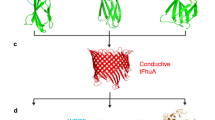
Abstract
Detection of specific proteins using nanopores is currently challenging. To address this challenge, we developed a collection of over twenty nanopore-addressable protein tags engineered as reporters (NanoporeTERs, or NTERs). NTERs are constructed with a secretion tag, folded domain and a nanopore-targeting C-terminal tail in which arbitrary peptide barcodes can be encoded. We demonstrate simultaneous detection of up to nine NTERs expressed in bacterial or human cells using MinION nanopore sensor arrays.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Data are available upon request and on can be found on github.com/uwmisl/NanoporeTERs.
Code availability
Codes are available upon request and can be found on github.com/uwmisl/NanoporeTERs. Custom MinION MinKNOW runscripts can also be obtained from Oxford Nanopore Technologies upon request.
References
Ghim, C. M., Lee, S. K., Takayama, S. & Mitchell, R. J. The art of reporter proteins in science: past, present and future applications. BMB Rep. 43, 451–460 (2010).
Rodriguez, E. A. et al. The growing and glowing toolbox of fluorescent and photoactive proteins. Trends Biochem. Sci 42, 111–129 (2017).
Martin, L., Che, A. & Endy, D. Gemini, a bifunctional enzymatic and fluorescent reporter of gene expression. PLoS ONE 4, e7569 (2009).
Parrello, D., Mustin, C., Brie, D., Miron, S. & Billard, P. Multicolor whole-cell bacterial sensing using a synchronous fluorescence spectroscopy-based approach. PLoS ONE 10, e0122848 (2015).
Shimo, T., Tachibana, K. & Obika, S. Construction of a tri-chromatic reporter cell line for the rapid and simple screening of splice-switching oligonucleotides targeting DMD exon 51 using high content screening. PLoS ONE 13, e0197373 (2018).
Wroblewska, A. et al. Protein barcodes enable high-dimensional single-cell CRISPR screens. Cell 175, 1141–1155 (2018).
He, W., Yuan, S., Zhong, W. H., Siddikee, M. A. & Dai, C. C. Application of genetically engineered microbial whole-cell biosensors for combined chemosensing. Appl. Microbiol. Biotechnol. 100, 1109–1119 (2016).
Nielsen, A. A. K. et al. Genetic circuit design automation. Science 352, aac7341 (2016).
Shi, W., Friedman, A. K. & Baker, L. A. Nanopore sensing. Anal. Chem. 89, 157–188 (2017).
Jain, M., Olsen, H. E., Paten, B. & Akeson, M. The Oxford Nanopore MinION: delivery of nanopore sequencing to the genomics community. Genome Biol. 17, 239 (2016).
Garalde, D. R. et al. Highly parallel direct RNA sequencing on an array of nanopores. Nat. Methods 15, 201–206 (2018).
Nivala, J., Marks, D. B. & Akeson, M. Unfoldase-mediated protein translocation through an α-hemolysin nanopore. Nat. Biotechnol. 31, 247–250 (2013).
Nivala, J., Mulroney, L., Li, G., Schreiber, J. & Akeson, M. Discrimination among protein variants using an unfoldase-coupled nanopore. ACS Nano 8, 12365–12375 (2014).
Yim, H. H. & Villarejo, M. osmY, a new hyperosmotically inducible gene, encodes a periplasmic protein in Escherichia coli. J. Bacteriol. 174, 3637–3644 (1992).
Kotzsch, A. et al. A secretory system for bacterial production of high-profile protein targets. Protein Sci. 20, 597–609 (2011).
Goyal, P. et al. Structural and mechanistic insights into the bacterial amyloid secretion channel CsgG. Nature 516, 250–253 (2014).
Taylor, S. S. et al. PKA: a portrait of protein kinase dynamics.Biochim. Biophys. Acta Proteins Proteom. 1697, 259–269 (2004).
Román, R. et al. Enhancing heterologous protein expression and secretion in HEK293 cells by means of combination of CMV promoter and IFNα2 signal peptide. J. Biotechnol. 239, 57–60 (2016).
Peroutka, R. J., Elshourbagy, N., Piech, T. & Butt, T. R. Enhanced protein expression in mammalian cells using engineered SUMO fusions: secreted phospholipase A 2. Protein Sci. 17, 1586–1595 (2008).
Gorochowski, T. E. et al. Genetic circuit characterization and debugging using RNA‐seq. Mol. Syst. Biol. 13, 952 (2017).
Gach, P. C. et al. A droplet microfluidic platform for automating genetic engineering. ACS Synth. Biol. 5, 426–433 (2016).
Chao, R., Mishra, S., Si, T. & Zhao, H. Engineering biological systems using automated biofoundries. Metab. Eng. 42, 98–108 (2017).
Madison, A. C. et al. Scalable device for automated microbial electroporation in a digital micro fluidic platform. ACS Synth. Biol. 6, 1701–1709 (2017).
Chen, Z. & Elowitz, E. B. Programmable protein circuit design. Cell 184, 2284–2301 (2021).
Acknowledgements
We thank additional members of the Molecular Information Systems Lab for helpful discussion and feedback on this work. The OsmY expression plasmid was generously provided by C. Bryan and L. Carter (Institute for Protein Design, University of Washington). We also thank A. Heron and R. Gutierrez (Oxford Nanopore Technologies) for providing the configurable MinION run script and discussions on its use, and M. Jain (UCSC) for a custom Matlab script that facilitated visualization of the raw MinION data. This work was supported in part by NSF EAGER Award no. 1841188 and NSF CCF Award no. 2006864 to L.C. and J.N., an NIH/NCI Cancer Center Support Grant (no. P30 CA015704) Pilot Award and NSF Award 2021552 to J.N. and a sponsored research agreement from Oxford Nanopore Technologies.
Author information
Authors and Affiliations
Contributions
N.C., K.Z., A.N. and N.B. performed wet laboratory experiments. K.Z. and K.D. developed the data analysis pipeline and performed computational analyses. Z.S. implemented the machine learning approach. N.B., K.S., L.C. and J.N. supervised the project. J.N. conceived and directed the project. All authors contributed to writing and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
A provisional patent has been filed by the University of Washington covering aspects of this work (Patent Application no. 17/283,007). K.S. is an employee of Microsoft. J.N. is a consultant to Oxford Nanopore Technologies. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Biotechnology thanks Yi-Tao Long, Meni Wanunu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–13, Notes and References.
Rights and permissions
About this article
Cite this article
Cardozo, N., Zhang, K., Doroschak, K. et al. Multiplexed direct detection of barcoded protein reporters on a nanopore array. Nat Biotechnol 40, 42–46 (2022). https://doi.org/10.1038/s41587-021-01002-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-021-01002-6
This article is cited by
-
Nanopore DNA sequencing technologies and their applications towards single-molecule proteomics
Nature Chemistry (2024)
-
Enzyme-less nanopore detection of post-translational modifications within long polypeptides
Nature Nanotechnology (2023)
-
A nanopore interface for higher bandwidth DNA computing
Nature Communications (2022)
-
Nanopore-detectable reporter proteins
Nature Methods (2021)