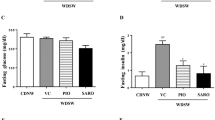
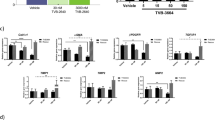
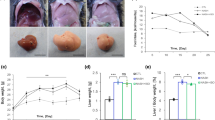
Abstract
Nonalcoholic steatohepatitis (NASH), a chronic liver disease without an approved therapy, is associated with lipotoxicity and insulin resistance and is a major cause of cirrhosis and hepatocellular carcinoma. Aramchol, a partial inhibitor of hepatic stearoyl-CoA desaturase (SCD1) improved steatohepatitis and fibrosis in rodents and reduced steatosis in an early clinical trial. ARREST, a 52-week, double-blind, placebo-controlled, phase 2b trial randomized 247 patients with NASH (n = 101, n = 98 and n = 48 in the Aramchol 400 mg, 600 mg and placebo arms, respectively; NCT02279524). The primary end point was a decrease in hepatic triglycerides by magnetic resonance spectroscopy at 52 weeks with a dose of 600 mg of Aramchol. Key secondary end points included liver histology and alanine aminotransferase (ALT). Aramchol 600 mg produced a placebo-corrected decrease in liver triglycerides without meeting the prespecified significance (−3.1, 95% confidence interval (CI) −6.4 to 0.2, P = 0.066), precluding further formal statistical analysis. NASH resolution without worsening fibrosis was achieved in 16.7% (13 out of 78) of Aramchol 600 mg versus 5% (2 out of 40) of the placebo arm (odds ratio (OR) = 4.74, 95% CI = 0.99 to 22.7) and fibrosis improvement by ≥1 stage without worsening NASH in 29.5% versus 17.5% (OR = 1.88, 95% CI = 0.7 to 5.0), respectively. The placebo-corrected decrease in ALT for 600 mg was −29.1 IU l−1 (95% CI = −41.6 to −16.5). Early termination due to adverse events (AEs) was <5%, and Aramchol 600 and 400 mg were safe, well tolerated and without imbalance in serious or severe AEs between arms. Although the primary end point of a reduction in liver fat did not meet the prespecified significance level with Aramchol 600 mg, the observed safety and changes in liver histology and enzymes provide a rationale for SCD1 modulation as a promising therapy for NASH and fibrosis and are being evaluated in an ongoing phase 3 program.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data supporting the findings of this study are owned by Galmed Research and Development Ltd. (Galmed) and contains potentially identifying or sensitive patient information since it includes, among others, human research participant data. Therefore, data are not publicly available due to patients’ right of privacy and confidentiality as well as ethical and commercial limitations imposed on Galmed. On request, Galmed will consider sharing certain datasets in accordance with applicable local laws as well as patient consent. Data sharing requests should include the type of data requested, the reason the data is requested and the intended use of the data.
References
Powell, E. E., Wong, V. W. & Rinella, M. Non-alcoholic fatty liver disease. Lancet 397, 2212–2224 (2021).
Samuel, V. T. & Shulman, G. I. Nonalcoholic fatty liver disease as a nexus of metabolic and hepatic diseases. Cell Metab. 27, 22–41 (2018).
Lambert, J. E., Ramos-Roman, M. A., Browning, J. D. & Parks, E. J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 146, 726–735 (2014).
Miyazaki, M. et al. Stearoyl-CoA desaturase 1 gene expression is necessary for fructose-mediated induction of lipogenic gene expression by sterol regulatory element-binding protein-1c-dependent and -independent mechanisms. J. Biol. Chem. 279, 25164–25171 (2004).
Gutiérrez-Juárez, R. et al. Critical role of stearoyl-CoA desaturase-1 (SCD1) in the onset of diet-induced hepatic insulin resistance. J. Clin. Invest. 116, 1686–1695 (2006).
Dobrzyn, P. et al. Stearoyl-CoA desaturase 1 deficiency increases fatty acid oxidation by activating AMP-activated protein kinase in liver. Proc. Natl Acad. Sci. USA 101, 6409–6414 (2004).
Bhattacharya, D. et al. Aramchol downregulates stearoyl CoA-desaturase 1 in hepatic stellate cells to attenuate cellular fibrogenesis. JHEP Rep. 3, 100237 (2021).
Powell, D. A. An overview of patented small molecule stearoyl coenzyme-A desaturase inhibitors (2009–2013). Expert Opin. Ther. Pat. 24, 155–175 (2014).
Leikin-Frenkel, A. et al. Fatty acid bile acid conjugate inhibits hepatic stearoyl coenzyme A desaturase and is non-atherogenic. Arch. Med. Res. 41, 397–404 (2010).
Gilat, T. et al. Prevention of diet-induced fatty liver in experimental animals by the oral administration of a fatty acid bile acid conjugate (FABAC). Hepatology 38, 436–442 (2003).
Leikin-Frenkel, A. et al. Treatment of preestablished diet-induced fatty liver by oral fatty acid-bile acid conjugates in rodents. Eur. J. Gastroenterol. Hepatol. 20, 1205–1213 (2008).
Golan-Gerstl, R. et al. SAT-454 - the anti-fibrotic effect of aramchol on fibrosis in TAA animal model. J. Hepatol. 66, S655–S656 (2017).
Iruarrizaga-Lejarreta, M. et al. Role of Aramchol in steatohepatitis and fibrosis in mice. Hepatol. Commun. 1, 911–927 (2017).
Safadi, R. et al. The fatty acid-bile acid conjugate Aramchol reduces liver fat content in patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 12, 2085–2091.e1 (2014).
Dulai, P. S., Sirlin, C. B. & Loomba, R. MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: clinical trials to clinical practice. J. Hepatol. 65, 1006–1016 (2016).
Loomba, R. et al. Multicenter validation of association between decline in MRI-PDFF and histologic response in NASH. Hepatology 72, 1219–1229 (2020).
Bril, F., Barb, D., Lomonaco, R., Lai, J. & Cusi, K. Change in hepatic fat content measured by MRI does not predict treatment-induced histological improvement of steatohepatitis. J. Hepatol. 72, 401–410 (2020).
Harrison, S. A. et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 394, 2012–2024 (2019).
Middleton, M. S. et al. Agreement between magnetic resonance imaging proton density fat fraction measurements and pathologist-assigned steatosis grades of liver biopsies from adults with nonalcoholic steatohepatitis. Gastroenterology 153, 753–761 (2017).
Sanyal, A. et al. Pegbelfermin (BMS-986036), a PEGylated fibroblast growth factor 21 analogue, in patients with non-alcoholic steatohepatitis: a randomised, double-blind, placebo-controlled, phase 2a trial. Lancet 392, 2705–2717 (2019).
Loomba, R. et al. GS-0976 reduces hepatic steatosis and fibrosis markers in patients with nonalcoholic fatty liver disease. Gastroenterology 155, 1463–1473.e.6 (2018).
Harrison, S. A. et al. Efficacy and safety of aldafermin, an engineered FGF19 analog, in a randomized, double-blind, placebo-controlled trial of patients with nonalcoholic steatohepatitis. Gastroenterology 160, 219–231.e1 (2021).
European Medicines Agency. Reflection Paper on Regulatory Requirements for the Development of Medicinal Products for Chronic Non-infectious Liver Diseases (PBC, PSC, NASH) (2018); https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-regulatory-requirements-development-medicinal-products-chronic-non-infectious-liver_en.pdf
Anania, F. A., Dimick-Santos, L., Mehta, R., Toerner, J. & Beitz, J. Nonalcoholic steatohepatitis: current thinking from the division of hepatology and nutrition at the Food and Drug Administration. Hepatology 73, 2023–2027 (2021).
Ratziu, V. et al. Elafibranor, an agonist of the peroxisome proliferator-activated receptor-α and -δ, induces resolution of nonalcoholic steatohepatitis without fibrosis worsening. Gastroenterology 150, 1147–1159.e.5 (2016).
Younossi, Z. M. et al. Obeticholic acid for the treatment of non-alcoholic steatohepatitis: interim analysis from a multicentre, randomised, placebo-controlled phase 3 trial. Lancet 394, 2184–2196 (2019).
Newsome, P. N. et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N. Engl. J. Med. 384, 1113–1124 (2021).
Harrison, S. A. et al. Insulin sensitizer MSDC-0602K in non-alcoholic steatohepatitis: a randomized, double-blind, placebo-controlled phase IIb study. J. Hepatol. 72, 613–626 (2020).
Taylor, R. S. et al. Association between fibrosis stage and outcomes of patients with nonalcoholic fatty liver disease: a systematic review and meta-analysis. Gastroenterology 158, 1611–1625.e12 (2020).
Neuschwander-Tetri, B. A. et al. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet 385, 956–965 (2015).
Sanyal, A. J. et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 362, 1675–1685 (2010).
Loomba, R. et al. Factors associated with histologic response in adult patients with nonalcoholic steatohepatitis. Gastroenterology 156, 88–95.e.5 (2019).
Chalasani, N. et al. Relationship between three commonly used non-invasive fibrosis biomarkers and improvement in fibrosis stage in patients with non-alcoholic steatohepatitis. Liver Int. 39, 924–932 (2019).
Siddiqui, M. S. et al. Diagnostic accuracy of noninvasive fibrosis models to detect change in fibrosis stage. Clin. Gastroenterol. Hepatol. 17, 1877–1885.e5 (2019).
Brown, J. M. & Rudel, L. L. Stearoyl-coenzyme A desaturase 1 inhibition and the metabolic syndrome: considerations for future drug discovery. Curr. Opin. Lipidol. 21, 192–197 (2010).
Warensjö, E., Ohrvall, M. & Vessby, B. Fatty acid composition and estimated desaturase activities are associated with obesity and lifestyle variables in men and women. Nutr. Metab. Cardiovasc. Dis. 16, 128–136 (2006).
Ntambi, J. M. et al. Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc. Natl Acad. Sci. USA 99, 11482–11486 (2002).
American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of Medical Care in Diabetes–2018. Diabetes Care 41, S13–S27 (2018).
Fernández-Ramos, D. et al. Arachidyl amido cholanoic acid improves liver glucose and lipid homeostasis in nonalcoholic steatohepatitis via AMPK and mTOR regulation. World J. Gastroenterol. 26, 5101–5117 (2020).
Rahman, S. M. et al. Stearoyl-CoA desaturase 1 deficiency elevates insulin-signaling components and down-regulates protein-tyrosine phosphatase 1B in muscle. Proc. Natl Acad. Sci. USA 100, 11110–11115 (2003).
Poletto, A. C. et al. Oleic and linoleic fatty acids downregulate Slc2a4/GLUT4 expression via NFKB and SREBP1 in skeletal muscle cells. Mol. Cell. Endocrinol. 401, 65–72 (2015).
Dobrzyn, A. et al. Stearoyl-CoA desaturase-1 deficiency reduces ceramide synthesis by downregulating serine palmitoyltransferase and increasing β-oxidation in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 288, E599–E607 (2005).
Li, Z. Z., Berk, M., McIntyre, T. M. & Feldstein, A. E. Hepatic lipid partitioning and liver damage in nonalcoholic fatty liver disease: role of stearoyl-CoA desaturase. J. Biol. Chem. 284, 5637–5644 (2009).
Liu, X., Burhans, M. S., Flowers, M. T. & Ntambi, J. M. Hepatic oleate regulates liver stress response partially through PGC-1α during high-carbohydrate feeding. J. Hepatol. 65, 103–112 (2016).
Kurikawa, N. et al. A novel inhibitor of stearoyl-CoA desaturase-1 attenuates hepatic lipid accumulation, liver injury and inflammation in model of nonalcoholic steatohepatitis. Biol. Pharm. Bull. 36, 259–267 (2013).
Trépo, E. & Valenti, L. Update on NAFLD genetics: from new variants to the clinic. J. Hepatol. 72, 1196–1209 (2020).
American Diabetes Association. American Diabetes Association Standards of Medical care in Diabetes–2014. Diabetes Care 37, S14–S80 (2014).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41, 1313–1321 (2005).
Bedossa, P. et al. Utility and appropriateness of the fatty liver inhibition of progression (FLIP) algorithm and steatosis, activity, and fibrosis (SAF) score in the evaluation of biopsies of nonalcoholic fatty liver disease. Hepatology 60, 565–575 (2014).
Acknowledgements
Financial support for medical editorial assistance was provided by Galmed Pharmaceuticals. We thank medical writer S Diskin (Bioforum Group) for editorial assistance with this manuscript. This study was sponsored by Galmed Pharmaceuticals. The protocol was written by a panel of experts and sponsor representatives. Authors participated actively in drafting and reviewing the manuscript. The corresponding author had full access to all data in the study and had final responsibility for the decision to submit for publication.
Author information
Authors and Affiliations
Consortia
Contributions
V.R. designed the study, was responsible for patient recruitment and treatment, data analysis and interpretation, is a member of the study advisory committee and wrote the manuscript. L.d.G. was a principal investigator responsible for patient recruitment and treatment and is a member of the study advisory committee. R.S., F.P., F.F., A.L.F. and J.F-F. recruited and treated patients. M.A. recruited and treated patients and is a member of the study advisory committee. D.B.B. performed the Central MRI Reading. K.L. performed the Central Pathology Reading. T.G. helped with the data analysis and interpretation and manuscript writing. S.K. helped with the statistical analysis plan, data analysis and interpretation. R.O. helped with study design, data review and interpretation. M.H. helped with study planning and design. L.H. helped with the data analysis and interpretation. R.L. helped with patient recruitment and treatment and is a member of the study advisory committee. S.F. helped with data review and interpretation. A.J.S. was the US lead principal investigator, helped with study design, data analysis and interpretation, supported the setup of the MRS Central Reading and is a member of the study advisory committee. Members of the ARREST investigator study group helped in patient recruitment and treatment.
Corresponding author
Ethics declarations
Competing interests
V.R. and R.L. are Galmed consultants and investigators in the Galmed-sponsored study described in the article. S.F. and A.J.S. are Galmed consultants, T.G., M.H., R.O. and L.H. are current or former Galmed employees, D.B.B. was responsible for central lab services for the Galmed-sponsored study described in the article. K.L. was responsible for the histological analysis services for the Galmed-sponsored study described in the article. S.K. is the Galmed statistician. L.d.G., R.S., F.P., F.F., J.F-F., M.A. and A.L.F. were Investigators in the Galmed-sponsored study described in the article. The ARREST investigator study group members were investigators or sub-investigators in the Galmed-sponsored study described in the article.
Additional information
Peer review information Nature Medicine thanks Vincent Wong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Jennifer Sargent was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and Methods.
Rights and permissions
About this article
Cite this article
Ratziu, V., de Guevara, L., Safadi, R. et al. Aramchol in patients with nonalcoholic steatohepatitis: a randomized, double-blind, placebo-controlled phase 2b trial. Nat Med 27, 1825–1835 (2021). https://doi.org/10.1038/s41591-021-01495-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-021-01495-3
This article is cited by
-
Advances in management of metabolic dysfunction-associated steatotic liver disease: from mechanisms to therapeutics
Lipids in Health and Disease (2024)
-
A patient-based iPSC-derived hepatocyte model of alcohol-associated cirrhosis reveals bioenergetic insights into disease pathogenesis
Nature Communications (2024)
-
Emerging Drug Therapies for Metabolic Dysfunction-Associated Steatotic Liver Disease: A Glimpse into the Horizon
Current Hepatology Reports (2024)
-
Identification of novel SCD1 inhibitor alleviates nonalcoholic fatty liver disease: critical role of liver-adipose axis
Cell Communication and Signaling (2023)
-
Pharmacotherapies of NAFLD: updated opportunities based on metabolic intervention
Nutrition & Metabolism (2023)