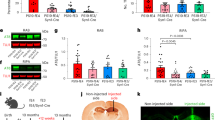
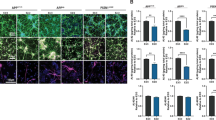
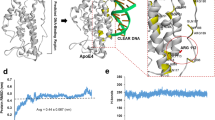
Abstract
Efforts to develop drugs for Alzheimer's disease (AD) have shown promise in animal studies, only to fail in human trials, suggesting a pressing need to study AD in human model systems. Using human neurons derived from induced pluripotent stem cells that expressed apolipoprotein E4 (ApoE4), a variant of the APOE gene product and the major genetic risk factor for AD, we demonstrated that ApoE4-expressing neurons had higher levels of tau phosphorylation, unrelated to their increased production of amyloid-β (Aβ) peptides, and that they displayed GABAergic neuron degeneration. ApoE4 increased Aβ production in human, but not in mouse, neurons. Converting ApoE4 to ApoE3 by gene editing rescued these phenotypes, indicating the specific effects of ApoE4. Neurons that lacked APOE behaved similarly to those expressing ApoE3, and the introduction of ApoE4 expression recapitulated the pathological phenotypes, suggesting a gain of toxic effects from ApoE4. Treatment of ApoE4-expressing neurons with a small-molecule structure corrector ameliorated the detrimental effects, thus showing that correcting the pathogenic conformation of ApoE4 is a viable therapeutic approach for ApoE4-related AD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Huang, Y. & Mucke, L. Alzheimer mechanisms and therapeutic strategies. Cell 148, 1204–1222 (2012).
Golde, T. E., Schneider, L. S. & Koo, E. H. Anti-Aβ therapeutics in Alzheimer's disease: the need for a paradigm shift. Neuron 69, 203–213 (2011).
Corder, E. H. et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer's disease in late-onset families. Science 261, 921–923 (1993).
Saunders, A. M. et al. Association of apolipoprotein E allele ε4 with late-onset familial and sporadic Alzheimer's disease. Neurology 43, 1467–1472 (1993).
Farrer, L. A. et al. APOE and Alzheimer Disease Meta Analysis Consortium. Effects of age, sex and ethnicity on the association between apolipoprotein E genotype and Alzheimer's disease. A meta-analysis. J. Am. Med. Assoc. 278, 1349–1356 (1997).
Genin, E. et al. APOE and Alzheimer's disease: a major gene with semi-dominant inheritance. Mol. Psychiatry 16, 903–907 (2011).
Huang, Y. Aβ-independent roles of apolipoprotein E4 in the pathogenesis of Alzheimer's disease. Trends Mol. Med. 16, 287–294 (2010).
Mahley, R. W. & Huang, Y. Apolipoprotein E sets the stage: response to injury triggers neuropathology. Neuron 76, 871–885 (2012).
Ashe, K. H. & Zahs, K. R. Probing the biology of Alzheimer's disease in mice. Neuron 66, 631–645 (2010).
Israel, M. A. et al. Probing sporadic and familial Alzheimer's disease using induced pluripotent stem cells. Nature 482, 216–220 (2012).
Yahata, N. et al. Anti-Aβ drug screening platform using human iPS-cell-derived neurons for the treatment of Alzheimer's disease. PLoS One 6, e25788 (2011).
Yagi, T. et al. Modeling familial Alzheimer's disease with induced pluripotent stem cells. Hum. Mol. Genet. 20, 4530–4539 (2011).
Almeida, S. et al. Induced pluripotent stem cell models of progranulin-deficient frontotemporal dementia uncover specific reversible neuronal defects. Cell Rep. 2, 789–798 (2012).
Kondo, T. et al. Modeling Alzheimer's disease with iPSCs reveals stress phenotypes associated with intracellular Aβ and differential drug responsiveness. Cell Stem Cell 12, 487–496 (2013).
Liu, Q. et al. Effect of potent γ-secretase modulator in human neurons derived from multiple presenilin-1 induced pluripotent stem cell mutant carriers. JAMA Neurol. 71, 1481–1489 (2014).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Brecht, W. J. et al. Neuron-specific apolipoprotein E4 proteolysis is associated with increased tau phosphorylation in brains of transgenic mice. J. Neurosci. 24, 2527–2534 (2004).
Harris, F. M. et al. Carboxyl-terminal-truncated apolipoprotein E4 causes Alzheimer's-disease-like neurodegeneration and behavioral deficits in transgenic mice. Proc. Natl Acad. Sci. USA 100, 10966–10971 (2003).
Hoover, B. R. et al. Tau mislocalization to dendritic spines mediates synaptic dysfunction independently of neurodegeneration. Neuron 68, 1067–1081 (2010).
Thies, E. & Mandelkow, E. M. Missorting of tau in neurons causes degeneration of synapses that can be rescued by the kinase MARK2 (Par-1). J. Neurosci. 27, 2896–2907 (2007).
Woodruff, G. et al. The presenilin-1 ΔE9 mutation results in reduced γ-secretase activity, but not total loss of PS1 function, in isogenic human stem cells. Cell Rep. 5, 974–985 (2013).
Castellano, J. M. et al. Human APOE isoforms differentially regulate brain amyloid-β peptide clearance. Sci. Transl. Med. 3, 89ra57 (2011).
Busciglio, J., Lorenzo, A., Yeh, J. & Yankner, B. A. β-amyloid fibrils induce tau phosphorylation and loss of microtubule binding. Neuron 14, 879–888 (1995).
Jin, M. et al. Soluble amyloid β-protein dimers isolated from Alzheimer cortex directly induce Tau hyperphosphorylation and neuritic degeneration. Proc. Natl. Acad. Sci. USA 108, 5819–5824 (2011).
Cunningham, M. et al. hPSC-derived maturing GABAergic interneurons ameliorate seizures and abnormal behavior in epileptic mice. Cell Stem Cell 15, 559–573 (2014).
Nicholas, C. R. et al. Functional maturation of hPSC-derived forebrain interneurons requires an extended timeline and mimics human neural development. Cell Stem Cell 12, 573–586 (2013).
Ahn, S., Kim, T. G., Kim, K. S. & Chung, S. Differentiation of human pluripotent stem cells into medial ganglionic eminence versus caudal ganglionic eminence cells. Methods 101, 103–112 (2016).
Fong, H. et al. Genetic correction of tauopathy phenotypes in neurons derived from human induced pluripotent stem cells. Stem Cell Rep. 1, 226–234 (2013).
Mak, A. C. et al. Effects of the absence of apolipoprotein E on lipoproteins, neurocognitive function and retinal function. JAMA Neurol. 71, 1228–1236 (2014).
Xu, Q. et al. Profile and regulation of apolipoprotein E (APOE) expression in the CNS in mice with targeting of green fluorescent protein gene to the Apoe locus. J. Neurosci. 26, 4985–4994 (2006).
Xu, Q. et al. Intron-3 retention and splicing controls neuronal expression of apolipoprotein E in the CNS. J. Neurosci. 28, 1452–1459 (2008).
Xu, P.-T. et al. Specific regional transcription of apolipoprotein E in human brain neurons. Am. J. Pathol. 154, 601–611 (1999).
Ulrich, J. D. et al. In vivo measurement of apolipoprotein E from the brain interstitial fluid using microdialysis. Mol. Neurodegener. 8, 13 (2013).
Mahley, R. W., Weisgraber, K. H. & Huang, Y. Apolipoprotein E4: a causative factor and therapeutic target in neuropathology, including Alzheimer's disease. Proc. Natl. Acad. Sci. USA 103, 5644–5651 (2006).
Chen, H. K. et al. Small-molecule structure correctors abolish detrimental effects of apolipoprotein E4 in cultured neurons. J. Biol. Chem. 287, 5253–5266 (2012).
Brodbeck, J. et al. Structure-dependent impairment of intracellular apolipoprotein E4 trafficking and its detrimental effects are rescued by small-molecule structure correctors. J. Biol. Chem. 286, 17217–17226 (2011).
Mahley, R. W. & Huang, Y. Small-molecule structure correctors target abnormal protein structure and function: structure corrector rescue of apolipoprotein-E4-associated neuropathology. J. Med. Chem. 55, 8997–9008 (2012).
Bien-Ly, N., Gillespie, A. K., Walker, D., Yoon, S. Y. & Huang, Y. Reducing human apolipoprotein E levels attenuates age-dependent Aβ accumulation in mutant human amyloid precursor protein transgenic mice. J. Neurosci. 32, 4803–4811 (2012).
Kim, J., Basak, J. M. & Holtzman, D. M. The role of apolipoprotein E in Alzheimer's disease. Neuron 63, 287–303 (2009).
Huynh, T. V. et al. Age-dependent effects of apoE reduction using antisense oligonucleotides in a model of β-amyloidosis. Neuron 96, 1013–1023.e4 (2017).
Liu, C. C. et al. ApoE4 accelerates early seeding of amyloid pathology. Neuron 96, 1024–1032.e3 (2017).
Huang, Y. A., Zhou, B., Wernig, M. & Südhof, T. C. ApoE2, ApoE3 and ApoE4 differentially stimulate APP transcription and Aβ secretion. Cell 168, 427–441.e21 (2017).
Kamenetz, F. et al. APP processing and synaptic function. Neuron 37, 925–937 (2003).
Cirrito, J. R. et al. Synaptic activity regulates interstitial fluid amyloid-β levels in vivo. Neuron 48, 913–922 (2005).
LaDu, M. J. et al. Self-assembly of HEK-cell-secreted ApoE particles resembles ApoE enrichment of lipoproteins as a ligand for the LDL-receptor-related protein. Biochemistry 45, 381–390 (2006).
Shi, Y. et al. ApoE4 markedly exacerbates Tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549, 523–527 (2017).
Andrews-Zwilling, Y. et al. Apolipoprotein E4 causes age- and Tau-dependent impairment of GABAergic interneurons, leading to learning and memory deficits in mice. J. Neurosci. 30, 13707–13717 (2010).
Leung, L. et al. Apolipoprotein E4 causes age- and sex-dependent impairments of hilar GABAergic interneurons and learning and memory deficits in mice. PLoS One 7, e53569 (2012).
Li, G. et al. GABAergic interneuron dysfunction impairs hippocampal neurogenesis in adult apolipoprotein E4 knock-in mice. Cell Stem Cell 5, 634–645 (2009).
Tong, L. M. et al. Inhibitory interneuron progenitor transplantation restores normal learning and memory in ApoE4 knock-in mice without or with Aβ accumulation. J. Neurosci. 34, 9506–9515 (2014).
Nuriel, T. et al. Neuronal hyperactivity due to loss of inhibitory tone in APOE4 mice lacking Alzheimer's-disease-like pathology. Nat. Commun. 8, 1464 (2017).
Knoferle, J. et al. Apolipoprotein E4 produced in GABAergic interneurons causes learning and memory deficits in mice. J. Neurosci. 34, 14069–14078 (2014).
Gillespie, A. K. et al. Apolipoprotein E4 causes age-dependent disruption of slow γ-oscillations during hippocampal sharp-wave ripples. Neuron 90, 740–751 (2016).
Liu, C. C., Liu, C. C., Kanekiyo, T., Xu, H. & Bu, G. Apolipoprotein E and Alzheimer's disease: risk, mechanisms and therapy. Nat. Rev. Neurol. 9, 106–118 (2013).
Takahashi, K., Okita, K., Nakagawa, M. & Yamanaka, S. Induction of pluripotent stem cells from fibroblast cultures. Nat. Protoc. 2, 3081–3089 (2007).
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat. Biotechnol. 27, 275–280 (2009).
Hu, B. Y. & Zhang, S. C. Differentiation of spinal motor neurons from pluripotent human stem cells. Nat. Protoc. 4, 1295–1304 (2009).
Krencik, R., Weick, J. P., Liu, Y., Zhang, Z. J. & Zhang, S. C. Specification of transplantable astroglial subtypes from human pluripotent stem cells. Nat. Biotechnol. 29, 528–534 (2011).
Acknowledgements
We thank A. Karydas for assistance with skin biopsies, S. Ordway and T. Pak for editorial assistance and P. Davies (Albert Einstein College of Medicine) for the PHF1 antibody. This work was supported by funding from the US National Institutes of Health (grant nos. AG048030 (Y.H.), AG048017 (Y.H.), AG056305 (Y.H.) and AG023501 (B.L.M. and Y.H.)) and the California Institute for Regenerative Medicine (grant nos. RN2-00952 and TRAN1-09394; both to Y.H.) and by a gift from the Roddenberry Foundation. C.W. was partially supported by a fellowship from the California Institute for Regenerative Medicine.
Author information
Authors and Affiliations
Contributions
C.W. and Y.H. designed and coordinated the study; C.W. performed most of the studies and data analyses; R.N. performed all MGE studies and related data analysis; Q.X. and D.J. helped with off-target analysis of gene editing and work with APOE-expressing lentiviral vectors and performed some western blots; D.W. designed and prepared the APOE-ε3 donor DNA for gene editing; D.W. and S.Y.Y. helped with genetic screening; M.E.B. helped with the cell culture; H.Y. helped with MGE studies and image collection; G.L. helped with miPSC studies; Z.A.M., B.L.M. and M.J.M. provided the APOE-null human skin biopsy; and C.W. and Y.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Y.H. is a cofounder and scientific advisory board member of E-Scape Bio, Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, C., Najm, R., Xu, Q. et al. Gain of toxic apolipoprotein E4 effects in human iPSC-derived neurons is ameliorated by a small-molecule structure corrector. Nat Med 24, 647–657 (2018). https://doi.org/10.1038/s41591-018-0004-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0004-z
This article is cited by
-
Synaptic vesicle glycoprotein 2 A in serum is an ideal biomarker for early diagnosis of Alzheimer’s disease
Alzheimer's Research & Therapy (2024)
-
Promoting Alzheimer’s disease research and therapy with stem cell technology
Stem Cell Research & Therapy (2024)
-
Simple modeling of familial Alzheimer’s disease using human pluripotent stem cell-derived cerebral organoid technology
Stem Cell Research & Therapy (2024)
-
Cell type-specific roles of APOE4 in Alzheimer disease
Nature Reviews Neuroscience (2024)
-
Thy1-ApoE4/C/EBPβ double transgenic mice act as a sporadic model with Alzheimer’s disease
Molecular Psychiatry (2024)