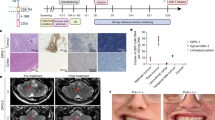
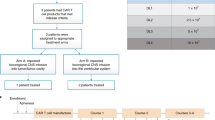
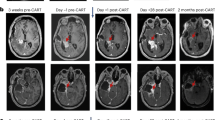
Abstract
Diffuse intrinsic pontine glioma (DIPG) and other diffuse midline gliomas (DMGs) with mutated histone H3 K27M (H3-K27M)1,2,3,4,5 are aggressive and universally fatal pediatric brain cancers6. Chimeric antigen receptor (CAR)-expressing T cells have mediated impressive clinical activity in B cell malignancies7,8,9,10, and recent results suggest benefit in central nervous system malignancies11,12,13. Here, we report that patient-derived H3-K27M-mutant glioma cell cultures exhibit uniform, high expression of the disialoganglioside GD2. Anti-GD2 CAR T cells incorporating a 4-1BBz costimulatory domain14 demonstrated robust antigen-dependent cytokine generation and killing of DMG cells in vitro. In five independent patient-derived H3-K27M+ DMG orthotopic xenograft models, systemic administration of GD2-targeted CAR T cells cleared engrafted tumors except for a small number of residual GD2lo glioma cells. To date, GD2-targeted CAR T cells have been well tolerated in clinical trials15,16,17. Although GD2-targeted CAR T cell administration was tolerated in the majority of mice bearing orthotopic xenografts, peritumoral neuroinflammation during the acute phase of antitumor activity resulted in hydrocephalus that was lethal in a fraction of animals. Given the precarious neuroanatomical location of midline gliomas, careful monitoring and aggressive neurointensive care management will be required for human translation. With a cautious multidisciplinary clinical approach, GD2-targeted CAR T cell therapy for H3-K27M+ diffuse gliomas of pons, thalamus and spinal cord could prove transformative for these lethal childhood cancers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Paugh, B. S. et al. Novel oncogenic PDGFRA mutations in pediatric high-grade gliomas. Cancer Res. 73, 6219–6229 (2013).
Wu, G. et al. Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat. Genet. 44, 251–253 (2012).
Buczkowicz, P. et al. Genomic analysis of diffuse intrinsic pontine gliomas identifies three molecular subgroups and recurrent activating ACVR1 mutations. Nat. Genet. 46, 451–456 (2014).
Taylor, K. R. et al. Recurrent activating ACVR1 mutations in diffuse intrinsic pontine glioma. Nat. Genet. 46, 457–461 (2014).
Wu, G. et al. The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat. Genet. 46, 444–450 (2014).
Jones, C. et al. Pediatric high-grade glioma: biologically and clinically in need of new thinking. Neuro Oncol. 19, 153–161 (2016).
Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
Davila, M. L. et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci. Transl. Med. 6, 224ra25 (2014).
Maude, S. L. et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl. J. Med. 371, 1507–1517 (2014).
Gardner, R. A. et al. Intent-to-treat leukemia remission by CD19 CAR T cells of defined formulation and dose in children and young adults. Blood 129, 3322–3331 (2017).
Brown, C. E. et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N. Engl. J. Med. 375, 2561–2569 (2016).
Ahmed, N. et al. HER2-specific chimeric antigen receptor-modified virus-specific T cells for progressive glioblastoma: a phase 1 dose-escalation Trial. JAMA Oncol. 3, 1094–1101 (2017).
O’Rourke, D.M. et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci. Transl. Med. 9, eaaa0984 (2017).
Long, A. H. et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat. Med. 21, 581–590 (2015).
Pule, M. A. et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat. Med. 14, 1264–1270 (2008).
Louis, C. U. et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood 118, 6050–6056 (2011).
Heczey, A. et al. CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol. Ther. 25, 2214–2224 (2017).
Majzner, R. G., Heitzeneder, S. & Mackall, C. L. Harnessing the immunotherapy revolution for the treatment of childhood cancers. Cancer Cell 31, 476–485 (2017).
Khuong-Quang, D. A. et al. K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol. 124, 439–447 (2012).
Schwartzentruber, J. et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482, 226–231 (2012).
Thomas, S., Straathof, K., Himoudi, N., Anderson, J. & Pule, M. An optimized GD2-targeting retroviral cassette for more potent and safer cellular therapy of neuroblastoma and other cancers. PLoS One 11, e0152196 (2016).
Long, A. H. et al. Reduction of MDSCs with all-trans retinoic acid improves CAR therapy efficacy for sarcomas. Cancer Immunol. Res. 4, 869–880 (2016).
Yu, A. L. et al. Anti-GD2 antibody with GM-CSF, interleukin-2, and isotretinoin for neuroblastoma. N. Engl. J. Med. 363, 1324–1334 (2010).
Perez Horta, Z., Goldberg, J. L. & Sondel, P. M. Anti-GD2 mAbs and next-generation mAb-based agents for cancer therapy. Immunotherapy 8, 1097–1117 (2016).
Hong, J. J. et al. Successful treatment of melanoma brain metastases with adoptive cell therapy. Clin. Cancer Res. 16, 4892–4898 (2010).
Suzuki, K. The pattern of mammalian brain gangliosides. II. Evaluation of the extraction procedures, postmortem changes and the effect of formalin preservation. J. Neurochem. 12, 629–638 (1965).
Kramer, K. et al. Compartmental intrathecal radioimmunotherapy: results for treatment for metastatic CNS neuroblastoma. J. Neurooncol. 97, 409–418 (2010).
Kramer, K. et al. A phase II study of radioimmunotherapy with intraventricular 131 I-3F8 for medulloblastoma. Pediatr. Blood Cancer 65, e26754 (2018).
Monje, M. et al. Hedgehog-responsive candidate cell of origin for diffuse intrinsic pontine glioma. Proc. Natl Acad. Sci. USA 108, 4453–4458 (2011).
Qin, E. Y. et al. Neural precursor-derived pleiotrophin mediates subventricular zone invasion by glioma. Cell 170, 845–859 (2017).
Grasso, C. S. et al. Functionally defined therapeutic targets in diffuse intrinsic pontine glioma. Nat. Med. 21, 555–559 (2015).
Fry, T. J. et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat. Med. 24, 20–28 (2018).
Walker, A. J. et al. Tumor antigen and receptor densities regulate efficacy of a chimeric antigen receptor targeting anaplastic lymphoma kinase. Mol. Ther. 25, 2189–2201 (2017).
Ali, N. et al. Xenogeneic graft-versus-host-disease in NOD-scid IL-2Rγnull mice display a T-effector memory phenotype. PLoS One 7, e44219 (2012).
Nagaraja, S. et al. Transcriptional dependencies in diffuse intrinsic pontine glioma. Cancer Cell 31, 635–652 (2017).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Louis, D. N. et al. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 131, 803–820 (2016).
Wolchok, J. D. et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin. Cancer Res. 15, 7412–7420 (2009).
Gust, J. et al. Endothelial activation and blood–brain barrier disruption in neurotoxicity after adoptive immunotherapy with CD19 CAR-T cells. Cancer Discov. 7, 1404–1419 (2017).
Lin, G. L. & Monje, M. A protocol for rapid post-mortem cell culture of diffuse intrinsic pontine glioma (DIPG). J. Vis. Exp. 7, e55360 (2017).
Lynn, R. C. et al. Targeting of folate receptor β on acute myeloid leukemia blasts with chimeric antigen receptor-expressing T cells. Blood 125, 3466–3476 (2015).
Haso, W. et al. Anti-CD22-chimeric antigen receptors targeting B-cell precursor acute lymphoblastic leukemia. Blood 121, 1165–1174 (2013).
Sen, G., Chakraborty, M., Foon, K. A., Reisfeld, R. A. & Bhattacharya-Chatterjee, M. B. Induction of IgG antibodies by an anti-idiotype antibody mimicking disialoganglioside GD2. J. Immunother. 21, 75–83 (1998).
Jena, B. et al. Chimeric antigen receptor (CAR)-specific monoclonal antibody to detect CD19-specific T cells in clinical trials. PLoS One 8, e57838 (2013).
Hendel, A. et al. Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat. Biotechnol. 33, 985–989 (2015).
Chu, V. T. et al. Increasing the efficiency of homology-directed repair for CRISPR–Cas9-induced precise gene editing in mammalian cells. Nat. Biotechnol. 33, 543–548 (2015).
Brinkman, E. K., Chen, T., Amendola, M. & van Steensel, B. Easy quantitative assessment of genome editing by sequence trace decomposition. Nucleic Acids Res. 42, e168 (2014).
Acknowledgements
We thank the following for generously providing cell cultures: A. Moore (University of Queensland) and C. Jones (Institute of Cancer Research) for QCTB R059, R. Seeger (Children's Hospital Los Angeles) for CHLA136, 255, C. Khanna (National Cancer Institute) for MG63-3 and L. Helman (Children's Hospital Los Angeles) for EW8 and TC32.
This work was supported by a Stand Up To Cancer–St. Baldrick’s–National Cancer Institute Pediatric Dream Team Translational Cancer Research Grant (C.L.M.). Stand Up To Cancer is a program of the Entertainment Industry Foundation administered by the American Association for Cancer Research. C.L.M is a member of the Parker Institute for Cancer Immunotherapy, which supports the Stanford University Cancer Immunotherapy Program. The authors gratefully acknowledge support from the National Institute of Neurological Disorders and Stroke (F31NS098554 to C.W.M. and R01NS092597 to M.M.), Abbie’s Army Foundation (M.M.), Unravel Pediatric Cancer (M.M.), Maiy’s Miracle Foundation (E.P.A.), Stella S. Jones Foundation (M.M.), McKenna Claire Foundation (M.M.), Alex’s Lemonade Stand Foundation (M.M.), Izzy's Infantry Foundation (M.M.), The Cure Starts Now Foundation and DIPG Collaborative (M.M.), Lyla Nsouli Foundation (M.M.), Declan Gloster Memorial Fund (M.M.), N8 Foundation (M.M.), Fly a Kite Foundation (M.M.), Liwei Wang Research Fund (M.M.), Virginia and D.K. Ludwig Fund for Cancer Research (M.M. and C.L.M.), Sam Jeffers Foundation (M.M.), Michael Mosier DEFEAT DIPG Foundation (M.M.), ChadTough Foundation (M.M.), Reller Family Research Fund, Child Health Research Institute at Stanford and SPARK program (M.M.) and the Anne T. and Robert M. Bass Endowed Faculty Scholarship in Pediatric Cancer and Blood Diseases (M.M.).
Author information
Authors and Affiliations
Contributions
C.W.M. and E.P.A. performed the antibody array screening. C.W.M. and M.M. identified GD2 as a target in H3-K27M+ DMGs. C.W.M. and S.S. performed immunohistochemistry and immunofluorescence microscopy on primary and xenograft tissue. L.L. and R.G.M. designed CAR constructs. R.G.M. and M.K. prepared CAR T cells for in vivo experiments. C.W.M. and P.J.W. conducted in vivo experiments. R.G.M., M.K. and S.P.R. performed in vitro T cell experiments and flow cytometry. E.H. contributed VUMC-DIPG10 and data on ganglioside synthesis pathway expression. H.V. performed neuropathological review of brain tissue. S.H. performed CRISPR–Cas9-mediated gene editing. C.W.M., R.G.M., H.V., M.M. and C.L.M. contributed to data analysis and interpretation. C.W.M., M.M., R.G.M. and C.L.M wrote the manuscript. C.W.M. and R.G.M. made the figures. M.M. and C.L.M. supervised all aspects of the work.
Corresponding authors
Ethics declarations
Competing interests
C.L.M, M.M., R.G.M. and C.W.M. are inventors on a patent application for GD2-directed CAR use for H3-K27M DMG.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Tables 2 and 3
Supplementary Table 1
Surface Panel Flow Cytometry Screening Data
Rights and permissions
About this article
Cite this article
Mount, C.W., Majzner, R.G., Sundaresh, S. et al. Potent antitumor efficacy of anti-GD2 CAR T cells in H3-K27M+ diffuse midline gliomas. Nat Med 24, 572–579 (2018). https://doi.org/10.1038/s41591-018-0006-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0006-x
This article is cited by
-
GMP-manufactured CRISPR/Cas9 technology as an advantageous tool to support cancer immunotherapy
Journal of Experimental & Clinical Cancer Research (2024)
-
Biopsy of diffuse midline glioma is safe and impacts targeted therapy: a systematic review and meta-analysis
Child's Nervous System (2024)
-
Novel Clinical Trials and Approaches in the Management of Glioblastoma
Current Oncology Reports (2024)
-
The landscape of current research on pediatric diffuse midline glioma: a quantitative analysis of shifts, leaders, and future avenues
Child's Nervous System (2024)
-
Glioblastoma stem cell metabolism and immunity
Cancer and Metastasis Reviews (2024)