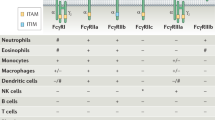
Abstract
Recent Ebola virus disease epidemics have highlighted the need for effective vaccines and therapeutics to prevent future outbreaks. Antibodies are clearly critical for control of this deadly disease; however, the specific mechanisms of action of protective antibodies have yet to be defined. In this Perspective we discuss the antibody features that correlate with in vivo protection during infection with Ebola virus, based on the results of a systematic and comprehensive study of antibodies directed against this virus. Although neutralization activity mediated by the Fab domains of the antibody is strongly correlated with protection, recruitment of immune effector functions by the Fc domain has also emerged as a complementary, and sometimes alternative, route to protection. For a subset of antibodies, Fc-mediated clearance and killing of infected cells seems to be the main driver of protection after exposure and mirrors observations in vaccination studies. Continued analysis of antibodies that achieve protection partially or wholly through Fc-mediated functions, the precise functions required, the intersection with specificity and the importance of these functions in different animal models is needed to identify and begin to capitalize on Fc-mediated protection in vaccines and therapeutics alike.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Carter, P. J. & Lazar, G. A. Next generation antibody drugs: pursuit of the ‘high-hanging fruit’. Nat. Rev. Drug Discov. 17, 197–223 (2018).
Salazar, G., Zhang, N., Fu, T.-M. & An, Z. Antibody therapies for the prevention and treatment of viral infections. NPJ Vaccines 2, 19 (2017).
Lu, L. L., Suscovich, T. J., Fortune, S. M. & Alter, G. Beyond binding: antibody effector functions in infectious diseases. Nat. Rev. Immunol. 18, 46–61 (2018).
Casadevall, A. Antibody-based therapies for emerging infectious diseases. Emerg. Infect. Dis. 2, 200–208 (1996).
Corti, D. et al. Prophylactic and postexposure efficacy of a potent human monoclonal antibody against MERS coronavirus. Proc. Natl. Acad. Sci. USA 112, 10473–10478 (2015).
Burton, D. R. & Saphire, E. O. Swift antibodies to counter emerging viruses. Proc. Natl. Acad. Sci. USA 112, 10082–10083 (2015).
Davey, R. T. Jr. et al. PREVAIL II Writing Group et al. A randomized, controlled trial of ZMapp for Ebola virus infection. N. Engl. J. Med. 375, 1448–1456 (2016).
Sondermann, P. & Szymkowski, D. E. Harnessing Fc receptor biology in the design of therapeutic antibodies. Curr. Opin. Immunol. 40, 78–87 (2016).
Nimmerjahn, F. Translating inhibitory Fc receptor biology into novel therapeutic approaches. J. Clin. Immunol. 36, 83–87 (2016).
Hogarth, P. M. & Pietersz, G. A. Fc receptor-targeted therapies for the treatment of inflammation, cancer and beyond. Nat. Rev. Drug Discov. 11, 311–331 (2012).
Howes, S. C., Koning, R. I. & Koster, A. J. Correlative microscopy for structural microbiology. Curr. Opin. Microbiol. 43, 132–138 (2018).
Hampton, C. M. et al. Correlated fluorescence microscopy and cryo-electron tomography of virus-infected or transfected mammalian cells. Nat. Protoc. 12, 150–167 (2017).
Kellner, C., Derer, S., Valerius, T. & Peipp, M. Boosting ADCC and CDC activity by Fc engineering and evaluation of antibody effector functions. Methods 65, 105–113 (2014).
Bournazos, S., DiLillo, D. J. & Ravetch, J. V. The role of Fc-FcγR interactions in IgG-mediated microbial neutralization. J. Exp. Med. 212, 1361–1369 (2015).
Lewis, G. K. Role of Fc-mediated antibody function in protective immunity against HIV-1. Immunology 142, 46–57 (2014).
Treffers, L. W. et al. Genetic variation of human neutrophil Fcγ receptors and SIRPα in antibody-dependent cellular cytotoxicity towards cancer cells. Eur. J. Immunol. 48, 344–354 (2018).
Bakema, J. E. & van Egmond, M. Fc receptor-dependent mechanisms of monoclonal antibody therapy of cancer. Curr. Top. Microbiol. Immunol. 382, 373–392 (2014).
Ram, S. et al. Utilizing complement evasion strategies to design complement-based antibacterial immunotherapeutics: Lessons from the pathogenic Neisseriae. Immunobiology 221, 1110–1123 (2016).
Bournazos, S. et al. Broadly neutralizing anti-HIV-1 antibodies require Fc effector functions for in vivo activity. Cell 158, 1243–1253 (2014).
Almagro, J. C., Daniels-Wells, T. R., Perez-Tapia, S. M. & Penichet, M. L. Progress and challenges in the design and clinical development of antibodies for cancer therapy. Front. Immunol. 8, 1751 (2018).
Saphire, E. O. et al. Viral Hemorrhagic Fever Immunotherapeutic Consortium. Systematic analysis of monoclonal antibodies against Ebola virus GP defines features that contribute to protection. Cell 174, 938–952 (2018).
Maruyama, T. et al. Ebola virus can be effectively neutralized by antibody produced in natural human infection. J. Virol. 73, 6024–6030 (1999).
Lee, J. E. et al. Structure of the Ebola virus glycoprotein bound to an antibody from a human survivor. Nature 454, 177–182 (2008).
Parren, P. W. H. I., Geisbert, T. W., Maruyama, T., Jahrling, P. B. & Burton, D. R. Pre- and postexposure prophylaxis of Ebola virus infection in an animal model by passive transfer of a neutralizing human antibody. J. Virol. 76, 6408–6412 (2002).
Oswald, W. B. et al. Neutralizing antibody fails to impact the course of Ebola virus infection in monkeys. PLoS Pathog. 3, e9 (2007).
Dye, J. M. et al. Postexposure antibody prophylaxis protects nonhuman primates from filovirus disease. Proc. Natl. Acad. Sci. USA 109, 5034–5039 (2012).
Qiu, X. et al. Ebola GP-specific monoclonal antibodies protect mice and guinea pigs from lethal Ebola virus infection. PLoS Negl. Trop. Dis. 6, e1575 (2012).
Qiu, X. et al. Successful treatment of Ebola virus-infected cynomolgus macaques with monoclonal antibodies. Sci. Transl. Med. 4, 138ra81 (2012).
Murin, C. D. et al. Structures of protective antibodies reveal sites of vulnerability on Ebola virus. Proc. Natl. Acad. Sci. USA 111, 17182–17187 (2014).
Audet, J. et al. Molecular characterization of the monoclonal antibodies composing ZMAb: a protective cocktail against Ebola virus. Sci. Rep. 4, 6881 (2014).
Olinger, G. G. Jr. et al. Delayed treatment of Ebola virus infection with plant-derived monoclonal antibodies provides protection in rhesus macaques. Proc. Natl. Acad. Sci. USA 109, 18030–18035 (2012).
Pettitt, J. et al. Therapeutic intervention of Ebola virus infection in rhesus macaques with the MB-003 monoclonal antibody cocktail. Sci. Transl. Med. 5, 199ra113 (2013).
Pallesen, J. et al. Structures of Ebola virus GP and sGP in complex with therapeutic antibodies. Nat. Microbiol. 1, 16128 (2016).
Wilson, J. A. et al. Epitopes involved in antibody-mediated protection from Ebola virus. Science 287, 1664–1666 (2000).
Davidson, E. et al. Mechanism of binding to Ebola virus glycoprotein by the ZMapp, ZMAb, and MB-003 cocktail antibodies. J. Virol. 89, 10982–10992 (2015).
Hood, C. L. et al. Biochemical and structural characterization of cathepsin L-processed Ebola virus glycoprotein: implications for viral entry and immunogenicity. J. Virol. 84, 2972–2982 (2010).
Schornberg, K. et al. Role of endosomal cathepsins in entry mediated by the Ebola virus glycoprotein. J. Virol. 80, 4174–4178 (2006).
Zeitlin, L. et al. Enhanced potency of a fucose-free monoclonal antibody being developed as an Ebola virus immunoprotectant. Proc. Natl. Acad. Sci. USA 108, 20690–20694 (2011).
Strasser, R. et al. Generation of glyco-engineered Nicotiana benthamiana for the production of monoclonal antibodies with a homogeneous human-like N-glycan structure. Plant Biotechnol. J. 6, 392–402 (2008).
Ackerman, M. E. et al. Natural variation in Fc glycosylation of HIV-specific antibodies impacts antiviral activity. J. Clin. Invest. 123, 2183–2192 (2013).
Mahan, A. E. et al. Antigen-specific antibody glycosylation is regulated via vaccination. PLoS Pathog. 12, e1005456 (2016).
Nimmerjahn, F., Anthony, R. M. & Ravetch, J. V. Agalactosylated IgG antibodies depend on cellular Fc receptors for in vivo activity. Proc. Natl. Acad. Sci. USA 104, 8433–8437 (2007).
Arnold, J. N., Wormald, M. R., Sim, R. B., Rudd, P. M. & Dwek, R. A. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu. Rev. Immunol. 25, 21–50 (2007).
Anthony, R. M. & Nimmerjahn, F. The role of differential IgG glycosylation in the interaction of antibodies with FcγRs in vivo. Curr. Opin. Organ Transplant. 16, 7–14 (2011).
Jefferis, R. Glycosylation of recombinant antibody therapeutics. Biotechnol. Prog. 21, 11–16 (2005).
Popp, O. et al. Development of a pre-glycoengineered CHO-K1 host cell line for the expression of antibodies with enhanced Fc mediated effector function. MAbs 10, 290–303 (2018).
Jiang, X.-R. et al. Advances in the assessment and control of the effector functions of therapeutic antibodies. Nat. Rev. Drug Discov. 10, 101–111 (2011).
Schuster, M. et al. Improved effector functions of a therapeutic monoclonal Lewis Y-specific antibody by glycoform engineering. Cancer Res. 65, 7934–7941 (2005).
Yamane-Ohnuki, N. & Satoh, M. Production of therapeutic antibodies with controlled fucosylation. MAbs 1, 230–236 (2009).
Qiu, X. et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 514, 47–53 (2014).
Corti, D. et al. Protective monotherapy against lethal Ebola virus infection by a potently neutralizing antibody. Science 351, 1339–1342 (2016).
Pascal, K. E. et al. Development of clinical-stage human monoclonal antibodies that treat advanced Ebola virus disease in non-human primates. J. Infect. Dis. https://doi.org/10.1093/infdis/jiy285 (2018).
Saphire, E. O. et al. How to turn competitors into collaborators. Nature 541, 283–285 (2017).
Halfmann, P. et al. Generation of biologically contained Ebola viruses. Proc. Natl. Acad. Sci. USA 105, 1129–1133 (2008).
Wong, A. C., Sandesara, R. G., Mulherkar, N., Whelan, S. P. & Chandran, K. A forward genetic strategy reveals destabilizing mutations in the Ebolavirus glycoprotein that alter its protease dependence during cell entry. J. Virol. 84, 163–175 (2010).
Ackerman, M. E., Barouch, D. H. & Alter, G. Systems serology for evaluation of HIV vaccine trials. Immunol. Rev. 275, 262–270 (2017).
Alter, G., Malenfant, J. M. & Altfeld, M. CD107a as a functional marker for the identification of natural killer cell activity. J. Immunol. Methods 294, 15–22 (2004).
Shedlock, D. J. et al. Antibody-mediated neutralization of Ebola virus can occur by two distinct mechanisms. Virology 401, 228–235 (2010).
Flyak, A. I. et al. Mechanism of human antibody-mediated neutralization of Marburg virus. Cell 160, 893–903 (2015).
King, L. B. et al. The Marburgvirus-neutralizing human monoclonal antibody MR191 targets a conserved site to block virus receptor binding. Cell Host Microbe 23, 101–109.e4 (2018).
Hashiguchi, T. et al. Structural basis for Marburg virus neutralization by a cross-reactive human antibody. Cell 160, 904–912 (2015).
Chandran, K., Sullivan, N. J., Felbor, U., Whelan, S. P. & Cunningham, J. M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science 308, 1643–1645 (2005).
Miller, E. H. et al. Ebola virus entry requires the host-programmed recognition of an intracellular receptor. EMBO J. 31, 1947–1960 (2012).
Gong, X. et al. Structural Insights into the Niemann-Pick C1 (NPC1)-mediated cholesterol transfer and Ebola infection. Cell 165, 1467–1478 (2016).
Kaletsky, R. L., Simmons, G. & Bates, P. Proteolysis of the Ebola virus glycoproteins enhances virus binding and infectivity. J. Virol. 81, 13378–13384 (2007).
Mohan, G. S., Li, W., Ye, L., Compans, R. W. & Yang, C. Antigenic subversion: a novel mechanism of host immune evasion by Ebola virus. PLoS Pathog. 8, e1003065 (2012).
Barba-Spaeth, G. et al. Structural basis of potent Zika-dengue virus antibody cross-neutralization. Nature 536, 48–53 (2016).
Corti, D. et al. A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science 333, 850–856 (2011).
Corti, D. et al. Cross-neutralization of four paramyxoviruses by a human monoclonal antibody. Nature 501, 439–443 (2013).
de Alwis, R. et al. Identification of human neutralizing antibodies that bind to complex epitopes on dengue virions. Proc. Natl. Acad. Sci. USA 109, 7439–7444 (2012).
Huang, J. et al. Identification of a CD4-binding-site antibody to HIV that evolved near-pan neutralization breadth. Immunity 45, 1108–1121 (2016).
Burton, D. R. & Mascola, J. R. Antibody responses to envelope glycoproteins in HIV-1 infection. Nat. Immunol. 16, 571–576 (2015).
Zhou, Y. & Sullivan, N. J. Immunology and evolvement of the adenovirus prime, MVA boost Ebola virus vaccine. Curr. Opin. Immunol. 35, 131–136 (2015).
Stanley, D. A. et al. Chimpanzee adenovirus vaccine generates acute and durable protective immunity against ebolavirus challenge. Nat. Med. 20, 1126–1129 (2014).
Qiu, X. et al. Mucosal immunization of cynomolgus macaques with the VSVΔG/ZEBOVGP vaccine stimulates strong ebola GP-specific immune responses. PLoS One 4, e5547 (2009).
Liu, Q. et al. Antibody-dependent-cellular-cytotoxicity-inducing antibodies significantly affect the post-exposure treatment of Ebola virus infection. Sci. Rep. 7, 45552 (2017).
Cimini, E. et al. Different features of Vδ2 T and NK cells in fatal and non-fatal human Ebola infections. PLoS Negl. Trop. Dis. 11, e0005645 (2017).
Gillis, C. M. et al. Mechanisms of anaphylaxis in human low-affinity IgG receptor locus knock-in mice. J. Allergy Clin. Immunol. 139, 1253–1265.e14 (2017).
Gillis, C., Gouel-Chéron, A., Jönsson, F. & Bruhns, P. Contribution of human FcγRs to disease with evidence from human polymorphisms and transgenic animal studies. Front. Immunol. 5, 254 (2014).
Smith, P., DiLillo, D. J., Bournazos, S., Li, F. & Ravetch, J. V. Mouse model recapitulating human Fcγ receptor structural and functional diversity. Proc. Natl. Acad. Sci. USA 109, 6181–6186 (2012).
Bournazos, S., DiLillo, D. J. & Ravetch, J. V. humanized mice to study FcγR function. Curr. Top. Microbiol. Immunol. 382, 237–248 (2014).
Nimmerjahn, F. & Ravetch, J. V. Fcγ receptors as regulators of immune responses. Nat. Rev. Immunol. 8, 34–47 (2008).
Sheeley, D. M., Merrill, B. M. & Taylor, L. C. Characterization of monoclonal antibody glycosylation: comparison of expression systems and identification of terminal alpha-linked galactose. Anal. Biochem. 247, 102–110 (1997).
Hills, A. E., Patel, A., Boyd, P. & James, D. C. Metabolic control of recombinant monoclonal antibody N-glycosylation in GS-NS0 cells. Biotechnol. Bioeng. 75, 239–251 (2001).
Baker, K. N. et al. Metabolic control of recombinant protein N-glycan processing in NS0 and CHO cells. Biotechnol. Bioeng. 73, 188–202 (2001).
Gunn, B. M. et al. Dissecting the role of Fc and Fab mediated functions in Ebola virus specific immunity. Cell Host Microbe 24, 221–233 (2018).
Shields, R. L. et al. Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human Fcgamma RIII and antibody-dependent cellular toxicity. J. Biol. Chem. 277, 26733–26740 (2002).
Chung, A. W. et al. Identification of antibody glycosylation structures that predict monoclonal antibody Fc-effector function. AIDS 28, 2523–2530 (2014).
Mahan, A. E. et al. Correction: antigen-specific antibody glycosylation is regulated via vaccination. PLoS Pathog. 12, e1005694 (2016).
Sneddon, L. U., Halsey, L. G. & Bury, N. R. Considering aspects of the 3Rs principles within experimental animal biology. J. Exp. Biol. 220, 3007–3016 (2017).
Jefferis, R. Isotype and glycoform selection for antibody therapeutics. Arch. Biochem. Biophys. 526, 159–166 (2012).
Hessell, A. J. et al. Fc receptor but not complement binding is important in antibody protection against HIV. Nature 449, 101–104 (2007).
Horton, H. M. et al. Antibody-mediated coengagement of FcγRIIb and B cell receptor complex suppresses humoral immunity in systemic lupus erythematosus. J. Immunol. 186, 4223–4233 (2011).
Chu, S. Y. et al. Reduction of total IgE by targeted coengagement of IgE B-cell receptor and FcγRIIb with Fc-engineered antibody. J. Allergy Clin. Immunol. 129, 1102–1115 (2012).
Nose, M. & Wigzell, H. Biological significance of carbohydrate chains on monoclonal antibodies. Proc. Natl. Acad. Sci. USA 80, 6632–6636 (1983).
Leatherbarrow, R. J. et al. Effector functions of a monoclonal aglycosylated mouse IgG2a: binding and activation of complement component C1 and interaction with human monocyte Fc receptor. Mol. Immunol. 22, 407–415 (1985).
Tobinai, K., Klein, C., Oya, N. & Fingerle-Rowson, G. A review of obinutuzumab (GA101), a novel type II anti-CD20 monoclonal antibody, for the treatment of patients with B-cell malignancies. Adv. Ther. 34, 324–356 (2017).
Beck, A. & Reichert, J. M. Marketing approval of mogamulizumab: a triumph for glyco-engineering. MAbs 4, 419–425 (2012).
Hoy, S. M. Obinutuzumab: a review of its use in patients with chronic lymphocytic leukaemia. Drugs 75, 285–296 (2015).
Ghazi, A., Trikha, A. & Calhoun, W. J. Benralizumab–a humanized mAb to IL-5Rα with enhanced antibody-dependent cell-mediated cytotoxicity–a novel approach for the treatment of asthma. Expert Opin. Biol. Ther. 12, 113–118 (2012).
Robak, T. GA-101, a third-generation, humanized and glyco-engineered anti-CD20 mAb for the treatment of B-cell lymphoid malignancies. Curr. Opin. Investig. Drugs 10, 588–596 (2009).
Lazar, G. A. et al. Engineered antibody Fc variants with enhanced effector function. Proc. Natl. Acad. Sci. USA 103, 4005–4010 (2006).
Diebolder, C. A. et al. Complement is activated by IgG hexamers assembled at the cell surface. Science 343, 1260–1263 (2014).
Shields, R. L. et al. High resolution mapping of the binding site on human IgG1 for FcγRI, FcγRII, FcγRIII, and FcRn and design of IgG1 variants with improved binding to the FcγR. J. Biol. Chem. 276, 6591–6604 (2001).
Stavenhagen, J. B. et al. Fc optimization of therapeutic antibodies enhances their ability to kill tumor cells in vitro and controls tumor expansion in vivo via low-affinity activating Fcγ receptors. Cancer Res. 67, 8882–8890 (2007).
Richards, J. O. et al. Optimization of antibody binding to FcγRIIa enhances macrophage phagocytosis of tumor cells. Mol. Cancer Ther. 7, 2517–2527 (2008).
Jurczak, W. et al. Phase IIa study of single-agent MOR208 in patients with relapsed or refractory B-cell non-Hodgkinas lymphoma. Blood 126, 1528 (2015).
Kellner, C. et al. The Fc-engineered CD19 antibody MOR208 (XmAb5574) induces natural killer cell-mediated lysis of acute lymphoblastic leukemia cells from pediatric and adult patients. Leukemia 27, 1595–1598 (2013).
Zalevsky, J. et al. The impact of Fc engineering on an anti-CD19 antibody: increased Fcγ receptor affinity enhances B-cell clearing in nonhuman primates. Blood 113, 3735–3743 (2009).
Kumar, A. et al. A phase 1 dose-escalation study of XmAb® 2513 in patients with relapsed or refractory Hodgkin lymphoma. Br. J. Haematol. 168, 902–904 (2015).
Lee, E. M. et al. Efficacy of an Fc-modified anti-CD123 antibody (CSL362) combined with chemotherapy in xenograft models of acute myelogenous leukemia in immunodeficient mice. Haematologica 100, 914–926 (2015).
Li, G. et al. Dual functional monoclonal antibody PF-04605412 targets integrin α5β1 and elicits potent antibody-dependent cellular cytotoxicity. Cancer Res. 70, 10243–10254 (2010).
de Jong, R. N. et al. A novel platform for the potentiation of therapeutic antibodies based on antigen-dependent formation of IgG hexamers at the cell surface. PLoS Biol. 14, e1002344 (2016).
Dall’Acqua, W. F., Kiener, P. A. & Wu, H. Properties of human IgG1s engineered for enhanced binding to the neonatal Fc receptor (FcRn). J. Biol. Chem. 281, 23514–23524 (2006).
Borrok, M. J. et al. pH-dependent binding engineering reveals an FcRn affinity threshold that governs IgG recycling. J. Biol. Chem. 290, 4282–4290 (2015).
Yu, X.-Q. et al. Safety, tolerability, and pharmacokinetics of MEDI4893, an investigational, extended-half-life, anti-Staphylococcus aureus Alpha-toxin human monoclonal antibody, in healthy adults. Antimicrob. Agents Chemother. 61, (2016).
Gaudinski, M. R. et al. Safety and pharmacokinetics of the Fc-modified HIV-1 human monoclonal antibody VRC01LS: a Phase 1 open-label clinical trial in healthy adults. PLoS Med. 15, e1002493 (2018).
Lewis, G. K. et al. Beyond viral neutralization. AIDS Res. Hum. Retroviruses 33, 760–764 (2017).
Corey, L. et al. Immune correlates of vaccine protection against HIV-1 acquisition. Sci. Transl. Med. 7, 310rv7 (2015).
Henry Dunand, C. J. et al. Both neutralizing and non-neutralizing human H7N9 influenza vaccine-induced monoclonal antibodies confer protection. Cell Host Microbe 19, 800–813 (2016).
Bootz, A. et al. Protective capacity of neutralizing and non-neutralizing antibodies against glycoprotein B of cytomegalovirus. PLoS Pathog. 13, e1006601 (2017).
Horwitz, J. A. et al. Non-neutralizing antibodies alter the course of HIV-1 infection in vivo. Cell 170, 637–648.e10 (2017).
Lee, J. E. et al. Complex of a protective antibody with its ebola virus GP peptide epitope: unusual features of a Vλx light chain. J. Mol. Biol. 375, 202–216 (2008).
Misasi, J. et al. Structural and molecular basis for Ebola virus neutralization by protective human antibodies. Science 351, 1343–1346 (2016).
Acknowledgements
We acknowledge U19AI107962 (a Center of Excellence in Translational Research (CETR) award from NIAID of the US National Institutes of Health), and the members of the Viral Hemorrhagic Fever Immunotherapeutic Consortium who contributed antibodies (many of which were unpublished) and analytical efforts on the project, including K.G. Andersen, A.B. Ward and D.R. Burton; R. Ahmed and C.W. Davis (Emory University); M.J. Aman (Integrated BioTherapeutics); A. Bukreyev (University of Texas Medical Branch); K. Chandran and J.R. Lai (Albert Einstein College of Medicine); J.E. Crowe, Jr. (Vanderbilt University School of Medicine); J.M. Dye (USAMRIID); B. Doranz (Integral Molecular); H. Feldmann (NIAID Rocky Mountain Lab); G. Georgiou (University of Texas); G.G. Olinger and L. Hensley (NIAID Integrated Research Facility); Y. Kawaoka (University of Wisconsin); G.P. Kobinger (Université Laval); K. Wagh and B. Korber (Los Alamos National Labs); F. Krammer (Icahn School of Medicine at Mt. Sinai); C.A. Kyratsous (Regeneron); C. Nykiforuk (Emergent Biosciences); X. Qiu (Public Health Agency of Canada); A. Sprecher (Médecins Sans Frontières); A.R. Townsend (University of Oxford); V. Volchkov (Université Lyon); L.M. Walker (Adimab); C.-I. Wang (A*STAR); L. Zeitlin (Mapp Biopharmaceutical); and the students, postdoctoral fellows, technicians and staff scientists in their labs, as well as J.V. Ravetch (Rockefeller University) for discussions. This is manuscript #29662 from Scripps Research.
Author information
Authors and Affiliations
Contributions
E.O.S., S.L.S., B.M.G. and J.C.M. prepared the figures; E.O.S., S.L.S., B.M.G. and G.A. analyzed data and drafted and edited the manuscript; B.M.G. performed the glycan clustering analysis; and E.O.S. and G.A. secured funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saphire, E.O., Schendel, S.L., Gunn, B.M. et al. Antibody-mediated protection against Ebola virus. Nat Immunol 19, 1169–1178 (2018). https://doi.org/10.1038/s41590-018-0233-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-018-0233-9
This article is cited by
-
Direct enhancement of viral neutralising antibody potency by the complement system: a largely forgotten phenomenon
Cellular and Molecular Life Sciences (2024)
-
NIAID/SMB Workshop on Multiscale Modeling of Infectious and Immune-Mediated Diseases
Bulletin of Mathematical Biology (2024)
-
Recombinant vaccines in 2022: a perspective from the cell factory
Microbial Cell Factories (2022)
-
A single immunization with a modified vaccinia Ankara vectored vaccine producing Sudan virus-like particles protects from lethal infection
npj Vaccines (2022)
-
Glycan shield of the ebolavirus envelope glycoprotein GP
Communications Biology (2022)