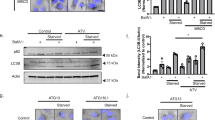
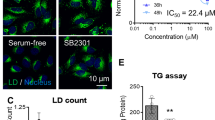
Abstract
Autophagy mediates the degradation of damaged proteins, organelles and pathogens, and plays a key role in health and disease. Thus, the identification of new mechanisms involved in the regulation of autophagy is of major interest. In particular, little is known about the role of lipids and lipid-binding proteins in the early steps of autophagosome biogenesis. Using target-agnostic, high-content, image-based identification of indicative phenotypic changes induced by small molecules, we have identified autogramins as a new class of autophagy inhibitor. Autogramins selectively target the recently discovered cholesterol transfer protein GRAM domain-containing protein 1A (GRAMD1A, which had not previously been implicated in autophagy), and directly compete with cholesterol binding to the GRAMD1A StART domain. GRAMD1A accumulates at sites of autophagosome initiation, affects cholesterol distribution in response to starvation and is required for autophagosome biogenesis. These findings identify a new biological function of GRAMD1A and a new role for cholesterol in autophagy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files. Additional raw data associated with all figures are available from the corresponding authors upon reasonable request. The atomic structure of the StART domain of GRAMD1C was deposited in the Protein Data Bank under the accession number 6GN5.
References
Mizushima, N., Levine, B., Cuervo, A. M. & Klionsky, D. J. Autophagy fights disease through cellular self-digestion. Nature 451, 1069–1075 (2008).
Mizushima, N., Yoshimori, T. & Ohsumi, Y. The role of Atg proteins in autophagosome formation. Ann. Rev. Cell. Dev. Biol. 27, 107–132 (2011).
Rubinsztein, D. C., Codogno, P. & Levine, B. Autophagy modulation as a potential therapeutic target for diverse diseases. Nat. Rev. Drug Discov. 11, 709–730 (2012).
White, E. Deconvoluting the context-dependent role for autophagy in cancer. Nat. Rev. Cancer 12, 401–410 (2012).
Laraia, L. et al. Discovery of novel cinchona-alkaloid-inspired oxazatwistane autophagy inhibitors. Angew. Chem. Int. Ed. 56, 2145–2150 (2017).
Robke, L. et al. Phenotypic identification of a novel autophagy inhibitor chemotype targeting lipid kinase VPS34. Angew. Chem. Int. Ed. 56, 8153–8157 (2017).
Robke, L. et al. Discovery of the novel autophagy inhibitor aumitin that targets mitochondrial complex I. Chem. Sci. 9, 3014–3022 (2018).
Dall’Armi, C., Devereaux, K. A. & Di Paolo, G. The role of lipids in the control of autophagy. Curr. Biol. 23, R33–R45 (2013).
Nascimbeni, A. C., Codogno, P. & Morel, E. Phosphatidylinositol‐3‐phosphate in the regulation of autophagy membrane dynamics. FEBS J. 284, 1267–1278 (2017).
Petiot, A., Ogier-Denis, E., Blommaart, E. F. C., Meijer, A. J. & Codogno, P. Distinct classes of phosphatidylinositol 3′-kinases are involved in signaling pathways that control macroautophagy in HT-29 cells. J. Biol. Chem. 275, 992–998 (2000).
Kihara, A., Noda, T., Ishihara, N. & Ohsumi, Y. Two distinct Vps34 phosphatidylinositol 3-kinase complexes function in autophagy and carboxypeptidase Y sorting in Saccharomyces cerevisiae. J. Cell Biol. 152, 519–530 (2001).
Dooley, H. C. et al. WIPI2 links LC3 conjugation with PI3P, autophagosome formation, and pathogen clearance by recruiting Atg12–5-16L1. Mol. Cell 55, 238–252 (2014).
Konstantinidis, G., Sievers, S. & Wu, Y.-W. in Autophagy in Differentiation and Tissue Maintenance. Methods in Molecular Biology Vol. 1854 (ed. Turksen, K.) 1–9 (Humana Press, 2018).
Balgi, A. D. et al. Screen for chemical modulators of autophagy reveals novel therapeutic inhibitors of mTORC1 signaling. PLoS ONE 4, e7124 (2009).
Peppard, J. V. et al. Identifying small molecules which inhibit autophagy: a phenotypic screen using image-based high-content cell analysis. Curr. Chem. Genom. Trans. Med. 8, 3–15 (2014).
Liu, J. et al. Beclin1 controls the levels of p53 by regulating the deubiquitination activity of USP10 and USP13. Cell 147, 223–234 (2011).
Wong, L. H. & Levine, T. P. Lipid transfer proteins do their thing anchored at membrane contact sites… but what is their thing? Biochem. Soc. Trans. 44, 517–527 (2016).
Khafif, M., Cottret, L., Balagué, C. & Raffaele, S. Identification and phylogenetic analyses of VASt, an uncharacterized protein domain associated with lipid-binding domains in eukaryotes. BMC Bioinformatics 15, 222 (2014).
Doerks, T., Strauss, M., Brendel, M. & Bork, P. GRAM, a novel domain in glucosyltransferases, myotubularins and other putative membrane-associated proteins. Trends Biochem. Sci. 25, 483–485 (2000).
Begley, M. J. et al. Crystal structure of a phosphoinositide phosphatase, MTMR2: insights into myotubular myopathy and Charcot-Marie-Tooth syndrome. Mol. Cell 12, 1391–1402 (2003).
Horenkamp, F. A., Valverde, D. P., Nunnari, J. & Reinisch, K. M. Molecular basis for sterol transport by StART‐like lipid transfer domains. EMBO J. 37, e98002 (2018).
Tong, J., Manik, M. K. & Im, Y. J. Structural basis of sterol recognition and nonvesicular transport by lipid transfer proteins anchored at membrane contact sites. Proc. Natl Acad. Sci. USA 115, E856–E865 (2018).
Sandhu, J. et al. Aster proteins facilitate nonvesicular plasma membrane to ER cholesterol transport in mammalian cells. Cell 175, 514–529 (2018).
Robers, M. B. et al. Target engagement and drug residence time can be observed in living cells with BRET. Nat. Commun. 6, 10091 (2015).
Jafari, R. et al. The cellular thermal shift assay for evaluating drug target interactions in cells. Nat. Protoc. 9, 2100–2122 (2014).
Holm, L. & Rosenström, P. Dali server: conservation mapping in 3D. Nucleic Acids Res. 38, W545–W549 (2010).
Gatta, A. T. et al. A new family of StART domain proteins at membrane contact sites has a role in ER–PM sterol transport. eLife 4, e07253 (2015).
Kimura, S., Noda, T. & Yoshimori, T. Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3. Autophagy 3, 452–460 (2007).
Blommaart, E. F. C., Krause, U., Schellens, J. P., Vreeling‐Sindelárová, H. & Meijer, A. J. The phosphatidylinositol 3‐kinase inhibitors wortmannin and LY294002 inhibit autophagy in isolated rat hepatocytes. Eur. J. Biochem. 243, 240–246 (2004).
Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat. Rev. Mol. Cell Biol. 9, 125–138 (2008).
Wijdeven, R. H. et al. Cholesterol and ORP1L-mediated ER contact sites control autophagosome transport and fusion with the endocytic pathway. Nat. Commun. 7, 11808 (2016).
Sarkar, S. et al. Impaired autophagy in the lipid-storage disorder Niemann-Pick type C1 disease. Cell Rep. 5, 1302–1315 (2013).
Murley, A. et al. Ltc1 is an ER-localized sterol transporter and a component of ER–mitochondria and ER–vacuole contacts. J. Cell Biol. 209, 539–548 (2015).
Elbaz-Alon, Y. et al. Lam6 regulates the extent of contacts between organelles. Cell Rep. 12, 7–14 (2015).
Murley, A. et al. Sterol transporters at membrane contact sites regulate TORC1 and TORC2 signaling. J. Cell Biol. 216, 2679–2689 (2017).
Besprozvannaya, M. et al. GRAM domain proteins specialize functionally distinct ER–PM contact sites in human cells. eLife 7, e31019 (2018).
Punnonen, E.-L., Pihakaski, K., Mattila, K., Lounatmaa, K. & Hirsimäki, P. Intramembrane particles and filipin labelling on the membranes of autophagic vacuoles and lysosomes in mouse liver. Cell. Tissue Res. 258, 269–276 (1989).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Tyanova, S. & Cox, J. in Cancer Systems Biology: Methods and Protocols (ed. von Stechow, L.) 133–148 (Springer, 2018).
Hashimoto, Y., Zhang, S. & Blissard, G. W. Ao38, a new cell line from eggs of the black witch moth, Ascalapha odorata (Lepidoptera: Noctuidae), is permissive for AcMNPV infection and produces high levels of recombinant proteins. BMC Biotechnol. 10, 50 (2010).
Oishi, H., Takaoka, Y., Nishimaki-Mogami, T., Saito, H. & Ueda, M. A novel nuclear receptor ligand, digoxigenin, is a selective antagonist of liver-X-receptors. Chem. Lett. 46, 313–314 (2016).
Kernstock, R. M. & Girotti, A. W. Lipid transfer protein binding of unmodified natural lipids as assessed by surface plasmon resonance methodology. Anal. Biochem. 365, 111–121 (2007).
Fang, J., Rand, K. D., Beuning, P. J. & Engen, J. R. False EX1 signatures caused by sample carryover during HX MS analyses. Int. J. Mass Spec. 302, 19–25 (2011).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Šali, A. & Blundell, T. L. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 234, 779–815 (1993).
Jones, G., Willett, P., Glen, R. C., Leach, A. R. & Taylor, R. Development and validation of a genetic algorithm for flexible docking. J. Mol. Biol. 267, 727–748 (1997).
Laskowski, R. A. & Swindells, M. B. LigPlot+: multiple ligand–protein interaction diagrams for drug discovery. J. Chem. Inf. Mod. 51, 2778–2786 (2011).
Itakura, E. & Mizushima, N. Characterization of autophagosome formation site by a hierarchical analysis of mammalian Atg proteins. Autophagy 6, 764–776 (2010).
de Chaumont, F. et al. Icy: an open bioimage informatics platform for extended reproducible research. Nat. Methods 9, 690–696 (2012).
Acknowledgements
This work was supported by the Max Planck Society (H.W.), DFG grant SPP 1623, ERC (ChemBioAP), Vetenskapsrådet (2018-04585) and the Knut and Alice Wallenberg Foundation (Y.-W.W). L.Laraia was supported by a fellowship from the Alexander von Humboldt Stiftung. D.P.C. is supported by a fellowship from the Canadian Institute of Health Research (MFE-152550). We thank S. Sievers and the Compound Management and Screening Center (COMAS), Dortmund, Germany, for compound screening. We thank R. Gasper-Schönenbrücher, K. Estel and the beamline staff for help with data collection at the SLS, Villigen, Switzerland. We thank S. Tooze for the kind gift of EGFP–WIPI2b cells. We acknowledge the Biochemical Imaging Center (BICU) at Umeå University and the National Microscopy Infrastructure, NMI (VR-RFI 2016-00968) for providing assistance in microscopy.
Author information
Authors and Affiliations
Contributions
L.Laraia carried out the analog and probe synthesis, the structure–activity relationship analysis, the initial biological compound validation and the proteomic target identification and initial target validation. A.F. carried out the cloning, expression and purification of all recombinant proteins as well as all fluorescence polarization, DSF, PIP-binding and crystallography experiments. A.F., A.B. and M.M. performed HDX-MS experiments. D.P.C. carried out cell biological characterization of GRAMD1A. G.K. and L.K. carried out autogramin validation experiments. W.H. and H.K. carried out the synthesis of the fluorescent probe. B.S. performed nanoBRET experiments. N.E. and L.Li carried out the cholesterol transfer experiments. M.D. performed AFM experiments. R.W. supervised cholesterol transfer and AFM experiments. P.J. carried out the MS proteomics analysis. I.R.V. analyzed crystallographic data and performed homology modeling and docking experiments. S.Z., P.R.-M. and M.K. provided reagents. All authors analyzed data. L.Laraia, A.F., D.P.C., G.K., Y.-W.W. and H.W. wrote the paper with comments from all other co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and Supplementary Figures 1–13
Supplementary Dataset 1
Mass spectrometry-based proteomics, raw data.
Supplementary Dataset 2
Kinase panel, complete data.
Supplementary Video 1
Live-cell imaging of HeLa cells simultaneously transfected with EGFP–WIPI1 and GRAMD1A–mCherry under starvation conditions.
Supplementary Video 2
Live-cell imaging of HeLa cells simultaneously transfected with EGFP–LC3 and GRAMD1A–mCherry under starvation conditions.
Supplementary Video 3
Additional live-cell imaging of HeLa cells simultaneously transfected with EGFP–LC3 and GRAMD1A–mCherry under starvation conditions.
Rights and permissions
About this article
Cite this article
Laraia, L., Friese, A., Corkery, D.P. et al. The cholesterol transfer protein GRAMD1A regulates autophagosome biogenesis. Nat Chem Biol 15, 710–720 (2019). https://doi.org/10.1038/s41589-019-0307-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-019-0307-5