Abstract
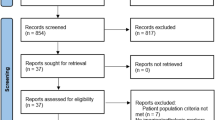
Nonalcoholic steatohepatitis (NASH) is a common chronic liver disease that may advance to fibrosis and lead to mortality; however, no pharmacotherapy is currently available. We tested the hypothesis that inhibition of both the sodium–glucose cotransporters 1 and 2 with licogliflozin would lead to improvement in NASH. A total of 107 patients with phenotypic or histologic NASH were randomized (1:2:2) to receive oral administration of either placebo (n = 21), licogliflozin 30 mg (n = 43) or 150 mg (n = 43) once daily for 12 weeks. Licogliflozin 150 mg showed a significant 32% (80% confidence interval (CI): 21–43%; P = 0.002) placebo-adjusted reduction in serum alanine aminotransferase after 12 weeks of treatment, the primary endpoint of the study. However, the 30 mg dose of licogliflozin did not meet the primary endpoint (placebo-adjusted reduction 21% (80% CI: 7–32%; P = 0.061)). Diarrhea occurred in 77% (33 of 43), 49% (21 of 43) and 43% (9 of 21) of patients treated with licogliflozin 150 mg, 30 mg and placebo, respectively, which was mostly mild in severity. No other major safety concerns were identified. Treatment with 150 mg licogliflozin led to reductions in serum alanine aminotransferase in patients with NASH. Studies of longer duration and in combination with drugs that have different mechanisms of action are needed to validate these findings and to define a role of licogliflozin as a therapeutic option for NASH. ClinicalTrials.gov identifier: NCT03205150.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this analysis are available within the article and its appendix. Requests for access to aggregate data and supporting clinical documents will be reviewed and approved by an independent review panel on the basis of scientific merit. All data provided are anonymized to respect the privacy of patients who have participated in the trial, in line with applicable laws and regulations. Availability of trial data is according to the criteria and process described at www.clinicalstudydatarequest.com.
References
Younossi, Z. et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 15, 11–20 (2018).
Chalasani, N. et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology 67, 328–357 (2018).
Williams, C. D. et al. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology 140, 124–131 (2011).
Wong, V. W. et al. Pathogenesis and novel treatment options for non-alcoholic steatohepatitis. Lancet Gastroenterol. Hepatol. 1, 56–67 (2016).
Younossi, Z. M. Non-alcoholic fatty liver disease - A global public health perspective. J. Hepatol. 70, 531–544 (2019).
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M. & Sanyal, A. J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 24, 908–922 (2018).
Vilar-Gomez, E. et al. Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology 149, 367–378 e365 (2015). quiz e314-365.
Lassailly, G. et al. Bariatric surgery provides long-term resolution of nonalcoholic steatohepatitis and regression of fibrosis. Gastroenterology 159, 1290–1301 e1295 (2020).
European Association for the Study of the Liver, European Association for the Study of Diabetes & European Association for the Study of Obesity EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 64, 1388–1402 (2016)..
Hallsworth, K. & Adams, L. A. Lifestyle modification in NAFLD/NASH: facts and figures. JHEP Rep. 1, 468–479 (2019).
Raj, H. et al. SGLT-2 inhibitors in non-alcoholic fatty liver disease patients with type 2 diabetes mellitus: a systematic review. World J. Diabetes 10, 114–132 (2019).
Hsiang, J. C. & Wong, V. W. SGLT2 inhibitors in liver patients. Clin. Gastroenterol. Hepatol. 18, 2168–2172 e2162 (2020).
Shimizu, M. et al. Evaluation of the effects of dapagliflozin, a sodium-glucose co-transporter-2 inhibitor, on hepatic steatosis and fibrosis using transient elastography in patients with type 2 diabetes and non-alcoholic fatty liver disease. Diabetes Obes. Metab. 21, 285–292 (2019).
Taheri, H. et al. Effect of empagliflozin on liver steatosis and fibrosis in patients with non-alcoholic fatty liver disease without diabetes: a randomized, double-blind, placebo-controlled trial. Adv. Ther. 37, 4697–4708 (2020).
He, Y. L. et al. The effects of licogliflozin, a dual SGLT1/2 inhibitor, on body weight in obese patients with or without diabetes. Diabetes Obes. Metab. 21, 1311–1321 (2019).
Poulsen, S. B., Fenton, R. A. & Rieg, T. Sodium-glucose cotransport. Curr. Opin. Nephrol. Hypertens. 24, 463–469 (2015).
Harrison, S. A. et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 394, 2012–2024 (2019).
Lawitz, E. J. et al. Acetyl-CoA carboxylase inhibitor GS-0976 for 12 weeks reduces hepatic de novo lipogenesis and steatosis in patients with nonalcoholic steatohepatitis. Clin. Gastroenterol. Hepatol. 16, 1983–1991 e1983 (2018).
Xie, Q. et al. The performance of enhanced liver fibrosis (ELF) test for the staging of liver fibrosis: a meta-analysis. PLoS ONE 9, e92772 (2014).
Kuchay, M. S. et al. Effect of empagliflozin on liver fat in patients with type 2 diabetes and nonalcoholic fatty liver disease: a randomized controlled trial (E-LIFT trial). Diabetes Care 41, 1801–1808 (2018).
Seko, Y. et al. Efficacy and safety of canagliflozin in type 2 diabetes mellitus patients with biopsy-proven nonalcoholic steatohepatitis classified as stage 1-3 fibrosis. Diabetes Metab. Syndr. Obes. 11, 835–843 (2018).
Stenlof, K. et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes. Metab. 15, 372–382 (2013).
Eriksson, J. W. et al. Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: a double-blind randomised placebo-controlled study. Diabetologia 61, 1923–1934 (2018).
Smith, G. I. et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J. Clin. Invest. 130, 1453–1460 (2020).
Loomba, R. et al. GS-0976 reduces hepatic steatosis and fibrosis markers in patients with nonalcoholic fatty liver disease. Gastroenterology 155, 1463–1473 e1466 (2018).
Harrison, S. A. et al. NGM282 for treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 391, 1174–1185 (2018).
Bays, H. E., Kozlovski, P., Shao, Q., Proot, P. & Keefe, D. Licogliflozin, a novel SGLT1 and 2 inhibitor: body weight effects in a randomized trial in adults with overweight or obesity. Obes. (Silver Spring) 28, 870–881 (2020).
He, Y., Schofield, J., Mahling, P., Mendonza, A. E. & Hinder, M. Investigation and management of stool frequency and consistency associated with SGLT1 inhibition by reducing dietary carbohydrate: a randomized trial. Clin. Pharmacol. Ther. 108, 995–1002 (2020).
Yokoo, T. et al. Nonalcoholic fatty liver disease: diagnostic and fat-grading accuracy of low-flip-angle multiecho gradient-recalled-echo MR imaging at 1.5T. Radiology 251, 67–76 (2009).
Yokoo, T. et al. Estimation of hepatic proton-density fat fraction by using MR imaging at 3.0T. Radiology 258, 749–759 (2011).
Mashhood, A. et al. Reproducibility of hepatic fat fraction measurement by magnetic resonance imaging. J. Magn. Reson. Imaging 37, 1359–1370 (2013).
Neuschwander-Tetri, B. A. et al. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet 385, 956–965 (2015).
Acknowledgments
We thank the patients for their participation in the study. The senior scientific writers, S. Kaur, Z. Birajdar and A. Meka of Novartis, provided medical writing and editorial support, which was funded by Novartis Institutes for BioMedical Research, Cambridge, MA, USA, in accordance with Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3). The study and these analyses were funded by Novartis Institutes for BioMedical Research, Cambridge, MA, USA, which was the sponsor of the study.
Author information
Authors and Affiliations
Contributions
All authors were involved in the preparation and critical review of the manuscript and approved the final version for submission. S.A.H., F.P.M., W.B.S., D. Alpenidze, D. Aizenberg, N.K., C.Y.C., E.Z., E.R., P.C., P.N.C., H.K., S.K., Z.B.A. and E.S., were involved in the acquisition of clinical data, and served as investigators in the clinical study from which data are reported in the manuscript. S.A.H. contributed to study design and review of protocol. A.E.M., Y.Z., M.M., S.M. S.K., S.T., A.P., M.K.B., Y.L.H. and C.U. were involved in the conception, design, statistical analysis and/or clinical conduct of the trial. All authors were involved in the interpretation of data in the manuscript. All authors agreed to be accountable for all aspects of the work and attest to the accuracy and integrity of the work.
Corresponding author
Ethics declarations
Competing interests
S.A.H. reports advisory board fees, consultation and research grants from Akero, Altimmune, Axcella, CirU/Ls, Cymabay, Galectin, Genfit, Hepion, Hightide, Intercept, Madrigal, Metacrine, NGM and Northsea; consultation and research grants from CIVI, ENYO, Gilead, Novartis and Viking; also consultation for Canfite, Echosens, Foresite, Fortress, Kowa, Medpace, Prometic and Ridgeline. W.B.S. is an employee of Alliance for Multispecialty Research. S.K. reports advisory board fees and consultation work for Boehringer Ingelheim, Novo Nordisk and Merck, also grants and research support from Johnson & Johnson. E.R. reports funding from Novartis for the present study. E.S., F.P.M., D. Alpenidze, D. Aizenberg, N.K., C.Y.C., E.Z., P.C., P.N.C., H.K. and Z.B.A. have nothing to disclose. A.M., Y.Z., M.M., S.M., S.K., M.K.B. and Y.L.H. are employees of Novartis. A.P. and C.U. were employees of Novartis until August 2021 and February 2021, respectively. S.T. was an employee of Novartis until 2021.
Peer review
Peer review information
Nature Medicine thanks Kenneth Cusi, Raj Vuppalanchi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling editors: Jennifer Sargent and Javier Carmona, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Overview of the study design.
Patients were screened during a 28-day window, followed by a baseline run-in period of 14-days prior to the day of first treatment. Patients were then randomized in a 1:2:2 ratio to receive a placebo or licogliflozin at 30 mg or 150 mg daily. The study drug was stopped after 12-weeks of treatment and patients were subsequently followed up for 28-days.
Extended Data Fig. 2 Change in absolute liver fat content after 12 weeks of treatment with licogliflozin.
a, Absolute decrease in liver fat content (mean change from baseline); b, proportion of patients (%) with an ≥5% absolute reduction in liver fat content. Baseline is defined as the last available measurement prior to the first dose. P values based on two-sided ANCOVA test. Error bars represent s.e. n, number of patients in each treatment group.
Extended Data Fig. 3 Liver fat in subgroup of patients with baseline percentage liver fat content ≥10%.
a, Absolute reduction in liver fat content (mean change from baseline); b, relative reduction in liver fat content (% change from baseline). Baseline is defined as the last available measurement prior to the first dose. P values based on two-sided ANCOVA test. n, number of patients in each treatment group.
Extended Data Fig. 4 Change in serum AST and GGT levels after 12 weeks of treatment with licogliflozin.
a, AST; b, GGT. Data presented as geometric mean ratio to baseline. Baseline is defined as the mean of measurements taken at the screening and baseline visits. A repeated-measures ANCOVA was performed for the log-transformed ratio to baseline. P values based on two-sided ANCOVA test: *P < 0.05, **P < 0.01, and ***P < 0.001. Error bars represent s.e. n for placebo, licogliflozin 30 mg, and licogliflozin 150 mg for AST: 21, 42, 38 (day 7); 21, 43, 36 (day 14); 21, 41, 33 (day 28); 20, 40, 33 (day 56); 20, 40, 34 (day 84); 19, 39, 34 (EOS); GGT: 21, 42, 38 (day 7); 21, 43, 36 (day 14); 21, 42, 34 (day 28); 20, 42, 34 (day 56); 20, 40, 34 (day 84); 19, 39, 34 (EOS). Dotted gray line represents end-of-treatment time point.
Extended Data Fig. 5 Changes in ELF panel and its components after 12 weeks of treatment.
a, ELF; b, HA; c, PIIINP; d, TIMP1; e, ELF (patient ≥ 8.5); f, HA (patient ≥ 8.5); g, PIIINP (patient ≥ 8.5); h, TIMP1 (patient ≥ 8.5). All data presented as geometric mean ratio to baseline. Baseline is defined as the last available measurement prior to the first dose. P values based on two-sided ANCOVA test. N for placebo, licogliflozin 30 mg, and 150 mg are 18, 34, and 29 (ELF, HA, PIIINP, TIMP1); 14, 40, and 26 (ELF ≥ 8.5, HA ≥ 8.5, PIIINP ≥ 8.5, TIMP1 ≥ 8.5). n, number of patients in each treatment group. Error bars represent s.e.
Extended Data Fig. 6 Change in metabolic parameters from baseline up to week 12.
a, HbA1c; b, HOMA-IR. *Ten patients who received exogenous insulin were excluded from HOMA-IR calculation (placebo (n = 3), licogliflozin 30 mg (n = 5), and licogliflozin 150 mg (n = 2)). Data presented as geometric mean ratio to baseline. P values based on two-sided ANCOVA test. Baseline is defined as the last available measurement prior to the first dose. n, number of patients in each treatment group. N for placebo, licogliflozin 30 mg, and licogliflozin 150 mg for HbA1c are 20, 40, and 34; N for HOMA-IR are placebo (n = 16), licogliflozin 30 mg (n = 32), and licogliflozin 150 mg (n = 31). Error bars represent s.e.
Extended Data Fig. 7 Mean HbA1c by treatment at baseline and week 12.
Data presented as mean (%). Error bars represent SD. n for placebo, licogliflozin 30 mg, and licogliflozin 150 mg for baseline: 21, 43, 41; week 12: 20, 40, 34.
Extended Data Fig. 8 Changes in biomarkers of liver fibrosis after 12 weeks of treatment with licogliflozin.
Data presented in subfigures (a–c) as geometric mean ratio to baseline, and in subfigure d, as mean change from baseline. a, PRO-C3; b, APRI; c, FIB4; d, NAFLD fibrosis score. Baseline is defined as the last available measurement prior to the first dose. P values based on two-sided ANCOVA test. N for placebo, licogliflozin 30 mg, and 150 mg are 21, 43, and 37 (APRI, FIB4, NAFLD fibrosis score) 20, 42, and 34 (Pro-C3). Error bars represent s.e.
Extended Data Fig. 9 Study outcomes with licogliflozin treatment in patients with or without diarrhea.
a, Bodyweight; b, ALT; c, placebo-adjusted decrease in LFC. P values based on two-sided ANCOVA test: *P < 0.05, **P < 0.01, and ***P < 0.001. Dotted gray line represents end of treatment time point. Data in subfigure a presented as mean % change from baseline, b, as geometric mean ratio to baseline, and c, as mean (%) reduction. Error bars represent s.e. n for placebo, licogliflozin 30 mg, and licogliflozin 150 mg for bodyweight in subgroup with diarrhea: 12, 21, 8 (day 7); 12, 22, 8 (day 14); 11, 21, 5 (day 28); 11, 21, 7 (day 42); 11, 21, 7 (day 56); 10, 19, 7 (day 84); 10, 19, 7 (EOS); bodyweight in subgroup without diarrhea: 9, 21, 30 (day 7); 9, 21, 28 (day 14); 9, 21, 25 (day 28); 9, 21, 27 (day 42); 9, 21, 26 (day 56); 9, 21, 27 (day 84); 9, 21, 27 (EOS). ALT in subgroup with diarrhea: 12, 21, 8 (day 7); 12, 22, 8 (day 14); 12, 21, 7 (day 28); 11, 21, 7 (day 56); 11, 19, 6 (day 84); 10, 18, 7 (EOS); ALT in subgroup without diarrhea: 9, 21, 30 (day 7); 9, 21, 28 (day 14); 9, 21, 27 (day 28); 9, 21, 27 (day 56); 9, 21, 26 (day 84); 9, 21, 27 (EOS). LFC in subgroup with diarrhea: 11, 18, 7 (day 84) and in subgroup without diarrhea: 8, 21, 26 (day 84). LFC, liver fat content.
Extended Data Fig. 10 Summary of findings of licogliflozin in NASH.
1He, Y.L. et al. Diabetes Obes. Metab. 21, 1311–1321 (2019). 2Current study data. ALT, alanine aminotransferase; GLP-1, glucagon-like peptide 1; NASH, nonalcoholic steatohepatitis; SGLT, sodium–glucose cotransporters; wt, weight.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Rights and permissions
About this article
Cite this article
Harrison, S.A., Manghi, F.P., Smith, W.B. et al. Licogliflozin for nonalcoholic steatohepatitis: a randomized, double-blind, placebo-controlled, phase 2a study. Nat Med 28, 1432–1438 (2022). https://doi.org/10.1038/s41591-022-01861-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-022-01861-9