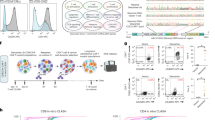
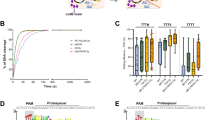
Abstract
Inefficient knock-in of transgene cargos limits the potential of cell-based medicines. In this study, we used a CRISPR nuclease that targets a site within an exon of an essential gene and designed a cargo template so that correct knock-in would retain essential gene function while also integrating the transgene(s) of interest. Cells with non-productive insertions and deletions would undergo negative selection. This technology, called SLEEK (SeLection by Essential-gene Exon Knock-in), achieved knock-in efficiencies of more than 90% in clinically relevant cell types without impacting long-term viability or expansion. SLEEK knock-in rates in T cells are more efficient than state-of-the-art TRAC knock-in with AAV6 and surpass more than 90% efficiency even with non-viral DNA cargos. As a clinical application, natural killer cells generated from induced pluripotent stem cells containing SLEEK knock-in of CD16 and mbIL-15 show substantially improved tumor killing and persistence in vivo.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. Unprocessed flow cytometry and microscopy images for Figs. 1–5 are provided as Source Data File 1, and numerical values for plotted data in Figs. 1–5 are provided as Source Data File 2. DNA constructs, gRNAs, primers, ddPCR probes, Digenome-Seq results and rhAmp-Seq panel of potential off-target candidate sites are listed in the Supplementary Table. All numerical data values used to generate figures in the Supplementary Information can be found in Supplementary Data. Raw flow cytometry plots and representative gating strategies, as well as uncropped microscopy and gel images, are provided in the Supplementary Information under Supplementary Notes. High-throughput sequencing data have been deposited in the National Center for Biotechnology Informationʼs Sequence Read Archive database (accession code PRJNA947757) and can be found at http://www.ncbi.nlm.nih.gov/bioproject/947757 (ref. 62). Source data are provided with this paper.
Code availability
Custom code used to analyze Digenome-Seq data is available at the Editas Medicine GitHub page at https://github.com/editasmedicine/digenomitas. Custom code used to identify candidate in silico off-target sites is also available at the Editas Medicine GitHub (https://github.com/editasmedicine/calitas).
References
Bailey, S. R. & Maus, M. V. Gene editing for immune cell therapies. Nat. Biotechnol. 37, 1425–1434 (2019).
Raffin, C., Vo, L. T. & Bluestone, J. A. Treg cell-based therapies: challenges and perspectives. Nat. Rev. Immunol. 20, 158–172 (2020).
Wilkinson, A. C., Igarashi, K. J. & Nakauchi, H. Haematopoietic stem cell self-renewal in vivo and ex vivo. Nat. Rev. Genet. 21, 541–554 (2020).
Raje, N. et al. Anti-BCMA CAR T-cell therapy bb2121 in relapsed or refractory multiple myeloma. N. Engl. J. Med. 380, 1726–1737 (2019).
Abramson, J. S. Anti-CD19 CAR T-cell therapy for B-cell non-Hodgkin lymphoma. Transfus. Med. Rev. 34, 29–33 (2020).
Frigault, M. J. et al. Tisagenlecleucel CAR T-cell therapy in secondary CNS lymphoma. Blood 134, 860–866 (2019).
Naeimi Kararoudi, M. et al. Optimization and validation of CAR transduction into human primary NK cells using CRISPR and AAV. Cell Rep. Methods 2, 100236 (2022).
Xie, G. et al. CAR-NK cells: a promising cellular immunotherapy for cancer. EBioMedicine 59, 102975 (2020).
Liu, E. et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N. Engl. J. Med. 382, 545–553 (2020).
Depil, S., Duchateau, P., Grupp, S. A., Mufti, G. & Poirot, L. ‘Off-the-shelf’ allogeneic CAR T cells: development and challenges. Nat. Rev. Drug. Discov. 19, 185–199 (2020).
Srpan, K. et al. Shedding of CD16 disassembles the NK cell immune synapse and boosts serial engagement of target cells. J. Cell Biol. 217, 3267–3283 (2018).
Snyder, K. M. et al. Expression of a recombinant high affinity IgG Fc receptor by engineered NK cells as a docking platform for therapeutic mAbs to target cancer cells. Front. Immunol. 9, 2873 (2018).
Hu, W., Wang, G., Huang, D., Sui, M. & Xu, Y. Cancer immunotherapy based on natural killer cells: current progress and new opportunities. Front. Immunol. 10, 1205 (2019).
Szmania, S. et al. Ex vivo-expanded natural killer cells demonstrate robust proliferation in vivo in high-risk relapsed multiple myeloma patients. J. Immunother. 38, 24–36 (2015).
Giron-Michel, J. et al. Membrane-bound and soluble IL-15/IL-15Rα complexes display differential signaling and functions on human hematopoietic progenitors. Blood 106, 2302–2310 (2005).
Weber, E. W., Maus, M. V. & Mackall, C. L. The emerging landscape of immune cell therapies. Cell 181, 46–62 (2020).
Eyquem, J. et al. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543, 113–117 (2017).
Vakulskas, C. A. et al. A high-fidelity Cas9 mutant delivered as a ribonucleoprotein complex enables efficient gene editing in human hematopoietic stem and progenitor cells. Nat. Med. 24, 1216–1224 (2018).
Zhang, L. et al. AsCas12a ultra nuclease facilitates the rapid generation of therapeutic cell medicines. Nat. Commun. 12, 3908 (2021).
Bulcha, J. T., Wang, Y., Ma, H., Tai, P. W. L. & Gao, G. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target. Ther. 6, 53 (2021).
Jasin, M. & Rothstein, R. Repair of strand breaks by homologous recombination. Cold Spring Harb. Perspect. Biol. 5, a012740 (2013).
Kao, T. et al. GAPTrap: a simple expression system for pluripotent stem cells and their derivatives. Stem Cell Rep. 7, 518–526 (2016).
Liu, Z. et al. Systematic comparison of 2A peptides for cloning multi-genes in a polycistronic vector. Sci. Rep. 7, 2193 (2017).
Dobosy, J. R. et al. RNase H-dependent PCR (rhPCR): improved specificity and single nucleotide polymorphism detection using blocked cleavable primers. BMC Biotechnol. 11, 80 (2011).
Park, J. B., Park, H., Son, J., Ha, S. J. & Cho, H. S. Structural study of monomethyl fumarate-bound human GAPDH. Mol. Cells 42, 597–603 (2019).
Yilmaz, A., Peretz, M., Aharony, A., Sagi, I. & Benvenisty, N. Defining essential genes for human pluripotent stem cells by CRISPR–Cas9 screening in haploid cells. Nat. Cell Biol. 20, 610–619 (2018).
Eisenberg, E. & Levanon, E. Y. Human housekeeping genes, revisited. Trends Genet. 29, 569–574 (2013).
Laoharawee, K. et al. Genome engineering of primary human B cells using CRISPR/Cas9. J. Vis. Exp. 10.3791/61855 (2020).
Pomeroy, E. J. et al. A genetically engineered primary human natural killer cell platform for cancer immunotherapy. Mol. Ther. 28, 52–63 (2020).
Robert, F., Barbeau, M., Ethier, S., Dostie, J. & Pelletier, J. Pharmacological inhibition of DNA-PK stimulates Cas9-mediated genome editing. Genome Med. 7, 93 (2015).
Roth, T. L. et al. Reprogramming human T cell function and specificity with non-viral genome targeting. Nature 559, 405–409 (2018).
Nguyen, D. N. et al. Polymer-stabilized Cas9 nanoparticles and modified repair templates increase genome editing efficiency. Nat. Biotechnol. 38, 44–49 (2020).
Oh, S. A. et al. High-efficiency nonviral CRISPR/Cas9-mediated gene editing of human T cells using plasmid donor DNA. J. Exp. Med. 219, e20211530 (2022).
Shy, B. R. et al. High-yield genome engineering in primary cells using a hybrid ssDNA repair template and small-molecule cocktails. Nat. Biotechnol. (2022).
Fennell, T. et al. CALITAS: a CRISPR–Cas-aware ALigner for In silico off-TArget Search. CRISPR J. 4, 264–274 (2021).
Tsai, S. Q. et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR–Cas nucleases. Nat. Biotechnol. 33, 187–197 (2015).
Kim, D. et al. Genome-wide target specificities of CRISPR RNA-guided programmable deaminases. Nat. Biotechnol. 35, 475–480 (2017).
Kosicki, M., Tomberg, K. & Bradley, A. Repair of double-strand breaks induced by CRISPR–Cas9 leads to large deletions and complex rearrangements. Nat. Biotechnol. 36, 765–771 (2018).
Bothmer, A. et al. Detection and modulation of DNA translocations during multi-gene genome editing in T cells. CRISPR J. 3, 177–187 (2020).
Ivancic, D. et al. INSERT-seq enables high-resolution mapping of genomically integrated DNA using Nanopore sequencing. Genome Biol. 23, 227 (2022).
Wang, Y., Zhao, Y., Bollas, A., Wang, Y. & Au, K. F. Nanopore sequencing technology, bioinformatics and applications. Nat. Biotechnol. 39, 1348–1365 (2021).
Sondka, Z. et al. The COSMIC Cancer Gene Census: describing genetic dysfunction across all human cancers. Nat. Rev. Cancer 18, 696–705 (2018).
Pickup, M., Novitskiy, S. & Moses, H. L. The roles of TGFβ in the tumour microenvironment. Nat. Rev. Cancer 13, 788–799 (2013).
Gornalusse, G. G. et al. HLA-E-expressing pluripotent stem cells escape allogeneic responses and lysis by NK cells. Nat. Biotechnol. 35, 765–772 (2017).
Zhang, L. et al. Author Correction: AsCas12a ultra nuclease facilitates the rapid generation of therapeutic cell medicines. Nat. Commun. 12, 4500 (2021).
Leibowitz, M. L. et al. Chromothripsis as an on-target consequence of CRISPR–Cas9 genome editing. Nat. Genet. 53, 895–905 (2021).
Ihry, R. J. et al. p53 inhibits CRISPR–Cas9 engineering in human pluripotent stem cells. Nat. Med. 24, 939–946 (2018).
Keppel, M. P., Saucier, N., Mah, A. Y., Vogel, T. P. & Cooper, M. A. Activation-specific metabolic requirements for NK cell IFN-γ production. J. Immunol. 194, 1954–1962 (2015).
Stadtmauer, E. A. et al. CRISPR-engineered T cells in patients with refractory cancer. Science 367, eaba7365 (2020).
Ho, J. Y. et al. Promoter usage regulating the surface density of CAR molecules may modulate the kinetics of CAR-T cells in vivo. Mol. Ther. Methods Clin. Dev. 21, 237–246 (2021).
Cichocki, F. et al. iPSC-derived NK cells maintain high cytotoxicity and enhance in vivo tumor control in concert with T cells and anti-PD-1 therapy. Sci. Transl. Med. 12, eaaz5618 (2020).
Kath, J. et al. Pharmacological interventions enhance virus-free generation of TRAC-replaced CAR T cells. Mol. Ther. Methods Clin. Dev. 25, 311–330 (2022).
Ng, E. S., Davis, R. P., Azzola, L., Stanley, E. G. & Elefanty, A. G. Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood 106, 1601–1603 (2005).
Decker, T. & Lohmann-Matthes, M. L. A quick and simple method for the quantitation of lactate dehydrogenase release in measurements of cellular cytotoxicity and tumor necrosis factor (TNF) activity. J. Immunol. Methods 115, 61–69 (1988).
Allicotti, G., Borras, E. & Pinilla, C. A time-resolved fluorescence immunoassay (DELFIA) increases the sensitivity of antigen-driven cytokine detection. J. Immunoassay Immunochem. 24, 345–358 (2003).
Uhlen, M. et al. Proteomics. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Maeder, M. L. et al. Development of a gene-editing approach to restore vision loss in Leber congenital amaurosis type 10. Nat. Med. 25, 229–233 (2019).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620 (2014).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Li, H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34, 3094–3100 (2018).
Marco, E. A highly efficient transgene knock-in technology in clinically relevant cell types. National Center for Biotechnology Information. http://www.ncbi.nlm.nih.gov/bioproject/947757 (2023).
Acknowledgements
We would like to thank additional members of the Editas Computational Biology, Informatics and Sequencing groups for generation and pipeline analysis of sequencing data. We thank R. Naines, C. Wang, J. Yao and H. An for providing primary cells for studies. We would like to thank J. Getgano, K. Gareau, E. Goncz, S. Zhang, J. Moon, K. Tsiounis and J. Schafer for support in the development of key assays and protocols. We would like to thank members of BlueRock Therapeutics LP for their support and collaboration related to engineering and culturing iPSCs. We would like to thank A. Dee for paper preparation support. Several graphics in the figures (cells in Fig. 1a, cells in Fig. 2a,h, cells in Fig. 3a,d,g, mouse schematic in Fig. 5a, cells in Supplementary Fig. 5c, cells in Supplementary Fig. 9c,e, cells in Supplementary Fig. 10a, cells in Supplementary Fig. 11a–c, cells in Supplementary Fig. 12c, cells in Supplementary Fig. 13a and cells in Supplementary Fig. 14a,b) were created with BioRender. We would like to thank Porterhouse Medical for graphic design support.
Author information
Authors and Affiliations
Contributions
A.G.A, S.Q.K., C.M.M., R.V., S.L., S.N.S., K.M.I., A.G., R.P., N.R.C., C.S.H., A.H.Z., S.E.S., M.C.J., M.W., A.C.W., D.M.C., D.Z., Y.H., J.D.N., P.Z. and P.M. performed experiments and analyzed data. L.B., X.S., J.A.F. and J.A.Z. analyzed data. A.G.A., S.Q.K., C.M.M., R.V., S.L., L.B., S.E.S., M.C.J., X.S., G.G., E.M., M.N., J.A.F., K.Z., K.-H.C., M.S.S., C.J.W. and J.A.Z. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
All authors were employees and shareholders of Editas Medicine at the time the work was performed. J.A.Z. and C.M.M. are inventors on patent WO2021226151A2 that has been filed by Editas Medicine relating to this work.
Peer review
Peer review information
Nature Biotechnology thanks Fyodor Urnov and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–18 as well as raw images of supplementary figure flow cytometry plots, microscopy images and western blot uncropped gels.
Supplementary Table 1
List of plasmid constructs, gRNAs, ddPCR and RT–qPCR primers, Digenome-Seq and rHampSeq specificity data.
Supplementary Data 1
Numerical data used to generate graphed figures in the Supplementary Information file.
Source data
Source Data Fig. 1, Fig. 2, Fig. 3 and Fig. 5
Contains all raw flow cytometry plots, microscopy images, IVIS images and histology images from main text figures.
Source Data Fig. 1, Fig. 2, Fig. 3, Fig. 4, Fig. 5
Contains raw numerical data for all graphs in main text figures.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Allen, A.G., Khan, S.Q., Margulies, C.M. et al. A highly efficient transgene knock-in technology in clinically relevant cell types. Nat Biotechnol 42, 458–469 (2024). https://doi.org/10.1038/s41587-023-01779-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-023-01779-8
This article is cited by
-
Chimeric antigen receptor-based natural killer cell immunotherapy in cancer: from bench to bedside
Cell Death & Disease (2024)
-
Enrichment strategies to enhance genome editing
Journal of Biomedical Science (2023)