Abstract
Here we report the identification of human CD66b−CD64dimCD115− neutrophil-committed progenitor cells (NCPs) within the SSCloCD45dimCD34+ and CD34dim/− subsets in the bone marrow. NCPs were either CD45RA+ or CD45RA−, and in vitro experiments showed that CD45RA acquisition was not mandatory for their maturation process. NCPs exclusively generated human CD66b+ neutrophils in both in vitro differentiation and in vivo adoptive transfer experiments. Single-cell RNA-sequencing analysis indicated NCPs fell into four clusters, characterized by different maturation stages and distributed along two differentiation routes. One of the clusters was characterized by an interferon-stimulated gene signature, consistent with the reported expansion of peripheral mature neutrophil subsets that express interferon-stimulated genes in diseased individuals. Finally, comparison of transcriptomic and phenotypic profiles indicated NCPs represented earlier neutrophil precursors than the previously described early neutrophil progenitors (eNePs), proNeus and COVID-19 proNeus. Altogether, our data shed light on the very early phases of neutrophil ontogeny.
Similar content being viewed by others
Main
Studies performed by single-cell sorting, transcriptional analysis, single-cell transplants and clonal tracking analysis1,2,3,4,5,6,7,8,9 have challenged the classical hierarchical model of hematopoiesis10,11. It has been ascertained that hematopoiesis occurs as a continuous process along developmental trajectories following early erythroid/megakaryocyte/eosinophil/basophil and lympho/myeloid lineages to generate mature, terminally differentiated cells. Transcriptomic profiling has shown that intermediate compartments of progenitors, such as common myeloid progenitors (CMPs) or granulocyte-macrophage progenitors (GMPs)10, are largely composed of clusters of cells displaying lineage-selective commitment. However, even though single-cell RNA-sequencing (scRNA-seq) experiments reveal the heterogeneity of various cell subpopulations12, flow-sorting and immunophenotyping approaches are still needed for their identification.
Conventional GMPs (cGMPs) represent the most restricted subset of myeloid progenitors in humans13,14. Based on flow cytometry approaches15, cGMPs have been fractionated into three main compartments: granulocyte-monocyte-dendritic cell (DC) progenitors (GMDPs), which generate CD66b+ granulocytes, CD14+ monocytes, conventional DCs (cDC1s, cDC2s) and plasmacytoid DCs (pDCs); monocyte-DC progenitors (MDPs), which generate DCs and CD14+ monocytes; and common dendritic progenitors (CDPs), which are exclusively committed to DCs. A different panel of markers which includes CD64 identified, within cGMPs, the common monocyte progenitors (cMoPs), which exclusively differentiate into pre-monocytes and subsequently CD14+ monocytes, and the revised GMPs (rGMPs), which generate only CD66b+ granulocytes and CD14+ monocytes16. Although neutrophil precursors are included in cGMPs3,4,6,17,18,19, little progress has been made to characterize the ontogeny of human neutrophils. A CD34+CD115−CD64+ subpopulation that generates granulocytes was identified in the fetal bone marrow (BM)20, without discriminating between eosinophil, basophil or neutrophil potential. Similarly, the phenotypic and/or morphological features of the GMDPs15 and rGMPs16-derived CD66b+ granulocytes were not pursued in detail. Various human neutrophil precursors, preNeus21, human Neutrophil Progenitors (hNePs)22, proNeus23 and early neutrophil progenitors (eNePs)24, have been described recently. However, they all express CD66b and CD15 and thus resemble the promyelocyte (PM), or more mature, neutrophil precursors. In contrast, the CD34+Ly6C+CD115− proNeu1s in mice are found within GMPs23, and represent the earliest identifiable precursors along the neutrophil maturation trajectory.
Here we report the identification, phenotypic characterization, single-cell transcriptomic analysis, and in vitro and in vivo functions of previously undescribed human uni-lineage CD34+ and CD34dim/− NCPs, and show they are upstream of eNePs, proNeus and COVID-19 proNeus23,24,25.
Results
CD123, CD115 and CD64 identify myeloid progenitors within cGMPs
To identify precursors of human neutrophils at earlier stages than PMs, we analyzed the SSCloCD45dim cells present in the low-density fraction of the BM (BM low-density cells (BM-LDCs)) (Fig. 1a and Extended Data Fig. 1a). SSCloCD45dim cells were lineage negative (Extended Data Fig. 1b) and included CD34+CD38− hematopoietic stem cells (HSCs) as well as CD34+ and CD34dim/− myeloid-lymphoid progenitors26,27, with the CD34dim/− cells representing a transitional progenitor stage4. To analyze their potential ability to generate neutrophils, we stained BM-LDCs with an antibody panel including CD34, CD38, CD10, CD123, CD45RA, CD64 and CD115 (refs. 13,14,20,28). We identified an SSCloLin−CD45dimCD10−CD38+ subset with variable expression of CD34 and CD45RA (Fig. 1a), including CD34+CD45RA−, CD34+CD45RA+, CD34dim/−CD45RA+ and CD34dim/−CD45RA− cell subsets. The CD34+CD45RA+ cells were also CD135+, and thus represented cGMPs (not shown)14, while CD34+CD45RA− cells (Fig. 1a) included more immature progenitors, such as CMPs and megakaryocyte-erythroid progenitors (MEPs)13,14. Based on expression of CD123, CD115 and CD64, cGMPs were subdivided into five discrete cell populations (Fig. 1a): CDPs; CD64+CD115+ cells, which were also CLEC12A+ and resembled the monocyte-committed progenitor population20, as well as cMoPs16; CD64−CD115+ cells, resembling MDPs15; CD64−CD115− cells, resembling the conventional GMDPs15; and CD64dimCD115− cells, resembling previously described granulocyte progenitors20.
a, Flow cytometry strategy for the identification of Lin−SSCloCD45dimCD10−CD38+ BM-LDCs; CD34+CD45RA−, CD34dim/−CD45RA+ and CD34dim/−CD45RA− subsets; and CD34+CD45RA+ cGMPs. CD123+CD115− CDPs (yellow); CD123dim/−CD64+CD115+ cMoPs (light blue), CD123dim/−CD64−CD115+ MDPs (brown); CD123dim/−CD64−CD115− GMDPs (CD64− GMDPs, dark blue) and CD123dim/−CD64dimCD115− cells (CD64dimCD115−, green) are displayed within cGMPs (one representative experiment out of 20 is shown). b, Flow cytometry strategy for the identification of CD66b+ neutrophils (green), CD141+CD14− cDC1s (magenta), CD303+CD14− pDCs/pre-DCs/DC5s (orange), CD14+CD1c− monocytes (light blue) and CD14−CD1c+ cDC2s (pink) generated by CD34+CD45RA+CD64dimCD115− cells, cMoPs or MDPs incubated with SFGc for 7 d. c, Bar graphs reporting the percentages of the leukocyte types generated, as defined in b, by CD34+CD45RA+CD64dimCD115− cells (n = 10), cMoPs (n = 8) or MDPs (n = 5). Data represent mean ± s.e.m. d,f, Flow cytometry strategy for the identification of CD66b+CD10−CD11b−CD16− PMs (gray), CD66b+CD10−CD11bdim/+CD16− MYs (pink), CD66b+CD10−CD11b+CD16+ MMs (light red), CD66b+CD10−CD11b+CD16++CD10− BCs (red) and CD66b+CD11b+CD16++CD10+ SNs (dark red) derived from CD34+CD45RA+CD64dimCD115− cells cultured with SFGc for 7 d (d, n = 15) and 14 d (f, n = 3). e,g, Morphology of neutrophils derived from CD34+CD45RA+CD64dimCD115− cells after 7 d (e) and 14 d (g) of culture (n = 3) K, thousand.
Collectively, the CD123, CD115 and CD64 combination of markers discriminates five different myeloid progenitor subsets within cGMPs.
CD34+CD45RA+CD64dimCD115− cells are neutrophil precursors
It was reported that neutrophils, but not basophils or eosinophils, derive from cGMPs4,17,18,19. To investigate whether CD34+CD45RA+CD64dimCD115− cells represented neutrophil-restricted progenitors, we cultured them with MS-5 cells29 for 7 d in the presence of stem cell factor (SCF), FMS-like tyrosine kinase 3 ligand (Flt3L) and granulocyte-colony stimulating factor (G-CSF) (hereafter, SFGc). CD34+CD45RA+CD64dimCD115− cells gave rise mostly to CD66b+ cells, and very few CD14+ monocytes (Fig. 1b,c). By contrast, SFGc-cultured cMoPs differentiated mainly into CD14+ monocytes, and minimally to CD66b+ cells and DCs (Fig. 1b,c), confirming their commitment to monocytes16. Conversely, SFGc-treated MDPs gave rise to CD141+CD14− cDC1s (which were also CLEC9A+), CD303+CD14− pDCs/pre-DCs/DC5 (refs. 30,31), CD14dim/−CD1c+ cDC2s and CD14+ monocytes (Fig. 1b,c), consistent with their predicted heterogeneous composition15,32. Moreover, SFGc fully maintained the viability of progenitor-generated cells after 7 d (Extended Data Fig. 2a) and promoted the expansion of CD34+CD45RA+CD64dimCD115− cells compared with cMoPs and MDPs (Extended Data Fig. 2b), consistent with the highest expression of CD114/G-CSFR in CD34+CD45RA+CD64dimCD115− cells (Extended Data Fig. 2c). Phenotypic (Fig. 1d) and morphologic (Fig. 1e) analysis of CD66b+ cells derived from SFGc-treated CD34+CD45RA+CD64dimCD115− cells indicated that they were mostly immature neutrophils, composed of CD11b−CD16− PMs, CD11bdim/+CD16− myelocytes (MYs), CD11b+CD16+ metamyelocytes (MMs), CD11b+CD16++CD10− band cells (BCs) and CD11b+CD16++CD10+ segmented neutrophils (SNs) (Fig. 2d). Consistently, CD34+CD45RA+CD64dimCD115− cells incubated with SFGc for 14 d generated more mature SNs (Fig. 1f), as confirmed by nucleus morphology (Fig. 1g). When CD34+CD45RA+CD64dimCD115− cells were cultured with SCF, Flt3L and granulocyte-macrophage colony-stimulating factor (GM-CSF) (SFG), which favors monocyte or monocyte and DC differentiation from cMoPs or MDPs, respectively16,29, the main progeny was, again, constituted by neutrophils (Extended Data Fig. 2d), confirming the uni-lineage commitment of CD34+CD45RA+CD64dimCD115− cells. Collectively, these experiments formally identified CD34+CD45RA+CD64dimCD115− cells as NCPs.
A CD64dimCD115− phenotype defines other CD34+ or CD34dim/− NCPs
Because NCPs have been described within the CD45RA− compartment3,19, we tested whether other, not yet described, neutrophil-restricted progenitors could be identified within the SSCloCD45dim region (Fig. 2a). A search for cells expressing the CD64dimCD115− phenotype in all the fractions delimited by the CD34 and CD45RA marker combination identified a total of four subsets of NCPs: CD34+CD45RA− NCP1s, CD34+CD45RA+ NCP2s, CD34dim/−CD45RA+ NCP3s and CD34dim/−CD45RA− NCP4s (Fig. 2a). Notably, we were able to identify all four NCPs in cord blood (CB) samples, spleen biopsies (not shown) and in reconstituting BM samples (day +21) from patients undergoing allogeneic hematopoietic stem cell transplant (alloHSCT) for hematological malignancies (Extended Data Fig. 3).
a, Flow cytometry strategy for the identification of CD64dimCD115−CD34+CD45RA− NCP1s (orange), CD64dimCD115−CD34+CD45RA+ NCP2s (green), CD64dimCD115−CD34dim/−CD45RA+ NCP3s (magenta), CD64dimCD115−CD34dim/−CD45RA− NCP4s (turquoise), cMoPs (light blue), MDPs (brown) and CD34dim/−CD45RA+CD64++CD115+ pre-monocytes (black) within SSCloCD45dimCD38+CD10−CD123dim/− BM-LDCs (one representative experiment out of 15 is shown). b, Morphology of purified NCP populations and PMs. Red arrows point to visible nucleoli, while blue arrows point to granules (n = 3). c, Bar graphs depicting the fold expansion of purified NCPs treated with SFGc for 7 d (NCP1s, NCP3s and NCP4s, n = 7; NCP2s, n = 8). Data represent mean ± s.e.m. Analysis was performed using one-way ANOVA and Tukey post hoc test. d, Flow cytometry strategy for identification of the phenotype of PMs (gray), MYs (pink), MMs (light red), BCs (red) and SNs (dark red) derived from NCPs cultured with SFGc for 7 d (left plots), as well as their morphology (middle panels). Right bar graphs report the percentages of the various neutrophil precursors. Data represent mean ± s.e.m. (NCP1s, NCP3s and NCP4s, n = 5; NCP2s, n = 6). Analysis was performed by using Friedman test and two-sided Dunn post hoc test.
Next, we sorted CD34+ and CD34dim/− NCPs to analyze their morphology and developmental potential. Morphologically, NCPs were found to display a high nuclear/cytoplasmic ratio (Fig. 2b) compatible with a progenitor identity. Moreover, granules were evident only in NCP3s and NCP4s, but at lower levels than in PMs (Fig. 2b). In addition, SFGc-treated NCP1s and NCP2s were found to display a comparable (Fig. 2c), but higher, proliferative potential than that exhibited by SFGc-treated NCP3s and NCP4s (Fig. 2c). Notably, NCPs were found to produce almost exclusively neutrophils at different stages of maturation, as determined by their phenotype and morphology (Fig. 2d). By the latter criteria, we found no eosinophils and/or basophils among the SFGc-generated cells (not shown), even if NCPs were incubated in SFG plus IL-3 and/or IL-5 (ref. 17). Moreover, NCPs generated no DCs when cultured in SFGc (not shown), while NCP2s and NCP3s exclusively generated a few CD14+ monocytes (not shown), which we subsequently found to derive from contaminating cMoP and pre-monocyte populations (not shown). In accordance with the results observed from the in vitro cultures, colony forming unit (CFU) assay experiments indicated that the colonies derived from NCPs were almost entirely granulocyte CFU (CFU-G), with a few macrophage CFU (CFU-M) arising from NCP2 and NCP3 only (Extended Data Fig. 4a). Of note, neutrophils generated by NCPs displayed respiratory burst ability (Extended Data Fig. 4b) and phagocytosis capacity (Extended Data Fig. 4c), both comparable to those displayed by peripheral mature neutrophils (PMNs). Finally, we found that NCP3s and NCP4s, unlike NCP1s and NCP2s, expressed CD15, although at lower levels than PMs and mature SNs (Extended Data Fig. 4d). However, a fraction of NCP3s and NCP4s expressed CD15 at levels comparable to those expressed by cMoPs, pre-monocytes and mature monocytes (Extended Data Fig. 4e), suggesting that CD15 alone does not specifically identify neutrophil precursors. Thus, these data exclude that NCPs correspond to SSCintLin−CD34−CD15intCD11b−CD16− early PMs32, which partially overlap with pre-monocytes. These results indicated that CD34+ and CD34dim/− NCPs were upstream of PMs in the neutrophil maturation pathway, were characterized by a CD64dimCD115− phenotype and differentially expressed CD45RA (Supplementary Table 1 and Extended Data Fig. 5).
NCP1s and NCP2s independently differentiate into neutrophils
Because the acquisition of CD45RA conventionally represents a mandatory step for myeloid progenitor maturation that occurs at the GMP stage10, we investigated the maturation path of CD34+ and CD34dim/− NCPs in in vitro culture with SFGc. Assuming an NCP1 to NCP2 to NCP3 to NCP4 sequential progression, we found that NCP2s downregulated the expression of CD34, acquired an CD34dim/−CD45RA+ phenotype and then progressively decreased the expression of CD45RA to become NCP4s (Fig. 3a). NCP3s matured into NCP4s by progressively decreasing CD45RA expression (Fig. 3a), while NCP4s remained CD34− and CD45RA− throughout the entire 7 d of culture (Fig. 3a). NCP1s downregulated the expression of CD34 without upregulating CD45RA, and directly matured into NCP4s (Fig. 3a). As expected, the frequency of NCPs progressively decreased between 4 d and 7 d, in line with their maturation into CD66b+ cells (Fig. 3b), and NCP3s and NCP4s differentiated through kinetics remarkably faster than those of NCP1s and NCP2s (Fig. 3b). As such, although NCP1s and NCP2s shared a CD34+CD64dimCD115− phenotype, they maintained the differential expression of CD45RA until their maturation into NCP4s.
a, Differentiation of CD64dimCD115− NCPs based on the changes of CD34 and CD45RA expression during culture with SFGc for 1, 2, 4 and 7 d. Plots at day 0 show the phenotype of sorted NCPs, while plots relative to day 1 to day 7 show live, CD45+CD66b−CD14− cells (n = 3). b, Bar graphs depicting the percentages of live CD66b+ neutrophils generated by SFGc-treated NCPs. Data represent mean ± s.e.m. (n = 3). Analysis was performed using one-way ANOVA and Tukey post hoc test.
Because NCP1s did not directly develop into NCP2s, the acquisition of CD45RA by the neutrophil precursors, which occurred exclusively along the NCP2 maturation path, could occur before the acquisition of CD64, implying that neutrophil precursors more immature than NCP1s and NCP2s would differ in their CD45RA expression. Hence, we sorted SSCloCD45dimCD10−CD123dim/−CD34+CD45RA−CD64−CD115− cells (hereafter, CD45RA−CD64− subset) and CD64− GMDPs and cultured them with SFGc to test whether precursors contained within CD64− GMDPs (Extended Data Fig. 6a) gave rise to NCP2s, as well as whether the CD45RA−CD64− subset (Extended Data Fig. 6a) generated both NCP1s and CD64− GMDPs, and, in turn, NCP2s. We noticed that the CD45RA−CD64− subset concomitantly generated both CD34+CD45RA+ and CD34−CD45RA− populations following two maturation routes (Extended Data Fig. 6b). Conversely, CD64− GMDPs differentiated into CD34−CD45RA− cells through an intermediate CD34dim/−CD45RA+ stage, therefore never giving rise to CD34+CD45RA− cells (Extended Data Fig. 6c). Analysis of the culture-derived cells indicated that the CD45RA−CD64− subset restored both NCP1s and NCP2s (Extended Data Fig. 6b), while CD64− GMDPs generated, as expected, only NCP2s (Extended Data Fig. 6c). Importantly, the CD45RA−CD64− subset generated CD64− GMDPs as early as day 2 (Extended Data Fig. 6b), while NCP2s emerged only at day 4, thus confirming that NCP2s derive from CD64− GMDPs. In addition, both the CD45RA−CD64− subset and the CD64− GMDPs gave rise to both NCP3s and NCP4s as well as to PMNs and DCs/monocytes (not shown). These observations indicated that NCP1s and NCP2s differentiated through separate maturation routes and were derived from CD34+ progenitors that bifurcate into CD45RA+ and CD45RA− NCPs before acquiring CD64 (Extended Data Fig. 6d).
NCPs precede PMs along the neutrophil maturation cascade
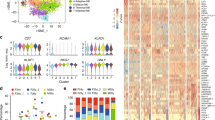
Next, we used RNA-sequencing (RNA-seq) to profile NCPs, HSCs/multipotent progenitors (MPPs), CMPs, PMs, MYs, MMs, BCs, SNs and PMNs. Both principal component analysis (PCA) (Fig. 4a) and hierarchical clustering analysis performed by optimal leaf ordering (Extended Data Fig. 7a) indicated that NCPs not only clustered by themselves but were also placed along the HSCs/MPPs and CMPs, PMs, MYs, MMs, BCs and SNs maturation trajectory, which ends in PMNs. Analysis of CD34 and PTPRC (which encodes CD45) transcript expression (Fig. 4b,c) corroborated the flow cytometry data. Furthermore, by performing k-means clustering of differentially expressed genes (DEGs), ten main gene groups (g1–g10) were identified among all samples (Fig. 4d and Supplementary Table 2). Genes encoding markers of immature cells (such as CD34, HOXA9, MYC, SOX4 and KIT), expressed only by HSCs/MPPs, CMPs and NCPs, were detected in g1 or g2 (Fig. 4d). Gene ontology (GO) analysis of the DEGs indicated the ‘MHC class II protein complex’ term was among the most significantly over-represented in g2 (Extended Data Fig. 7b), in line with the previously described expression of MHC class II in very immature neutrophil progenitors33. Similarly, DEGs mainly involved in ribosome assembly, mitochondria formation and cell cycle regulation, characterizing g3, g4 and g5, respectively, were expressed in NCPs, PMs, MYs and MMs, but not in nonproliferating BCs and SNs (Fig. 4d, Extended Data Fig. 7b,c and Supplementary Table 2).
a, PCA scatter plot based on the DEGs identified from bulk RNA-seq analyses of NCP1s (orange), NCP2s (green), NCP3s (magenta) and NCP4s (turquoise), as well as HSCs/MPPs (pink), CMPs (purple), PMs (salmon), MYs (blue violet), MMs (blue), BCs (light green), SNs (light blue) and PMNs (dark red) (n = 3–5). b,c, Schemes represent genome-browser views of RNA-seq signals at the CD34 (b) and PTPRC/CD45 (c) loci of NCPs. Exons 3 and 5 of PTPRC, whose transcription is essential for the expression of the CD45RA variants, are highlighted by red boxes in c. d, Heatmap displaying the expression patterns of the gene groups (g1–g10) resulting from the k-means analysis of DEGs identified among the various neutrophil-lineage cells and CMPs. The median gene expression levels of biological replicates were calculated, and data were represented as z score. e, Expression of antigenic MPO, ELANE, DEFA and LTF by immunocytochemical staining of NCPs, PMs and MYs/MMs. Three representative stained cells for each population are shown. Original magnification, ×600.
Importantly, g5 contained DEGs encoding messenger RNAs for the azurophilic granule (AG) proteins (such as MPO, AZU1, PRTN3 or ELANE), which are transcribed first during granulopoiesis, as well as transcription factors known to be typically expressed in immature neutrophils, such as GFI1 and CEBPE (ref. 34) (Fig. 4d). A more focused analysis revealed that AG genes started to be expressed in CMPs, progressively increased in NCP1s/NCP2s and NCP3s/NCP4s, were maximally transcribed in PMs and MYs, and then gradually disappeared (Extended Data Fig. 7c). Defensin genes such as DEFA1, DEFA1B, DEFA3 and DEFA4, which encode AG-stored proteins35, were very poorly expressed in NCPs, and included in g6 (Fig. 4d and Supplementary Table 2). Moreover, g6, but also g7 and g8, included other DEGs absent in NCPs, and were enriched for mRNAs encoding specific granule (SG) proteins (for example, LYZ, LTF, LCN2 and CEACAM8/CD66b), gelatinase granule (GG) proteins (for example, MMP9, ARG1 and CD177), secretory vesicle (SV) proteins (for example, MMP25 and ANXA3) and cell membrane (CM) proteins (for example, CXCR1, CXCR2 and ICAM3) (Fig. 4d and Extended Data Fig. 7c), and thus associated to GO terms such as ‘neutrophil degranulation’ and ‘neutrophil activation’ (Extended Data Fig. 7b). Furthermore, g9 and g10 were enriched in interferon-stimulated genes (ISGs), as well as other genes associated to the GO terms ‘response to bacterium’ and ‘cytokine production’, present in BCs, SNs and PMNs (Fig. 4d and Extended Data Fig. 7b). Consistent with the literature35, mRNAs encoding proteins involved in the production of reactive oxygen species, phagocytosis and chemotaxis (Extended Data Fig. 7c) were transcribed starting from the MM–BC stages and were highly expressed in PMNs.
To get more insights into the specific transcriptomic differences among the NCPs, we performed DEG analysis by using the likelihood ratio test36 and identified 1,114 DEGs. NCPs distinctly segregated from each other by PCA, indicating remarkable differences among their gene expression profiles (Extended Data Fig. 7d). However, while PC1 differences were mostly determined by cell cycle and AG genes, which were expressed more in NCP1s/NCP2s and NCP3s/NCP4s, respectively (Extended Data Fig. 7d), mRNAs that mostly contributed to the PC2 variations, namely PRG2, CLC, EPX and IL5RA, found in NCP1s, or IRF8, ANXA2, SAMHD1, SLAMF7, LYZ and F13A1, found in NCP2s and NCP3s (Extended Data Fig. 7d), are typically expressed in progenitors of eosinophils or monocytes, respectively. Immunocytochemistry analysis of cytospins from NCPs, PMs and MYs/MMs indicated that NCPs were myeloperoxidase (MPO)+ and that NCP3/NCP4s, but not NCP1/NCP2s, consistently expressed neutrophil elastase (ELANE) (Fig. 4e). Moreover, NCPs were defensin−, unlike PMs and MY/MM, with the latter also lactoferrin (LTF)+, as reported37, and confirming the RNA-seq data (Fig. 4d and Supplementary Table 2). These observations indicated that NCPs were committed to the neutrophil lineage, and that they are placed upstream of PMs along the neutrophil maturation cascade.
NCPs are upstream of eNePs, human proNeu1/2s and COVID-19 proNeus
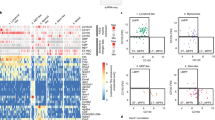
We next asked whether NCPs were upstream of the recently described Lin−CD66b+ eNePs24, Lin−CD66b+ proNeu1/2s (ref. 23) and other cells found in the blood of patients with COVID-19 (ref. 25) defined as proNeus, based on the constitutive expression of mRNAs encoding markers for proNeu1/2s (including CD15, CD43, CD63, CD66b, CD81)23. We compared publicly available RNA-seq datasets for eNePs and more mature precursors (N1 without eNeP, N2 and mixed N3/N4/N5)24, as well as of scRNA-seq datasets from COVID-19 proNeus and (more mature) preNeus25, with the gene expression profiles of NCPs by PCA (Fig. 5a). Human proNeu1/2 transcriptomes were not available23. eNePs clustered with PMs, while N1 without eNePs and N2 localized close to MYs and MMs, respectively (Fig. 5a). Moreover, in accordance with their more mature phenotypes, N3/N4/N5 grouped with BCs (Fig. 5a). Similarly, according to RNA-seq pseudo-bulk computing, COVID-19 proNeus and preNeus clustered before and after MMs, respectively (Fig. 5b). Examination of the mRNA accumulation encoding for molecules distinctively associated to discrete neutrophil maturation stages (Fig. 5b), indicated that only NCP1s and NCP2s expressed CD34 mRNA, while all NCPs, but not eNePs or COVID-19 proNeus, accumulated mRNAs associated to stemness, such as KIT, MYC and MHC class II genes. Moreover, NCPs, eNePs and COVID-19 proNeus, as well as PMs and MYs, expressed AG genes (that is, ELANE, PRTN3 and MPO), albeit at variable levels. By contrast, only eNePs and COVID-19 proNeus expressed PM-associated genes, including defensins and BPI (that is, AG proteins), or SLPI and CEACAM8/CD66b (that is, SG proteins). Finally, and in agreement with PCA results (Fig. 5a), only COVID-19 proNeus, but not NCPs or eNePs, expressed SG and GG genes, such as LCN2, LTF and ARG1, at levels comparable to MYs (Fig. 5b).
a, PCA scatter plot based on the top 500 most variable genes identified across: (1) bulk RNA-seq analyses of our HSCs/MPPs, CMPs, NCP1s, NCP2s, NCP3s, NCP4s, PMs, MYs, MMs, BCs, SNs and PMNs; (2) bulk RNA-seq analyses of eNePs, N1 without (w/o) eNeP, N2 and mixed N3/N4/N5 from the report by Dinh et al.24 (cyan); (3) pseudo-bulk RNA-seq analyses of COVID-19 proNeus and preNeus from the report by Schulte-Schrepping et al.25 (red). b, Heatmap displaying the expression grade of genes encoding for molecules distinctively associated to discrete neutrophil maturation stages by the cells represented in the PCA of panel a. Relative expression levels are represented as z score. c, Flow cytometry strategy for the identification of SSChiCD66b+Lin−CD71+CD117+ eNePs (red) within SSChiCD66b+Lin− N1s. d, Overlay plot of eNePs (red) with SSChiCD66b+Lin− neutrophils (gray). Cells were analyzed based on CD11b and CD16 expression. e, Overlay plot of NCPs (orange, green, magenta and turquoise) and eNePs (red). Cells were analyzed based on SSC parameter and CD66b expression. f, Histogram showing the expression of CD71, CD117, CD49d and CD66b by NCPs (orange, green, magenta and turquoise) and eNePs (red), as compared with the fluorescence-negative control (light gray). g, Flow cytometry strategy for the identification of CD66b+CD15+Lin−CD11b−CD49dhiSSClo proNeu1s (red) and CD66b+CD15+Lin−CD11b−CD49dintSSChi proNeu2s (black) within CD66b+CD15+Lin− proNeus. h, Histogram overlays showing the expression of CD66b, CD15 and CD34 by NCPs (orange, green, magenta and turquoise), proNeu1s (red) and proNeu2s (black), as compared with fluorescence-negative control (light gray). For panels c–h, one representative experiment out of three is shown.
Next, we reproduced the gating strategies used to identify eNePs24 and human proNeu1/2s (ref. 23) to directly compare their phenotypes with those of NCPs. eNePs were defined as a CD117+ subset of the Lin−CD66b+CD71+ N1 cells (Fig. 5c). However, according to their CD11b, CD16 and CD66b expression (Fig. 5d–f), as well as side scatter (SSC) properties (Fig. 5e), eNePs corresponded mostly to a more immature fraction of PMs24 (Fig. 5a). By contrast, NCPs were CD66b− and exclusively located in the SSClo fraction of immature BM cells (Fig. 5e,f and Extended Data Fig. 5). In addition, NCPs were positive for markers not specifically associated to neutrophil precursors26,38,39,40, namely CD71, CD117 and CD49d (Fig. 5f).
Because a detailed gating strategy for their identification and isolation, or a definitive phenotype, is not available for proNeus, we distinguished them as Lin−CD66b+CD15+CD11b−CD49dhiSSClo proNeu1s and Lin−CD66b+CD15+CD11b−CD49intSSChi proNeu2s (ref. 23). proNeu1/2s (Fig. 5g) differed from NCPs in terms of expression of CD66b, CD15 and CD34 (Fig. 5h), but not CD64 and CD115 (not shown). Differently from NCPs, proNeu1s and proNeu2s expressed CD66b at mid/low and mid/high levels, respectively (Fig. 5h). Similarly, while NCP1s and NCP2s were CD15− and NCP3/NCP4s were CD15dim/+, proNeu1/2s had a higher expression of CD15 than NCPs (Fig. 5h). Furthermore, we found that proNeu1s were CD34−, similarly to proNeu2s, NCP3s and NCP4s (Fig. 5h), in contrast with the Infinity Flow approach identifying proNeu1s as CD34+ (ref. 24). This phenotypic analysis supported the notion that proNeu1s consisted of highly immature PMs, similarly to eNePs, while proNeu2s corresponded to PMs. Together, these data suggested that NCPs were more immature than eNePs and COVID-19 proNeus at the molecular level, as well as than eNePs and proNeu1/2s at the phenotypic level, while COVID-19 proNeus were more mature than originally assumed25.
NCPs generate neutrophils in vivo
To investigate their contribution to granulopoiesis in vivo, NCPs were adoptively transferred into immunodeficient mice by different experimental approaches. In a first set of experiments, total NCPs were sorted and injected intravenously into AAVNOG mice. Human CD66b+ neutrophils were recovered in the peripheral blood (Fig. 6a), but not in the BM, of recipient AAVNOG mice 11 d after NCP transfer. In a second set of experiments, NCPs sorted from the BM of HLA-A2+ donors were transferred by intratibial injection into AAVHIR mice (that is, NOG mice engrafted with human HLA-A2−CD34+ cells, then treated with adeno-associated virus serotype 9 (AAV9) as before, and finally pre-conditioned with a nonmyeloablative dose of busulfan). CD66b+ neutrophils, although undetectable in the BM (Fig. 6b), represented the only human HLA-A2+ cells that were detected in the peripheral blood of recipient AAVHIR mice 14 d following NCP transfer (Fig. 6b–d). The remaining human cells consisted of HLA-A2− monocytes and B lymphocytes (Fig. 6b,c), which likely survived the busulfan conditioning. Altogether, these in vivo adoptive transfer experiments indicated the neutrophil-specific commitment of NCPs and supported their biological role in neutropoiesis.
a, Flow cytometry strategy for the identification of human SSChiCD66b+CD45+CD16+ neutrophils (green), by excluding SSChiCD66b+CD45hiCD16− eosinophils within the peripheral blood from AAVNOG mice, 11 d after intraocular injection of total NCPs. One representative mouse out of three with similar results is shown. b, Flow cytometry strategy for the identification of human SSChiCD66b+CD45+CD16+ neutrophils (green) in BM and peripheral blood of HLA-A2− AAVHIR mouse, 14 d following intratibial injection of total HLA-A2+ NCPs. CD14+HLA-DR+ monocytes (magenta) and CD14−HLA-DR+ B lymphocytes (light blue) are also detectable within the SSCloCD66b− region. One representative mouse out of six with similar results is shown. c, Histogram overlays showing the expression of HLA-A2 by monocytes (magenta), B lymphocytes (light blue) and neutrophils (green) identified within peripheral blood of HLA-A2− AAVHIR mouse, before (left) and after (right) HLA-A2+ NCP injection (n = 6). d, Bar graphs depicting the percentage of human HLA-A2+CD66b+ neutrophils (right) within total HLA-A2+ cells (left). Percentages were calculated by analyzing peripheral bloods of HLA-A2− AAVHIR mice, 14 d following intratibial injection of HLA-A2+ NCPs. Data represent mean ± s.e.m. (n = 6).
NCPs fall into multiple scRNA-seq cell clusters
To unequivocally dissect their transcriptional heterogeneity, we performed scRNA-seq in sorted NCP1s, NCP2s, NCP3s, NCP4s and cMoPs. Dimensionality reduction by Uniform Manifold Approximation and Projection (UMAP)41 on the 17,902 cells sequenced indicated that NCPs clearly segregate from cMoPs, while NCP1s mostly overlap with NCP2s and NCP3s with NCP4s (Fig. 7a and Extended Data Fig. 8a). Unbiased, graph-based clustering by Seurat42 identified nine discrete cell clusters (Fig. 7b), of which three (c7, c8 and c9) were excluded from all subsequent analyses because they consisted of eosinophil precursors (c7, identified by EPX mRNA expression), megakaryocyte/erythroid precursors (c8, identified by PF4 and HBB mRNA expression) or cells with an unclear ontogeny and high expression of apoptosis-related genes (c9) (Fig. 7b). Of the six remaining cell clusters, four were unequivocally attributable to the neutrophil lineage (c1, c2, c3 and c4), and two to the monocyte lineage (c5 and c6) (Fig. 7b). Such segregation was also confirmed by hierarchical clustering analysis of their DEGs (Extended Data Fig. 8b), as genes typical of the neutrophil (such as CEBPE, GFI1 or ELANE) and monocyte (such as IRF8, CSF1R or FLT3) lineages were exclusively expressed in c1–c4 and c5–c6, respectively (Fig. 7c). Moreover, distribution analysis of scRNA-seq cell clusters in NCPs and cMoPs evidenced that c1–c4 were completely absent in cMoPs (Fig. 7c), indicating that CD115 expression efficiently discriminates NCPs. By contrast, c5 cells were found to contaminate NCP2s and NCP3s, but not NCP1s and NCP4s (Fig. 7d), therefore explaining the PCA results on bulk RNA-seq (Extended Data Fig. 7d).
a, UMAP visualization of scRNA-seq profiles of NCP1s (red), NCP2s (orange), NCP3s (light blue), NCP4s (blue) and, for comparison, cMoPs (green), isolated from three different BMs. b, Identification of the clusters (c1–c9) in the UMAP shown in a, by Louvain clustering analysis. c, Violin plots showing the mRNA expression levels (as ln(UMI)) of neutrophil-lineage-specific genes (that is, CEBPE, GFI1 and ELANE) and monocyte lineage-specific genes (that is, CSF1R, IRF8 and FLT3), across c1–c6 cells. Cluster colors are the same of those shown in Fig. 7b. d, Stacked bar graph shows the relative abundances of c1–c6 cells in NCP1s, NCP2s, NCP3s, NCP4s and cMoPs.
The distribution of c1–c4 cells in NCP1s and NCP4s is specular to that of NCP2s and NCP3s, respectively (Fig. 8a). Moreover, while NCP1s and NCP2s were enriched mainly in c1 cells, NCP3s and NCP4s were preferentially accumulated in c3 and c4 cells (Fig. 8a). By contrast, c2 cells were equally distributed among the four NCPs (Fig. 8a). Calculation of the pseudotime value for every cell by Destiny43 (Fig. 8b), to define their maturation state, showed that c1 contained the more immature cells, while c3 and c4 contained relatively more mature cells (Fig. 8c). Accordingly, CD34 was preferentially expressed in c1 and c2 cells (Fig. 8d), consistent with their relatively more abundant presence in NCP1s and NCP2s (Fig. 8a). c2 cells, instead, displayed variable pseudotime values (Fig. 8c), in line with their presence in all NCPs (Fig. 8a). Seurat analysis of the DEGs in c1–c4 cells resulted in the identification of 136 genes (Fig. 8e and Supplementary Table 3). c1 cells had high expression of typical genes of immature proliferating cells (such as CD34, TOP2A, TUBB and HIST1H4C) (Fig. 8e and Extended Data Fig. 8c,d), consistent with their lowest pseudotime values (Fig. 8c). c1 cells also expressed genes associated to the ‘MHC class II protein complex’ GO term (Extended Data Fig. 8e), consistent with the g2 genes from bulk RNA-seq data (Extended Data Fig. 7a). By contrast, c3 and c4 cells had high expression of AG genes (Fig. 8e and Extended Data Fig. 8c,d), and displayed GO terms mostly enriched for ‘neutrophil activation’ and ‘neutrophil degranulation’ (Extended Data Fig. 8e), in accordance with their elevated pseudotime values (Fig. 8c), and their prevalent correspondence with NCP3s and NCP4s (Fig. 8a). Notably, c4 cells specifically expressed high mRNA levels of BEX1 (Fig. 8e and Extended Data Fig. 8c), while c2 cells had high expression of ISGs, such as ISG15, IFI6, IFIT3 and many more (Fig. 8e and Extended Data Fig. 8c), in line with the enrichment of the GO term ‘defense response to virus’ in this cluster (Extended Data Fig. 8e). No SG or GG genes were present in scRNA-seq datasets (Extended Data Fig. 8d), confirming the RNA-seq data. Finally, using Destiny43 to assess their potential developmental trajectories, we found that c1–c4 cells distributed along three branches. The c1 branch included the majority of c1 cells (Fig. 8f,g and Extended Data Fig. 8f), together with a minor quote of the c2, c3 and c4 cells, and branched into two different trajectories (Fig. 8f): one, defined as the ‘conventional trajectory’, was mainly characterized by cells expressing increased AG mRNA (c3 and c4 cells), but also included BEX1+ cells (c4 cells); the other, defined as the ‘ISG trajectory’ (Fig. 8f), was characterized by ISGhi cells (c2 cells) (Fig. 8h). As such, scRNA-seq clustering analysis identified four clusters of neutrophil precursor cells at different stages of maturation, distributed along two maturation routes, and indicated that NCP1s and NCP2s on one hand, and NCP3s and NCP4s on the other hand, were identical in terms of cluster distribution.
a, Stacked bar graph shows the relative abundance of c1–c4 cells in NCP1s, NCP2s, NCP3s and NCP4s, after exclusion of cells from monocyte progenitors (c5 and c6). b,c, UMAP (b) and violin (c) plots restricted to c1–c4 cells showing the pseudotime value for every cell, as calculated by the Destiny algorithm. d, Expression patterns of CD34 mRNA projected on a UMAP plot restricted to c1–c4 cells. e, Heatmap showing scaled expression of discriminative gene sets (Bonferroni-corrected P < 0.05; two-sided Wilcoxon Rank Sum test) by c1–c4 cells, as defined in Fig. 7b. The most specific genes of each cluster are depicted on the right y axis. The color scheme is based on z score distribution from −4 (purple) to 4 (yellow). f,g, Trajectory plots showing the distribution of c1–c4 cells (f), and their pseudotime values (g). In f, the arrows indicate both the ‘conventional’, and the ‘ISG’, developmental trajectories, along the neutrophil lineage. h, mRNA expression patterns of genes characteristic of c1 (that is, TOP2A), c2 (that is, IFI6), c3 (that is, CTSG) and c4 (that is, BEX1) cells, projected on the trajectory plots from f.
Discussion
In this study, we identified the phenotypes of four different CD64dimCD115− NCPs within the SSCloLin−CD45dim region of human BM. Based on their differential CD34 and CD45RA expression, NCPs were subdivided into CD34+CD45RA− NCP1s, CD34+CD45RA+ NCP2s, CD34dim/−CD45RA+ NCP3s and CD34dim/−CD45RA− NCP4s. Within cGMPs, we also defined CD64+CD115+ cMoPs (refs. 16,20), CD64−CD115+ MDPs and CD64−CD115− GMDPs (ref. 15). Because GMDPs generate neutrophils, but not eosinophils and basophils4,17,18,19, we propose to rename GMDPs as NMDPs (that is, neutrophil-monocyte-DC progenitors).
Our experiments uncovered that, while NCP1s originated from CD45RA− progenitors and directly matured into NCP4s, NCP2s developed from NMDPs and matured into NCP4s through NCP3s. Hence, our data not only confirmed that the transition of neutrophil precursors from HSCs to cGMPs was associated to their CD45RA upregulation10, but also uncovered the existence of a second developmental route, independent from CD45RA upregulation, which might be explained by the necessity to increase the number of peripheral neutrophils during emergency granulopoiesis. Alternatively, it could be that CD45RA may serve either for the localization of CD34+ and CD34dim/− NCPs in specific BM niches, or for a yet unknown function under pathological conditions.
Using transcriptomic dataset and phenotypic analysis comparisons, our data showed that NCP neutrophil precursors represent an earlier stage of differentiation compared with the recently described eNePs24, human proNeu1/2s (ref. 23) and COVID-19 proNeus25. In fact, clustering analysis of DEGs among CD34+ and CD34dim/− NCPs evidenced a remarkable enrichment of genes related to cell proliferation by NCP1s and NCP2s, as well as of AG-encoding genes by NCP3s and NCP4s.
scRNA-seq experiments uncovered that the CD34+ and CD34dim/− NCPs consisted of four cell clusters (c1–c4) distributed at substantially identical ratios in NCP1s and NCP2s, as well as in NCP3s and NCP4s. One of them (c2) was enriched in ISGs, consistent with the recent identification of PMN subsets expressing ISGs during health44,45 or pathological conditions25,46,47,48,49. Treatment of patients with severe COVID-19 with dexamethasone was reported to reduce the peripheral ISG+ neutrophil subset on the one hand, but to expand the subset of immature immunosuppressive cells on the other hand48, suggesting neutrophil polarization could be modulated by pharmacological interventions.
Clusters of neutrophil precursors displaying levels of maturation similar to c1–c4 cells have been identified by scRNA-seq of HSPCs from human BM, even though the sorting strategy for their isolation was not described. For instance, four clusters of neutrophil precursors (N0–N3) were found in CMPs (N0) and cGMPs (N1, N2 and N3)3. N1, N2 and N3 might correspond in part to the c1 cells given their expression of CD34 protein and AG mRNA. However, N3 could be ascribed to a monocyte progenitor due to its expression of LYZ, SAMHD1, CSF1R, ANXA2, KLF4 and IRF8 mRNA3, which we found to be peculiar to cMoPs. Similarly, it remains uncertain whether N0, which only expresses MPO among the AG genes3, corresponds to a neutrophil progenitor, because MPO can also be expressed by cMoPs9. However, none of these studies3,19 detected specific ISG- or BEX1-containing clusters (as we did), likely because of the much lower number of neutrophil precursors investigated compared with our scRNA-seq set.
In summary, we identified CD34+ and CD34dim/− NCPs within human BMs, all of them characterized by an SSCloLin−CD45dimCD64dimCD115− phenotype. As such, CD34+ and CD34dim/− NCPs were easily sortable. We expect that future work will uncover specific membrane markers that would allow the sorting of the cell clusters within the CD34+ and CD34dim/− NCPs, to allow an improved characterization and to evaluate if and how they expand under pathological conditions.
Methods
Isolation of cells from human BM and peripheral blood samples
BM samples were obtained from healthy donors (n = 35; Supplementary Table 4) selected for donation purposes according to the Italian Bone Marrow Donor Registry criteria and following informed, written consent. Fresh BM samples (1.5/2 ml) were collected under aseptic conditions in heparinized sterile syringes, processed using endotoxin-free polypropylene tubes (Greiner Bio-One) and subjected to density gradient centrifugation onto Ficoll-Paque (GE Healthcare). BM-LDCs were then collected, and either immediately processed for flow cytometry analysis, or suspended in αMEM growth medium (Corning) supplemented with 10% low-endotoxin FBS (<0.5 endotoxin unit (EU) ml−1), in the presence of penicillin/streptomycin (from now on termed ‘culture medium’)29, to be finally distributed in tissue-culture plates (Corning) and preincubated for 20 h at 37 °C in CO2 (ref. 20). Occasionally, fresh aliquots of BM-LDCs were frozen in 90% FBS (Sigma-Aldrich) plus 10% dimethylsulfoxide (Sigma-Aldrich) and stored in liquid nitrogen, since results obtained by using thawed or unfrozen BM-LDC samples were found fully comparable. Neutrophils from healthy donors were purified by the EasySep Human Neutrophil Enrichment Kit (Stem Cell Technologies; >99.7% purity) after centrifuging buffy coats onto Ficoll-Paque gradient density50. BM samples, obtained from three patients undergoing alloHSCT for hematological malignancies, were collected at day +21 after alloHSCT (when hematopoiesis is actively reconstituting from donor-derived HSCs). At this time-point, all three patients were in complete remission and had achieved full donor chimerism, according to variable number tandem repeats analysis (available from the authors). The study has been cleared by the Ethics Committee of the Azienda Ospedaliera Universitaria Integrata di Verona (Italy) (CMRI/55742).
Flow cytometry and FACS experiments
For flow cytometry experiments, BM-LDCs, sorted progenitor subpopulations or culture-derived cells were counted, resuspended in 50 µl of PBS buffer (Corning) plus 2% FBS and 2 mM EDTA (Sigma-Aldrich) (from now on termed ‘staining buffer’), and subsequently incubated for 10 min in the presence of 5% human serum (Sigma-Aldrich). Cells were then stained for 30 min on ice by fluorochrome-conjugated monoclonal antibodies (mAbs) for either 8-color MACSQuant10 Analyzer (Miltenyi Biotec), or 14-color LSRFortessa X-20 (BD) and MACSQuant16 Analyzer (Miltenyi Biotec) flow-cytometers, as listed in Supplementary Tables 5 and 6. Viability of both fresh and 20-h-preincubated BM-LDC samples was assessed by either propidium iodide (PI) (Sigma-Aldrich) or alternatively Sytox Blue (Invitrogen) exclusion, and reproducibly found >95%. The mean fluorescence intensity (MFI) relative to selected markers was obtained by subtracting either the MFI of the correspondent isotype control mAbs, or the cell autofluorescence (fluorescence minus one, FMO), to their fluorescence value. FlowJo software v.10.7.1 was used for data analysis. For FACS of the myeloid progenitors, 50–100 × 106 BM-LDCs were resuspended at 100 × 106 per ml and labeled for 45 min at 4 °C (in the dark) with the mAbs reported in Supplementary Table 7. Anti-CD14 and anti-IL5Rα mAbs were always included in the sorting panel to exclude, respectively, mature monocytes and eosinophil-committed progenitors. Conversely, we omitted the use of antiCD38 and antiCD117 antibodies since NCPs were found as CD38+ and CD117+. Cells were then washed and resuspended at 30 × 106 per ml in staining buffer, to be ultimately filtered through a 0.35-µM nylon mesh. Cells were finally sorted by using a FACSAria Fusion (BD) cell sorter equipped with 85-μm nozzle, immediately centrifuged, resuspended in αMEM medium, counted and used for experiments. Alternatively, sorted cells were lysed in RLT buffer (Qiagen) for RNA extraction51. Sorted cell populations displayed a >95% purity, as verified by flow cytometry analysis.
In vitro differentiation assay
To evaluate the differentiation potential of sorted BM progenitors, the latter cells were cultured on the MS-5 stromal cell line (ACC 441, Leibniz-Institut DSMZ), according to protocols previously described29. Briefly, the day before the coculture experiments, MS-5 cells cultured in αMEM medium at 95% confluence were incubated with 10 µg ml−1 mitomycin C (Sigma-Aldrich) for 3 h at 37 °C. Then, after treatment with 0.05% trypsin/EDTA (Corning), MS-5 cells were collected and resuspended in αMEM medium at 0.25 × 106 per ml, to be finally seeded in round-bottom 96-well tissue-culture plates for 24 h. Thus, 1–3 × 103 of the various sorted BM progenitors were resuspended in 100 μl of αMEM medium, seeded on top of MS-5 cells and incubated with 20 international units (IU) ml−1 Flt3L (Miltenyi Biotec), 10 IU ml−1 SCF (Miltenyi Biotec) and either 100 IU ml−1 GM-CSF (Peprotech)29 or, as in previous studies52,53, 6,500 IU ml−1 G-CSF (Myelostim, Italfarmaco Spa), to compose, respectively, the SFG29 or SFGc cocktail. Cells were then collected from the MS-5 cells starting at day 1, and up to day 14, of the culture, either for flow cytometry staining, or for functional studies. Expansion of purified progenitor populations is reported as mean fold-change of live, CD45+ output cells from the number of cells plated as input at time 0.
Morphological analysis of neutrophil precursors and culture-derived cell populations
Cytospins prepared from either sorted neutrophil precursors or culture-derived cell populations were stained by the May-Grunwald Giemsa procedure. Slides were analyzed by a Leica DM 6000 B microscope, equipped with a Leica DFC 300FX Digital Color Camera (Leica Microsystems).
O2 − production and phagocytosis assay
Either progenitor-derived neutrophils collected from NCPs cultured for 7 d, or freshly isolated healthy donor neutrophils, were washed and resuspended at 0.25 × 106 per ml in HBSS supplemented with 10% FBS, containing 1 mM CaCl2 and 5 mM glucose. O2− production in response to 20 ng ml−1 PMA (Sigma-Aldrich) was assessed by the Cytochrome C reduction assay54. To assess phagocytosis capacity, 0.025 × 106 cells per 100 µl were incubated in Eppendorf tubes at 37 °C, in the absence/presence of 20 µg ml−1 unopsonized zymosan particles. Cells were intermittently resuspended and, after 30 min, treated with an excess of cold HBSS and centrifuged to stop phagocytosis. Cytospin preparations of these samples were stained by the May-Grunwald Giemsa procedure, and slides analyzed by a Leica DM 6000 B microscope, equipped with a Leica DFC 300FX Digital Color Camera (Leica Microsystem).
Immunocytochemistry
Sorted cells were spotted on polarized slides and immediately fixed by 95% ethanol. Before the immunostaining procedure, slides were placed for 10 min in methanol containing 0.3% H2O2 for endogenous peroxidase quenching. Slides were rehydrated for 3 min in ethanol at 95 °C, and then washed in tap and distilled water. Slides were then washed in pH 7.4/7.6 Tris-HCl for 5 min before incubation for 2 h at room temperature with antibodies towards MPO (Rb polyclonal, dilution 1:5,000, A0398 Agilent Technologies), ELANE (1:200 dilution, clone NP57, Agilent Technologies), α-Defensins (DEFA; dilution 1:70, clone H-2, Santa Cruz Biotechnology) and LTF (Rb polyclonal, dilution 1:500, hpa059976, Sigma-Aldrich). Reactions were revealed by using the EnVision Detection System HRP, followed by diaminobenzidine for 5 min. Subsequently, slides were counterstained by Meyer’s hematoxylin for 10 min and coverslipped. Immunostained slides were photographed using a DP-73 Olympus digital camera, mounted on an Olympus BX60 microscope (×600 magnification, square side: 20 μm).
CFU assay
The CFU assay was performed by using the ‘complete Stem MACS HSC-CFU medium’ (containing 20 ng ml−1 G-CSF, 20 ng ml−1 GM-CSF, 20 ng ml−1 IL-3, 20 ng ml−1 IL-6 and 3 U ml−1 Epo, from Miltenyi Biotec), according to the manufacturer’s protocol.
Adoptive transfer experiments in mice
NOD.Cg-PrkdcscidIl2rgtm1Sug/JicTac (NOG) mice were purchased from Taconic Biosciences and maintained under specific-pathogen-free conditions at the Verona University animal facility. All animal experiments were approved by the local Ethics Committee (https://www.univr.it/it/cirsal), and conducted according to the guidelines of the Federation of European Laboratory Animal Science Associations. Animal experiments were performed according to national (protocol number 12722, approved by the Ministerial Decree Number 14/2012-B of 18 January 2012, and protocol number BR15/08, approved by the Ministerial Decree Number 925/2015-PR of 28 August 2015), as well as European, laws and regulations. All animals were housed in conditions of 22 °C and 45–65% humidity with 12-h light cycle. For adoptive transfer into NOG mice, animals (4-week-old females) were intramuscularly injected with 108 multiplicity of infection (MOI) of recombinant AAV9 vectors coding for human CSF2, CSF3, IL-3 and CXCL8, to favor the expansion of the myeloid compartment. In brief, cytokine complementary DNAs were purchased from GeneScript and AAV9 vectors were produced by Prof. M. Giacca’s laboratory (International Centre for Genetic Engineering and Biotechnology (ICGEB), Trieste and King’s College, London) by co-transfection of HEK293T cells (ACC 635, Leibniz-Institut DSMZ). After 2 weeks, mice were γ-irradiated (1.2 Gy), and 6 h later engrafted with a total of 105 pooled NCPs (to have enough cells) via intraocular injection. After 11 d, mice were euthanized, and BM and blood analyzed by FACS (3 out of 3 mice were successfully engrafted). For adoptive transfer into Human Immune Reconstituted (HIR) mice, animals (4-week-old females) were γ-irradiated (1.2 Gy) and, after 6 h, engrafted with 105 hHLA-A2−CD34+ cells (catalog no.: 4M-105, Lonza) via tail vein injection (intravenously). AAV9 vectors coding for human CSF2, CSF3, IL-3 and CXCL8 were intramuscularly administered at week 4 after engraftment, as already detailed. Hematopoietic engraftment was considered adequate when 25% of human CD45-expressing cells could be detected in peripheral blood of mice by FACS, according to the formula: frequency of hCD45+ cells/(frequency of hCD45+ + frequency of mCD45+ cells) × 100. At week 12 post-engraftment, mice were treated with a single nonmyeloablative dose of busulfan (10 mg kg−1) and, 24 h later, animals received an intratibial injection of a total of 1.5 × 104 pooled HLA-A2+ NCPs. After 14 d, mice were euthanized, and BM and blood were analyzed by FACS (6 out of 6 mice were successfully engrafted by HLA-A2+ NCPs).
RNA-seq
Total RNA was extracted by the RNeasy Mini Kit (Qiagen) after cell lysis55. Libraries for transcriptome analysis were prepared using the Smart-seq2 protocol56. Briefly, 2 ng of total RNA was copied into a first-strand cDNA by reverse transcription and template-switching, using oligo (dT) primers (5′–AAGCAGTGGTATCAACGCAGAGTACT30VN-3′, from Thermo Fisher) and a locked nucleic acid (LNA)-containing template-switching oligo (5′-AAGCAGTGGTATCAACGCAGAGTACATrGrG+G-3′, from Exicon). The resulting cDNA was pre-amplified, purified and tagmented with Tn5 transposase. cDNA fragments generated after tagmentation were gap-repaired, enriched by PCR and purified to create the final cDNA library. Libraries were sequenced on the Illumina NextSeq 500 in single-read mode (1 × 75 cycles) at the Centro Piattaforme Tecnologiche of the University of Verona.
scRNA-seq
Sorted cells were labeled by using the BD Single-Cell Multiplexing Kit (BD Biosciences), strictly following the manufacturer’s protocol. Such a procedure allowed us to combine five samples (NCP1s, NCP2s, NCP3s, NCP4s and cMoPs) into a single pool. Each sample was washed twice in FACS buffer and resuspended in cold BD Sample Buffer (BD Biosciences). Only samples with high viability (>85%, as evaluated by the BD Rhapsody scanner) were used for sequencing. Samples from the same donor were pooled in equal amounts to achieve approximately 15,000 cells in 620 µl, and loaded onto a BD Rhapsody cartridge for an incubation of 20 min at room temperature. Then, Cell Capture Beads (BD Biosciences) were added to the cartridge and incubated at room temperature for 3 min, and thereafter cartridges were washed. Cells were then lysed, and the released mRNAs were captured by Cell Capture Beads. mRNAs were then retrieved, to be washed before performing reverse transcription and treatment with Exonuclease I. cDNA libraries were prepared by using the BD Rhapsody Whole Transcriptome Analysis Amplification kit and BD Single-Cell Multiplexing kit (BD Biosciences). Size-distribution (quality) of final libraries was assessed by Agilent 2200 TapeStation with High Sensitivity D5000 ScreenTape and quantified using a Qubit Fluorometer using the Qubit dsDNA HS Kit (Thermo Fisher, no. Q32854). Sequencing was performed in paired-end mode (2 × 75 cycles) on a NextSeq 500 System (Illumina). This procedure was utilized for three different BMs, whose data were integrated as outlined below.
RNA-seq computational analysis
Computational analysis of transcriptome datasets generated by Smart-seq2 was performed by using the bioinformatic pipeline, as previously described57. Briefly, after quality filtering, according to the Illumina pipeline, removal of contaminant adapters and base quality trimming were performed using Trim Galore! (http://www.bioinformatics.babraham.ac.uk/projects/trim_galore/) script. Gene counts were normalized among various samples using DESeq2 (ref. 36), and only genes coding for protein and long noncoding RNA were retained for downstream analysis. DEGs were identified using DESeq2, by using as selection parameter adjusted P value lower than 0.01 and likelihood ratio test36. Batch effects were removed using the limma package’s ‘removeBatchEffect’ function before performing PCA. PCA was performed on DEGs by using Bioconductor/R package pcaExplorer v.2.10.0.
Computational inference of developmental path
Hierarchical clustering was performed using the Euclidean distance and Ward aggregation as criteria. To find a suitable linear order within the hierarchical clustering dendrogram (HSCs/MPPs, CMPs, NCPs, PMs, MYs, MMs, BCs, SCs and mature blood neutrophils), we used the optimal leaf ordering seriation method of R package seriation, v.1.2-9 (ref. 58).
Seven Bridges processing for scRNA-seq data
After demultiplexing of bcl files by using Bcl2fastq2 v.2.20 from Illumina and assessment of reads quality, paired-end scRNA-seq reads were then filtered for valid cell barcodes using the barcode whitelist provided by BD. Then, sequenced reads were aligned to the hg38 human transcriptome and the expression of transcripts in each cell was quantified via the standard Rhapsody analysis pipeline (BD Biosciences) on Seven Bridges (https://www.sevenbridges.com), following manufacturer’s recommendations.
Seurat workflow for scRNA-seq data analysis
The R package Seurat59 was utilized for all downstream analysis. For each single-cell dataset, the number of detected genes, the number of unique molecular identifiers (UMIs), as well as the fraction of UMIs corresponding to mitochondrial features, which altogether reflect the transcriptome quality of each cell, were calculated. Only cells that transcribed at least 200 genes, and only genes that were expressed in at least 10 cells, were included in the analysis. In sum, 15,969 cells and 17,398 genes were obtained. Next, cells with more than 25% mitochondrial transcripts and with a number of genes above the 99th percentile or below the 1st percentile were removed for downstream analysis. Therefore, after such a rigorous quality control, a total of 15,057 cells were analyzed (4,465, 6,958 and 3,634 from BMs of, respectively, donor 1, donor 2 and donor 3). To examine cell cycle variation in our data, by using CellCycleScoring function, we assigned a score to each cell, based on its expression of G2/M and S phase markers. To remove batch effects across data from different donors, we performed dataset integration using SCTransform integration workflow (https://satijalab.org/seurat/v3.2/integration.html). Normalization and detection of highly variable genes were performed using the difference between the S and G2M cell cycle scores and the percentage of mitochondrial UMIs as covariates. To identify the integration of anchor genes among the three datasets from different BMs, the FindIntegrationAnchors() function was used applying default parameters. Using Seurat’s IntegrateData(), samples were combined into one object. These ‘integrated’ batch-corrected values were then set as the ‘default assay’, and gene expression values were scaled before running PCA. The dimensional reduction of the integrated dataset was computed by summarizing the first 50 principal components and visualized in a two-dimensional UMAP representation. Clustering was conducted using the FindNeighbors() and FindClusters() functions using the same 50 principal components, and a resolution parameter set to 0.3. Differential expression tests were performed using the FindAllMarkers() function. DEGs were identified using the nonparametrical Wilcoxon rank sum test, based on normalized data. P value adjustment was performed using Bonferroni correction based on the total number of genes in the dataset. Genes with >0.25 log-fold changes, at least 25% expressed in tested groups and Bonferroni-corrected P < 0.01 were considered as significant DEGs. Euclidean distance between clusters was calculated on DEGs and hierarchical clustering was performed in R, using the ‘ward.D’ linkage clustering. The average gene expression of clusters was calculated using the function AverageExpression().
GO of DEGs from bulk RNA-seq and scRNA-seq
GO analysis was performed on DEGs from bulk RNA-seq and scRNA-seq for the cellular component, biological process and molecular function ontology domains by using the Bioconductor/R package clusterProfiler (v.3.14.3)60.
Analytic comparison among transcriptomes obtained from NCPs, eNePs and COVID-19 proNeus
RNA-seq datasets from eNePs and other more mature neutrophil precursors (that is, N1 without eNeP, N2 and mixed N3/N4/N5)24 were analyzed exactly as our bulk RNA-seq analyses. For COVID-19 proNeus and preNeus25, cells categorized as ‘pre-Neutrophils’ and ‘pro-Neutrophils’ (that is, clusters 6 and 8, respectively, from Fig. 7b of Schulte-Schrepping et al. paper25) were retrieved from scRNA-seq datasets and then normalized to count per million, by using the NormalizeData() function of Seurat R packages and applying the following settings: ‘normalization.method = RC’ and ‘scale.factor = 1e6’. Subsequently, pseudo-bulk RNA-seq analyses of proNeus and preNeus from selected donors (‘BN-12’, ‘BN-18’, ‘BN-19’) were computed by evaluating the mean expression for each gene. Each of the two datasets24,25 was merged with ours, and only genes present in all three datasets were included in the analysis and transformed to log2 pseudo-count values for downstream assays.
Trajectory analysis and pseudotime calculation
Trajectory analysis was performed by using the destiny algorithm v.3.01 (ref. 43). In brief, the neutrophil precursor space was subsetted and the diffusion map was calculated based on the top 2,000 variable genes with a sum of at least 10 counts over all cells. Based on the diffusion map, a diffusion pseudotime was calculated to infer a transition probability between the different cell states of the neutrophils.
Statistical analysis
Data are expressed as mean ± s.d. or s.e.m. of the number of indicated experiments. Where applicable, normality distribution was estimated using the D’Agostino–Pearson or Shapiro–Wilk normality test. Statistical evaluation for normally distributed data was performed by using one-way or two-way analysis of variance (ANOVA), followed by Tukey’s or Bonferroni’s post hoc test, respectively. Non-normally distributed data were assessed with Mann–Whitney test, or, for multiple group comparison, with Kruskal–Wallis or Friedman test followed by Dunn’s multiple comparison test. The tests used are indicated in the respective figure legends. Values of P < 0.05 were considered statistically significant. Statistical analysis was performed by GraphPad Prism v.7.0 software. No randomization was performed in experiments with BM cells since this was not relevant. Samples were allocated into experimental groups based on gating strategy. In animal experiments, mice were randomly assigned to each group. No statistical methods were used to predetermine sample sizes, but our sample sizes are similar to those reported in previous publications24,44,45
Reporting Summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Raw datasets have been submitted to the Gene Expression Omnibus database (http://www.ncbi.nlm.nih.gov/geo/) and are available under the accession number GSE164687. Bulk RNA-seq datasets of eNePs and HSCs/CMPs were downloaded from GSE157103 and GSE113182, respectively. COVID-19 proNeus and preNeus scRNA-seq datasets were downloaded from https://www.fastgenomics.org. Source data are provided with this paper.
Code availability
R scripts for data processing are available through https://github.com/fbianchetto/Neutrophil-Committed-Progenitors.
References
Notta, F. et al. Distinct routes of lineage development reshape the human blood hierarchy across ontogeny. Science 351, aab2116 (2016).
Paul, F. et al. Transcriptional heterogeneity and lineage commitment in myeloid progenitors. Cell 163, 1663–1677 (2015).
Velten, L. et al. Human haematopoietic stem cell lineage commitment is a continuous process. Nat. Cell Biol. 19, 271–281 (2017).
Pellin, D. et al. A comprehensive single cell transcriptional landscape of human hematopoietic progenitors. Nat. Commun. 10, 2395 (2019).
Yanez, A. et al. Granulocyte-monocyte progenitors and monocyte-dendritic cell progenitors independently produce functionally distinct monocytes. Immunity 47, 890–902.e4 (2017).
Zheng, S. et al. Molecular transitions in early progenitors during human cord blood hematopoiesis. Mol. Syst. Biol. 14, e8041 (2018).
Karamitros, D. et al. Single-cell analysis reveals the continuum of human lympho-myeloid progenitor cells. Nat. Immunol. 19, 85–97 (2018).
Weinreb, C., Rodriguez-Fraticelli, A., Camargo, F. D. & Klein, A. M. Lineage tracing on transcriptional landscapes links state to fate during differentiation. Science 367, eaaw3381 (2020).
Muench, D. E. et al. Mouse models of neutropenia reveal progenitor-stage-specific defects. Nature 582, 109–114 (2020).
Doulatov, S., Notta, F., Laurenti, E. & Dick, J. E. Hematopoiesis: a human perspective. Cell Stem Cell 10, 120–136 (2012).
Watcham, S., Kucinski, I. & Gottgens, B. New insights into hematopoietic differentiation landscapes from single-cell RNA sequencing. Blood 133, 1415–1426 (2019).
Jacobsen, S. E. W. & Nerlov, C. Haematopoiesis in the era of advanced single-cell technologies. Nat. Cell Biol. 21, 2–28 (2019).
Manz, M. G., Miyamoto, T., Akashi, K. & Weissman, I. L. Prospective isolation of human clonogenic common myeloid progenitors. Proc. Natl Acad. Sci. USA 99, 11872–11877 (2002).
Doulatov, S. et al. Revised map of the human progenitor hierarchy shows the origin of macrophages and dendritic cells in early lymphoid development. Nat. Immunol. 11, 585–593 (2010).
Lee, J. et al. Restricted dendritic cell and monocyte progenitors in human cord blood and bone marrow. J. Exp. Med. 212, 385–399 (2015).
Kawamura, S. et al. Identification of a human clonogenic progenitor with strict monocyte differentiation potential: a counterpart of mouse cMoPs. Immunity 46, 835–848.e4 (2017).
Mori, Y. et al. Identification of the human eosinophil lineage-committed progenitor: revision of phenotypic definition of the human common myeloid progenitor. J. Exp. Med. 206, 183–193 (2009).
Gorgens, A. et al. Revision of the human hematopoietic tree: granulocyte subtypes derive from distinct hematopoietic lineages. Cell Rep. 3, 1539–1552 (2013).
Drissen, R., Thongjuea, S., Theilgaard-Monch, K. & Nerlov, C. Identification of two distinct pathways of human myelopoiesis. Sci. Immunol. 4, eaau7148 (2019).
Olweus, J., Thompson, P. A. & Lund-Johansen, F. Granulocytic and monocytic differentiation of CD34hi cells is associated with distinct changes in the expression of the PU.1-regulated molecules, CD64 and macrophage colony-stimulating factor receptor. Blood 88, 3741–3754 (1996).
Evrard, M. et al. Developmental analysis of bone marrow neutrophils reveals populations specialized in expansion, trafficking, and effector functions. Immunity 48, 364–379.e8 (2018).
Zhu, Y. P. et al. Identification of an early unipotent neutrophil progenitor with pro-tumoral activity in mouse and human bone marrow. Cell Rep. 24, 2329–2341.e8 (2018).
Kwok, I. et al. Combinatorial single-cell analyses of granulocyte-monocyte progenitor heterogeneity reveals an early uni-potent neutrophil progenitor. Immunity 53, 303–318.e5 (2020).
Dinh, H. Q. et al. Coexpression of CD71 and CD117 identifies an early unipotent neutrophil progenitor population in human bone marrow. Immunity 53, 319–334.e6 (2020).
Schulte-Schrepping, J. et al. Severe COVID-19 is marked by a dysregulated myeloid cell compartment. Cell 182, 1419–1440.e23 (2020).
van Lochem, E. G. et al. Immunophenotypic differentiation patterns of normal hematopoiesis in human bone marrow: reference patterns for age-related changes and disease-induced shifts. Cytom. B Clin. Cytom. 60, 1–13 (2004).
Gorczyca, W. et al. Immunophenotypic pattern of myeloid populations by flow cytometry analysis. Methods Cell. Biol. 103, 221–266 (2011).
Olweus, J. et al. Dendritic cell ontogeny: a human dendritic cell lineage of myeloid origin. Proc. Natl Acad. Sci. USA 94, 12551–12556 (1997).
Breton, G., Lee, J., Liu, K. & Nussenzweig, M. C. Defining human dendritic cell progenitors by multiparametric flow cytometry. Nat. Protoc. 10, 1407–1422 (2015).
Villani, A. C. et al. Single-cell RNA-seq reveals new types of human blood dendritic cells, monocytes, and progenitors. Science 356, eaah4573 (2017).
See, P. et al. Mapping the human DC lineage through the integration of high-dimensional techniques. Science 356, eaag3009 (2017).
Lee, J. et al. Lineage specification of human dendritic cells is marked by IRF8 expression in hematopoietic stem cells and multipotent progenitors. Nat. Immunol. 18, 877–888 (2017).
Olweus, J., Lund-Johansen, F. & Terstappen, L. W. Expression of cell surface markers during differentiation of CD34+, CD38−/lo fetal and adult bone marrow cells. Immunomethods 5, 179–188 (1994).
Cowland, J. B. & Borregaard, N. Granulopoiesis and granules of human neutrophils. Immunol. Rev. 273, 11–28 (2016).
Cassatella, M. A., Ostberg, N. K., Tamassia, N. & Soehnlein, O. Biological roles of neutrophil-derived granule proteins and cytokines. Trends Immunol. 40, 648–664 (2019).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Mora-Jensen, H. et al. Technical advance: immunophenotypical characterization of human neutrophil differentiation. J. Leukoc. Biol. 90, 629–634 (2011).
Elghetany, M. T. Surface antigen changes during normal neutrophilic development: a critical review. Blood Cells Mol. Dis. 28, 260–274 (2002).
Olweus, J., Lund-Johansen, F. & Terstappen, L. W. CD64/Fc gamma RI is a granulo-monocytic lineage marker on CD34+ hematopoietic progenitor cells. Blood 85, 2402–2413 (1995).
Gross, S. et al. Characterization and phenotypic analysis of differentiating CD34+ human bone marrow cells in liquid culture. Eur. J. Haematol. 59, 318–326 (1997).
Becht, E. et al. Dimensionality reduction for visualizing single-cell data using UMAP. Nat. Biotechnol. 37, 38–44 (2018).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Angerer, P. et al. destiny: diffusion maps for large-scale single-cell data in R. Bioinformatics 32, 1241–1243 (2016).
Xie, X. et al. Single-cell transcriptome profiling reveals neutrophil heterogeneity in homeostasis and infection. Nat. Immunol. 21, 1119–1133 (2020).
Gupta, S. et al. Sex differences in neutrophil biology modulate response to type I interferons and immunometabolism. Proc. Natl Acad. Sci. USA 117, 16481–16491 (2020).
Zilionis, R. et al. Single-cell transcriptomics of human and mouse lung cancers reveals conserved myeloid populations across individuals and species. Immunity 50, 1317–1334.e1310 (2019).
Mistry, P. et al. Transcriptomic, epigenetic, and functional analyses implicate neutrophil diversity in the pathogenesis of systemic lupus erythematosus. Proc. Natl Acad. Sci. USA 116, 25222–25228 (2019).
Sinha, S. et al. Dexamethasone modulates immature neutrophils and interferon programming in severe COVID-19. Nat. Med. 28, 201–211 (2022).
Bost, P. et al. Deciphering the state of immune silence in fatal COVID-19 patients. Nat. Commun. 12, 1428 (2021).
Calzetti, F. et al. The importance of being “pure” neutrophils. J. Allergy Clin. Immunol. 139, 352–355.e6 (2017).
Zimmermann, M. et al. IFNɑ enhances the production of IL-6 by human neutrophils activated via TLR8. Sci. Rep. 6, 19674 (2016).
Tura, O. et al. Optimal ex vivo expansion of neutrophils from PBSC CD34+ cells by a combination of SCF, Flt3-L and G-CSF and its inhibition by further addition of TPO. J. Transl. Med. 5, 53 (2007).
Jie, Z. et al. Large-scale ex vivo generation of human neutrophils from cord blood CD34+ cells. PLoS ONE 12, e0180832 (2017).
Serra, M. C., Calzetti, F., Ceska, M. & Cassatella, M. A. Effect of substance P on superoxide anion and IL-8 production by human PMNL. Immunology 82, 63–69 (1994).
Tamassia, N. et al. The MyD88-independent pathway is not mobilized in human neutrophils stimulated via TLR4. J. Immunol. 178, 7344–7356 (2007).
Picelli, S. et al. Full-length RNA-seq from single cells using Smart-seq2. Nat. Protoc. 9, 171–181 (2014).
Bianchetto-Aguilera, F. et al. Deciphering the fate of slan+-monocytes in human tonsils by gene expression profiling. FASEB J. 34, 9269–9284 (2020).
Hahsler, M., Hornik, K. & Buchta, C. Getting things in order: an introduction to the R package seriation. J. Stat. Softw. 25, 1–34 (2008).
Butler, A. et al. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Acknowledgements
This work was supported by grants to M.A.C. (from AIRC grant no. IG-20339; MIUR-PRIN grant no. 20177J4E75_004; and Fondazione Cariverona) and to N.T. (grant no. GR-2016-02361263). We thank P. Mazzi for preparing cytospins, E. Pietronigro for slide acquisition, S. Pasqualato (European Institute of Oncology, Milan, Italy) for providing Tn5 transposase and A. Carraro (Centro Trapianti Fegato, AOUI Verona, Italy) and V. Bergamini (UOC Ostetricia e Ginecologia B, AOUI Verona, Italy) for providing spleen and cord blood samples. We thank G. Zini (University Cattolica, Hematology Department, Rome) for her professional consultancy about NCP morphology. Centro Piattaforme Tecnologiche (CPT) of the University of Verona has been instrumental for the access to the flow cytometry/cell analysis and genomic/transcriptomic platforms.
Author information
Authors and Affiliations
Contributions
F.C. and G.F. performed and analyzed flow cytometry, cell sorting and in vitro differentiation experiments. C.C. and A.M. were involved in cell sorting experiments. N.T., M.C. and S.G. prepared samples for RNA-seq and scRNA-seq. N.T. and F.B.-A. analyzed RNA-seq and scRNA-seq data. F.C. and I.S. performed CFU assay. S.L. and W.V. performed and analyzed ICC. F.C., G.F., S.C., S.U., A.M. and V.B. were involved in adoptive transfer experiments. F.B., M.B. and C.T. provided patient samples. F.C., G.F., N.T., P.S. and M.A.C. designed and wrote the manuscript. M.A.C. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks Pierre Guermonprez and Hongbo Luo for their contribution to the peer review of this work. Ioana Visan was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Gating strategy to identify lineage-positive cells and immature SSCloCD45dim myeloid/lymphoid progenitors within BM-LDCs.
a, Flow cytometry workflow for the identification of mature leukocyte populations and SSCloCD45dim immature myeloid/lymphoid progenitors within BM-LDCs. Mature cell populations were identified as: SSChiCD66b+CD16−CD45hi eosinophils (pink); SSChiCD66b+CD16-/+CD45+ neutrophils (green); SSCloCD141+CLEC9A+ cDC1s (magenta); SSCloFcεRI+CD1c− basophils (dark red); SSCloFcεRIdimCD1c+ cDC2s (yellow); SSCloCD45RA+CD33+CD123+CD303+ pDCs (green); SSCloCD3−CD19−CD56+ NK cells (light blue); SSCloCD3−CD19−CD56−CD14+CD16−/CD14+/-CD16+/- total monocytes (light blue); SSCloCD3+CD19− T cells (brown); SSCloCD3−CD19+ B cells (orange). Immature myeloid/lymphoid progenitors were identified as SSCloCD45dim cells (purple). b, Dot plot overlays of lineage-positive/mature cells and SSCloCD45dim immature progenitors.
Extended Data Fig. 2 Biological features of CD34+CD45RA+CD64dimCD115− cells, cMoPs and MDPs prior to, and after treatment with, either SFGc, or SFG.
a, Bar graphs depicting the percentage of PI−, live cells generated by CD34+CD45RA+CD64dimCD115−cells (n = 8), cMoPs (n = 7) and MDPs (n = 4) treated with SFGc for 7 days. Data represent means ± s.e.m. b, Bar graphs showing the fold expansion of CD34+CD45RA+CD64dimCD115−cells (n = 8), cMoPs (n = 6) and MDPs (n = 4) treated with SFGc for 7 days. Data represent means ± s.e.m. Analysis was performed by using Kruskal-Wallis test, Dunn’s post-hoc test. c, Representative histogram overlays showing the expression of CD114 by CD34+CD45RA+CD64dimCD115−cells (green), cMoPs (light blue) and MDP (brown) populations. Gray profile corresponds to isotype control (n = 3). (d) Bar graphs reporting the percentages of neutrophils (green), cDC1s (magenta), pDCs/preDCs/DC5s (orange), monocytes (light blue) and cDC2s (pink) generated by CD34+CD45RA+CD64dimCD115− cells (n = 8), cMoPs (n = 9) and MDPs (n = 6) treated with SFG for 7 days. Data represent means ± s.e.m.
Extended Data Fig. 3 Reconstitution of NCPs and mature neutrophils in BMs of allogeneic HSC-transplanted patients.
Flow cytometry strategy for the identification of reconstituting neutrophils (green), eosinophils (pink), NCP1s (orange), NCP2s (green), NCP3s (magenta), NCP4s (turquoise), cMoPs (light blue), MDPs (brown) and pre-monocytes (black) within BM-LDCs of reconstituting BM (at day +21) from a patient undergoing allogeneic hematopoietic stem cell transplantation (alloHSCT) (n = 3).
Extended Data Fig. 4 Functionality of neutrophils derived from NCPs, as well as CD15 expression by both neutrophil and monocyte precursors among BM-LDCs and their mature counterparts.
a, Histogram plot showing the percentages and the type of colonies generated after 14 days of NCP and BM-LDC culture (n = 3). Data represent means ± s.e.m. b, Representative plot showing the time-course of O2− production by neutrophils derived from NCPs cultured for 7 days with SFGc (orange, green, magenta and turquoise) as compared to blood neutrophils (red) (n = 3). c, Representative cytospins showing the phagocytosis of zymosan particles by neutrophils derived from NCPs cultured for 7 days with SFGc (n = 3). d,e, Histogram overlays showing the expression of CD15 by NCPs (orange, green, magenta and turquoise), PMs (purple), SNs (black), cMoPs (light blue), pre-monocytes (blue) and CD14+ monocytes (red) as compared to the fluorescence-negative control (light gray) (n = 5).
Extended Data Fig. 5 14-color flow cytometry antibody panel to identify NCPs, monocyte/DC progenitors and immature neutrophil populations in human BMs.
a, Flow cytometry workflow (Supplementary Table 6) showing the gating strategy to identify myeloid progenitors within the SSCloCD66b−Lin(CD3/CD19/CD1c/CD141)-CD14−CD16−CD56−CD45dim region of BM-LDCs (dark red): CD34+CD45RA+ cGMPs, CD34+CD45RA+CD123+CD115− CDPs (yellow), CD34+CD45RA+CD123dim/-CD64dimCD115− NCP2s (green), CD34+CD45RA+CD123dim/-CD64+CD115+ cMoPs (light blue), CD34+CD45RA+CD123dim/-CD64−CD115+MDPs (brown), CD34+CD45RA−CD123dim/-CD64dimCD115− NCP1s (orange), CD34dim/-CD45RA+CD123dim/-CD64dimCD115− NCP3s (magenta), CD34dim/-CD45RA+CD123dim/-CD64++CD115+ pre-monocytes (blue), CD34dim/-CD45RA-CD123dim/-CD64dimCD115− NCP4s (turquoise). The same flow cytometry workflow identify SSChiCD66b+CD16−CD45hi eosinophils (black) as well as all the neutrophil precursors within the SSChiCD66b+Lin(CD3/CD19/CD1c/CD141)- region (green): CD66b+CD10−CD11b−CD16−PMs (grey), CD66b+CD10−CD11bdim/+CD16−MYs (light red), CD66b+CD10−CD11b+CD16+ MMs (purple), CD66b+CD10−CD11b+CD16++CD10− BCs (light yellow), and CD66b+CD11b+CD16++CD10+ SNs (pink). b, Dot plot overlays depicting the phenotype variation (black arrows) of the populations composing the neutrophil maturation cascade in terms of SSC parameter and CD66b, CD15 and CD45 marker modulation.
Extended Data Fig. 6 NCP1s and NCP2s derive from phenotypically distinct CD34+ progenitors.
a, Flow cytometry strategy for the identification of SSCloCD45dimCD10−CD123dim/-CD34+CD45RA−CD64− subset (CD45RA−CD64− subset, pink) and SSCloCD45dimCD10−CD123dim/-CD38+CD34+CD45RA+CD64−CD115−GMDPs (CD64−GMDPs, dark blue). b,c, Plots showing the differentiation potential of CD45RA−CD64− subset (b) and CD64−GMDPs (c) based on the changes of CD34 and CD45RA, as well as CD64 and CD115, expression by the generated cells during culture with SFGc for 2, 4 or 7 days (n = 3). d Hypothetic model of myeloid cell ontogeny according to our results. The scheme shows that the acquisition of CD45RA represents a very premature event (that occurs prior to that of CD64) along the maturation trajectories of early progenitors of neutrophils, monocytes and DCs, occurring from the multilineage CD34+CD45RA−CD64−CD115−progenitor subset (including HSCs, CMPs, MEPs) into transitional multilineage progenitor pools. The latter pools include GMDPs (also named as neutrophil-, monocyte- and DC-committed progenitors, NMDPs), MDPs and CDPs, that subsequently mature into uni-lineage precursors, including NCP2s/NCP3s and cMoPs/pre-monocytes. The scheme also shows that the acquisition of CD45RA does not occur in those progenitors present within CD34+CD45RA−CD64−CD115− subset that directly upregulates CD64 expression and generates NCP1s. As shown in the scheme, NCP1s directly differentiate into NCP4s, while NCP2s originate NCP4s via NCP3s.
Extended Data Fig. 7 Characterization of NCP transcriptomes as determined by RNA-seq.
a, Developmental path of NCP1s, NCP2s, NCP3s and NCP4s, as well as HSCs/MPPs, CMPs, PMs, MYs, MMs, BCs, SNs and mature neutrophils (PMN), computationally determined from bulk RNA-seq datasets by using the optimal leaf ordering (OLO) algorithm. b, GO terms enriched by genes associated with the ten gene groups (g1-g10) identified by K-means analysis, as shown in Fig. 4d. The top five GO terms with Benjamini-Hochberg-corrected P values <0.05 (one-sided Fisher’s exact test) are shown for every gene group. ‘Gene ratio’ indicate the fraction of DEGs present in the given GO term. (c), Box plots showing the distribution of mRNA expression levels [as log2(FPKM + 1)] for genes associated to cell cycle, AG, SG, GG, SV and GM, as well as ROS biosynthetic process, phagocytosis, and chemotaxis. The box plot shows the median with the lower and upper quartiles representing a 25th to 75th percentile range and whiskers extending to 1.5 × interquartile range (IQR). LOESS fitting of the data with relative confidence interval is represented by a blue line with a shadow area. d, PCA biplots based on the DEGs identified by LRT among bulk RNA-seq of NCP1s (orange), NCP2s (green), NCP3s (magenta) and NCP4s (turquiose). The graph lists the ten most relevant genes contributing to sample variations (indicated by vectors) for both PC1 and PC2, under both positive and negative directions. Vector lengths correlate with the weight of the given gene within the components.
Extended Data Fig. 8 Additional characterization of NCP and cMoP scRNA-seqs.
a, Density plots of NCP1s, NCP2s, NCP3s, NCP4s and cMoPs overlaid on the UMAP of Fig. 7a. Density of cells in each plot is depicted according to the indications of the color bar. b, Hierarchical clustering dendrogram based on the DEGs identified among the neutrophilic and monocytic cell clusters shown in Fig. 7b. The vertical axis of the dendrogram represents the dissimilarity between clusters (that is, Euclidean distance). c, Violin plots showing the mRNA expression levels [as ln (UMI)] of selected genes across the four neutrophil clusters (c1-c4) chosen among the top defining genes indicated in Fig. 8e. Clusters are colored according to Fig. 7bd, Expression patterns of cell-cycle, AG, SG, and GG genes projected on the UMAP plot restricted to neutrophil progenitor clusters (c1-c4). e, Gene Ontology analysis of DEGs for c2-c4. For every cluster (x-axis), the top ten Gene Ontology terms with Benjamini-Hochberg-corrected P values <0.05 (one-sided Fisher’s exact test) are shown. For cluster 1 no enrichment of biological processes GO term was identified. f, Trajectory plots of c1-c4 cells as defined in Fig. 7b. In each plot is depicted the density of cells according to the color bar placed at the right bottom corner of the panel.
Supplementary information
Supplementary Tables
The file contains Supplementary Tables 1–7.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Rights and permissions
About this article
Cite this article
Calzetti, F., Finotti, G., Tamassia, N. et al. CD66b−CD64dimCD115− cells in the human bone marrow represent neutrophil-committed progenitors. Nat Immunol 23, 679–691 (2022). https://doi.org/10.1038/s41590-022-01189-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-022-01189-z
This article is cited by
-
Small-molecule α-lipoic acid targets ELK1 to balance human neutrophil and erythrocyte differentiation
Stem Cell Research & Therapy (2024)
-
Comparative transcriptomics coupled to developmental grading via transgenic zebrafish reporter strains identifies conserved features in neutrophil maturation
Nature Communications (2024)
-
An immunophenotype-coupled transcriptomic atlas of human hematopoietic progenitors
Nature Immunology (2024)
-
Neutrophil diversity and plasticity: Implications for organ transplantation
Cellular & Molecular Immunology (2023)
-
Cellular and transcriptional dynamics of human neutrophils at steady state and upon stress
Nature Immunology (2022)