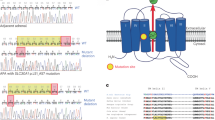
Abstract
Most aldosterone-producing adenomas (APAs) have gain-of-function somatic mutations of ion channels or transporters. However, their frequency in aldosterone-producing cell clusters of normal adrenal gland suggests a requirement for codriver mutations in APAs. Here we identified gain-of-function mutations in both CTNNB1 and GNA11 by whole-exome sequencing of 3/41 APAs. Further sequencing of known CTNNB1-mutant APAs led to a total of 16 of 27 (59%) with a somatic p.Gln209His, p.Gln209Pro or p.Gln209Leu mutation of GNA11 or GNAQ. Solitary GNA11 mutations were found in hyperplastic zona glomerulosa adjacent to double-mutant APAs. Nine of ten patients in our UK/Irish cohort presented in puberty, pregnancy or menopause. Among multiple transcripts upregulated more than tenfold in double-mutant APAs was LHCGR, the receptor for luteinizing or pregnancy hormone (human chorionic gonadotropin). Transfections of adrenocortical cells demonstrated additive effects of GNA11 and CTNNB1 mutations on aldosterone secretion and expression of genes upregulated in double-mutant APAs. In adrenal cortex, GNA11/Q mutations appear clinically silent without a codriver mutation of CTNNB1.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Source data for Figs. 2a–f and 3a–c,e,g are provided with the paper. The raw RNA-seq dataset analyzed to generate Fig. 4a,b, Supplementary Table 3 and Supplementary Fig. 4 is available upon request from the Science for Life Laboratory Data Centre through the link https://doi.org/10.17044/NBIS/G000007. Regulations by the service provider may make access technically restricted to PIs at Swedish organizations. The microarray datasets analyzed to generate Fig. 4a,b are deposited in the Gene Expression Omnibus database (GSE64957) or are available from the corresponding author on reasonable request. The WES raw data of the 41 APAs and controls investigated for recurrent pathogenic somatic mutation are available from the Sequence Read Archive under accession nos. PRJNA732946 and PRJNA729738. All other raw data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Choi, M. et al. K+ channel mutations in adrenal aldosterone-producing adenomas and hereditary hypertension. Science 331, 768–772 (2011).
Beuschlein, F. et al. Somatic mutations in ATP1A1 and ATP2B3 lead to aldosterone-producing adenomas and secondary hypertension. Nat. Genet. 45, 440–444 (2013).
Scholl, U. I. et al. Somatic and germline CACNA1D calcium channel mutations in aldosterone-producing adenomas and primary aldosteronism. Nat. Genet. 45, 1050–1054 (2013).
Azizan, E. A. et al. Somatic mutations in ATP1A1 and CACNA1D underlie a common subtype of adrenal hypertension. Nat. Genet. 45, 1055–1060 (2013).
Azizan, E. A. et al. Microarray, qPCR and KCNJ5 sequencing of aldosterone-producing adenomas reveal differences in genotype and phenotype between zona glomerulosa- and zona fasciculata-like tumors. J. Clin. Endocrinol. Metab. 97, E819–E829 (2012).
Monticone, S. et al. Immunohistochemical, genetic and clinical characterization of sporadic aldosterone-producing adenomas. Mol. Cell Endocrinol. 411, 146–154 (2015).
Akerstrom, T. et al. Novel somatic mutations and distinct molecular signature in aldosterone-producing adenomas. Endocr. Relat. Cancer 22, 735–744 (2015).
De Sousa, K. et al. Genetic, cellular, and molecular heterogeneity in adrenals with aldosterone-producing adenoma. Hypertension 75, 1034–1044 (2020).
Nanba, K. et al. Targeted molecular characterization of aldosterone-producing adenomas in White Americans. J. Clin. Endocrinol. Metab. 103, 3869–3876 (2018).
Wu, V. C. et al. The prevalence of CTNNB1 mutations in primary aldosteronism and consequences for clinical outcomes. Sci. Rep. 7, 39121 (2017).
Nishimoto, K. et al. Aldosterone-stimulating somatic gene mutations are common in normal adrenal glands. Proc. Natl Acad. Sci. USA 112, E4591–E4599 (2015).
Williams, T. A. et al. Visinin-like 1 is upregulated in aldosterone-producing adenomas with KCNJ5 mutations and protects from calcium-induced apoptosis. Hypertension 59, 833–839 (2012).
Akerstrom, T. et al. Activating mutations in CTNNB1 in aldosterone producing adenomas. Sci. Rep. 6, 19546 (2016).
Tadjine, M., Lampron, A., Ouadi, L. & Bourdeau, I. Frequent mutations of beta-catenin gene in sporadic secreting adrenocortical adenomas. Clin. Endocrinol. (Oxf.) 68, 264–270 (2008).
Omata, K. et al. Cellular and genetic causes of idiopathic hyperaldosteronism. Hypertension 72, 874–880 (2018).
Teo, A. E. et al. Pregnancy, primary aldosteronism, and adrenal CTNNB1 mutations. N. Engl. J. Med. 373, 1429–1436 (2015).
Kalinec, G., Nazarali, A. J., Hermouet, S., Xu, N. & Gutkind, J. S. Mutated alpha subunit of the Gq protein induces malignant transformation in NIH 3T3 cells. Mol. Cell Biol. 12, 4687–4693 (1992).
Gutowski, S. et al. Antibodies to the alpha q subfamily of guanine nucleotide-binding regulatory protein alpha subunits attenuate activation of phosphatidylinositol 4,5-bisphosphate hydrolysis by hormones. J. Biol. Chem. 266, 20519–20524 (1991).
Backman, S. et al. RNA sequencing provides novel insights into the transcriptome of aldosterone producing adenomas. Sci. Rep. 9, 6269 (2019).
Wiese, M. et al. The beta-catenin/CBP-antagonist ICG-001 inhibits pediatric glioma tumorigenicity in a Wnt-independent manner. Oncotarget 8, 27300–27313 (2017).
Zhou, L. et al. Multiple genes of the renin-angiotensin system are novel targets of Wnt/beta-catenin signaling. J. Am. Soc. Nephrol. 26, 107–120 (2015).
Doghman, M., Cazareth, J. & Lalli, E. The T cell factor/beta-catenin antagonist PKF115-584 inhibits proliferation of adrenocortical carcinoma cells. J. Clin. Endocrinol. Metab. 93, 3222–3225 (2008).
Zhou, T. et al. CTNNB1 knockdown inhibits cell proliferation and aldosterone secretion through inhibiting Wnt/beta-catenin signaling in H295R cells. Technol. Cancer Res. Treat. 19, 1533033820979685 (2020).
Jeppesen, J. V. et al. LH-receptor gene expression in human granulosa and cumulus cells from antral and preovulatory follicles. J. Clin. Endocrinol. Metab. 97, E1524–E1531 (2012).
Breen, S. M. et al. Ovulation involves the luteinizing hormone-dependent activation of G(q/11) in granulosa cells. Mol. Endocrinol. 27, 1483–1491 (2013).
Gazdar, A. F. et al. Establishment and characterization of a human adrenocortical carcinoma cell line that expresses multiple pathways of steroid biosynthesis. Cancer Res. 50, 5488–5496 (1990).
Tissier, F. et al. Mutations of beta-catenin in adrenocortical tumors: activation of the Wnt signaling pathway is a frequent event in both benign and malignant adrenocortical tumors. Cancer Res. 65, 7622–7627 (2005).
Boulkroun, S. et al. Aldosterone-producing adenoma formation in the adrenal cortex involves expression of stem/progenitor cell markers. Endocrinology 152, 4753–4763 (2011).
Shaikh, L. H. et al. LGR5 activates noncanonical Wnt signaling and inhibits aldosterone production in the human adrenal. J. Clin. Endocrinol. Metab. 100, E836–E844 (2015).
Zhou, J. et al. Transcriptome pathway analysis of pathological and physiological aldosterone-producing human tissues. Hypertension 68, 1424–1431 (2016).
Taylor, M. J. et al. Chemogenetic activation of adrenocortical Gq signaling causes hyperaldosteronism and disrupts functional zonation. J. Clin. Invest. 130, 83–93 (2020).
Leng, S. et al. beta-Catenin and FGFR2 regulate postnatal rosette-based adrenocortical morphogenesis. Nat. Commun. 11, 1680 (2020).
Schwindinger, W. F., Francomano, C. A. & Levine, M. A. Identification of a mutation in the gene encoding the alpha subunit of the stimulatory G protein of adenylyl cyclase in McCune–Albright syndrome. Proc. Natl Acad. Sci. USA 89, 5152–5156 (1992).
Weinstein, L. S. et al. Activating mutations of the stimulatory G protein in the McCune–Albright syndrome. N. Engl. J. Med. 325, 1688–1695 (1991).
Idowu, B. D. et al. A sensitive mutation-specific screening technique for GNAS1 mutations in cases of fibrous dysplasia: the first report of a codon 227 mutation in bone. Histopathology 50, 691–704 (2007).
Vasilev, V. et al. McCune–Albright syndrome: a detailed pathological and genetic analysis of disease effects in an adult patient. J. Clin. Endocrinol. Metab. 99, E2029–E2038 (2014).
Rey, R. A. et al. Unexpected mosaicism of R201H-GNAS1 mutant-bearing cells in the testes underlie macro-orchidism without sexual precocity in McCune–Albright syndrome. Hum. Mol. Genet. 15, 3538–3543 (2006).
Wu, D. Q., Lee, C. H., Rhee, S. G. & Simon, M. I. Activation of phospholipase C by the alpha subunits of the Gq and G11 proteins in transfected Cos-7 cells. J. Biol. Chem. 267, 1811–1817 (1992).
Ayturk, U. M. et al. Somatic activating mutations in GNAQ and GNA11 are associated with congenital hemangioma. Am. J. Hum. Genet. 98, 789–795 (2016).
Van Raamsdonk, C. D. et al. Mutations in GNA11 in uveal melanoma. N. Engl. J. Med. 363, 2191–2199 (2010).
Shirley, M. D. et al. Sturge–Weber syndrome and port-wine stains caused by somatic mutation in Gnaq. N. Engl. J. Med. 368, 1971–1979 (2013).
Thomas, A. C. et al. Mosaic activating mutations in GNA11 and GNAQ are associated with phakomatosis pigmentovascularis and extensive dermal melanocytosis. J. Invest. Dermatol. 136, 770–778 (2016).
Simon, D. P. & Hammer, G. D. Adrenocortical stem and progenitor cells: implications for adrenocortical carcinoma. Mol. Cell Endocrinol. 351, 2–11 (2012).
Berthon, A. et al. WNT/beta-catenin signalling is activated in aldosterone-producing adenomas and controls aldosterone production. Hum. Mol. Genet. 23, 889–905 (2014).
Lerario, A. M., Moraitis, A. & Hammer, G. D. Genetics and epigenetics of adrenocortical tumors. Mol. Cell Endocrinol. 386, 67–84 (2014).
Wang, J. J., Peng, K. Y., Wu, V. C., Tseng, F. Y. & Wu, K. D. CTNNB1 mutation in aldosterone producing adenoma. Endocrinol. Metab. (Seoul) 32, 332–338 (2017).
Assie, G. et al. Integrated genomic characterization of adrenocortical carcinoma. Nat. Genet. 46, 607–612 (2014).
Jakobsen, J. N., Santoni-Rugiu, E., Grauslund, M., Melchior, L. & Sorensen, J. B. Concomitant driver mutations in advanced EGFR-mutated non-small-cell lung cancer and their impact on erlotinib treatment. Oncotarget 9, 26195–26208 (2018).
Gainor, J. F. et al. ALK rearrangements are mutually exclusive with mutations in EGFR or KRAS: an analysis of 1,683 patients with non-small cell lung cancer. Clin. Cancer Res. 19, 4273–4281 (2013).
Nanba, K. et al. Genetic characteristics of aldosterone-producing adenomas in Blacks. Hypertension 73, 885–892 (2019).
Pignatti, E. et al. Beta-catenin causes adrenal hyperplasia by blocking zonal transdifferentiation. Cell Rep. 31, 107524 (2020).
Vouillarmet, J. et al. Aldosterone-producing adenoma with a somatic KCNJ5 mutation revealing APC-dependent familial adenomatous polyposis. J. Clin. Endocrinol. Metab. 101, 3874–3878 (2016).
Polak, P. et al. Cell-of-origin chromatin organization shapes the mutational landscape of cancer. Nature 518, 360–364 (2015).
Yeh, I. et al. Combined activation of MAP kinase pathway and beta-catenin signaling cause deep penetrating nevi. Nat. Commun. 8, 644 (2017).
Piaggio, F. et al. Secondary somatic mutations in G-protein-related pathways and mutation signatures in uveal melanoma. Cancers (Basel) 11, 1688 (2019).
Chen, X. et al. The melanoma-linked ‘redhead’ MC1R influences dopaminergic neuron survival. Ann. Neurol. 81, 395–406 (2017).
Cavlan, D., Storr, H. L., Berney, D., Evagora, C. & King, P. J. Adrenal pigmentation in PPNAD is a result of melanin deposition and associated with upregulation of the melanocortin 1 receptor. Endocr. Abstr. 38, 154 (2015).
Binder, J. X. et al. COMPARTMENTS: unification and visualization of protein subcellular localization evidence. Database (Oxf.) 2014, bau012 (2014).
de Lau, W. et al. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature 476, 293–297 (2011).
Vidal, V. et al. The adrenal capsule is a signaling center controlling cell renewal and zonation through Rspo3. Genes Dev. 30, 1389–1394 (2016).
Yi, H., Wang, Y., Kavallaris, M. & Wang, J. Y. Lgr4-mediated potentiation of Wnt/β-catenin signaling promotes MLL leukemogenesis via an Rspo3/Wnt3a-Gnaq pathway in leukemic stem cells. Blood 122, 887 (2013).
Carter, J. M. et al. CTNNB1 mutations and estrogen receptor expression in neuromuscular choristoma and its associated fibromatosis. Am. J. Surg. Pathol. 40, 1368–1374 (2016).
Crago, A. M. et al. Near universal detection of alterations in CTNNB1 and Wnt pathway regulators in desmoid-type fibromatosis by whole-exome sequencing and genomic analysis. Genes Chromosomes Cancer 54, 606–615 (2015).
Maria, A. G. et al. Mosaicism for KCNJ5 causing early-onset primary aldosteronism due to bilateral adrenocortical hyperplasia. Am. J. Hypertens. 33, 124–130 (2020).
Zhang, E. D. et al. Mutation spectrum in GNAQ and GNA11 in Chinese uveal melanoma. Precis. Clin. Med. 2, 213–220 (2019).
Gerstenblith, M. R., Goldstein, A. M., Fargnoli, M. C., Peris, K. & Landi, M. T. Comprehensive evaluation of allele frequency differences of MC1R variants across populations. Hum. Mutat. 28, 495–505 (2007).
Eguchi, K. et al. An adverse pregnancy-associated outcome due to overlooked primary aldosteronism. Intern. Med. 53, 2499–2504 (2014).
Saner-Amigh, K. et al. Elevated expression of luteinizing hormone receptor in aldosterone-producing adenomas. J. Clin. Endocrinol. Metab. 91, 1136–1142 (2006).
Gagnon, N. et al. Genetic characterization of GnRH/LH-responsive primary aldosteronism. J. Clin. Endocrinol. Metab. 103, 2926–2935 (2018).
Albiger, N. M. et al. A case of primary aldosteronism in pregnancy: do LH and GNRH receptors have a potential role in regulating aldosterone secretion? Eur. J. Endocrinol. 164, 405–412 (2011).
Berthon, A., Drelon, C. & Val, P. Pregnancy, primary aldosteronism, and somatic CTNNB1 mutations. N. Engl. J. Med. 374, 1493–1494 (2016).
Murtha, T. D., Carling, T. & Scholl, U. I. Pregnancy, primary aldosteronism, and somatic CTNNB1 mutations. N. Engl. J. Med. 374, 1492–1493 (2016).
Burton, T. J. et al. Evaluation of the sensitivity and specificity of (11)C-metomidate positron emission tomography (PET)-CT for lateralizing aldosterone secretion by Conn’s adenomas. J. Clin. Endocrinol. Metab. 97, 100–109 (2012).
Letavernier, E. et al. Blood pressure outcome of adrenalectomy in patients with primary hyperaldosteronism with or without unilateral adenoma. J. Hypertens. 26, 1816–1823 (2008).
Funder, J. W. et al. Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 93, 3266–3281 (2008).
Fernandes-Rosa, F. L. et al. Genetic spectrum and clinical correlates of somatic mutations in aldosterone-producing adenoma. Hypertension 54, 354–361 (2014).
Akerstrom, T. et al. Comprehensive re-sequencing of adrenal aldosterone producing lesions reveal three somatic mutations near the KCNJ5 potassium channel selectivity filter. PLoS ONE 7, e41926 (2012).
Gomez-Sanchez, C. E. et al. Development of monoclonal antibodies against human CYP11B1 and CYP11B2. Mol. Cell Endocrinol. 383, 111–117 (2014).
Bustin, S. A. Why the need for qPCR publication guidelines? The case for MIQE. Methods 50, 217–226 (2010).
Acknowledgements
The CTNNB1 plasmid was a kind gift of M. Bienz, Medical Research Council Laboratory of Molecular Biology, Cambridge. The 11C-metomidate positron emission tomography (PET) CT The project was funded in part by the British Heart Foundation through a Clinical Research Training Fellowship (no. FS/19/50/34566) and PhD Studentship (no. FS/14/75/31134), by the National Institute of Health Research (NIHR) through Senior Investigator award no. NF-SI-0512-10052 (all to M.J.B.) and by NIHR Efficacy and Mechanisms Evaluation Project (no. 14/145/09, to W.M.D., M.G. and M.J.B.) and Barts and the London Charity project (no. MGU0360), to W.M.D. and M.J.B. The project was further funded through institutional support from INSERM, Agence Nationale de la Recherche (no. ANR-15-CE14-0017-03) and Fondation pour la Recherche Médicale (no. EQU201903007864) to M.-C.Z. and the NIHR Advanced Fellowship (no. NIHR3000098) to H.L.S. E.A.B.A. is a Royal Society-Newton Advanced Research Fellow (no. NA170257/FF-2018-033). R.V.T. is supported by a Wellcome Trust Investigator Award (no. 106995/Z/15/Z) and the NIHR Oxford Biomedical Research Centre (BRC) Programme. C.P.C. is supported by the NIHR BRC at Barts and The London School of Medicine and Dentistry. M.G., A.M., and R.S. are supported by the NIHR Cambridge BRC (no. IS-BRC-1215-20014). The research of J.L.K., Z.T. and R.F. was supported by the National Medical Research Council and BRC of Singapore. Research in London and Cambridge, UK, was further supported by the NIHR Barts Cardiovascular BRC (no. IS-BRC-1215-20022) and the Cambridge BRC and BRC-funded Tissue Bank. The research utilized Queen Mary University of London’s Apocrita HPC facility, supported by QMUL Research-IT (https://doi.org/10.5281/zenodo.438045). Assistance from the Endocrine Unit Laboratory of the National University of Malaysia (UKM) Medical Centre, and from L. K. Chin and S. Khadijah (UKM) is acknowledged.
Author information
Authors and Affiliations
Contributions
C.P.C., E.A.B.A. and M.J.B. discovered the mutations in GNA11 and GNAQ, replicated by J.Z. and F.L.F.-R. J.Z., E.A.B.A., C.P.C., F.L.F.-R., S. Boulkroun, H.L.S., M.-C.Z. and M.J.B. conceived and designed the subsequent experiments/analyses. C.J., A.T., H.L.S., E.C., G.A., X.W., E.G., L.A., S. Backman, P.H., P.B., T.A., R.S., D.M.B., J.P.K., W.M.D., L.P. and F.E.K.F. contributed to cohort ascertainment, phenotypic characterization and recruitment. S. Backman, C.P.C., S.P., Z.T., L.A.M., T.A. and S.G. contributed to WES/RNA-seq production, validation, analysis and reanalysis. J.Z., F.L.F.-R., S. Boulkroun, X.W., A.E.D.T., E.A.B.A., E.C., S.G., G.A. and T.A. performed targeted sequencing and RT–qPCR analyses. J.Z. performed LCM and genotyping of adrenal zones and biopsy punches. S.J., S. Boulkroun, A.M. and J.Z. performed, and F.L.F.-R. and E.A.B.A. analyzed, IHC staining. C.E.G.S. developed antisera for use in IHC. J.Z., S.G., A.G., K.E.L. and R.V.T. contributed to plasmid construction for GNA11 and GNAQ. J.Z., E.A.B.A. and G.A. performed the functional experiments on transfected H295R and primary human adrenal cells. J.Z. and S.O. undertook confocal analyses. J.Z., E.A.B.A., F.L.F.-R., C.A.M., R.F., E.W., D.K., J.L.K., Z.T. and C.P.C. performed ddPCR, WES and NGS for genotyping of adjacent adrenal regions. C.P.C., J.Z., E.A.B.A. and M.J.B. contributed to statistical analyses. E.A.B.A. and M.J.B. drafted the manuscript, for which J.Z., E.A.B.A., C.P.C., F.L.F.-R., S. Boulkroun, T.A., A.M. and M.J.B. contributed figures. C.P.C., F.L.F.-R., S. Boulkroun, M.G., V.A.K. and M.-C.Z. critically reviewed the text. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Genetics thanks Lee Weinstein and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 High LHCGR expression in GNA11 and CTNNB1 double mutant co-transfected primary human adrenal cells.
a, APA 351 T cells transfected with CTNNB1 (untagged plasmid) and GNA11 (GFP-tagged plasmid) wild-type or Q209P (red boxed cell). LHCGR and CTNNB1 expression was visualized as in Fig. 3f using the primary antibody rabbit anti-LHCGR #NLS1436 (1:200; Novus Biologicals, UK) and the primary antibody mouse anti-CTNNB1 #610154 (1:100; BD transduction Lab, USA), respectively. Scale bars, 50 μm. b, Immunofluorescence of LHCGR in APA 351 T cells was quantified using corrected total cell fluorescence (CTCF). LHCGR expression was increased in cells expressing high CTNNB1 and GNA11 Q209P (the exact number, n, of cells quantified from two independent experiment are as indicated below the x-axis; the P-values indicated are according to Kolmogorov–Smirnov statistical test). High CTNNB1 was determined as CTCF > 10,000. Data are presented as mean values + /- s.e.m.
Extended Data Fig. 2 GNA11 somatic mutations were found in the adjacent adrenals to double-mutant APA of patient 6.
a, From six different regions (R1-5, at the edges of the adrenal cortex, R6 and APA, within the circled areas) in the formalin fixed paraffin embedded (FFPE) adjacent adrenal gland, genomic DNA samples of patient 6 were genotyped for CTNNB1 and GNA11 mutations. Immunohistochemistry of KCNJ5 and CYP11B2 were used for region selection. Scale bar, 10 mm and 50 μm as indicated. b, Sanger sequencing identified weak chromatogram peaks of CTNNB1 G34R and GNA11 Q209P somatic mutations in region 6 of the adjacent adrenal gland. c, Next generation sequencing confirmed the CTNNB1 G34R and GNA11 Q209P mutations in region 6 of the adjacent adrenal gland. d, qPCR of R1-6 and APA showed a 337-fold higher of TMEM132E, 38-fold higher of CYP11B2, 14-fold higher of DKK1 and 10-fold higher of LHCGR expression in region 6 compared to region 5. Regions 1-5 have similar expression of the above genes. The APA had the highest expression of CYP11B2, TMEM132E, DKK1, LHCGR and lowest expression of CYP11B1 and LGR5 compared to regions 1–6.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6 and Tables 1–9
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Rights and permissions
About this article
Cite this article
Zhou, J., Azizan, E.A.B., Cabrera, C.P. et al. Somatic mutations of GNA11 and GNAQ in CTNNB1-mutant aldosterone-producing adenomas presenting in puberty, pregnancy or menopause. Nat Genet 53, 1360–1372 (2021). https://doi.org/10.1038/s41588-021-00906-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-021-00906-y
This article is cited by
-
Primary aldosteronism: molecular medicine meets public health
Nature Reviews Nephrology (2023)
-
Somatic SLC30A1 mutations altering zinc transporter ZnT1 cause aldosterone-producing adenomas and primary aldosteronism
Nature Genetics (2023)
-
The diagnosis and management of pheochromocytoma and paraganglioma during pregnancy
Reviews in Endocrine and Metabolic Disorders (2023)
-
The mystery of transient pregnancy-induced cushing’s syndrome: a case report and literature review highlighting GNAS somatic mutations and LHCGR overexpression
Endocrine (2023)
-
[11C]metomidate PET-CT versus adrenal vein sampling for diagnosing surgically curable primary aldosteronism: a prospective, within-patient trial
Nature Medicine (2023)