Abstract
Neurofilament light chain (NfL) is a promising fluid biomarker of disease progression for various cerebral proteopathies. Here we leverage the unique characteristics of the Dominantly Inherited Alzheimer Network and ultrasensitive immunoassay technology to demonstrate that NfL levels in the cerebrospinal fluid (n = 187) and serum (n = 405) are correlated with one another and are elevated at the presymptomatic stages of familial Alzheimer’s disease. Longitudinal, within-person analysis of serum NfL dynamics (n = 196) confirmed this elevation and further revealed that the rate of change of serum NfL could discriminate mutation carriers from non-mutation carriers almost a decade earlier than cross-sectional absolute NfL levels (that is, 16.2 versus 6.8 years before the estimated symptom onset). Serum NfL rate of change peaked in participants converting from the presymptomatic to the symptomatic stage and was associated with cortical thinning assessed by magnetic resonance imaging, but less so with amyloid-β deposition or glucose metabolism (assessed by positron emission tomography). Serum NfL was predictive for both the rate of cortical thinning and cognitive changes assessed by the Mini–Mental State Examination and Logical Memory test. Thus, NfL dynamics in serum predict disease progression and brain neurodegeneration at the early presymptomatic stages of familial Alzheimer’s disease, which supports its potential utility as a clinically useful biomarker.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data that support the findings of this study are available from DIAN at https://dian.wustl.edu/our-research/observational-study/dian-observational-study-investigator-resources/.
References
Bateman, R. J. et al. Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. N. Engl. J. Med. 367, 795–804 (2012).
Gordon, B. A. et al. Spatial patterns of neuroimaging biomarker change in individuals from families with autosomal dominant Alzheimer’s disease: a longitudinal study. Lancet Neurol. 17, 241–250 (2018).
Jack, C. R. Jr. et al. NIA-AA Research Framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 14, 535–562 (2018).
Sperling, R. A., Karlawish, J. & Johnson, K. A. Preclinical Alzheimer disease: the challenges ahead. Nat. Rev. Neurol. 9, 54–58 (2013).
Fandos, N. et al. Plasma amyloid β 42/40 ratios as biomarkers for amyloid β cerebral deposition in cognitively normal individuals. Alzheimers Dement. (Amst) 8, 179–187 (2017).
Mattsson, N., Andreasson, U., Zetterberg, H. & Blennow, K. Association of plasma neurofilament light with neurodegeneration in patients with Alzheimer disease. JAMA Neurol. 74, 557–566 (2017).
Nakamura, A. et al. High performance plasma amyloid-β biomarkers for Alzheimer’s disease. Nature 554, 249–254 (2018).
Ovod, V. et al. Amyloid β concentrations and stable isotope labeling kinetics of human plasma specific to central nervous system amyloidosis. Alzheimers Dement. 13, 841–849 (2017).
Weston, P. S. J. et al. Serum neurofilament light in familial Alzheimer disease: a marker of early neurodegeneration. Neurology 89, 2167–2175 (2017).
Mielke, M. M. et al. Plasma phospho-tau181 increases with Alzheimer’s disease clinical severity and is associated with tau- and amyloid-positron emission tomography. Alzheimers Dement. 14, 989–997 (2018).
Petzold, A. Neurofilament phosphoforms: surrogate markers for axonal injury, degeneration and loss. J. Neurol. Sci. 233, 183–198 (2005).
Schlaepfer, W. W. & Lynch, R. G. Immunofluorescence studies of neurofilaments in the rat and human peripheral and central nervous system. J. Cell Biol. 74, 241–250 (1977).
Bacioglu, M. et al. Neurofilament light chain in blood and CSF as marker of disease progression in mouse models and in neurodegenerative diseases. Neuron 91, 56–66 (2016).
Barro, C. et al. Serum neurofilament as a predictor of disease worsening and brain and spinal cord atrophy in multiple sclerosis. Brain 141, 2382–2391 (2018).
Brureau, A. et al. NF-L in cerebrospinal fluid and serum is a biomarker of neuronal damage in an inducible mouse model of neurodegeneration. Neurobiol. Dis. 104, 73–84 (2017).
Kuhle, J. et al. Serum neurofilament light chain in early relapsing remitting MS is increased and correlates with CSF levels and with MRI measures of disease severity. Mult. Scler. 22, 1550–1559 (2016).
Zhou, W. et al. Plasma neurofilament light chain levels in Alzheimer’s disease. Neurosci. Lett. 650, 60–64 (2017).
Morris, J. C. et al. Developing an international network for Alzheimer research: the Dominantly Inherited Alzheimer Network. Clin. Investig. (Lond) 2, 975–984 (2012).
Moulder, K. L. et al. Dominantly Inherited Alzheimer Network: facilitating research and clinical trials. Alzheimers Res. Ther. 5, 48 (2013).
Ryman, D. C. et al. Symptom onset in autosomal dominant Alzheimer disease: a systematic review and meta-analysis. Neurology 83, 253–260 (2014).
Henriksen, K. et al. The future of blood-based biomarkers for Alzheimer’s disease. Alzheimers Dement. 10, 115–131 (2014).
McDade, E. et al. Longitudinal cognitive and biomarker changes in dominantly inherited Alzheimer disease. Neurology 91, e1295–e1306 (2018).
Sutphen, C. L. et al. Longitudinal decreases in multiple cerebrospinal fluid biomarkers of neuronal injury in symptomatic late onset Alzheimer’s disease. Alzheimers Dement. 14, 869–879 (2018).
Benzinger, T. L. S. et al. Regional variability of imaging biomarkers in autosomal dominant Alzheimer’s disease. Proc. Natl Acad. Sci. USA 110, E4502–E4509 (2013).
Burnham, S. C. et al. Clinical and cognitive trajectories in cognitively healthy elderly individuals with suspected non-Alzheimer’s disease pathophysiology (SNAP) or Alzheimer’s disease pathology: a longitudinal study. Lancet Neurol. 15, 1044–1053 (2016).
Jack, C. R. Jr. & Holtzman, D. M. Biomarker modeling of Alzheimer’s disease. Neuron 80, 1347–1358 (2013).
Lu, C. H. et al. Neurofilament light chain: a prognostic biomarker in amyotrophic lateral sclerosis. Neurology 84, 2247–2257 (2015).
Rojas, J. C. et al. Plasma neurofilament light chain predicts progression in progressive supranuclear palsy. Ann. Clin. Transl. Neurol. 3, 216–225 (2016).
Norgren, N., Karlsson, J. E., Rosengren, L. & Stigbrand, T. Monoclonal antibodies selective for low molecular weight neurofilaments. Hybrid. Hybridomics 21, 53–59 (2002).
Byrne, L. M. et al. Neurofilament light protein in blood as a potential biomarker of neurodegeneration in Huntington’s disease: a retrospective cohort analysis. Lancet Neurol. 16, 601–609 (2017).
Karran, E., Mercken, M. & De Strooper, B. The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat. Rev. Drug Discov. 10, 698–712 (2011).
Fagan, A. M. et al. Longitudinal change in CSF biomarkers in autosomal-dominant Alzheimer’s disease. Sci. Transl. Med. 6, 226ra230 (2014).
Kinnunen, K. M. et al. Presymptomatic atrophy in autosomal dominant Alzheimer’s disease: a serial magnetic resonance imaging study. Alzheimers Dement. 14, 43–53 (2018).
Shahim, P. et al. Serum neurofilament light protein predicts clinical outcome in traumatic brain injury. Sci. Rep. 6, 36791 (2016).
Jucker, M. & Walker, L. C. Propagation and spread of pathogenic protein assemblies in neurodegenerative diseases. Nat. Neurosci. 21, 1341–1349 (2018).
Bateman, R. J. et al. Autosomal-dominant Alzheimera’s disease: a review and proposal for the prevention of Alzheimera’s disease. Alzheimers Res. Ther. 3, 1 (2011).
Cairns, N. J. et al. Neuropathologic assessment of participants in two multi-center longitudinal observational studies: the Alzheimer Disease Neuroimaging Initiative (ADNI) and the Dominantly Inherited Alzheimer Network (DIAN). Neuropathology 35, 390–400 (2015).
Tang, M. et al. Neurological manifestations of autosomal dominant familial Alzheimer’s disease: a comparison of the published literature with the Dominantly Inherited Alzheimer Network observational study (DIAN-OBS). Lancet Neurol. 15, 1317–1325 (2016).
Thomas, J. B. et al. Functional connectivity in autosomal dominant and late-onset Alzheimer disease. JAMA Neurol. 71, 1111–1122 (2014).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 12, 189–198 (1975).
Morris, J. C. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43, 2412–2414 (1993).
Wechsler, D. Wechsler Memory Scale-Revised: Manual (The Psychological Corporation, San Antonio, 1987).
Kuhle, J. et al. A comparative study of CSF neurofilament light and heavy chain protein in MS. Mult. Scler. 19, 1597–1603 (2013).
Disanto, G. et al. Serum neurofilament light: a biomarker of neuronal damage in multiple sclerosis. Ann. Neurol. 81, 857–870 (2017).
Carpenter, B. et al. Stan: a probabilistic programming language. J. Stat. Softw. https://www.jstatsoft.org/article/view/v076i01 (2017).
Gelman, A., Lee, D. & Guo, J. Stan: a probabilistic programming language for Bayesian inference and optimization. J. Educ. Behav. Stat. 40, 530–543 (2015).
Acknowledgements
We would like to thank M. Staufenbiel and M. Eichner for support and helpful comments and C. Haass and M. Suarez (Munich) for experimental and logistic support. Data collection and sharing for this project was supported by DIAN (grant no. UF1AG032438) funded by the National Institute on Aging and the German Center for Neurodegenerative Diseases (DZNE). Additional support came from the National Institutes of Health-funded NINDS Center Core for Brain Imaging (grant no. P30NS098577), the National Science Foundation (grant no. DGE-1745038), National Institutes of Health (grant no. UL1TR001873 to J.M.N.), the Swiss National Science Foundation (grant no. 320030-160221 to J.K.), the National Institute for Health Research University College London Hospitals Biomedical Research Centre, and the MRC Dementias Platform UK (grant nos. MR/L023784/1 and MR/009076/1). We acknowledge the altruism of the participants and their families and input of the DIAN research and support staff at each of the participating sites for their contributions to this study.
Author information
Authors and Affiliations
Consortia
Contributions
All authors were involved in sample and data collection. J.K., O.P., A.A., S.A.K., and C.B. performed the immunoassay work. S.A.S., A.A., G.W., and B.A.G. performed the statistical analyses. M.J., O.P., S.A.S., A.A., and B.A.G. designed the study and wrote the manuscript with the help of the coauthors J.K., S.A.K., C.B., S.G., E.K.-B., C. LaFougere, C. Laske, J.V., J.L., C.L.M., R.M., P.R.S., M.N.R., N.R.G.-R., S.S., B.G., J.M.R., J.M.N., J.C., A.M.G., T.L.S.B., J.C.M., R.J.B., G.W., A.M.F., and E.M.M.
Corresponding author
Ethics declarations
Competing interests
A.M.G. has consulted for Cognition Therapeutics, Biogen, GlaxoSmithKline, Illumina, Eisai, AbbVie, and Pfizer and served on the Scientific Advisory Board for Denali Therapeutics. A.M.F. is a member of the Scientific Advisory Boards for AbbVie, Genentech and Roche Diagnostics and provides consultation for Araclon/Grifols and DiamiR.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Difference distribution curve for baseline (cross-sectional) CSF and serum NfL levels in mutation carriers and non-carriers.
a,b, Difference of posterior distribution for baseline CSF NfL (n = 187) (a) and baseline serum NfL (n = 405) (b) as a function of EYO. The solid red lines depict the median of the difference distribution; the shaded area represents the 99% equal-tailed credible intervals. EYO was considered statistically significant if the 99% equal-tailed credible intervals of the posterior distribution did not overlap 0 (6.8 years before EYO for both baseline CSF and serum NfL). For the absolute values of baseline CSF and serum NfL, see Fig. 1a,b.
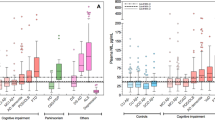
Extended Data Fig. 2 No difference in baseline CSF and serum NfL levels among APP, PSEN1, and PSEN2 mutation carriers.
a, Two-tailed pairwise Student’s t-test comparisons of CSF NfL levels of carriers of a mutation in APP (n = 14), PSEN1 (n = 82), or PSEN2 (n = 11). b, Same analysis, using a two-tailed pairwise Student’s t-test for the serum NfL of carriers of a mutation in APP (n = 39), PSEN1 (n = 185), or PSEN2 (n = 19). No differences in log(CSF NfL) or log(serum NfL) were found between the groups (F(2, 104) = 1.8108, P = 0.1686 and F(2, 240) = 1.9205, P = 0.1488, respectively). Similarly, no differences were found by two-tailed pairwise Student’s t-test when age and disease status (presymptomatic, symptomatic) were treated as covariates. The boxes map to the median, 25th and 75th quintiles, and the whiskers extend to the 1.5 × IQR.
Extended Data Fig. 3 Longitudinal serum NfL and bifurcation of mutation carriers from non-carriers.
a, Spaghetti plot showing longitudinal serum NfL for non-carriers (NC, n = 63, blue) and mutation carriers (MC, n = 133, red) as a function of EYO. These are the same data as in Fig. 2a but with a logarithmic scale on the y axis to better appreciate the changes during the presymptomatic stage (for details, see Fig. 2a). b, Difference of posterior distribution for serum NfL rate of change between mutation carriers and non-carriers, as a function of EYO (n = 196). The solid red line depicts the median of the difference distribution, and the shaded area represents the 99% equal-tailed credible intervals. EYO was considered statistically significant if the 99% equal-tailed credible intervals of the posterior distribution did not overlap 0 (16.2 years before EYO). c, Individual estimated rate of change in serum NfL (same data as in Fig. 2b, n = 63 for non-carriers and n = 133 for mutation carriers). A regression analysis was performed with two breaks of slope (see Methods for calculation). With this model the first bifurcation point was found at −18.6 years before EYO, the second at −5.8 years before EYO.
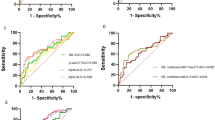
Extended Data Fig. 4 Rate of change per year of serum NfL is a better parameter to distinguish presymptomatic and symptomatic mutation carriers from non-carriers compared to single cross-sectional serum NfL.
Receiver operating characteristic analysis for non-carriers (NC) versus presymptomatic mutation carriers (MC) and non-carriers versus symptomatic mutation carriers with cross-sectional (baseline serum NfL) and longitudinal (serum NfL rate of change per year) data. The true positive fraction (sensitivity) is on the y axis and the false positive fraction (1-specificity) on the x axis. The area under the curve (AUC, accuracy), as well as the cutoff value and χ2 P value from the logistic regression are shown. The chance level of the area under the curve is 0.50. Converters (for rate of change, see Fig. 2c) were considered presymptomatic mutation carriers.
Extended Data Fig. 5 No difference in serum NfL rate of change among APP, PSEN1, and PSEN2 mutation carriers and no association with estimated age of onset.
a, Using two-tailed pairwise Student’s t-tests, no differences in the rate of change of log(serum NfL) (year−1) levels among APP (n = 24), PSEN1 (n = 104), and PSEN2 (n = 5) mutation carriers (F(2, 130) = 0.4678, P = 0.6274) was found. Similarly, no differences were found when age and disease status (presymptomatic, symptomatic) were treated as covariates in a two-tailed pairwise Student’s t-test. b, No difference between an individual’s deviation from the EYO-adjusted median rate of change in NfL and their expected age of symptom onset using LMEMs. Individuals were grouped in 4 categories with expected symptom onset at 20–39 (n = 17), 40–49 (n = 54), 50–59 (n = 56), and over 60 years of age (n = 6); group comparisons, P > 0.146. See Methods for the calculations. The boxes map to the median, 25th and 75th quintiles, and the whiskers extend to the 1.5 × IQR.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2 and Supplementary Statistical Analysis
Rights and permissions
About this article
Cite this article
Preische, O., Schultz, S.A., Apel, A. et al. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat Med 25, 277–283 (2019). https://doi.org/10.1038/s41591-018-0304-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0304-3
This article is cited by
-
Utilization of fluid-based biomarkers as endpoints in disease-modifying clinical trials for Alzheimer’s disease: a systematic review
Alzheimer's Research & Therapy (2024)
-
Childhood obesity and risk of Alzheimer’s disease: a Mendelian randomization study
Archives of Public Health (2024)
-
Blood-based CNS regionally and neuronally enriched extracellular vesicles carrying pTau217 for Alzheimer’s disease diagnosis and differential diagnosis
Acta Neuropathologica Communications (2024)
-
Synaptic vesicle glycoprotein 2 A in serum is an ideal biomarker for early diagnosis of Alzheimer’s disease
Alzheimer's Research & Therapy (2024)
-
Introducing neurofilament light chain measure in psychiatry: current evidence, opportunities, and pitfalls
Molecular Psychiatry (2024)