Abstract
Classic major histocompatibility complex class I (MHC-I) presentation relies on shuttling cytosolic peptides into the endoplasmic reticulum (ER) by the transporter associated with antigen processing (TAP). Viruses disable TAP to block MHC-I presentation and evade cytotoxic CD8+ T cells. Priming CD8+ T cells against these viruses is thought to rely solely on cross-presentation by uninfected TAP-functional dendritic cells. We found that protective CD8+ T cells could be mobilized during viral infection even when TAP was absent in all hematopoietic cells. TAP blockade depleted the endosomal recycling compartment of MHC-I molecules and, as such, impaired Toll-like receptor–regulated cross-presentation. Instead, MHC-I molecules accumulated in the ER–Golgi intermediate compartment (ERGIC), sequestered away from Toll-like receptor control, and coopted ER-SNARE Sec22b-mediated vesicular traffic to intersect with internalized antigen and rescue cross-presentation. Thus, when classic MHC-I presentation and endosomal recycling compartment–dependent cross-presentation are impaired in dendritic cells, cell-autonomous noncanonical cross-presentation relying on ERGIC-derived MHC-I counters TAP dysfunction to nevertheless mediate CD8+ T cell priming.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Source data and uncropped immunoblot images are provided with this paper. All other data supporting the findings of the paper are available from the corresponding author upon reasonable request.
References
Blum, J. S., Wearsch, P. A. & Cresswell, P. Pathways of antigen processing. Annu. Rev. Immunol. 31, 443–473 (2013).
Neefjes, J., Jongsma, M. L., Paul, P. & Bakke, O. Towards a systems understanding of MHC class I and MHC class II antigen presentation. Nat. Rev. Immunol. 11, 823–836 (2011).
Blees, A. et al. Structure of the human MHC-I peptide-loading complex. Nature 551, 525–528 (2017).
Mayerhofer, P. U. & Tampe, R. Antigen translocation machineries in adaptive immunity and viral immune evasion. J. Mol. Biol. 427, 1102–1118 (2015).
Praest, P., Liaci, A. M., Forster, F. & Wiertz, E. New insights into the structure of the MHC class I peptide-loading complex and mechanisms of TAP inhibition by viral immune evasion proteins. Mol. Immunol. 113, 103–114 (2019).
Blander, J. M. Regulation of the cell biology of antigen cross-presentation. Annu. Rev. Immunol. 36, 717–753 (2018).
Ackerman, A. L., Kyritsis, C., Tampe, R. & Cresswell, P. Early phagosomes in dendritic cells form a cellular compartment sufficient for cross presentation of exogenous antigens. Proc. Natl Acad. Sci. USA 100, 12889–12894 (2003).
Delamarre, L., Holcombe, H. & Mellman, I. Presentation of exogenous antigens on major histocompatibility complex (MHC) class I and MHC class II molecules is differentially regulated during dendritic cell maturation. J. Exp. Med. 198, 111–122 (2003).
Nair-Gupta, P. et al. TLR signals induce phagosomal MHC-I delivery from the endosomal recycling compartment to allow cross-presentation. Cell 158, 506–521 (2014).
Montealegre, S., Abramova, A., Manceau, V., de Kanter, A.F. & van Endert, P. The role of MHC class I recycling and Arf6 in cross-presentation by murine dendritic cells. Life Sci. Alliance 2, e201900464 (2019).
Oliveira, C. C. & van Hall, T. Alternative antigen processing for MHC class I: multiple roads lead to Rome. Front. Immunol. 6, 298 (2015).
Rock, K. L. & Shen, L. Cross-presentation: underlying mechanisms and role in immune surveillance. Immunol. Rev. 207, 166–183 (2005).
Sengupta, D., Graham, M., Liu, X. & Cresswell, P. Proteasomal degradation within endocytic organelles mediates antigen cross-presentation. EMBO J. 38, e99266 (2019).
Cruz, F. M., Colbert, J. D., Merino, E., Kriegsman, B. A. & Rock, K. L. The biology and underlying mechanisms of cross-presentation of exogenous antigens on MHC-I molecules. Annu. Rev. Immunol. 35, 149–176 (2017).
Heath, W. R. & Carbone, F. R. Cross-presentation in viral immunity and self-tolerance. Nat. Rev. Immunol. 1, 126–134 (2001).
Joffre, O. P., Segura, E., Savina, A. & Amigorena, S. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 12, 557–569 (2012).
Gutiérrez-Martínez, E. et al. Cross-presentation of cell-associated antigens by MHC class I in dendritic cell subsets. Front. Immunol. 6, 363 (2015).
Helft, J. et al. Cross-presenting CD103+ dendritic cells are protected from influenza virus infection. J. Clin. Investig. 122, 4037–4047 (2012).
Silvin, A. et al. Constitutive resistance to viral infection in human CD141+ dendritic cells. Sci. Immunol. 2, eaai8071 (2017).
Sigal, L. J., Crotty, S., Andino, R. & Rock, K. L. Cytotoxic T-cell immunity to virus-infected non-haematopoietic cells requires presentation of exogenous antigen. Nature 398, 77–80 (1999).
Whitney, P. G. et al. Effective priming of herpes simplex virus-specific CD8+ T cells in vivo does not require infected dendritic cells. J. Virol. 92, e01508–e01517 (2018).
Donaldson, J. G. & Williams, D. B. Intracellular assembly and trafficking of MHC class I molecules. Traffic 10, 1745–1752 (2009).
Nebenfuhr, A., Ritzenthaler, C. & Robinson, D. G. Brefeldin A: deciphering an enigmatic inhibitor of secretion. Plant Physiol. 130, 1102–1108 (2002).
Cebrian, I. et al. Sec22b regulates phagosomal maturation and antigen crosspresentation by dendritic cells. Cell 147, 1355–1368 (2011).
Hewitt, E. W. The MHC class I antigen presentation pathway: strategies for viral immune evasion. Immunology 110, 163–169 (2003).
Noriega, V., Redmann, V., Gardner, T. & Tortorella, D. Diverse immune evasion strategies by human cytomegalovirus. Immunol. Res. 54, 140–151 (2012).
Shen, L., Sigal, L. J., Boes, M. & Rock, K. L. Important role of cathepsin S in generating peptides for TAP-independent MHC class I crosspresentation in vivo. Immunity 21, 155–165 (2004).
Tiwari, N. et al. A transporter associated with antigen-processing independent vacuolar pathway for the MHC class I-mediated presentation of endogenous transmembrane proteins. J. Immunol. 178, 7932–7942 (2007).
Bertholet, S. et al. Leishmania antigens are presented to CD8+ T cells by a transporter associated with antigen processing-independent pathway in vitro and in vivo. J. Immunol. 177, 3525–3533 (2006).
Merzougui, N., Kratzer, R., Saveanu, L. & van Endert, P. A proteasome-dependent, TAP-independent pathway for cross-presentation of phagocytosed antigen. EMBO Rep. 12, 1257–1264 (2011).
Lawand, M., Abramova, A., Manceau, V., Springer, S. & van Endert, P. TAP-dependent and -independent peptide import into dendritic cell phagosomes. J. Immunol. 197, 3454–3463 (2016).
Blander, J. M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Huang, A. Y., Bruce, A. T., Pardoll, D. M. & Levitsky, H. I. In vivo cross-priming of MHC class I-restricted antigens requires the TAP transporter. Immunity 4, 349–355 (1996).
Lankat-Buttgereit, B. & Tampe, R. The transporter associated with antigen processing: function and implications in human diseases. Physiol. Rev. 82, 187–204 (2002).
Halenius, A. & Hengel, H. Human cytomegalovirus and autoimmune disease. Biomed. Res. Int. 2014, 472978 (2014).
Fu, Y., Yan, G., Shi, L. & Faustman, D. Antigen processing and autoimmunity. Evaluation of mRNA abundance and function of HLA-linked genes. Ann. N. Y. Acad. Sci. 842, 138–155 (1998).
Aldrich, C. J. et al. Positive selection of self- and alloreactive CD8+ T cells in Tap-1 mutant mice. Proc. Natl Acad. Sci. USA 91, 6525–6528 (1994).
Van Kaer, L., Ashton-Rickardt, P. G., Ploegh, H. L. & Tonegawa, S. TAP1 mutant mice are deficient in antigen presentation, surface class I molecules, and CD4–8+ T cells. Cell 71, 1205–1214 (1992).
Behar, S. M., Dascher, C. C., Grusby, M. J., Wang, C. R. & Brenner, M. B. Susceptibility of mice deficient in CD1D or TAP1 to infection with Mycobacterium tuberculosis. J. Exp. Med. 189, 1973–1980 (1999).
Alloatti, A. et al. Critical role for Sec22b-dependent antigen cross-presentation in antitumor immunity. J. Exp. Med. 214, 2231–2241 (2017).
Wu, S. J. et al. A critical analysis of the role of SNARE protein SEC22B in antigen cross-presentation. Cell Rep. 19, 2645–2656 (2017).
Sigal, L. J. & Rock, K. L. Bone marrow–derived antigen-presenting cells are required for the generation of cytotoxic T lymphocyte responses to viruses and use transporter associated with antigen presentation (Tap)-dependent and -independent pathways of antigen presentation. J. Exp. Med. 192, 1143–1150 (2000).
Geng, J., Zaitouna, A. J. & Raghavan, M. Selected HLA-B allotypes are resistant to inhibition or deficiency of the transporter associated with antigen processing (TAP). PLoS Pathog. 14, e1007171 (2018).
Cerundolo, V. & de la Salle, H. Description of HLA class I- and CD8-deficient patients: insights into the function of cytotoxic T lymphocytes and NK cells in host defense. Semin. Immunol. 18, 330–336 (2006).
de la Salle, H. et al. HLA class I deficiencies due to mutations in subunit 1 of the peptide transporter TAP1. J. Clin. Investig. 103, R9–R13 (1999).
Garrido, G. et al. Tumor-targeted silencing of the peptide transporter TAP induces potent antitumor immunity. Nat. Commun. 10, 3773 (2019).
Marijt, K. A., Van Der Burg, S. H. & van Hall, T. TEIPP peptides: exploration of unTAPped cancer antigens. Oncoimmunology 8, 1599639 (2019).
Marijt, K. A. & van Hall, T. To TAP or not to TAP: alternative peptides for immunotherapy of cancer. Curr. Opin. Immunol. 64, 15–19 (2020).
Uger, R. A. & Barber, B. H. Presentation of an influenza nucleoprotein epitope incorporated into the H-2Db signal sequence requires the transporter-associated with antigen presentation. J. Immunol. 158, 685–692 (1997).
Zhong, W., Reche, P. A., Lai, C. C., Reinhold, B. & Reinherz, E. L. Genome-wide characterization of a viral cytotoxic T lymphocyte epitope repertoire. J. Biol. Chem. 278, 45135–45144 (2003).
Perez, O. A. et al. CD169+ macrophages orchestrate innate immune responses by regulating bacterial localization in the spleen. Sci. Immunol. 2, eaah5520 (2017).
Suchin, E. J. et al. Quantifying the frequency of alloreactive T cells in vivo: new answers to an old question. J. Immunol. 166, 973–981 (2001).
Drutman, S. B. & Trombetta, E. S. Dendritic cells continue to capture and present antigens after maturation in vivo. J. Immunol. 185, 2140–2146 (2010).
Moltedo, B., Li, W., Yount, J. S. & Moran, T. M. Unique type I interferon responses determine the functional fate of migratory lung dendritic cells during influenza virus infection. PLoS Pathog. 7, e1002345 (2011).
Rodriguez-Madoz, J. R. et al. Inhibition of the type I interferon response in human dendritic cells by dengue virus infection requires a catalytically active NS2B3 complex. J. Virol. 84, 9760–9774 (2010).
Noriega, V. M. et al. Human cytomegalovirus modulates monocyte-mediated innate immune responses during short-term experimental latency in vitro. J. Virol. 88, 9391–9405 (2014).
Parham, P., Barnstable, C. J. & Bodmer, W. F. Use of a monoclonal antibody (W6/32) in structural studies of HLA-A,B,C, antigens. J. Immunol. 123, 342–349 (1979).
Bolte, S. & Cordelieres, F. P. A guided tour into subcellular colocalization analysis in light microscopy. J. Microsc. 224, 213–232 (2006).
Husebye, H. et al. The Rab11a GTPase controls Toll-like receptor 4-induced activation of interferon regulatory factor-3 on phagosomes. Immunity 33, 583–596 (2010).
Acknowledgements
We thank V. Gillespie for expert pathology on mouse lung tissues. We thank S. Trombetta (Boehringer Ingelheim); K. Rock (University of Massachusetts); P. Cresswell (Yale University); and W. Li, T. M. Moran, D. B. Rubiov and A. Fernandez-Sesma (Icahn School of Medicine at Mount Sinai) for reagents and technical advice. We are grateful to current Blander laboratory members and to H. Gupta, M. A. Blander and S. J. Blander for discussions and support. The ISMMS-Microscopy Shared Resource Facility was supported by grants no. NIH 5R24 CA095823-04, no. S10 RR0 9145-01 and no. NSF DBI-9724504. This work was supported by NIH grants no. AI073899 and no. AI123284 to J.M.B.; an NIH/NIAID Center for Research on Influenza Pathogenesis contract as part of the CEIRS Network no. HHSN266200700010C to A.G.-S., grant nos. AI101820 and AI112318 to D.T., and grant no. AI143861 to K.M.K.; German Research Foundation grants no. SFB 807–Membrane Transport and Communication and no. TA157/7; and by the European Research Council (ERC Advanced Grant no. 789121) to R.T. Support to J.M.B. was also provided by NIH grants no. DK111862 and no. AI127658, the Burroughs Wellcome Fund, and the Leukemia and Lymphoma Society. G.B. has been supported by a fellowship and is currently supported by a career development award from the Crohn’s and Colitis Foundation. T.G. was supported by an American Heart Association pre-doctoral fellowship and NIH grant no. F32CA224438.
Author information
Authors and Affiliations
Contributions
G.B., P.N.-G. and J.M.B. designed experiments, directed the study and wrote the manuscript. P.N.-G. and G.B. performed most in vitro and in vivo experiments, respectively, and curated data. M.S., G.M., A.C. and A.G.-S. provided influenza A virus animal model expertise and management and conducted animal weight loss and lung viral titer measurements. F.S. and R.T. prepared and provided recombinant soluble TAP inhibitor US6. J.M. and G.B. performed immunoblots for knockdown validation. T.G. and D.T. provided reagents and methodology for HCMV infections. S.T.Y. and K.M.K. sectioned, stained and imaged lung tissues by confocal microscopy. J.M.B. supervised and conceived of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Immunology thanks Malini Raghavan, Scheherazade Sadegh-Nasseri and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. L. A. Dempsey was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
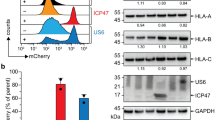
Extended Data Fig. 1 Steady state residual expression of MHC-I at the plasma membrane of Tap–/– DC.
a, Confocal micrographs of WT or Tap−⁄− resting DCs stained for cholera toxin B subunit (CTB), H2-Kb and ERGIC-53. b, FACS analyses of WT or Tap−⁄− DCs for surface H2-Kb. Scale bars represent 10 µm. Data represent at least three independent experiments.
Extended Data Fig. 2 HCMV protein expression is detected only in DC treated with live and not UV-inactivated virus.
Confocal micrographs of human DCs stained for immediate early (IE) viral protein along with CTB, 48h post infection with either HCMVTB40/E or its UV-irradiated counterpart at a MOI=10. Scale bars represent 10 µm. Data represent at least three independent experiments.
Extended Data Fig. 3 Impaired TAP function blocks classic MHC-I presentation but not cross-presentation.
a, Classic MHC-I presentation by WT DCs of SIINFEKL derived from influenza PR/8-OTI virus to OVA-specific CD8+ T cells. DCs were infected with influenza PR/8-OT-I virus for 5h and incubated in the presence of active or inactive US6. b, c, WT and Tap−⁄− DC cross-presentation of SIINFEKL from E. coli-OVA at several time points (b), or at 4.5h post-phagocytosis of either E. coli-OVA or 5h post-infection with influenza PR/8-OTI virus (c). Data represent at least three independent experiments except for (b) depicting two independent experiments. The mean ± SEM are presented and each symbol represents biological replicate. **P < 0.01; N.S.= non statistically significant (P>0.05) using an unpaired two-tailed t-test.
Extended Data Fig. 4 Classic MHC-I presentation of viral antigen by Wt DC is not affected by Sec22b or Rab11a inhibition.
a, Immunoblots for the expression of β-actin, Sec22b and Rab11a on whole cell extracts prepared from DCs seven days post-differentiation of bone marrow progenitors that had been transduced with lentiviruses encoding for control shRNA or shRNA targeting either Rab11a or Sec22b. Immunoblots were cropped to show indicated proteins. b, WT DC progenitors were transduced with recombinant lentiviruses expressing scrambled, Sec22b or Rab11a specific shRNA. Classic MHC-I presentation of SIINFEKL at 5h after infection with recombinant SIINFEKL-expressing Influenza-OTI (PR/8-OTI) (left panels, no secondary treatment) or cross-presentation of SIINFEKL from heat-inactivated Influenza-OTI given to DCs at 3h following infection by Influenza-OTI virus. Cross-presentation was assessed 2h later. Data represent at least three independent experiments. The mean ± SEM are presented and each symbol represents biological replicate. N.S.= non statistically significant (P>0.05) using an unpaired two-tailed t-test.
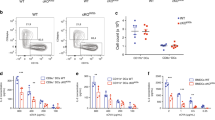
Extended Data Fig. 5 Lung CD8 T cells during lethal challenge with influenza A virus.
a, Percent of influenza A specific CD8+ T cells in the lungs of WT→WT and Tap⎯/⎯→WT mice at days 3 and 5 post lethal challenge with 75 p.f.u. influenza A PR8 virus. Data show two different H-2Dd tetramers loaded with either a polymerase acidic protein epitope (SSLENFRAYV) or a nucleoprotein epitope (ASNENMETM). b, Representative FACS plot and % CD8+CD44+ cells in the lung of naïve chimeric mice. c, Flow cytometry plots showing CD8+ T cells in the lungs or spleens of WT mice on days 1 and 2 following injection with anti-CD8α antibody (clone 2.43) intranasally (i.n., 200µg), intraperitoneally (i.p., 100µg), or via both routes (i.p.+i.n.). Percent of CD3+CD8β+ T cells remaining are indicated in the blue gates. The mean ± SEM are presented and each symbol represents a mouse. N.S.= non statistically significant (P>0.05) using an unpaired two-tailed t-test.
Extended Data Fig. 6 CD8 T cells mediate protection of Tap⎯/⎯ chimeric mice against lethal challenge with influenza A virus.
a, Confocal micrographs at 20X magnification of lung sections from indicated anti-CD8α-treated mice stained for CD8α, CD11c, EpCAM and influenza A virus. Legend to the right shows the color code for each antibody. Scale bar represents 50 µm. b, Lung PR8 viral titers at days 3 and 5 in WT→WT and Tap⎯/⎯→WT chimeric mice. Each symbol is one mouse. c, Pathology scores of infected mice at different time points post lethal PR8 challenge in WT→WT and Tap⎯/⎯→WT mice. Slides were scored by a blinded pathologist for perivascular, bronchiolar or alveolar inflammation, epithelial degeneration or necrosis, and intraluminal debris or hemorrhage. Each symbol is one mouse. d, Hematoxylin-and-eosin staining of lung sections from WT→WT and Tap⎯/⎯→WT mice on days 3 or 5 post PR8 challenge. Scale bar represents 100 μm. The mean ± SEM are presented and each symbol represents a mouse. Data represent 2 experiments (for a total n of 140 mice).
Supplementary information
Supplementary Information
Supplementary gating strategy.
Supplementary Video 1
A schematic illustrating the cell biology of noncanonical cross-presentation based on the data presented in this study. The model phagocytic cargo shown here is a bacterium that engages TLR signaling. Based on previous work, two pathways of vesicular traffic contribute to the cross-presenting phagosome: TLR-regulated traffic from the ERC to the phagosome mediated by TLR–MyD88–IKK2-dependent phosphorylation of SNAP23 (not shown, refer to ref. 9) which delivers MHC-I molecules, and TLR-independent traffic from the ERGIC to phagosomes requiring the ER-SNARE Sec22b, which delivers the components of the peptide-loading complex, including TAP and Sec61 (refs. 9,24). Accumulation of MHC-I molecules in the ERGIC upon TAP dysfunction enables their Sec22b-dependent recruitment to phagosomes to prime CD8+ T cells through noncanonical cross-presentation.
Supplementary Video 2
3D reconstruction of confocal stacks showing CD11c+ cells with infected epithelial cell inclusions (white merge) in the lungs of WT → WT at day 3 post influenza A/PR8 challenge. Staining for CD11c is in red, influenza in green, CD8 in cyan and EpCAM in magenta.
Supplementary Video 3
3D reconstruction of confocal stacks showing CD11c+ cells with infected epithelial cell inclusions (white merge) in the lungs of Tap⎯/⎯ → WT at day 3 post influenza A/PR8 challenge. Staining for CD11c is in red, influenza in green, CD8 in cyan and EpCAM in magenta.
Source data
Source Data Fig. 1
Numerical data used for plots.
Source Data Fig. 2
Numerical data used for plots.
Source Data Fig. 3
Numerical data used for plots.
Source Data Fig. 4
Numerical data used for plots.
Source Data Fig. 4
Unprocessed immunoblots.
Source Data Fig. 5
Numerical data used for plots.
Source Data Fig. 6
Numerical data used for plots.
Source Data Fig. 7
Numerical data used for plots.
Source Data Fig. 8
Numerical data used for plots.
Source Data Extended Data Fig. 3
Numerical data used for plots.
Source Data Extended Data Fig. 4
Numerical data used for plots.
Source Data Extended Data Fig. 4
Unprocessed immunoblots.
Source Data Extended Data Fig. 5
Numerical data used for plots.
Source Data Extended Data Fig. 6
Numerical data used for plots.
Rights and permissions
About this article
Cite this article
Barbet, G., Nair-Gupta, P., Schotsaert, M. et al. TAP dysfunction in dendritic cells enables noncanonical cross-presentation for T cell priming. Nat Immunol 22, 497–509 (2021). https://doi.org/10.1038/s41590-021-00903-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-021-00903-7
This article is cited by
-
A Novel Platinum Resistance-Related Immune Gene Signature for Overall Survival Prediction in Patients with Ovarian Cancer
Biochemical Genetics (2024)
-
A guide to antigen processing and presentation
Nature Reviews Immunology (2022)