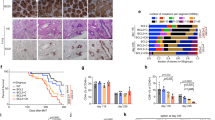
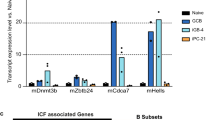
Abstract
During the germinal center (GC) reaction, B cells undergo extensive redistribution of cohesin complex and three-dimensional reorganization of their genomes. Yet, the significance of cohesin and architectural programming in the humoral immune response is unknown. Herein we report that homozygous deletion of Smc3, encoding the cohesin ATPase subunit, abrogated GC formation, while, in marked contrast, Smc3 haploinsufficiency resulted in GC hyperplasia, skewing of GC polarity and impaired plasma cell (PC) differentiation. Genome-wide chromosomal conformation and transcriptional profiling revealed defects in GC B cell terminal differentiation programs controlled by the lymphoma epigenetic tumor suppressors Tet2 and Kmt2d and failure of Smc3-haploinsufficient GC B cells to switch from B cell- to PC-defining transcription factors. Smc3 haploinsufficiency preferentially impaired the connectivity of enhancer elements controlling various lymphoma tumor suppressor genes, and, accordingly, Smc3 haploinsufficiency accelerated lymphomagenesis in mice with constitutive Bcl6 expression. Collectively, our data indicate a dose-dependent function for cohesin in humoral immunity to facilitate the B cell to PC phenotypic switch while restricting malignant transformation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Datasets generated in this manuscript were deposited in the NCBI Gene Expression Omnibus under the accession number GSE143852. Source data are provided with this paper.
References
Mesin, L., Ersching, J. & Victora, G. D. Germinal center B cell dynamics. Immunity 45, 471–482 (2016).
Mlynarczyk, C., Fontan, L. & Melnick, A. Germinal center-derived lymphomas: the darkest side of humoral immunity. Immunol. Rev. 288, 214–239 (2019).
Shapiro-Shelef, M. & Calame, K. Regulation of plasma-cell development. Nat. Rev. Immunol. 5, 230–242 (2005).
Nutt, S. L., Taubenheim, N., Hasbold, J., Corcoran, L. M. & Hodgkin, P. D. The genetic network controlling plasma cell differentiation. Semin. Immunol. 23, 341–349 (2011).
Dekker, J., Marti-Renom, M. A. & Mirny, L. A. Exploring the three-dimensional organization of genomes: interpreting chromatin interaction data. Nat. Rev. Genet. 14, 390–403 (2013).
Yu, M. & Ren, B. The three-dimensional organization of mammalian genomes. Annu. Rev. Cell Dev. Biol. 33, 265–289 (2017).
Dixon, J. R. et al. Topological domains in mammalian genomes identified by analysis of chromatin interactions. Nature 485, 376–380 (2012).
Jin, F. et al. A high-resolution map of the three-dimensional chromatin interactome in human cells. Nature 503, 290–294 (2013).
Kagey, M. H. et al. Mediator and cohesin connect gene expression and chromatin architecture. Nature 467, 430–435 (2010).
Losada, A. Cohesin in cancer: chromosome segregation and beyond. Nat. Rev. Cancer 14, 389–393 (2014).
Peters, J. M., Tedeschi, A. & Schmitz, J. The cohesin complex and its roles in chromosome biology. Genes Dev. 22, 3089–3114 (2008).
Ladurner, R. et al. Cohesin’s ATPase activity couples cohesin loading onto DNA with Smc3 acetylation. Curr. Biol. 24, 2228–2237 (2014).
Hu, B. et al. ATP hydrolysis is required for relocating cohesin from sites occupied by its Scc2/4 loading complex. Curr. Biol. 21, 12–24 (2011).
White, J. K. et al. Genome-wide generation and systematic phenotyping of knockout mice reveals new roles for many genes. Cell 154, 452–464 (2013).
Liu, J. & Krantz, I. D. Cornelia de Lange syndrome, cohesin, and beyond. Clin. Genet. 76, 303–314 (2009).
Casola, S. et al. Tracking germinal center B cells expressing germ-line immunoglobulin γ1 transcripts by conditional gene targeting. Proc. Natl Acad. Sci. USA 103, 7396–7401 (2006).
Viny, A. D. et al. Dose-dependent role of the cohesin complex in normal and malignant hematopoiesis. J. Exp. Med. 212, 1819–1832 (2015).
Beguelin, W. et al. EZH2 enables germinal centre formation through epigenetic silencing of CDKN1A and an Rb–E2F1 feedback loop. Nat. Commun. 8, 877 (2017).
Thomas-Claudepierre, A. S. et al. The cohesin complex regulates immunoglobulin class switch recombination. J. Exp. Med. 210, 2495–2502 (2013).
Zhang, X. et al. Fundamental roles of chromatin loop extrusion in antibody class switching. Nature 575, 385–389 (2019).
Ortega-Molina, A. et al. The histone lysine methyltransferase KMT2D sustains a gene expression program that represses B cell lymphoma development. Nat. Med. 21, 1199–1208 (2015).
Soro, P. G. et al. Differential involvement of the transcription factor Blimp-1 in T cell-independent and -dependent B cell differentiation to plasma cells. J. Immunol. 163, 611–617 (1999).
Scharer, C. D., Barwick, B. G., Guo, M., Bally, A. P. R. & Boss, J. M. Plasma cell differentiation is controlled by multiple cell division-coupled epigenetic programs. Nat. Commun. 9, 1698 (2018).
Klein, U. et al. Transcription factor IRF4 controls plasma cell differentiation and class-switch recombination. Nat. Immunol. 7, 773–782 (2006).
Victora, G. D. et al. Identification of human germinal center light and dark zone cells and their relationship to human B-cell lymphomas. Blood 120, 2240–2248 (2012).
Agirre, X. et al. Long non-coding RNAs discriminate the stages and gene regulatory states of human humoral immune response. Nat. Commun. 10, 821 (2019).
Cho, R. J. et al. Transcriptional regulation and function during the human cell cycle. Nat. Genet. 27, 48–54 (2001).
Whitfield, M. L. et al. Identification of genes periodically expressed in the human cell cycle and their expression in tumors. Mol. Biol. Cell 13, 1977–2000 (2002).
Su, A. I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl Acad. Sci. USA 101, 6062–6067 (2004).
Rosenwald, A. et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N. Engl. J. Med. 346, 1937–1947 (2002).
Zeller, K. I. et al. Global mapping of c-Myc binding sites and target gene networks in human B cells. Proc. Natl Acad. Sci. USA 103, 17834–17839 (2006).
Dominguez, P. M. et al. TET2 deficiency causes germinal center hyperplasia, impairs plasma cell differentiation, and promotes B-cell lymphomagenesis. Cancer Discov. 8, 1632–1653 (2018).
Victora, G. D. et al. Germinal center dynamics revealed by multiphoton microscopy with a photoactivatable fluorescent reporter. Cell 143, 592–605 (2010).
Ersching, J. et al. Germinal center selection and affinity maturation require dynamic regulation of mTORC1 kinase. Immunity 46, 1045–1058 (2017).
Dominguez-Sola, D. et al. The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry. Nat. Immunol. 13, 1083–1091 (2012).
Rao, S. S. et al. A 3D map of the human genome at kilobase resolution reveals principles of chromatin looping. Cell 159, 1665–1680 (2014).
Rickert, R. C., Roes, J. & Rajewsky, K. B lymphocyte-specific, Cre-mediated mutagenesis in mice. Nucleic Acids Res. 25, 1317–1318 (1997).
Jiang, Y. et al. CREBBP inactivation promotes the development of HDAC3-dependent lymphomas. Cancer Discov. 7, 38–53 (2017).
Hummel, M. et al. A biologic definition of Burkitt’s lymphoma from transcriptional and genomic profiling. N. Engl. J. Med. 354, 2419–2430 (2006).
Jais, J. P. et al. The expression of 16 genes related to the cell of origin and immune response predicts survival in elderly patients with diffuse large B-cell lymphoma treated with CHOP and rituximab. Leukemia 22, 1917–1924 (2008).
Lenz, G. et al. Stromal gene signatures in large-B-cell lymphomas. N. Engl. J. Med. 359, 2313–2323 (2008).
Shaknovich, R. et al. DNA methylation signatures define molecular subtypes of diffuse large B-cell lymphoma. Blood 116, e81–e89 (2010).
Lin, Y. C. et al. Global changes in the nuclear positioning of genes and intra- and interdomain genomic interactions that orchestrate B cell fate. Nat. Immunol. 13, 1196–1204 (2012).
Bunting, K. L. et al. Multi-tiered reorganization of the genome during B cell affinity maturation anchored by a germinal center-specific locus control region. Immunity 45, 497–512 (2016).
Johanson, T. M. et al. Transcription-factor-mediated supervision of global genome architecture maintains B cell identity. Nat. Immunol. 19, 1257–1264 (2018).
Ise, W. & Kurosaki, T. Plasma cell differentiation during the germinal center reaction. Immunol. Rev. 288, 64–74 (2019).
Mazumdar, C. et al. Leukemia-associated cohesin mutants dominantly enforce stem cell programs and impair human hematopoietic progenitor differentiation. Cell Stem Cell 17, 675–688 (2015).
Mullenders, J. et al. Cohesin loss alters adult hematopoietic stem cell homeostasis, leading to myeloproliferative neoplasms. J. Exp. Med. 212, 1833–1850 (2015).
Davidson, I. F. et al. DNA loop extrusion by human cohesin. Science 366, 1338–1345 (2019).
Kim, Y., Shi, Z., Zhang, H., Finkelstein, I. J. & Yu, H. Human cohesin compacts DNA by loop extrusion. Science 366, 1345–1349 (2019).
Zhang, J. et al. Disruption of KMT2D perturbs germinal center B cell development and promotes lymphomagenesis. Nat. Med. 21, 1190–1198 (2015).
Deng, W. et al. Controlling long-range genomic interactions at a native locus by targeted tethering of a looping factor. Cell 149, 1233–1244 (2012).
Groschel, S. et al. A single oncogenic enhancer rearrangement causes concomitant EVI1 and GATA2 deregulation in leukemia. Cell 157, 369–381 (2014).
Rao, S. S. P. et al. Cohesin loss eliminates all loop domains. Cell 171, 305–320 (2017).
Fudenberg, G., Abdennur, N., Imakaev, M., Goloborodko, A. & Mirny, L. A. Emerging evidence of chromosome folding by loop extrusion. Cold Spring Harb. Symp. Quant. Biol. 82, 45–55 (2017).
Cuartero, S. et al. Control of inducible gene expression links cohesin to hematopoietic progenitor self-renewal and differentiation. Nat. Immunol. 19, 932–941 (2018).
Sasca, D. et al. Cohesin-dependent regulation of gene expression during differentiation is lost in cohesin-mutated myeloid malignancies. Blood 134, 2195–2208 (2019).
De Koninck, M. & Losada, A. Cohesin mutations in cancer. Cold Spring Harb. Perspect. Med. 6, a026476 (2016).
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001).
Cattoretti, G. et al. Deregulated BCL6 expression recapitulates the pathogenesis of human diffuse large B cell lymphomas in mice. Cancer Cell 7, 445–455 (2005).
Silverman, J., Suckow, M. A. & Murthy, S. The IACUC Handbook 3rd edn (CRC Press, Taylor & Francis Group, 2014).
Harrow, J. et al. GENCODE: the reference human genome annotation for The ENCODE Project. Genome Res. 22, 1760–1774 (2012).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Li, B., Ruotti, V., Stewart, R. M., Thomson, J. A. & Dewey, C. N. RNA-seq gene expression estimation with read mapping uncertainty. Bioinformatics 26, 493–500 (2010).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Sergushichev, A. A. An algorithm for fast preranked gene set enrichment analysis using cumulative statistic calculation. Preprint at bioRxiv https://doi.org/10.1101/060012 (2016).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Lazaris, C., Kelly, S., Ntziachristos, P., Aifantis, I. & Tsirigos, A. HiC-bench: comprehensive and reproducible Hi-C data analysis designed for parameter exploration and benchmarking. BMC Genomics 18, 22 (2017).
Wolff, J. et al. Galaxy HiCExplorer: a web server for reproducible Hi-C data analysis, quality control and visualization. Nucleic Acids Res. 46, W11–W16 (2018).
Imakaev, M. et al. Iterative correction of Hi-C data reveals hallmarks of chromosome organization. Nat. Methods 9, 999–1003 (2012).
Zheng, X. & Zheng, Y. CscoreTool: fast Hi-C compartment analysis at high resolution. Bioinformatics 34, 1568–1570 (2018).
Crane, E. et al. Condensin-driven remodelling of X chromosome topology during dosage compensation. Nature 523, 240–244 (2015).
van Galen, P. et al. A multiplexed system for quantitative comparisons of chromatin landscapes. Mol. Cell 61, 170–180 (2016).
Girardot, C., Scholtalbers, J., Sauer, S., Su, S. Y. & Furlong, E. E. Je, a versatile suite to handle multiplexed NGS libraries with unique molecular identifiers. BMC Bioinformatics 17, 419 (2016).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Street, K. et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics 19, 477 (2018).
Acknowledgements
We thank J. Yan and B. Ren (University of California, San Diego) for their help in setting up the in situ Hi-C protocol. Sequencing was performed in the Epigenomics Core and the Genomics Core at Weill Cornell Medicine. The authors thank J. McCormick and T. Baumgartner from the Flow Cytometry Core Facility at Weill Cornell Medicine and G. Nanjangud from the Molecular Genetics Core at Memorial Sloan Kettering Cancer Center for expert assistance with protocols. A.M.M. is supported by NCI/NIH R35 CA220499, NCI/NIH P01 CA229086-01A1 (together with A.T.), LLS-SCOR 7012-16, LLS-TRP 6572-19, the Samuel Waxman Cancer Research Foundation, the Follicular Lymphoma Consortium and the Chemotherapy Foundation. M.A.R. was a recipient of a postdoctoral fellowship grant from the Lymphoma Research Foundation and is funded by an NHLBI 5 T32 HL135465-3 training grant. O.E. is supported by NIH grants UL1TR002384 and R01 CA194547 and LLS-SCOR grants 180078-02 and 7021-20. R.L.L. was supported by NCI P30 CA008748, NCI R35 CA197594-01A1, NCI R01 CA216421 and NCI PS-OC U54 CA143869-05. A.D.V. is supported by NCI K08 CA215317, the William Raveis Charitable Fund Fellowship of the Damon Runyon Cancer Research Foundation (DRG 117-15) and an Evans MDS Young Investigator grant from the Edward P. Evans Foundation. C.E.M. thanks the Scientific Computing Unit, XSEDE Supercomputing Resources as well as the Starr Cancer Consortium (I7-A765, I9-A9-071 and I13-0052) and acknowledges funding from the WorldQuant Foundation, The Pershing Square Sohn Cancer Research Alliance, the NIH (grants R01 CA249054, R01 AI151059, P01 CA214274, R01 AI125416-03 and R21 AI129851-02) and Leukemia and Lymphoma Society grants LLS 9238-16 and LLS MCL-982. A.T. is funded by American Cancer Society RSG-15-189-01-RMC, a St. Baldrick’s Scholar Award and NCI/NIH Cancer Center Support Grant P30 CA016087.
Author information
Authors and Affiliations
Contributions
M.A.R. conceptualized, designed and performed most experiments, with the help of M.F.C. and D.K., and analyzed and interpreted data, drafted the manuscript and supervised the study. C.M. analyzed and interpreted RNA-seq, Hi-C and Mint-ChIP data, and C.R.C. analyzed single-cell RNA-seq data with the help of M.R.T., X.W., A.K., B.B. and A.S.D. A.D.V. and R.L.L. provided the Smc3 animal model and contributed to the interpretation of results. D.R.M. has helped with the execution of experiments and analysis of data. W.B., M.T.C.F. and H.S. provided technical expertise and helped with the interpretation of results. Z.C. analyzed the clinical data. A.T., O.E. and C.E.M. provided expertise and resources for data analysis. A.M.M. participated in the study conceptualization, interpretation of data, drafting of the manuscript, acquisition of funds and supervision of the project. All authors have read and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
A.M.M. receives research funding from Janssen Pharmaceuticals, Sanofi and Daiichi Sankyo, has consulted for Epizyme and Constellation and is on the scientific advisory board of KDAC pharmaceuticals. R.L.L. is on the supervisory board of Qiagen and was on the scientific advisory board of Loxo, Imago, Mana, Auron, C4 Therapeutics and Isoplexis, which include equity interest. R.L.L. receives research support from and consulted for Celgene and Roche and consults for Incyte, Lilly, Janssen, Astellas, Morphosys and Novartis. R.L.L. receives research support from Prelude and has received honoraria from AstraZeneca, Roche, Lilly and Amgen for invited lectures and from Gilead for grant reviews. O.E. is supported by Janssen and Eli Lilly research grants. O.E. is a scientific advisor and equity holder in Freenome, Owkin, Volastra Therapeutics and One Three Biotech. C.E.M. is a cofounder and board member for Biotia and Onegevity Health as well as an advisor or compensated speaker for Abbvie, Acuamark Diagnostics, ArcBio, Bio-Rad, DNA Genotek, Genialis, Genpro, Karius, Illumina, New England BioLabs, Qiagen, Whole Biome and Zymo Research. C.M. is a consultant and analyst for Onegevity Health. M.A.R., C.R.C., M.F.C., D.K., B.B., A.K., A.D.V., M.R.T., D.R.M., A.S.D., X.W., W.B., M.T.C.F., H.S., Z.C. and A.T. have no relevant competing interests to declare.
Additional information
Peer review information Peer reviewer reports are available. L. A. Dempsey was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Smc3 knockout abrogates GC formation whereas Smc3 haploinsufficiency induces GC Hyperplasia.
a, Box plots showing transcripts per million (TPM) of cohesin core subunits (Smc3, Smc1a, Stag1, Stag2 and Rad21) and regulator proteins (Nipbl, Wapl, Pds5a, Pds5b) in mouse and human (b). NB: naive B cells, CB: centroblasts, CC: centrocytes, PC: splenic plasma cells, TPC: tonsillar plasma cells, BMPC: bone marrow plasma cells. c, Scheme depicting Smc3 conditional knockout model breeding strategy and d, genotyping strategy. e, Gating strategy used for flow cytometric analysis of GC B cells. f, Genotyping of sorted GC B cells. Representative gel of one experiment out of four performed. g, Representative flow cytometry plots showing total B cells (B220+ cells) and follicular B cells (B220+CD23hiCD21lo cells) and marginal zone B cell populations (B220+CD23loCD21hi cells) and quantitative data for one representative experiment out of 4 performed in Smc3wt/wt (n=6), Smc3wt/– (n=4) and Smc3–/– (n=3) as shown (right). One-way ANoVA followed by Tukey test for multiple comparisons was used. h, i, Quantification of GC B cells, defined as B220+Fas+CD38–, 4 days (h, p=0.002) or 15 days (I, p=0.0434) after SRBC immunization. Two-tail unpaired t-test was used. j, RT-qPCR comparing Smc3 mRNA expression in GC B cells (centroblasts, CB and centrocytes, CC), plasmablasts (PB) and plasma cells (PC) in Cγ1wt/cre;Smc3wt/wt and Cγ1wt/cre;Smc3wt/– mice as indicated. Two-tail unpaired t-test was used. CB, p=0.0039, CC: p=0.0005, PB: p=0.0041 and PC: p=0.0271. k, Capillary immunoblot analyses comparing Smc3, Smc1a, Rad21 and Sag2 protein levels in centroblasts (CB), centrocytes (CC) and naïve B cells (NBC) from Cγ1wt/cre;Smc3wt/wt (n=3) and Cγ1wt/cre;Smc3wt/– (n=3) mice as indicated. Representative gel our of two experiments performed. Right panels show quantifications of the cohesin complex subunits using β-actin protein as loading control. Two-way ANoVA was used. **p<0.01. In (a) and (b) box plots show the median as center, first and third quartiles as the box hinges, and whiskers extend to the smallest and largest value no further than the 1.5 × interquartile range (IQR) away from the hinges. In (g) (h), (i), (j) and (k) data are presented as mean +/- SD.
Extended Data Fig. 2 Smc3 haploinsufficiency confers proliferative advantage to GC B cells without chromosomal instability.
a, Cγ1wt/cre;Smc3wt/wt (n=5) and Cγ1wt/cre;Smc3wt/– (n=5) were immunized with SRBCs to induce GC formation. One hour prior to euthanasia, mice were injected with 50 mg/kg i.v. 5-ethynyl-2’-deoxyuridine (EdU). b, GC organoids were performed with splenocytes from Cγ1wt/cre;Smc3wt/wt and Cγ1wt/cre;Smc3wt/– mice. c, On day 0 and day 4 after organoid plating, the presence of B cells and GC-like cells (B220+Fas+GL7+) was determined by flow cytometry. d, Gating strategy followed to detect apoptotic active Caspase-3+ GC B cells.
Extended Data Fig. 3 GC hyperplasia induced by Smc3 haploinsufficiency is due to expansion of centrocytes.
(a) Mice were immunized with sheep red blood cells (SRBCs) to induce GC formation and euthanized 8 days after. (b) Gating strategy followed to identify DZ (B220+Fas+CD38–CXCR4+CD86–) and LZ (B220+Fas+CD38–CXCR4–CD86+) GC B cells. (c) Quantification of absolute numbers of DZ centroblasts (Cγ1wt/cre;Smc3wt/wt (n=7) and Cγ1wt/cre;Smc3wt/– (n=6) mice) or LZ centrocytes (Cγ1wt/cre;Smc3wt/wt (n=6) and Cγ1wt/cre;Smc3wt/– (n=6) mice). (d) Cγ1wt/cre;Smc3fl/fl were crossed to Rosa26YFP/YFP to produce mice that express the yellow fluorescent protein when expressing the cre recombinase. (e) Gating strategy used in Cγ1wt/cre;Rosa26YFP;Smc3wt/– and Cγ1wt/cre;Rosa26YFP;Smc3wt/wt mice to identify YFP+ GC B cells. (f) Plot showing YFP+ GC B cells in the reporter Cγ1wt/cre;Rosa26YFP;Smc3wt/– (n=4) and Cγ1wt/cre;Rosa26YFP;Smc3wt/wt (n=9) mice. In (c) and (f) data are presented as mean +/- SD.
Extended Data Fig. 4 Smc3 haploinsufficient mice have impaired terminal differentiation.
a, Mice were immunized on day 0 with 100 μg NP-KLH:Alum 1:1 i.p., and received a boost on day 21 of 100 μg NP-CGG: Alum 1:1 i.p. Mice were bleed on days 0, 14, 26, 35 and 70, at which time were euthanized. b, Representative flow cytometry gating strategy followed to determine the presence of bone marrow plasma cells (BMPC, CD138+B220– cells) and long-lived BMPCs (NPcyt+IgG1cyt+CD138+B220– cells). c, Representative flow cytometry gating strategy followed to determine the percentage of IgM- and IgG1-expressing GC B cells. d, Frequency of IgG1+, IgM+, or IgG1+/IgM+ GC B cells in 4, 8 or 15 day SRBC immunized Cγ1wt/cre;Smc3wt/wt (n=5) or Cγ1wt/cre; Smc3wt/– (n=5) mice. Two tail unpaired t-test. All differences are statistically non-significant. e, Splenocytes from Cγ1wt/cre;Rosa26YFP;Smc3wt/wt (n=5) and Cγ1wt/cre;Rosa26YFP;Smc3wt/– (n=5) mice were cultured in the presence of IL-4, LPS and anti-CD40 antibody and PB/PC followed by flow cytometry and f, levels of active Cspase-3 were detected by flow cytometry using the presented gating strategy. g, Active Caspase-3 levels in B cells, PBs and PCs from ex vivo cultures. h-i, 8, 15 and 25-day immunized mice were sacrificed and splenocytes stained for GC markers. Nuclear staining of Irf4 was done afterwards. (h) Representative flow cytometry plots of IRF4+ GC B cells and (i) plots depicting the percentage of IRF4+ GC B cells in Cγ1wt/cre;Smc3wt/wt (n=5) and Cγ1wt/cre;Smc3wt/– (n=5) mice (at every time point). j, At each time point, mice were injected with 50 μg/kg EdU i.p. and sacrificed one hour later. Quantification of IRF4+ GC B cells in S-phase in the spleens of SRBC immunized mice for the indicated times in Cγ1wt/cre;Smc3wt/wt (n=5) and Cγ1wt/cre;Smc3wt/– (n=5) mice at every time point. **p<0.01, ***p<0.001 and ****p<0.0001. In (d), (i) and (j) data are presented as mean +/- SD. (i) and (j) two-way ANoVA and Sidak multi-comparison test was used.
Extended Data Fig. 5 Smc3wt/– centrocytes fail to upregulate GC exit genes and transition towards the plasma cell lineage.
a, Gene set enrichment analysis (GSEA) plots for TET2_KO and shKMT2D_DOWN. b, Sorting strategy used for single cell RNA-seq by BDRhapsody.
Extended Data Fig. 6 Smc3 haploinsufficiency disrupts intra-TAD interactions and GC exit gene looping.
a, Genome wide chromosomal interactivity was mapped by in situ Hi-C in Cγ1wt/cre;Smc3wt/wt (n=2) or Cγ1wt/cre;Smc3wt/– (n=2) centrocytes (B220+Fas+CD38–CXCR4–CD86+) from 8 day SRBC immunized mice. b, Cartoon depicting the definition of intra- and inter-TAD connectivity. c, Log2 fold change of inter- and intra-TAD interactions in Smc3 haploinsufficient versus wild-type centrocytes. p-value expresses significant difference between inter- and intra-TAD interactions. Wilcoxon rank-sum test was used, p<2.2×10–16. d, TAD boundary strength for Smc3 haploinsufficient centrocytes versus wild-type centrocytes by Hi-C. Wilcoxon rank-sum test was used, p<2.2×10–16. e, z-score of promoter short-range interactivity change plotted for genes which are downregulated, non-changing or upregulated in centrocyte RNA-seq of Smc3 haploinsufficient cells versus control. Wilcoxon rank-sum test was used, Down vs NS p<1.6×10–7, NS vs Up p=0.055 and Down vs Up p=0.00024. f, Gene set enrichment of compartment changes in Smc3 haploinsufficient vs wild-type centrocytes. Compartment C-scores are calculated in 100 kb intervals and annotated to genes based on the location of their TSS within these bins. Genes are ranked by Δ c-score and analyzed by GSEA. g, Gene set enrichment of enhancer-promoter interactivity changes in Smc3 haploinsufficient vs wild-type centrocytes. Enhancers are annotated to the nearest TSS (as explained in Methods), normalized contacts between a gene and all its enhancers are summed to get an aggregate promoter-enhancer looping score. Genes are ranked by Δ promoter-enhancer score between Smc3 haploinsufficient and wild-type mice and analyzed by GSEA. In (c) and (e) box plots show the median as center, first and third quartiles as the box hinges, and whiskers extend to the smallest and largest value no further than the 1.5 × interquartile range (IQR) away from the hinges.
Extended Data Fig. 7 Smc3 dosage does not impair B cell differentiation and is only required for GC exit.
a, Scheme depicting Smc3 conditional knockout model breeding strategy. b, Early B cell differentiation stages and markers used for identification. c, Representative flow cytometry plots of splenic CD3+ T cells, B220+ B cells and naïve B cells in CD19wt/cre;Smc3wt/wt (n=4), C19wt/cre;Smc3wt/– (n=7) and C19wt/cre;Smc3–/– (n=4) and d, quantification of ratio between splenic B220+/CD3+ cells in Cd19wt/cre;Smc3wt/wt (n=4), Cd19wt/cre;Smc3wt/– (n=7) and Cd19wt/cre;Smc3–/– (n=4) (****p<0.0001) or e, naïve B cells in Cd19wt/cre;Smc3wt/wt (n=5) and Cd19wt/cre;Smc3wt/– (n=9). f, Quantification of the percentage of bone marrow pre-BI cells, immature B cells, and splenic follicular B cells in Cd19wt/cre;Smc3wt/wt (n=4) and Cd19wt/cre;Smc3wt/– (n=5). NS: non-significant differences. In (d), (e), and (f) data are presented as mean +/- SD. In (d) and (e) one-way ANoVA and Tukey multi-comparison test was used, and two-way unpaired t-test in (f).
Extended Data Fig. 8 Haploinsufficiency of Smc3 cooperates with Bcl6 to induce lymphoma.
a, Bone marrow transplantation experimental design. Bone marrow cells were isolated from Cγ1wt/cre;Smc3wt/wt, Cγ1wt/cre;Smc3wt/–, IμBcl6;Cγ1wt/cre;Smc3wt/w and IμBcl6;Cγ1wt/cre;Smc3wt/– CD45.2 donors, and transplanted through tail vein injection into lethally irradiated CD45.1 mice. After engraftment, mice were immunized with SRBCs every 3 weeks for the duration of the experiment. Mice were euthanized when presented with signs of disease. b, Representative flow cytometry plots performed 300 days after engraftment showing that all B220+ are CD45.2+ and CD45.1–, demonstrating that all B cells in these mice come from the donor and not from the recipient. c, Representative allele occurrence was showed by performing PCR in DNA from tumors from IμBcl6;Cγ1wt/cre;Smc3wt/w and IμBcl6;Cγ1wt/cre;Smc3wt/– mice. One representative gel is shown out of 2 experiments performed.
Supplementary information
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Unprocessed gels.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 7
Statistical source data.
Source Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed gels.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed gels.
Rights and permissions
About this article
Cite this article
Rivas, M.A., Meydan, C., Chin, C.R. et al. Smc3 dosage regulates B cell transit through germinal centers and restricts their malignant transformation. Nat Immunol 22, 240–253 (2021). https://doi.org/10.1038/s41590-020-00827-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-00827-8
This article is cited by
-
Loss of CREBBP and KMT2D cooperate to accelerate lymphomagenesis and shape the lymphoma immune microenvironment
Nature Communications (2024)
-
Three-dimensional genome organization in immune cell fate and function
Nature Reviews Immunology (2023)
-
3D chromatin architecture and transcription regulation in cancer
Journal of Hematology & Oncology (2022)
-
OCT2 pre-positioning facilitates cell fate transition and chromatin architecture changes in humoral immunity
Nature Immunology (2021)