Abstract
Mounting evidence suggests that function and connectivity of the striatum is disrupted in schizophrenia1,2,3,4,5. We have developed a new hypothesis-driven neuroimaging biomarker for schizophrenia identification, prognosis and subtyping based on functional striatal abnormalities (FSA). FSA scores provide a personalized index of striatal dysfunction, ranging from normal to highly pathological. Using inter-site cross-validation on functional magnetic resonance images acquired from seven independent scanners (n = 1,100), FSA distinguished individuals with schizophrenia from healthy controls with an accuracy exceeding 80% (sensitivity, 79.3%; specificity, 81.5%). In two longitudinal cohorts, inter-individual variation in baseline FSA scores was significantly associated with antipsychotic treatment response. FSA revealed a spectrum of severity in striatal dysfunction across neuropsychiatric disorders, where dysfunction was most severe in schizophrenia, milder in bipolar disorder, and indistinguishable from healthy individuals in depression, obsessive-compulsive disorder and attention-deficit hyperactivity disorder. Loci of striatal hyperactivity recapitulated the spatial distribution of dopaminergic function and the expression profiles of polygenic risk for schizophrenia. In conclusion, we have developed a new biomarker to index striatal dysfunction and established its utility in predicting antipsychotic treatment response, clinical stratification and elucidating striatal dysfunction in neuropsychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The dataset of UCLA Consortium for Neuropsychiatric Phenomics is available at the OpenfMRI database (https://openfmri.org/dataset/ds000030/). The [11C]raclopride PET template can be found in a previous study56. [18F]FDOPA PET and [123I]FP-CIT SPECT templates are available at the NITRC website (http://www.nitrc.org/projects/spmtemplates/). Human gene expression data from the post-mortem tissues from six donors are available at http://human.brain-map.org. Requests for other raw and analyzed data will be promptly reviewed by the Institute of Automation, Chinese Academy of Sciences to verify whether the request is subject to any intellectual property or confidentiality obligations.
Code availability
The SVM model (from all 1,100 subjects across the seven scanners) and code for calculating the personalized FSA score are available at https://github.com/BingLiu-Lab/FSA. A web-based research tool for the online calculation of personalized FSA score is available at https://www.szbiomarkers.net/fsa. Resting-state fMRI data preprocessing was implemented in an open MATLAB-based tool, BRANT version 3.3546 (http://brant.brainnetome.org/en/latest). Most data manipulation and machine learning on neuroimaging data were performed based on scikit-learn54 and nilearn (https://github.com/nilearn/nilearn), which are publicly available.
References
Simpson, E. H., Kellendonk, C. & Kandel, E. A possible role for the striatum in the pathogenesis of the cognitive symptoms of schizophrenia. Neuron 65, 585–596 (2010).
Howes, O. D. & Kapur, S. The dopamine hypothesis of schizophrenia: version III—the final common pathway. Schizophr. Bull. 35, 549–562 (2009).
Fornito, A. et al. Functional dysconnectivity of corticostriatal circuitry as a risk phenotype for psychosis. JAMA Psychiatry 70, 1143–1151 (2013).
Martino, M. et al. Abnormal resting-state connectivity in a substantia nigra-related striato-thalamo-cortical network in a large sample of first-episode drug-naive patients with schizophrenia. Schizophr. Bull. 44, 419–431 (2018).
McCutcheon, R. A., Abi-Dargham, A. & Howes, O. D. Schizophrenia, dopamine and the striatum: from biology to symptoms. Trends Neurosci. 42, 205–220 (2019).
van Os, J. & Kapur, S. Schizophrenia. Lancet 374, 635–645 (2009).
Kennedy, J. L., Altar, C. A., Taylor, D. L., Degtiar, I. & Hornberger, J. C. The social and economic burden of treatment-resistant schizophrenia: a systematic literature review. Int. Clin. Psychopharmacol. 29, 63–76 (2014).
McCutcheon, R. A., Reis Marques, T. & Howes, O. D. Schizophrenia—an overview. J. Am. Med. Assn. Psychiatry 77, 201–210 (2020).
Howes, O. D. & Kapur, S. A neurobiological hypothesis for the classification of schizophrenia: type A (hyperdopaminergic) and type B (normodopaminergic). Br. J. Psychiatry 205, 1–3 (2014).
Falkai, P. A desperate search for biomarkers in schizophrenia. What is going wrong? World Psychiatry 10, 38–39 (2011).
Sarpal, D. K. et al. Baseline striatal functional connectivity as a predictor of response to antipsychotic drug treatment. Am. J. Psychiatry 173, 69–77 (2016).
White, T. P. et al. Dysfunctional striatal systems in treatment-resistant schizophrenia. Neuropsychopharmacology 41, 1274–1285 (2016).
Lancaster, T. M. et al. Polygenic risk of psychosis and ventral striatal activation during reward processing in healthy adolescents. JAMA Psychiatry 73, 852–861 (2016).
Lancaster, T. M. et al. Structural and functional neuroimaging of polygenic risk for schizophrenia: a recall-by-genotype-based approach. Schizophr. Bull. 45, 405–414 (2019).
Zou, Q. H. et al. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J. Neurosci. Methods 172, 137–141 (2008).
Zang, Y., Jiang, T., Lu, Y., He, Y. & Tian, L. Regional homogeneity approach to fMRI data analysis. Neuroimage 22, 394–400 (2004).
Shirer, W. R., Ryali, S., Rykhlevskaia, E., Menon, V. & Greicius, M. D. Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb. Cortex 22, 158–165 (2012).
Fan, L. et al. The human Brainnetome Atlas: a new brain atlas based on connectional architecture. Cereb. Cortex 26, 3508–3526 (2016).
Abraham, A. et al. Deriving reproducible biomarkers from multi-site resting-state data: an autism-based example. Neuroimage 147, 736–745 (2017).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Lindenmayer, J. P., Bernstein-Hyman, R. & Grochowski, S. Five-factor model of schizophrenia initial validation. J. Nerv. Ment. Dis. 182, 631–638 (1994).
Kusomi, I., Boku, S. & Takahashi, Y. Psychopharmacology of atypical antipsychotic drugs: from the receptor binding profile to neuroprotection and neurogenesis. Psychiatry Clin. Neurosci. 69, 243–258 (2015).
Cross-Disorder Group of the Psychiatric Genomics Consortium et al. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat. Genet. 45, 984–994 (2013).
International Schizophrenia Consortium et al. Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature 460, 748–752 (2009).
Jauhar, S. et al. A test of the transdiagnostic dopamine hypothesis of psychosis using positron emission tomographic imaging in bipolar affective disorder and schizophrenia. JAMA Psychiatry 74, 1206–1213 (2017).
Lichtenstein, P. et al. Common genetic determinants of schizophrenia and bipolar disorder in Swedish families: a population-based study. Lancet 373, 234–239 (2009).
Howes, O. D. et al. The nature of dopamine dysfunction in schizophrenia and what this means for treatment. Arch. Gen. Psychiatry 69, 776–786 (2012).
Chen, K. C. et al. Striatal dopamine transporter availability in drug-naive patients with schizophrenia: a case-control SPECT study with [99mTc]-TRODAT-1 and a meta-analysis. Schizophr. Bull. 39, 378–386 (2013).
Schizophrenia Working Group of the Psychiatric Genomics Consortium Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Garcia-Gomez, F. J. et al. Elaboration of the SPM template for the standardization of SPECT images with 123I-ioflupane. Rev. Esp. Med. Nucl. Imagen Mol. 32, 350–356 (2013).
Grecchi, E., Doyle, O. M., Bertoldo, A., Pavese, N. & Turkheimer, F. E. Brain shaving: adaptive detection for brain PET data. Phys. Med. Biol. 59, 2517–2534 (2014).
Hawrylycz, M. J. et al. An anatomically comprehensive atlas of the adult human brain transcriptome. Nature 489, 391–399 (2012).
Watanabe, K., Taskesen, E., van Bochoven, A. & Posthuma, D. Functional mapping and annotation of genetic associations with FUMA. Nat. Commun. 8, 1826 (2017).
Howes, O. D. et al. Treatment-resistant schizophrenia: Treatment Response and Resistance in Psychosis (TRRIP) Working Group consensus guidelines on diagnosis and terminology. Am. J. Psychiatry 174, 216–229 (2017).
Roberts, D. L. & Velligan, D. I. Medication adherence in schizophrenia. Drug Discov. Today Ther. Strateg. 8, 11–15 (2011).
Zhang, J. P. et al. Schizophrenia polygenic risk score as a predictor of antipsychotic efficacy in first-episode psychosis. Am. J. Psychiatry 176, 21–28 (2019).
Frank, J. et al. Identification of increased genetic risk scores for schizophrenia in treatment-resistant patients. Mol. Psychiatry 20, 150–151 (2015).
Kaar, S. J., Natesan, S., McCutcheon, R. & Howes, O. D. Antipsychotics: mechanisms underlying clinical response and side-effects and novel treatment approaches based on pathophysiology. Neuropharmacology, 107704 (2019); https://doi.org/10.1016/j.neuropharm.2019.107704
Valenti, O., Cifelli, P., Gill, K. M. & Grace, A. A. Antipsychotic drugs rapidly induce dopamine neuron depolarization block in a developmental rat model of schizophrenia. J. Neurosci. 31, 12330–12338 (2011).
Jauhar, S. et al. The effects of antipsychotic treatment on presynaptic dopamine synthesis capacity in first-episode psychosis: a positron emission tomography study. Biol. Psychiatry 85, 79–87 (2019).
McNabb, C. B. et al. Functional network dysconnectivity as a biomarker of treatment resistance in schizophrenia. Schizophr. Res. 195, 160–167 (2018).
Hadley, J. A. et al. Ventral tegmental area/midbrain functional connectivity and response to antipsychotic medication in schizophrenia. Neuropsychopharmacology 39, 1020–1030 (2014).
Alonso-Sol¡s, A. et al. Resting-state functional connectivity alterations in the default network of schizophrenia patients with persistent auditory verbal hallucinations. Schizophr. Res. 161, 261–268 (2015).
Friston, K. J., Williams, S., Howard, R., Frackowiak, R. S. & Turner, R. Movement-related effects in fMRI time-series. Magn. Reson. Med. 35, 346–355 (1996).
Xu, K., Liu, Y., Zhan, Y., Ren, J. & Jiang, T. BRANT: a versatile and extendable resting-state fMRI toolkit. Front. Neuroinform. 12, 52 (2018).
Power, J. D., Plitt, M., Laumann, T. O. & Martin, A. Sources and implications of whole-brain fMRI signals in humans. Neuroimage 146, 609–625 (2017).
Duyn, J. H., Leopold, D. A., Maier, A., Scholvinck, M. L. & Ye, F. Q. Neural basis of global resting-state fMRI activity. Proc. Natl Acad. Sci. USA 107, 10238–10243 (2010).
Yang, G. J. et al. Altered global brain signal in schizophrenia. Proc. Natl Acad. Sci. USA 111, 7438–7443 (2014).
Tziortzi, A. C. et al. Connectivity-based functional analysis of dopamine release in the striatum using diffusion-weighted MRI and positron emission tomography. Cereb. Cortex 24, 1165–1177 (2014).
Arbabshirani, M. R., Plis, S., Sui, J. & Calhoun, V. D. Single subject prediction of brain disorders in neuroimaging: promises and pitfalls. Neuroimage 145, 137–165 (2017).
Abraham, A. et al. Machine learning for neuroimaging with scikit-learn. Front. Neuroinform. 8, 14 (2014).
Smyser, C. D. et al. Longitudinal analysis of neural network development in preterm infants. Cereb. Cortex 20, 2852–2862 (2010).
Power, J. D. et al. Functional network organization of the human brain. Neuron 72, 665–678 (2011).
Tzourio-Mazoyer, N. et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15, 273–289 (2002).
Poldrack, R. A. et al. A phenome-wide examination of neural and cognitive function. Sci. Data 3, 160110 (2016).
Rizzo, G., Veronese, M., Expert, P., Turkheimer, F. E. & Bertoldo, A. MENGA: a new comprehensive tool for the integration of neuroimaging data and the Allen human brain transcriptome atlas. PLoS One 11, e0148744 (2016).
Steiger, J. H. Tests for comparing elements of a correlation matrix. Psychol. Bull. 87, 245–251 (1980).
Romme, I. A. C., de Reus, M. A., Ophoff, R. A., Kahn, R. S. & van den Heuvel, M. P. Connectome disconnectivity and cortical gene expression in patients with schizophrenia. Biol. Psychiatry 81, 495–502 (2017).
Richiardi, J. et al. Correlated gene expression supports synchronous activity in brain networks. Science 348, 1241–1244 (2015).
Ripke, S. et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Acknowledgements
We thank C. Y. Wang, T. M. Si, G. L. Gong and M. Li for their critical discussions and comments, N. N. Tian for her assistance with figure drawing, and R. E. Perozzi and E. F. Perozzi for reviewing the English and content of this study. This work was supported by the National Key Basic Research and Development Program (973) (grant 2011CB707800 to T.J.), the National Key Research and Development Plan (grant 2016YFC0904300 to B.L.), the Strategic Priority Research Program of Chinese Academy of Science (grant XDB32020200 to B.L.) and the Natural Science Foundation of China (grant 81771451 to B.L.).
Author information
Authors and Affiliations
Contributions
T.J. and B.L. led the project. B.L., T.J. and A.L. were responsible for the study concept and the design of the study. W.Y. provided crucial advice for the study. K.X. carried out the neuroimaging data preprocessing. A.L. and B.L. analyzed the data, created the figures and wrote the manuscript. A.Z. and O.H. made substantial contributions to the manuscript and provided critical comments. A.Z., W.Y., X.W., O.H., H.Y., Y.L., L.F., K.J.W., J.L., S.L., M.W., Y. Sun, M.S. and T.J. participated in discussions of the results and the manuscript. G.R. developed the website. P.L., J.C., Y.C., W.Y., H.Y., H.N.W., W.L., Z.L., Y.Y., H.G., P.W., L. Lv, L. Lu, J.Y., Y. Song, H.L.W., H.Z., H.W., Y.N., Y.D., Y.C., J.X., X.X. and D.Z. contributed to the data acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Kate Gao was the primary editor on this article, and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Elevated regional homogeneity (ReHo) in schizophrenia.
a, Map of t-statistic values for between-group difference in ReHo (schizophrenia group minus healthy comparison group; unpaired two-sided t-test). n = 560 subjects with schizophrenia and n = 540 controls. Top: Data pooled across all seven scanners. The largest t-statistic values were for areas within the striatum, indicating that the striatum is a focal point of significantly increased ReHo in schizophrenia. Bottom: Data analyzed independently for each of the seven scanners. Only t-statistic values within the striatum are shown. Each axial slice (z = 5) corresponds to data acquired from an independent scanner. Images ordered according to Panel b. b, Scatter-box chart of the mean fALFF in the striatum of normal controls (NC, shown in blue) and schizophrenia groups (SZ, shown in red) across different scanners. From left: Peking University Sixth Hospital (SIMENS scanner, P < .0003; n = 92 SZ, n = 98 NC), Beijing Huilongguan Hospital (SIMENS scanner, P < 5 × 10−6; n = 83 SZ, n = 59 NC), Xijing Hospital (SIMENS scanner, P < 1 × 10−4; n = 90 SZ, n = 54 NC), Henan Mental Hospital (SIMENS scanner, P < 4 × 10−8; n = 81 SZ, n = 102 NC), Henan Mental Hospital (GE scanner, P < 6 × 10−7; n = 49 SZ, n = 69 NC), Renmin Hospital of Wuhan University (GE scanner, P = .00025; n = 82 SZ, n = 89 NC), and Zhumadian Psychiatric Hospital (GE scanner, P < 7 × 10−10; n = 83 SZ, n = 69 NC). Striatal hyperactivity indicated by increased ReHo in the schizophrenia group is evident in samples from all sites despite having been acquired using different scanners. Significance was assessed by unpaired two-sided t-test and there was no adjustment for multiple testing. The sample size and definition of boxplot are consistent with those in Fig. 2b.
Extended Data Fig. 2 Impacts of intra-scan head motion confound on FSA.
a, Boxplot of mean framewise distance (FD) for schizophrenia (red) and healthy comparison (blue) groups. As indicated by the asterisk, the mean FD of FSA was significantly higher in the schizophrenia group in PKU6 hospital (P < .05, unpaired two-sided t-test). No significant difference in FD between schizophrenia group and healthy controls in other cohorts (P values are shown in the figure; unpaired two-sided t-test). The sample size and definition of boxplot are consistent with those in Fig. 2b. b, Scatter plots showing the absence of a significant correlation between percentage change in PANSS symptom severity and mean FD (PKU6 hospital: P = .57, n = 37; ZMD hospital: P = .72, n = 58). c, Scatter plots showing the partial correlation between percentage change in PANSS symptom severity and the FSA score, controlling for the effect of mean FD (PKU6 hospital: P = .000034, n = 37; ZMD hospital: P = .0012, n = 58). d, Relationship between the FSA score and mean FD. e–g, Relationship between mean FD and the FSA score based on fALFF, intra-striatal FC and extra-striatal FC. Pearson correlation were used to assess significance in Panels b-g.
Extended Data Fig. 3 Impacts of baseline symptom on FSA and percentage symptom reduction.
a, b, No significant correlation between the FSA score and PANSS at baseline in longitudinal cohorts from either PKU6 hospital or ZMD hospital. c, d, No significant correlation between the FSA at baseline and the percentage reduction in PANSS in longitudinal cohorts from either PKU6 hospital or ZMD hospital. For Panels a-d: Ps > .05; Pearson correlation; n = 37 in PKU6 hospital and ZMD hospital n = 58 in ZMD hospital.
Extended Data Fig. 4 Prognostic utility of FSA stratified according to antipsychotic categories.
The relationship between percentage reduction in PANSS and the FSA score stratified according to antipsychotic category. All the antipsychotic categories with more than (or equal to) 5 patients were analyzed and Pearson’s r was represented. a, subjects with schizophrenia in PKU6 hospital, n = 10 Risperidone, n = 12 Olanzapine, n = 5 Amisulpride. b, subjects with schizophrenia in ZMD hospital, n = 19 Clozapine, n = 29 Risperidone, n = 6 Olanzapine. The single asterisk (*) and double asterisks (**) indicate P < .1 and P < .05, respectively. c, The linear relationship between the 5-TH2A / D2 affinity ratio and the negative of effect size (the natural logarithm of Pearson’s r) of FSA correlated with the antipsychotic response in specific medication group (n = 6, Pearson correlation).
Extended Data Fig. 5 Regional specificity of FSA by control brain regions.
a, The classification accuracy from inter-site validation across all regions of interest, using 1100 cross-section subjects described in Methods section. b, Pearson’s correlation coefficients between the PANSS percentage reduction and the FSA score in PKU6 (n = 37). c, Pearson’s correlation coefficients between the PANSS percentage reduction and the FSA score in ZMD (n = 58). All significant correlations (FDR adjusted, P < .05; Pearson correlation) are represented in red color. The results were that the striatum yielded the highest classification accuracy among all the control regions that we evaluated and that the striatum was the only region that enabled a reliable prediction of the antipsychotic response.
Extended Data Fig. 6 FSA in previously unused datasets characterized by different MR parameters, ethnicities or disease categories.
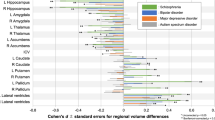
Density plots show FSA scores computed in five independent datasets (Validation datasets 1-5) comprising individuals with schizophrenia, bipolar disorder, depression, OCD, and ADHD. Groups comprising healthy comparison individuals are shown in blue. To aid visualization, FSA was normalized separately for each dataset. Specifically, we first standardized the FSA scores in individuals with schizophrenia and healthy controls, and then aligned the standardized model for FSA score in other diagnostic groups. Comparisons were made by unpaired two-sided t-test in Panels a-f. a, Dataset 1: FSA scores for individuals with schizophrenia (n = 30), but not bipolar disorders (BP, n = 25), OCD (n = 30) or depression (DEP, n = 27), significantly differed from those of healthy controls (n = 29). b, Dataset 2: FSA generalizes to a 1.5-T MRI scanner. The FSA scores for individuals with schizophrenia (n = 30) were significantly different from those of healthy controls (n = 29); Furthermore, individuals with bipolar disorder (n = 30) a trend toward significantly lower FSA scores compared to controls (P = .080). c, Dataset 3: Both individuals with schizophrenia (n = 81) and bipolar disorder (n = 41) showed significantly lower FSA scores compared to healthy controls (n = 102), and FSA scores did not differ significantly between the schizophrenia and bipolar groups. d, Dataset 4: FSA generalizes to non-Han Chinese ethnicities. Both individuals with bipolar disorder (n = 45) and schizophrenia (n = 47) showed significantly lower FSA scores compared to healthy controls (n = 115), whereas individuals with ADHD (n = 39) did not differ from controls. e, Dataset 5: FSA scores for individuals with schizophrenia (n = 92), but not OCD (n = 20), significantly differed from those of healthy controls (n = 98). f, Forest plots for FSA score of individuals with bipolar disorder compared with individuals with schizophrenia or healthy controls. The sample sizes in Datasets 1-4 were described above. SMD = standardized mean difference. The relative effect sizes were calculated as standardized mean differences (Hedges’ g) and reported with their 95% confidence interval (CI). An overall treatment effect is calculated as a weighted average of the individual effect sizes (95% CI shown). Based in the four validation datasets, SMD of FSA scores between individuals with bipolar disorders and healthy controls were significantly negative (overall effect size = 6.45, P < 2 × 10−10); SMD of FSA scores between individuals with bipolar disorders and schizophrenia were significantly positive (overall effect size = 3.83, P < .0002). Overall the result suggests that bipolar disorder is positioned between schizophrenia and healthy controls on the FSA spectrum. Moreover, we found that FSA scores for individuals with OCD, ADHD and depression did not significantly differ from healthy comparison individuals, indicating that there is less overlap between the three neuropsychiatric disorders and schizophrenia in striatal dysfunction.
Extended Data Fig. 7 Spatial correlation between [11C] Raclopride PET imaging and fALFF t-statistic maps within the striatum and its subregions.
[11C] Raclopride PET image (left) and fALFF t-statistic map (right) are represented on the top. Scatter plots show the association between the t-statistic map and the marker of D2/3 receptor density within distinct striatal subregions (Pearson’s r was represented below, in 1.5 mm resampled resolution): ventral caudate (orange red; left hemisphere r = .78, n = 1094 voxels; right hemisphere r = .83, n = 764 voxels), globus pallidus (burnt yellow; left r = .85 n = 758 voxels; right r = .72, n = 773 voxels), nucleus accumbens (leafy green; left r = .51, n = 776 voxels; right r = .79, n = 958 voxels), ventromedial putamen (lightish blue; left r = .82, n = 793 voxels; right r = .49, n = 607 voxels), dorsal caudate (bright lilac; left r = .81, n = 1178 voxels; right r = .84, n = 1588 voxels) and dorsolateral putamen (lightish red; left r = .57 n = 1079 voxels; right r = .25, n = 1111 voxels). The bottommost scatter plot is for the whole striatum.
Supplementary information
Supplementary Information
Supplementary Figs. 1–14, Tables 1–11 and STARD checklist.
Rights and permissions
About this article
Cite this article
Li, A., Zalesky, A., Yue, W. et al. A neuroimaging biomarker for striatal dysfunction in schizophrenia. Nat Med 26, 558–565 (2020). https://doi.org/10.1038/s41591-020-0793-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-0793-8
This article is cited by
-
Replication of a neuroimaging biomarker for striatal dysfunction in psychosis
Molecular Psychiatry (2024)
-
A distinctive subcortical functional connectivity pattern linking negative affect and treatment outcome in major depressive disorder
Translational Psychiatry (2024)
-
Frontostriatal circuitry and the tryptophan kynurenine pathway in major psychiatric disorders
Psychopharmacology (2024)
-
Using brain structural neuroimaging measures to predict psychosis onset for individuals at clinical high-risk
Molecular Psychiatry (2024)
-
Sampling inequalities affect generalization of neuroimaging-based diagnostic classifiers in psychiatry
BMC Medicine (2023)