Abstract
B cell maturation antigen (BCMA) is a target for various immunotherapies and a biomarker for tumor load in multiple myeloma (MM). We report a case of irreversible BCMA loss in a patient with MM who was enrolled in the KarMMa trial (NCT03361748) and progressed after anti-BCMA CAR T cell therapy. We identified selection of a clone with homozygous deletion of TNFRSF17 (BCMA) as the underlying mechanism of immune escape. Furthermore, we found heterozygous TNFRSF17 loss or monosomy 16 in 37 out of 168 patients with MM, including 28 out of 33 patients with hyperhaploid MM who had not been previously treated with BCMA-targeting therapies, suggesting that heterozygous TNFRSF17 deletion at baseline could theoretically be a risk factor for BCMA loss after immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials will be promptly reviewed by University Hospital Würzburg and Münchner Leukämielabor to verify if the request is subject to any confidentiality and data protection obligations. Any data and materials that can be shared will be released via a material transfer agreement. All raw and analyzed scRNA-seq data can be found in the National Center for Biotechnology Information Gene Expression Omnibus (accession no. GSE143317). Adherence to the Datenschutz-Grundverordnung (https://dsgvo-gesetz.de/ and https://data.consilium.europa.eu/doc/document/ST-5419-2016-REV-1/en/pdf) is mandatory for sharing WGS data. Thus, the complete whole-genome raw dataset of a single patient cannot be shared according to European law. Processed data from which the identification of a patient is not possible can be made available. WGS data derived from public datasets are deposited in the European Genome-phenome Archive with the references EGAS00001002111 (ref. 7) and EGAS00001001810 (ref. 9). Source data are provided with this paper.
References
Shah, N., Chari, A., Scott, E., Mezzi, K. & Usmani, S. Z. B-cell maturation antigen (BCMA) in multiple myeloma: rationale for targeting and current therapeutic approaches. Leukemia 34, 985–1005 (2020).
Raje, N. et al. Anti-BCMA CAR T-Cell therapy bb2121 in relapsed or refractory multiple myeloma. N. Engl. J. Med. 380, 1726–1737 (2019).
Cohen, A. D. et al. B cell maturation antigen-specific CAR T cells are clinically active in multiple myeloma. J. Clin. Invest. 129, 2210–2221 (2019).
Brudno, J. N. et al. T cells genetically modified to express an anti-B-cell maturation antigen chimeric antigen receptor cause remissions of poor-prognosis relapsed multiple myeloma. J. Clin. Oncol. 36, 2267–2280 (2018).
Cheng, J. et al. Pan-cancer analysis of homozygous deletions in primary tumours uncovers rare tumour suppressors. Nat. Commun. 8, 1221 (2017).
Tai, Y.-T. et al. APRIL and BCMA promote human multiple myeloma growth and immunosuppression in the bone marrow microenvironment. Blood 127, 3225–3236 (2016).
Rasche, L. et al. Spatial genomic heterogeneity in multiple myeloma revealed by multi-region sequencing. Nat. Commun. 8, 268 (2017).
Sawyer, J. R. et al. Hyperhaploidy is a novel high-risk cytogenetic subgroup in multiple myeloma. Leukemia 31, 637–644 (2017).
Weinhold, N. et al. Clonal selection and double-hit events involving tumor suppressor genes underlie relapse in myeloma. Blood 128, 1735–1744 (2016).
Orlando, E. J. et al. Genetic mechanisms of target antigen loss in CAR19 therapy of acute lymphoblastic leukemia. Nat. Med. 24, 1504–1506 (2018).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-Cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Bannerji, R. et al. Clinical activity of REGN1979, a bispecific human, anti-CD20 × anti-CD3 antibody, in patients with relapsed/refractory (R/R) B-cell non-Hodgkin lymphoma (B-NHL). Blood 134, 762 (2019).
Paul, M. R., Wong, V., Aristizabal, P. & Kuo, D. J. Treatment of recurrent refractory pediatric pre-B acute lymphoblastic leukemia using inotuzumab ozogamicin monotherapy resulting in CD22 antigen expression loss as a mechanism of therapy resistance. J. Pediatr. Hematol. Oncol. 41, e546–e549 (2019).
Martin, N. et al. Correlation of tumor BCMA expression with response and acquired resistance to idecabtagene vicleucel in the KarMMa study in relapsed and refractory multiple myeloma. HemaSphere https://doi.org/10.1097/HS9.0000000000000404 (2020).
Rustad, E. H. et al. Revealing the impact of structural variants in multiple myeloma. Blood Cancer Discov. 1, 258–273 (2020).
Fernández de Larrea, C. et al. Defining an optimal dual-targeted CAR T-cell therapy approach simultaneously targeting BCMA and GPRC5D to prevent BCMA escape-driven relapse in multiple myeloma. Blood Cancer Discov. 1, 146–154 (2020).
Garfall, A. L. & June, C. H. Trispecific antibodies offer a third way forward for anticancer immunotherapy. Nature 575, 450–451 (2019).
Pillarisetti, K. et al. A T-cell-redirecting bispecific G-protein-coupled receptor class 5 member D × CD3 antibody to treat multiple myeloma. Blood 135, 1232–1243 (2020).
Gogishvili, T. et al. SLAMF7-CAR T cells eliminate myeloma and confer selective fratricide of SLAMF7+ normal lymphocytes. Blood 130, 2838–2847 (2017).
Casucci, M. et al. CD44v6-targeted T cells mediate potent antitumor effects against acute myeloid leukemia and multiple myeloma. Blood 122, 3461–3472 (2013).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
McInnes, L. et al. UMAP: Uniform Manifold Approximation and Projection. J. Open Source Softw. 3, 861 (2018).
Waltman, L. & van Eck, N. J. A smart local moving algorithm for large-scale modularity-based community detection. Eur. Phys. J. B 86, 471 (2013).
Kowalczyk, M. S. et al. Single-cell RNA-seq reveals changes in cell cycle and differentiation programs upon aging of hematopoietic stem cells. Genome Res. 25, 1860–1872 (2015).
Erhard, F. Estimating pseudocounts and fold changes for digital expression measurements. Bioinformatics 34, 4054–4063 (2018).
Raczy, C. et al. Isaac: ultra-fast whole-genome secondary analysis on Illumina sequencing platforms. Bioinformatics 29, 2041–2043 (2013).
Chen, X. et al. Manta: rapid detection of structural variants and indels for germline and cancer sequencing applications. Bioinformatics 32, 1220–1222 (2016).
Kim, S. et al. Strelka2: fast and accurate calling of germline and somatic variants. Nat. Methods 15, 591–594 (2018).
Acknowledgements
We thank S. Roth for her excellent technical support. We acknowledge the work and contribution of T. Mack from Bristol Myers Squibb. L.R. was supported by the German Cancer Aid via the MSNZ program and IZKF Würzburg. H.E and M.H. were supported by the DFG via SFB/TR 221 project A3. A.-E.S. and O.D. were supported by the German Research Society graduate college 2157. Bristol Myers Squibb provided clinical trial data related to the presented case but no financial support for the study.
Author information
Authors and Affiliations
Contributions
L.R., M.C.D.V., K.H., A.-E.S. and H.E. contributed to study design and manuscript preparation. K.M.K., M.G., M.C., J.D., S. Kraus, A.H., X.Z., S.D., M.S.T., M.H. and S.P. contributed to the collection of clinical specimens. A.R. and V.F. performed the immunohistochemistry analysis. M.C.D.V., O.D., P.A., V.F. and A.-E.S. performed the scRNA-seq. O.D. performed the scRNA-seq data analysis. M.T., S.M., S.T., M.M. and C.H. performed whole-genome and bulk RNA sequencing. F.E. contributed to the bioinformatics analysis. N.W. analyzed the additional patent cohorts. S. Kaiser and K.H. provided clinical trial data. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
K.H. and S.K. are employees of Bristol Myers Squibb. M.H. is listed as an inventor on patent applications and granted patents related to CAR T cell therapy and CAR T cell technologies; he has received speaker honoraria from Celgene/Bristol Myers Squibb, Janssen and Kite/Gilead. H.E. received research funding from Janssen, Bristol Myers Squibb/Celgene, Amgen, Novartis, Sanofi, GSK and Janssen and has received speaker honoraria from Bristol Myers Squibb/Celgene, Amgen, Novartis, Takeda, Sanofi and GSK. L.R. has received speaker honoraria from Bristol Myers Squibb/Celgene, Sanofi, GSK, Oncopeptides and Janssen. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Medicine thanks Eric Smith, Premal Lulla and Alfred Garfall for their contribution to the peer review of this work. Saheli Sadanand was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Baseline characteristics and previous therapies.
Summary of patient baseline characteristics and previous therapies before inclusion on the KarMMa trial. Abbreviations: Rev, Lenalidomide; Dara, Daratumumab; SC mob, stem cell mobilization; auto SCT, autologous stem cell transplantation; Pom, Polmalidomide; Carf, Carfilzomib; Cyclo, Cyclophosphamide; Dexa, Dexamethasone; Thal, Thalidomide.
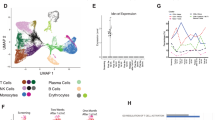
Extended Data Fig. 2 Cell type annotation in bone marrow biopsies.
a, Schematic of the experimental workflow. b, UMAP embedding of 5,592 single-cell transcriptomes from bone marrow biopsies before treatment (Baseline) and after relapse (Relapse). c, as b showing the annotated cell types. d, Dotplot showing the mean (of scaled log-normalized counts) expression (color) and the proportion in percentage of positive (non-zero expression) cells per cluster (size). MM, Multiple Myeloma. e, Log-normalized expression of marker genes specific for the identified cell types (MM: IGHG1+; B/DC: HLA-DRA+, CD79B+, LILRA4+; T/NK: CD3D+, NKG7+; Neutrophil: HLA-DRA-, FCGR3B+; Monocyte: HLA-DRA+, CD68+; Ery: ALAS2+) color coded on the UMAP embedding as panels b and c.
Extended Data Fig. 3 TNFRSF17 (BCMA) expression from baseline to relapse.
a, UMAP embedding of 2,471 single-cell transcriptomes of Multiple Myeloma cells (MM cells from Extended Data Fig. 2 extracted and re-analyzed) from bone marrow biopsies before treatment (45 MM single cells, Baseline, blue) and after relapse (2,426 MM single cells, Relapse, orange). b, As panel a colored by the log-normalized expression of TNFRSF17 (BCMA). c, As panel a colored by cluster. d, Dotplot of scaled, log-normalized expression (color) of marker genes identified for the different clusters shown in panel d and the proportion in percentage of positive (non-zero expression) cells per cluster (size). e, Violin plot showing the expression (log-normalized) of target antigens of immunotherapy between baseline and relapse. Horizontal line shows median of expression value. f, Expression of indicated genes shown in e color coded on top of the UMAP embedding as in panels a and c.
Extended Data Fig. 4 T cell state characterization at baseline and relapse.
a, UMAP embedding of 1,044 T cells from bone marrow biopsies before (635, blue dots) and at relapse (409, orange dots). b, UMAP embedding (as panel a) showing T cell subsets. c, Dotplot showing the scaled, log-normalized expression (color) of canonical marker genes for each cell subset and the proportion in percentage of positive (non-zero expression) cells per cluster (size). d, Violin-plots showing log-normalized expression of T cell exhaustion (LAG3, TIGIT, PDCD1, HAVCR2) and activation markers (IFNG, IFIT1, IFIT3, IFI16, HLA-DRA, HLA-DRB1, HLA-DRB5) between baseline (orange) and relapse (blue) across different cell subsets. P-values were calculated using the two-sided wilcoxon rank sum test, no adjustment for multiple comparisons was performed. Horizontal line shows median of expression value. Abbreviations: Th: Helper T cells; Treg: Regulatory T cells; NK: Natural killer cells; Prolif: Proliferating T cells.
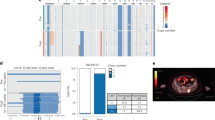
Extended Data Fig. 5 Genomic architecture of baseline sample.
Circos plot of CD138+ selected cells from the baseline sample including copy number variations, structural variants, and single nucleotide variations based on WGS data. Outer track runs clockwise from chromosome 1 to Y. Inner track shows gains > 1 Mb in blue and losses > 1 Mb in red. Red lines inside the circle represent interchromosomal reciprocal translocations with vaf > 0.1. Genes with mutations (NRAS, ERBB4 and TP53) are depicted in red and genes with detected variants of unknown significance (TTN) are shown in grey.
Extended Data Fig. 6 Copy number variant analysis.
a, Normalized Log2-ratio of gene expression within the cytogenetic band 16p13.13 between baseline and progression (n = number MM cells=45 MM cells at baseline and n = 2,426 MM cells at relapse). Error bars represent 90 % credible intervals. b, Total UMI count in non-malignant control cells at baseline vs. progression. We focused on the changes of expression levels from baseline to progression for genes located in the heterozygous deletion region. If the patient acquired the heterozygous deletion after treatment, systematic changes of gene expression are expected in this region. In contrast, this is not expected with the heterozygous deletion already present at baseline. In our patient, there were strong expression changes for genes in this region in malignant plasma cells (median > 3.7-fold difference) but not in monocytes (median < 1.3-fold difference) that we analyzed as negative control.
Extended Data Fig. 7 Interphase FISH of the 16p heterozygous deletion.
Interphase Fluorescent-in-situ-hybridization (FISH) using the CBFB/MYH11 probe set in a patient with heterozygous del(16p) and concomitant del(16q). Two aberrant cells show only 1 signal instead of 2 for the respective probes in line with two deletions encompassing the MYH11 (located on 16p13.1) and CBFB (located on 16q22) locus.
Extended Data Fig. 8 TNFRSF17 RNA-seq expression data from 28 relapsed/refractory multiple myeloma (R/R MM) patients.
TNFRSF17 (BCMA) expression levels in patients with and w/o BCMA heterozygous deletions. Bulk RNAseq data for 28 R/R MM patients with availability of WGS data. Two patients showed a heterozygous TNFRSF17 deletion (red dots), whereas the remaining patients did not show BCMA aberrations (grey dots). Expression levels did not differ between the two groups.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Source data
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Rights and permissions
About this article
Cite this article
Da Vià, M.C., Dietrich, O., Truger, M. et al. Homozygous BCMA gene deletion in response to anti-BCMA CAR T cells in a patient with multiple myeloma. Nat Med 27, 616–619 (2021). https://doi.org/10.1038/s41591-021-01245-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-021-01245-5
This article is cited by
-
Challenges and strategies associated with CAR-T cell therapy in blood malignancies
Experimental Hematology & Oncology (2024)
-
Bispecific CS1-BCMA CAR-T cells are clinically active in relapsed or refractory multiple myeloma
Leukemia (2024)
-
Real-world analysis of teclistamab in 123 RRMM patients from Germany
Leukemia (2024)
-
Fc receptor-like 5 (FCRL5)-directed CAR-T cells exhibit antitumor activity against multiple myeloma
Signal Transduction and Targeted Therapy (2024)
-
CAR T therapies in multiple myeloma: unleashing the future
Cancer Gene Therapy (2024)