risk of coronary heart disease, stroke and heart failure: a systematic review and dose–response meta-analysis of prospective studies. Crit. Rev. Food Sci. Nutr. 59, 1071–1090 (2019).
Stayner, L., Steenland, K., Dosemeci, M. & Hertz-Picciotto, I. Attenuation of exposure-response curves in occupational cohort studies at high exposure levels. Scand. J. Work Environ. Health 29, 317–324 (2003).
Kyu, H. H. et al. Physical activity and risk of breast cancer, colon cancer, diabetes, ischemic heart disease, and ischemic stroke events: systematic review and dose–response meta-analysis for the Global Burden of Disease Study 2013. Brit. Med. J. 354, i3857 (2016).
Mueller, M. et al. Methods to systematically review and meta-analyse observational studies: a systematic scoping review of recommendations. BMC Med. Res. Method. 18, 44 (2018).
Zheng, P. et al. The burden of proof studies: assessing the evidence of risk. Nat. Med. (in the press).
Zheng, P., Barber, R., Sorensen, R. J., Murray, C. J. & Aravkin, A. Y. Trimmed constrained mixed effects models: formulations and algorithms. J. Comput. Graph. Stat. https://doi.org/10.1080/10618600.2020.1868303 (2021).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Brit. Med. J. 372, n71 (2021).
Al-Shaar, L. et al. Red meat intake and risk of coronary heart disease among US men: prospective cohort study. Brit. Med. J. 371, m4141 (2020).
Bernstein, A. M. et al. Dietary protein sources and the risk of stroke in men and women. Stroke 43, 637–644 (2012).
Bernstein, A. M. et al. Major dietary protein sources and risk of coronary heart disease in women. Circulation 122, 876–883 (2010).
Diallo, A. et al. Red and processed meat intake and cancer risk: results from the prospective NutriNet-Santé cohort study. Int. J. Cancer 142, 230–237 (2018).
Egeberg, R. et al. Associations between red meat and risks for colon and rectal cancer depend on the type of red meat consumed. J. Nutr. 143, 464–472 (2013).
English, D. R. et al. Red meat, chicken, and fish consumption and risk of colorectal cancer. Cancer Epidemiol. Biomark. Prev. 13, 1509–1514 (2004).
Ericson, U. et al. Food sources of fat may clarify the inconsistent role of dietary fat intake for incidence of type 2 diabetes. Am. J. Clin. Nutr. 101, 1065–1080 (2015).
Etemadi, A. et al. Mortality from different causes associated with meat, heme iron, nitrates, and nitrites in the NIH-AARP Diet and Health study: population-based cohort study. Brit. Med. J. 357, j1957 (2017).
Fraser, G. E. Associations between diet and cancer, ischemic heart disease, and all-cause mortality in non-Hispanic white California Seventh-day Adventists. Am. J. Clin. Nutr. 70, 532s–538s (1999).
Fretts, A. M. et al. Associations of processed meat and unprocessed red meat intake with incident diabetes: the Strong Heart Family study. Am. J. Clin. Nutr. 95, 752–758 (2012).
Genkinger, J. M., Makambi, K. H., Palmer, J. R., Rosenberg, L. & Adams-Campbell, L. L. Consumption of dairy and meat in relation to breast cancer risk in the Black Women’s Health Study. Cancer Causes Control 24, 675–684 (2013).
Gilsing, A. M. J. et al. Vegetarianism, low meat consumption and the risk of lung, postmenopausal breast and prostate cancer in a population-based cohort study. Eur. J. Clin. Nutr. 70, 723–729 (2016).
Gilsing, A. M. J. et al. Vegetarianism, low meat consumption and the risk of colorectal cancer in a population based cohort study. Sci. Rep. 5, 13484 (2015).
Haring, B. et al. Dietary protein intake and coronary heart disease in a large community based cohort: results from the Atherosclerosis Risk in Communities (ARIC) study. PLoS ONE 9, e109552 (2014).
Inoue-Choi, M., Sinha, R., Gierach, G. L. & Ward, M. H. Red and processed meat, nitrite, and heme iron intakes and postmenopausal breast cancer risk in the NIH-AARP Diet and Health study. Int. J. Cancer 138, 1609–1618 (2016).
Järvinen, R., Knekt, P., Hakulinen, T., Rissanen, H. & Heliövaara, M. Dietary fat, cholesterol and colorectal cancer in a prospective study. Br. J. Cancer 85, 357–361 (2001).
Jones, R. R. et al. Ingested nitrate, disinfection by-products, and risk of colon and rectal cancers in the Iowa Women’s Health Study cohort. Environ. Int. 126, 242–251 (2019).
Kabat, G. C., Miller, A. B., Jain, M. & Rohan, T. E. Dietary iron and heme iron intake and risk of breast cancer: a prospective cohort study. Cancer Epidemiol. Biomark. Prev. 16, 1306–1308 (2007).
Key, T. J. et al. Consumption of meat, fish, dairy products, and eggs and risk of ischemic heart disease. Circulation 139, 2835–2845 (2019).
Knuppel, A. et al. Meat intake and cancer risk: prospective analyses in UK Biobank. Int. J. Epidemiol. 49, 1540–1552 (2020).
Kurotani, K. et al. Red meat consumption is associated with the risk of type 2 diabetes in men but not in women: a Japan Public Health Center-based Prospective study. Br. J. Nutr. 110, 1910–1918 (2013).
Lajous, M. et al. Processed and unprocessed red meat consumption and incident type 2 diabetes among French women. Diabetes Care 35, 128–130 (2011).
Larsson, S. C., Rafter, J., Holmberg, L., Bergkvist, L. & Wolk, A. Red meat consumption and risk of cancers of the proximal colon, distal colon and rectum: the Swedish Mammography Cohort. Int J. Cancer 113, 829–834 (2005).
Larsson, S. C., Virtamo, J. & Wolk, A. Red meat consumption and risk of stroke in Swedish women. Stroke 42, 324–329 (2011).
Larsson, S. C., Virtamo, J. & Wolk, A. Red meat consumption and risk of stroke in Swedish men. Am. J. Clin. Nutr. 94, 417–421 (2011).
Männistö, S., Kontto, J., Kataja-Tuomola, M., Albanes, D. & Virtamo, J. High processed meat consumption is a risk factor of type 2 diabetes in the α-Tocopherol, β-Carotene Cancer Prevention study. Br. J. Nutr. 103, 1817–1822 (2010).
Mehta, S. S. et al. A prospective analysis of red and processed meat consumption and risk of colorectal cancer in women. Cancer Epidemiol. Biomark. Prev. 29, 141–150 (2020).
Mejborn, H., Møller, S. P., Thygesen, L. C. & Biltoft-Jensen, A. Dietary intake of red meat, processed meat, and poultry and risk of colorectal cancer and all-cause mortality in the context of dietary guideline compliance. Nutrients 13, E32 (2020).
Mills, P. K., Beeson, W. L., Phillips, R. L. & Fraser, G. E. Dietary habits and breast cancer incidence among Seventh-day Adventists. Cancer 64, 582–590 (1989).
Møller, S. P., Mejborn, H., Christensen, A. I., Biltoft-Jensen, A. & Thygesen, L. C. Meat consumption, stratified by dietary quality, and risk of heart disease. Br. J. Nutr. 126, 1881–1887 (2021).
Montonen, J. et al. Food consumption and the incidence of type II diabetes mellitus. Eur. J. Clin. Nutr. 59, 441–448 (2005).
Ollberding, N. J., Wilkens, L. R., Henderson, B. E., Kolonel, L. N. & & Le Marchand, L. Meat consumption, heterocyclic amines and colorectal cancer risk: the Multiethnic Cohort Study. Int. J. Cancer 131, E1125–E1133 (2012).
Pala, V. et al. Meat, eggs, dairy products, and risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Am. J. Clin. Nutr. 90, 602–612 (2009).
Papier, K. et al. Meat consumption and risk of 25 common conditions: outcome-wide analyses in 475,000 men and women in the UK Biobank study. BMC Med. 19, 53 (2021).
Parr, C. L., Hjartåker, A., Lund, E. & Veierød, M. B. Meat intake, cooking methods and risk of proximal colon, distal colon and rectal cancer: the Norwegian Women and Cancer (NOWAC) cohort study. Int. J. Cancer 133, 1153–1163 (2013).
Pietinen, P. et al. Diet and risk of colorectal cancer in a cohort of Finnish men. Cancer Causes Control 10, 387–396 (1999).
Pouchieu, C. et al. Prospective association between red and processed meat intakes and breast cancer risk: modulation by an antioxidant supplementation in the SU.VI.MAX randomized controlled trial. Int. J. Epidemiol. 43, 1583–1592 (2014).
Rajabi, A. A. et al. Towards refining World Cancer Research Fund/American Institute for Cancer Research cancer prevention recommendations for red and processed meat intake: insights from Alberta’s Tomorrow Project cohort. Br. J. Nutr. 127, 607–618 (2022).
Singh, P. N. & Fraser, G. E. Dietary risk factors for colon cancer in a low-risk population. Am. J. Epidemiol. 148, 761–774 (1998).
Steinbrecher, A., Erber, E., Grandinetti, A., Kolonel, L. N. & Maskarinec, G. Meat consumption and risk of type 2 diabetes: the Multiethnic Cohort. Public Health Nutr. 14, 568–574 (2011).
Takata, Y. et al. Red meat and poultry intakes and risk of total and cause-specific mortality: results from cohort studies of Chinese adults in Shanghai. PLoS ONE 8, e56963 (2013).
Talaei, M., Wang, Y.-L., Yuan, J.-M., Pan, A. & Koh, W.-P. Meat, dietary heme iron, and risk of type 2 diabetes mellitus: the Singapore Chinese Health study. Am. J. Epidemiol. 186, 824–833 (2017).
Taylor, E. F., Burley, V. J., Greenwood, D. C. & Cade, J. E. Meat consumption and risk of breast cancer in the UK Women’s Cohort study. Br. J. Cancer 96, 1139–1146 (2007).
The InterAct Consortium. Association between dietary meat consumption and incident type 2 diabetes: the EPIC-InterAct study. Diabetologia 56, 47–59 (2013).
Tiemersma, E. W. et al. Meat consumption, cigarette smoking, and genetic susceptibility in the etiology of colorectal cancer: results from a Dutch prospective study. Cancer Causes Control 13, 383–393 (2002).
Tong, T. Y. N. et al. The associations of major foods and fibre with risks of ischaemic and haemorrhagic stroke: a prospective study of 418 329 participants in the EPIC cohort across nine European countries. Eur. Heart J. 41, 2632–2640 (2020).
van Woudenbergh, G. J. et al. Meat consumption and its association with C-reactive protein and incident type 2 diabetes: the Rotterdam study. Diabetes Care 35, 1499–1505 (2012).
Villegas, R. et al. The association of meat intake and the risk of type 2 diabetes may be modified by body weight. Int. J. Med Sci. 3, 152–159 (2006).
Virtanen, H. E. K. et al. Intake of different dietary proteins and risk of type 2 diabetes in men: the Kuopio Ischaemic Heart Disease Risk Factor Study. Br. J. Nutr. 117, 882–893 (2017).
Ward, H. A. et al. Pre-diagnostic meat and fibre intakes in relation to colorectal cancer survival in the European prospective investigation into cancer and nutrition. Br. J. Nutr. 116, 316–325 (2016).
Wei, E. K. et al. Comparison of risk factors for colon and rectal cancer. Int. J. Cancer 108, 433–442 (2004).
Whiteman, D., Muir, J., Jones, L., Murphy, M. & Key, T. Dietary questions as determinants of mortality: the OXCHECK experience. Public Health Nutr. 2, 477–487 (1999).
Yiannakou, I. et al. A prospective analysis of red and processed meat intake in relation to colorectal cancer in the Black Women’s Health study. J. Nutr. 152, 1254–1262 (2022).
World Cancer Research Fund/American Institute for Cancer Research. Diet, Nutrition, Physical Activity and Colorectal Cancer. https://www.wcrf.org/wp-content/uploads/2021/02/Colorectal-cancer-report.pdf
Farvid, M. S. et al. Consumption of red meat and processed meat and cancer incidence: a systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 36, 937–951 (2021).
Anderson, J. J. et al. Red and processed meat consumption and breast cancer: UK Biobank cohort study and meta-analysis. Eur. J. Cancer 90, 73–82 (2018).
Abete, I., Romaguera, D., Vieira, A. R., Lopez de Munain, A. & Norat, T. Association between total, processed, red and white meat consumption and all-cause, CVD and IHD mortality: a meta-analysis of cohort studies. Br. J. Nutr. 112, 762–775 (2014).
Micha, R., Wallace, S. K. & Mozaffarian, D. Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus. Circulation 121, 2271–2283 (2010).
Yang, C. et al. Red meat consumption and the risk of stroke: a dose–response meta-analysis of prospective cohort studies. J. Stroke Cerebrovasc. Dis. 25, 1177–1186 (2016).
Chen, G.-C., Lv, D.-B., Pang, Z. & Liu, Q.-F. Red and processed meat consumption and risk of stroke: a meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 67, 91–95 (2013).
Kim, K. et al. Role of total, red, processed, and white meat consumption in stroke incidence and mortality: a systematic review and meta‐analysis of prospective cohort studies. J. Am. Heart Assoc. 6, e005983 (2017).
Kaluza, J., Wolk, A. & Larsson, S. C. Red meat consumption and risk of stroke: a meta-analysis of prospective studies. Stroke 43, 2556–2560 (2012).
Guasch-Ferré, M. et al. Meta-analysis of randomized controlled trials of red meat consumption in comparison with various comparison diets on cardiovascular risk factors. Circulation 139, 1828–1845 (2019).
Schünemann, H. et al. (eds) GRADE Handbook (GRADE Working Group, 2013).
Higgins, J. P. et al. Cochrane Handbook for Systematic Reviews of Interventions (John Wiley & Sons, 2019).
Morgan, J., Taylor, A. & Fewtrell, M. Meat consumption is positively associated with psychomotor outcome in children up to 24 months of age. J. Pediatr. Gastroenterol. Nutr. 39, 493–498 (2004).
Tang, M. & Krebs, N. F. High protein intake from meat as complementary food increases growth but not adiposity in breastfed infants: a randomized trial. Am. J. Clin. Nutr. 100, 1322–1328 (2014).
Zheng, P. limetr: linear mixed effects model with trimming. https://github.com/zhengp0/limetr.
Zheng, P. xspline: advanced spline tools. https://github.com/zhengp0/xspline.
Stevens, G. A. et al. Guidelines for Accurate and Transparent Health Estimates Reporting: the GATHER statement. Lancet 388, e19–e23 (2016).
Farvid, M. S. et al. Consumption of red and processed meat and breast cancer incidence: a systematic review and meta-analysis of prospective studies. Int. J. Cancer 143, 2787–2799 (2018).
Alexander, D. D., Weed, D. L., Cushing, C. A. & Lowe, K. A. Meta-analysis of prospective studies of red meat consumption and colorectal cancer. Eur. J. Cancer Prev. 20, 293–307 (2011).
Chan, D. S. M. et al. Red and processed meat and colorectal cancer incidence: meta-analysis of prospective studies. PLoS ONE 6, e20456 (2011).
de Medeiros, G. C. B. S. et al. Associations of the consumption of unprocessed red meat and processed meat with the incidence of cardiovascular disease and mortality, and the dose–response relationship: a systematic review and meta-analysis of cohort studies. Crit. Rev. Food Sci. Nutr. 0, 1–14 (2022).
Han, M. A. et al. Reduction of red and processed meat intake and cancer mortality and incidence. Ann. Intern. Med. 171, 711–720 (2019).
Kaluza, J., Wolk, A. & Larsson, S. C. Red meat consumption and risk of stroke. Stroke 43, 2556–2560 (2012).
Papier, K., Knuppel, A., Syam, N., Jebb, S. A. & Key, T. J. Meat consumption and risk of ischemic heart disease: a systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 0, 1–12 (2021).
World Cancer Research Fund & American Institute for Cancer Research. Diet, Nutrition, Physical Activity and Colorectal Cancer. http://dietandcancerreport.org (2018).
Wu, J. et al. Dietary protein sources and incidence of breast cancer: a dose–response meta-analysis of prospective studies. Nutrients 8, 730 (2016).
Zeraatkar, D. et al. Red and processed meat consumption and risk for all-cause mortality and cardiometabolic outcomes. Ann. Intern. Med. 171, 703–710 (2019).
Zhang, R., Fu, J., Moore, J. B., Stoner, L. & Li, R. Processed and unprocessed red meat consumption and risk for type 2 diabetes mellitus: an updated meta-analysis of cohort studies. Int. J. Environ. Res. Public Health 18, 10788 (2021).
Pan, A. et al. Red meat consumption and mortality: results from 2 prospective cohort studies. Arch. Intern. Med. 172, 555–563 (2012).
Aravkin, A. & Davis, D. Trimmed statistical estimation via variance reduction. Math. Oper. Res. 45, 292–322 (2020).
Efron, B., Hastie, T., Johnstone, I. & Tibshirani, R. Least angle regression. Ann. Stat. 32, 407–451 (2004).
Tibshirani, R. Regression shrinkage and selection via the lasso. J. R. Stat. Soc. Ser. B Methodol. 58, 267–288 (1996).
Biggerstaff, B. J. & Tweedie, R. L. Incorporating variability in estimates of heterogeneity in the random effects model in meta-analysis. Stat. Med. 16, 753–768 (1997).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. Brit. Med. J. 315, 629–634 (1997).
Shi, L. & Lin, L. The trim-and-fill method for publication bias: practical guidelines and recommendations based on a large database of meta-analyses. Medicine 98, e15987 (2019).
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 36, 1–48 (2010).
Crippa, A., Discacciati, A., Bottai, M., Spiegelman, D. & Orsini, N. One-stage dose–response meta-analysis for aggregated data. Stat. Methods Med. Res. 28, 1579–1596 (2019).
Liu, Q., Cook, N. R., Bergström, A. & Hsieh, C.-C. A two-stage hierarchical regression model for meta-analysis of epidemiologic nonlinear dose–response data. Comput. Stat. Data Anal. 53, 4157–4167 (2009).
Crippa, A. & Orsini, N. Multivariate dose-response meta-analysis: the dosresmeta R package. J. Stat. Softw. 72, 1–15 (2016).
Acknowledgements
Research reported in this publication was supported by the Bill & Melinda Gates Foundation; Bloomberg Philanthropies; the University of Melbourne; Queensland Department of Health, Australia; the National Health and Medical Research Council, Australia; Public Health England; the Norwegian Institute of Public Health; St Jude Children’s Research Hospital; the Cardiovascular Medical Research and Education Fund; the National Institute on Ageing of the National Institutes of Health (award P30AG047845); and the National Institute of Mental Health of the National Institutes of Health (award R01MH110163). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funders. The funders of the study had no role in study design, data collection, data analysis, data interpretation, writing of the final report or the decision to publish.
Author information
Authors and Affiliations
Contributions
A.A., C.B., S.I.H., L.B.M., E.C.M. and C.J.L.M. managed the estimation or publications process. H.L., A.A., C.B., G.F., L.B.M. and C.J.L.M. wrote the first draft of the manuscript. H.L. and M.C.P. had primary responsibility for applying analytical methods to produce estimates. H.L., C.A., G.F., V.I., M.C.P. and A.L.S. had primary responsibility for seeking, cataloging, extracting or cleaning data, and for designing or coding figures and tables. A.A. and M.B. provided data or critical feedback on data sources. H.L., A.A., J.H., R.J.D.S., A.Y.A. and P.Z. developed methods or computational machinery. A.A., M.B., S.I.H., R.J.D.S., A.Y.A., P.Z. and C.J.L.M. provided critical feedback on methods or results. H.L., A.A., C.B., M.B., S.I.H., S.A.M., M.C.P., R.J.D.S., A.Y.A., P.Z. and C.J.L.M. drafted the work or revised it critically for important intellectual content. A.A., C.A., S.I.H., E.C.M. and A.Y.A. managed the overall research enterprise.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Sabita Soedamah-Muthu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Jennifer Sargent and Ming Yang, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 PRISMA flow diagram of unprocessed red meat data seeking approach.
The PRISMA flow diagram covering unprocessed red meat and all 6 outcomes. Template is from: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi: 10.1136/bmj.n71. For more information, visit: http://www.prisma-statement.org/. * EMBASE and Web of Science were searched for recent records (from January 1, 2020, to May 10, 2022) to augment the date-unrestricted PubMed search (records up to May 10, 2022).
Extended Data Fig. 2 Red meat consumption and type 2 diabetes.
a, log-relative risk function. b, relative risk function. c, A modified funnel plot showing the residuals (relative to 0) on the x-axis and the estimated standard deviation (SD) that includes reported SD and between-study heterogeneity on the y-axis.
Extended Data Fig. 3 Red meat consumption and ischemic stroke.
a, log-relative risk function. b, relative risk function. c, A modified funnel plot showing the residuals (relative to 0) on the x-axis and the estimated standard deviation (SD) that includes reported SD and between-study heterogeneity on the y-axis.
Extended Data Fig. 4 Red meat consumption and hemorrhagic stroke.
a, log-relative risk function. b, relative risk function. c, A modified funnel plot showing the residuals (relative to 0) on the x-axis and the estimated standard deviation (SD) that includes reported SD and between-study heterogeneity on the y-axis.
Supplementary information
Supplementary Information
Supplementary Sections 1–7, Supplementary Tables 1–8 and Supplementary Figs. 1–8
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lescinsky, H., Afshin, A., Ashbaugh, C. et al. Health effects associated with consumption of unprocessed red meat: a Burden of Proof study. Nat Med 28, 2075–2082 (2022). https://doi.org/10.1038/s41591-022-01968-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-022-01968-z
This article is cited by
-
Estimating effects of whole grain consumption on type 2 diabetes, colorectal cancer and cardiovascular disease: a burden of proof study
Nutrition Journal (2024)
-
Health effects associated with exposure to secondhand smoke: a Burden of Proof study
Nature Medicine (2024)
-
Swiss agriculture can become more sustainable and self-sufficient by shifting from forage to grain legume production
Communications Earth & Environment (2024)
-
Health effects associated with chewing tobacco: a Burden of Proof study
Nature Communications (2024)
-
Unacceptable use of substandard metrics in policy decisions which mandate large reductions in animal-source foods
npj Science of Food (2024)