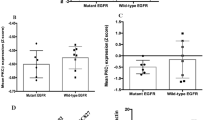
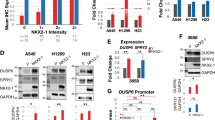
Abstract
RAS mutations are frequent in human cancer, especially in pancreatic, colorectal and non-small-cell lung cancers (NSCLCs)1,2,3. Inhibition of the RAS oncoproteins has proven difficult4, and attempts to target downstream effectors5,6,7 have been hampered by the activation of compensatory resistance mechanisms8. It is also well established that KRAS-mutant tumors are insensitive to inhibition of upstream growth factor receptor signaling. Thus, epidermal growth factor receptor antibody therapy is only effective in KRAS wild-type colon cancers9,10. Consistently, inhibition of SHP2 (also known as PTPN11), which links receptor tyrosine kinase signaling to the RAS–RAF–MEK–ERK pathway11,12, was shown to be ineffective in KRAS-mutant or BRAF-mutant cancer cell lines13. Our data also indicate that SHP2 inhibition in KRAS-mutant NSCLC cells under normal cell culture conditions has little effect. By contrast, SHP2 inhibition under growth factor–limiting conditions in vitro results in a senescence response. In vivo, inhibition of SHP2 in KRAS-mutant NSCLC also provokes a senescence response, which is exacerbated by MEK inhibition. Our data identify SHP2 inhibition as an unexpected vulnerability of KRAS-mutant NSCLC cells that remains undetected in cell culture and can be exploited therapeutically.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cancer Genome Atlas Research Network. The Cancer Genome Atlas Pan-Cancer analysis project. Nat. Genet. 45, 1113–1120 (2013).
Molina, J. R., Yang, P., Cassivi, S. D., Schild, S. E. & Adjei, A. A. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin. Proc. 83, 584–594 (2008).
Forbes, S. A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2011).
Stephen, A. G., Esposito, D., Bagni, R. K. & McCormick, F. Dragging Ras back in the ring. Cancer Cell 25, 272–281 (2014).
Flaherty, K. T. et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N. Engl. J. Med. 367, 107–114 (2012).
Zhao, Y. & Adjei, A. A. The clinical development of MEK inhibitors. Nat. Rev. Clin. Oncol. 11, 385–400 (2014).
Wang, D., Boerner, S. A., Winkler, J. D. & LoRusso, P. M. Clinical experience of MEK inhibitors in cancer therapy. Biochim. Biophys. Acta. 1773, 1248–1255 (2007).
Bernards, R. A missing link in genotype-directed cancer therapy. Cell 151, 465–468 (2012).
Karapetis, C. S. et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N. Engl. J. Med. 359, 1757–1765 (2008).
De Roock, W. et al. Effects of KRAS, BRAF, NRAS, and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy in chemotherapy-refractory metastatic colorectal cancer: a retrospective consortium analysis. Lancet Oncol. 11, 753–762 (2010).
Grossmann, K. S., Rosario, M., Birchmeier, C. & Birchmeier, W. The tyrosine phosphatase Shp2 in development and cancer. Adv. Cancer Res. 106, 53–89 (2010).
Chan, R. J. & Feng, G. S. PTPN11 is the first identified proto-oncogene that encodes a tyrosine phosphatase. Blood 109, 862–867 (2007).
Chen, Y. N. et al. Allosteric inhibition of SHP2 phosphatase inhibits cancers driven by receptor tyrosine kinases. Nature 535, 148–152 (2016).
Sun, C. et al. Intrinsic resistance to MEK inhibition in KRAS mutant lung and colon cancer through transcriptional induction of ERBB3. Cell Rep. 7, 86–93 (2014).
Vogel, W., Lammers, R., Huang, J. & Ullrich, A. Activation of a phosphotyrosine phosphatase by tyrosine phosphorylation. Science 259, 1611–1614 (1993).
Lu, W., Shen, K. & Cole, P. A. Chemical dissection of the effects of tyrosine phosphorylation of SHP-2. Biochemistry 42, 5461–5468 (2003).
Feng, G. S., Hui, C. C. & Pawson, T. SH2-containing phosphotyrosine phosphatase as a target of protein-tyrosine kinases. Science 259, 1607–1611 (1993).
Wu, T. R. et al. SHP-2 is a dual-specificity phosphatase involved in Stat1 dephosphorylation at both tyrosine and serine residues in nuclei. J. Biol. Chem. 277, 47572–47580 (2002).
Taylor, S. J., Resnick, R. J. & Shalloway, D. Nonradioactive determination of Ras-GTP levels using activated ras interaction assay. Methods Enzymol. 333, 333–342 (2001).
Waters, A. M. et al. Evaluation of the selectivity and sensitivity of isoform- and mutation-specific RAS antibodies. Sci. Signal 10, eaao3332 (2017).
Coppe, J. P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Kuilman, T. et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133, 1019–1031 (2008).
Eggert, T. et al. Distinct functions of senescence-associated immune responses in liver tumor surveillance and tumor progression. Cancer Cell 30, 533–547 (2016).
Hunter, J. C. et al. Biochemical and structural analysis of common cancer-associated KRAS mutations. Mol. Cancer Res. 13, 1325–1335 (2015).
Lu, S., Jang, H., Nussinov, R. & Zhang, J. The structural basis of oncogenic mutations G12, G13 and Q61 in small GTPase K-Ras4B. Sci. Rep. 6, 21949 (2016).
Fridman, A. L. & Tainsky, M. A. Critical pathways in cellular senescence and immortalization revealed by gene expression profiling. Oncogene 27, 5975–5987 (2008).
Purcell, M., Kruger, A. & Tainsky, M. A. Gene expression profiling of replicative and induced senescence. Cell Cycle 13, 3927–3937 (2014).
Hernandez-Segura, A. et al. Unmasking transcriptional heterogeneity in senescent cells. Curr. Biol. 27, 2652–2660 (2017).
Byrne, A. T. et al. Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat. Rev. Cancer 17, 254–268 (2017).
Jackson, E. L. et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 15, 3243–3248 (2001).
Ruess, D. A. et al. Mutant KRAS-driven cancers depend on PTPN11/SHP2 phosphatase. Nat. Med. https://doi.org/10.1038/s41591-018-0024-8 (2018).
Lan, L. et al. Shp2 signaling suppresses senescence in PyMT-induced mammary gland cancer in mice. EMBO J. 34, 1493–1508 (2015).
Garcia Fortanet, J. et al. Allosteric inhibition of SHP2: identification of a potent, selective, and orally efficacious phosphatase inhibitor. J. Med. Chem. 59, 7773–7782 (2016).
Prahallad, A. et al. PTPN11 is a central node in intrinsic and acquired resistance to targeted cancer drugs. Cell Rep. 12, 1978–1985 (2015).
Herold, M. J., van den Brandt, J., Seibler, J. & Reichardt, H. M. Inducible and reversible gene silencing by stable integration of an shRNA-encoding lentivirus in transgenic rats. Proc. Natl. Acad. Sci. USA 105, 18507–18512 (2008).
Ambrogio, C. et al. Combined inhibition of DDR1 and Notch signaling is a therapeutic strategy for KRAS-driven lung adenocarcinoma. Nat. Med. 22, 270–277 (2016).
Jonkers, J. et al. Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat. Genet. 29, 418–425 (2001).
Acknowledgements
We thank the Intervention Unit of the Mouse Clinic for Cancer and Aging (MCCA) of the Netherlands Cancer Institute, in particular, M. v. d. Ven and N. Proost, for the technical support with in vivo studies. We are grateful to pathologist H. Horlings for evaluation and scoring of in vivo samples. We are grateful to the NCI RAS Initiative for the kind gift of the panel of Rasless cells reconstituted with mutant KRAS alleles. This work was supported by grants from the Center for Cancer Genomics (CGC.NL) and the Dutch Cancer Society (KWF). S.M. was financially supported by an EMBO Long-Term Fellowship (ALTF 1184-2014) co-funded by the European Commission FP7 (Marie Curie Actions, LTFCOFUND2013, GA-2013-609409). G.G. was supported by an iCare fellowship by the Associazione Italiana per la Ricerca sul Cancro (AIRC) co-funded by the European Union. A. Bardelli was supported by the European Community’s Seventh Framework Programme under grant agreement no. 602901 MErCuRIC, Horizon 2020 grant agreement no. 635342-2 MoTriColor, IMI contract no. 115749 CANCER-ID, AIRC 2010 Special Program Molecular Clinical Oncology 5 per mille, Project no. 9970 Extension program, AIRC IG no. 16788, Fondazione Piemontese per la Ricerca sul Cancro-ONLUS 5 per mille 2011 e 2014 Ministero della Salute. A.V. and E.N. were supported by the Fondo de Investigaciones Sanitarias, FIS (PI16-01898 (to A.V.) and PI14-01109 (to E.N.)), and by the Spanish Association Against Cancer, AECC (CGB14142035THOM) (to A.V.).
Author information
Authors and Affiliations
Contributions
R.B. supervised the work. R.B., S.M. and A.P. designed the experiments. S.M., A.M.-S., A.P. and A. Bosma performed the experiments and analyzed the data. C.L. analyzed the data. P.K. designed the genetically engineered mouse model experiments. J.D.S. and N.d.W. acquired and analyzed the MRI data. A. Bardelli designed and G.G. carried out the xenograft experiments. A.V., E.N. and S.G.-R. designed and carried out the PDX and PDOX experiments. R.B. and S.M. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Table 1
Rights and permissions
About this article
Cite this article
Mainardi, S., Mulero-Sánchez, A., Prahallad, A. et al. SHP2 is required for growth of KRAS-mutant non-small-cell lung cancer in vivo. Nat Med 24, 961–967 (2018). https://doi.org/10.1038/s41591-018-0023-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0023-9
This article is cited by
-
From bench to bedside: current development and emerging trend of KRAS-targeted therapy
Acta Pharmacologica Sinica (2024)
-
Combinatorial strategies to target RAS-driven cancers
Nature Reviews Cancer (2024)
-
Re-expression of epigenetically silenced PTPRR by histone acetylation sensitizes RAS-mutant lung adenocarcinoma to SHP2 inhibition
Cellular and Molecular Life Sciences (2024)
-
SHP2 inhibitors maintain TGFβ signalling through SMURF2 inhibition
npj Precision Oncology (2023)
-
Targeting small GTPases: emerging grasps on previously untamable targets, pioneered by KRAS
Signal Transduction and Targeted Therapy (2023)