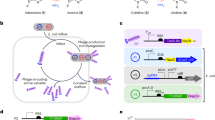
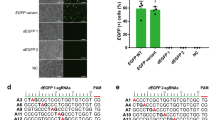
Abstract
Applications of adenine base editors (ABEs) have been constrained by the limited compatibility of the deoxyadenosine deaminase component with Cas homologs other than SpCas9. We evolved the deaminase component of ABE7.10 using phage-assisted non-continuous and continuous evolution (PANCE and PACE), which resulted in ABE8e. ABE8e contains eight additional mutations that increase activity (kapp) 590-fold compared with that of ABE7.10. ABE8e offers substantially improved editing efficiencies when paired with a variety of Cas9 or Cas12 homologs. ABE8e is more processive than ABE7.10, which could benefit screening, disruption of regulatory regions and multiplex base editing applications. A modest increase in Cas9-dependent and -independent DNA off-target editing, and in transcriptome-wide RNA off-target editing can be ameliorated by the introduction of an additional mutation in the TadA-8e domain. Finally, we show that ABE8e can efficiently install natural mutations that upregulate fetal hemoglobin expression in the BCL11A enhancer or in the the HBG promoter in human cells, targets that were poorly edited with ABE7.10. ABE8e augments the effectiveness and applicability of adenine base editing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
HTS data have been deposited in the NCBI Sequence Read Archive database (PRJNA589228). All plasmids encoding ABE8e variants in this study will be available through Addgene. A subset of selection plasmids used in this study will be available through Addgene. Other materials are available upon reasonable request.
Code availability
Custom script used to analyze processivity is available in Supplementary Note 1.
Change history
20 May 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41587-020-0562-8
References
Gaudelli, N. M. et al. Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Rees, H. A. & Liu, D. R. Base editing: precision chemistry on the genome and transcriptome of living cells. Nat. Rev. Genet. 19, 770–788 (2018).
Nishida, K. et al. Targeted nucleotide editing using hybrid prokaryotic and vertebrate adaptive immune systems. Science 353, aaf8729 (2016).
Komor, A. C. et al. Improved base excision repair inhibition and bacteriophage Mu Gam protein yields C:G-to-T:A base editors with higher efficiency and product purity. Sci. Adv. 3, eaao4774 (2017).
Koblan, L. W. et al. Improving cytidine and adenine base editors by expression optimization and ancestral reconstruction. Nat. Biotechnol. 36, 843–846 (2018).
Landrum, M. J. et al. ClinVar: public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 44, D862–D868 (2016).
Landrum, M. J. et al. ClinVar: public archive of relationships among sequence variation and human phenotype. Nucleic Acids Res. 42, D980–D985 (2014).
Komor, A. C., Badran, A. H. & Liu, D. R. Editing the genome without double-stranded DNA breaks. ACS Chem. Biol. 13, 383–388 (2018).
Villiger, L. et al. Treatment of a metabolic liver disease by in vivo genome base editing in adult mice. Nat. Med. 24, 1519–1525 (2018).
Ryu, S. M. et al. Adenine base editing in mouse embryos and an adult mouse model of Duchenne muscular dystrophy. Nat. Biotechnol. 36, 536–539 (2018).
Yeh, W. H., Chiang, H., Rees, H. A., Edge, A. S. B. & Liu, D. R. In vivo base editing of post-mitotic sensory cells. Nat. Commun. 9, 2184 (2018).
Tanaka, S. et al. In vivo targeted single-nucleotide editing in zebrafish. Sci. Rep. 8, 11423 (2018).
Zhang, Y. et al. Programmable base editing of zebrafish genome using a modified CRISPR-Cas9 system. Nat. Commun. 8, 118 (2017).
Ma, Y. et al. Highly efficient and precise base editing by engineered dCas9-guide tRNA adenosine deaminase in rats. Cell Discov. 4, 39 (2018).
Zafra, M. P. et al. Optimized base editors enable efficient editing in cells, organoids and mice. Nat. Biotechnol. 36, 888–893 (2018).
Liu, Z. et al. Efficient generation of mouse models of human diseases via ABE- and BE-mediated base editing. Nat. Commun. 9, 2338 (2018).
Liu, Z. et al. Highly efficient RNA-guided base editing in rabbit. Nat. Commun. 9, 2717 (2018).
Song, C.-Q. et al. Adenine base editing in an adult mouse model of tyrosinaemia. Nat. Biomed. Eng. 4, 125–130 (2019).
Hu, J. H. et al. Evolved Cas9 variants with broad PAM compatibility and high DNA specificity. Nature 556, 57–63 (2018).
Kim, Y. B. et al. Increasing the genome-targeting scope and precision of base editing with engineered Cas9-cytidine deaminase fusions. Nat. Biotechnol. 35, 371–376 (2017).
Huang, T. P. et al. Circularly permuted and PAM-modified Cas9 variants broaden the targeting scope of base editors. Nat. Biotechnol. 37, 626–631 (2019).
Kleinstiver, B. P. et al. Engineered CRISPR-Cas12a variants with increased activities and improved targeting ranges for gene, epigenetic and base editing. Nat. Biotechnol. 37, 276–282 (2019).
Li, X. et al. Base editing with a Cpf1-cytidine deaminase fusion. Nat. Biotechnol. 36, 324–327 (2018).
Nishimasu, H. et al. Engineered CRISPR-Cas9 nuclease with expanded targeting space. Science 361, 1259–1262 (2018).
Yourik, P., Fuchs, R. T., Mabuchi, M., Curcuru, J. L. & Robb, G. B. Staphylococcus aureus Cas9 is a multiple-turnover enzyme. RNA 25, 35–44 (2019).
Bryson, D. I. et al. Continuous directed evolution of aminoacyl-tRNA synthetases. Nat. Chem. Biol. 13, 1253–1260 (2017).
Carlson, J. C., Badran, A. H., Guggiana-Nilo, D. A. & Liu, D. R. Negative selection and stringency modulation in phage-assisted continuous evolution. Nat. Chem. Biol. 10, 216–222 (2014).
Dickinson, B. C., Leconte, A. M., Allen, B., Esvelt, K. M. & Liu, D. R. Experimental interrogation of the path dependence and stochasticity of protein evolution using phage-assisted continuous evolution. Proc. Natl Acad. Sci. USA 110, 9007–9012 (2013).
Dickinson, B. C., Packer, M. S., Badran, A. H. & Liu, D. R. A system for the continuous directed evolution of proteases rapidly reveals drug-resistance mutations. Nat. Commun. 5, 5352 (2014).
Hubbard, B. P. et al. Continuous directed evolution of DNA-binding proteins to improve TALEN specificity. Nat. Methods 12, 939–942 (2015).
Leconte, A. M. et al. A population-based experimental model for protein evolution: effects of mutation rate and selection stringency on evolutionary outcomes. Biochemistry 52, 1490–1499 (2013).
Packer, M. S., Rees, H. A. & Liu, D. R. Phage-assisted continuous evolution of proteases with altered substrate specificity. Nat. Commun. 8, 956 (2017).
Wang, T., Badran, A. H., Huang, T. P. & Liu, D. R. Continuous directed evolution of proteins with improved soluble expression. Nat. Chem. Biol. 14, 972–980 (2018).
Roth, T. B., Woolston, B. M., Stephanopoulos, G. & Liu, D. R. Phage-Assisted evolution of Bacillus methanolicus methanol Dehydrogenase 2. ACS Synth. Biol. 8, 796–806 (2019).
Thuronyi, B. W. et al. Continuous evolution of base editors with expanded target compatibility and improved activity. Nat. Biotechnol. 37, 1070–1079 (2019).
Badran, A. H. & Liu, D. R. In vivo continuous directed evolution. Curr. Opin. Chem. Biol. 24, 1–10 (2015).
Esvelt, K. M., Carlson, J. C. & Liu, D. R. A system for the continuous directed evolution of biomolecules. Nature 472, 499–503 (2011).
Badran, A. H. et al. Continuous evolution of Bacillus thuringiensis toxins overcomes insect resistance. Nature 533, 58–63 (2016).
Badran, A. H. & Liu, D. R. Development of potent in vivo mutagenesis plasmids with broad mutational spectra. Nat. Commun. 6, 8425 (2015).
Grunewald, J. et al. CRISPR DNA base editors with reduced RNA off-target and self-editing activities. Nat. Biotechnol. 37, 1041–1048 (2019).
Swarts, D. C., van der Oost, J. & Jinek, M. Structural basis for guide RNA processing and seed-dependent DNA targeting by CRISPR-Cas12a. Mol. Cell 66, 221–233 (2017).
Liang, P. et al. Genome-wide profiling of adenine base editor specificity by EndoV-seq. Nat. Commun. 10, 67 (2019).
Tsai, S. Q. et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat. Biotechnol. 33, 187–197 (2015).
Rees, H. A. et al. Improving the DNA specificity and applicability of base editing through protein engineering and protein delivery. Nat. Commun. 8, 15790 (2017).
Grunewald, J. et al. Transcriptome-wide off-target RNA editing induced by CRISPR-guided DNA base editors. Nature 569, 433–437 (2019).
Rees, H. A., Wilson, C., Doman, J. L. & Liu, D. R. Analysis and minimization of cellular RNA editing by DNA adenine base editors. Sci. Adv. 5, eaax5717 (2019).
Zhou, C. et al. Off-target RNA mutation induced by DNA base editing and its elimination by mutagenesis. Nature 571, 275–278 (2019).
Doman, J. L., Raguram, A., Newby, G. A. & Liu, D. R. Evaluation and minimization of Cas9-independent off-target DNA editing by cytosine base editors. Nat. Biotechnol. https://doi.org/10.1038/s41587-020-0414-6 (2020).
Amato, A. et al. Interpreting elevated fetal hemoglobin in pathology and health at the basic laboratory level: new and known ɣ- gene mutations associated with hereditary persistence of fetal hemoglobin. Int. J. Lab. Hematol. 36, 13–19 (2014).
Canver, M. C. et al. BCL11A enhancer dissection by Cas9-mediated in situ saturating mutagenesis. Nature 527, 192–197 (2015).
Wu, Y. et al. Highly efficient therapeutic gene editing of human hematopoietic stem cells. Nat. Med. 25, 776–783 (2019).
Vierstra, J. et al. Functional footprinting of regulatory DNA. Nat. Methods 12, 927–930 (2015).
Powars, D. R., Weiss, J. N., Chan, L. S. & Schroeder, W. A. Is there a threshold level of fetal hemoglobin that ameliorates morbidity in sickle cell anemia? Blood 63, 921–926 (1984).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Yao, M., Hatahet, Z., Melamede, R. J. & Kow, Y. W. Purification and characterization of a novel deoxyinosine-specific enzyme, deoxyinosine 3’ endonuclease, from Escherichia coli. J. Biol. Chem. 269, 16260–16268 (1994).
Dalhus, B. et al. Structures of endonuclease V with DNA reveal initiation of deaminated adenine repair. Nat. Struct. Mol. Biol. 16, 138–143 (2009).
Clement, K. et al. CRISPResso2 provides accurate and rapid genome editing sequence analysis. Nat. Biotechnol. 37, 224–226 (2019).
Acknowledgements
This work was supported by US National Institutes of Health awards U01 AI142756, RM1 HG009490, R01 EB022376 and R35 GM118062, St. Jude Collaborative Research Consortium, the Bill and Melinda Gates Foundation and the Howard Hughes Medical Institute (HHMI). M.F.R. was supported by an HHMI Hanna Gray Fellowship. K.T.Z. was supported by Harvard Chemical Biology Training Grant (T32 GM095450). G.A.N was supported by the Helen Hay Whitney Fellowship. C.W. was supported as a Marlon Abbe Fellow of the Damon Runyon Cancer Research Foundation (DRG-2343-18). L.W.K. was supported by a National Science Foundation Graduate Research Fellowship Program. D.E.B. was supported by the National Heart, Lung, and Blood Institute (P01HL053749), Burroughs Wellcome Fund and the St. Jude Children’s Research Hospital Collaborative Research Consortium. We thank S. Miller and T. Wang for providing sgRNA plasmids. We thank A. Raguram for help with computational analyses. We thank J. Doman and A. Raguram for plasmids used during the orthogonal R-loop assay.
Author information
Authors and Affiliations
Contributions
M.F.R. and K.T.Z. conducted the experiments, performed analyses, and wrote the manuscript. E.E., G.A.N, A.L., B.W.T, C.W. and L.W.K. conducted the experiments and performed analyses. J.Z. and D.E.B. provided information on disease loci. J.A.D. provided feedback on biochemical analyses. D.R.L supervised the research and wrote the manuscript. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare competing financial interests. D.R.L. is a consultant and co-founder of Editas Medicine, Pairwise Plants, Beam Therapeutics, and Prime Medicine, companies that use genome editing. The authors have filed patent applications on evolved ABEs. The Regents of the University of California have patents issued and pending for CRISPR technologies on which J.A.D. is an inventor. J.A.D. is a co-founder of Caribou Biosciences, Editas Medicine, Scribe Therapeutics, and Mammoth Biosciences, and a scientific advisory board member of Caribou Biosciences, Intellia Therapeutics, eFFECTOR Therapeutics, Scribe Therapeutics, Mammoth Biosciences, Synthego, and Inari; she is a director at Johnson & Johnson. The authors declare no competing non-financial interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Mutation table of variants from PANCE and PACE.
Data were obtained by sequencing individual plaques. Conserved mutations are bolded. Mutations that are highlighted in the structure in Fig. 2b are highlighted to match the amino acid positions in the structure. Genotypes in red were tested for base editing activity in mammalian cells.
Extended Data Fig. 2 PACE schedule for deoxyadenosine deaminase evolution.
Lagoon L1 contains host cells harboring P1, P2, and P3e. Lagoons L2 and L3 contain host cells harboring P1, P2, and P3g, which form a more stringent selection circuit than the circuit in lagoon L1. For details on plasmids, see Supplementary Table 1. The stringency of the ABE selection was further modulated by increasing the lagoon flow rate (dashed lines). For the first 12 hours, gene III was expressed by the addition of anhydrotetracycline to enable genetic drift in the absence of selection pressure12,13.
Extended Data Fig. 3 Titration data at eight editor doses comparing base editing efficiencies for ABE8e and ABE8e-dimer at three sites in HEK293T cells.
Base editing with ABE8e and ABE8e-dimer in HEK293T cells at three genomic sites in HEK293T cells. Transfections were performed with constant amount of sgRNA plasmid but eight varying doses of ABE plasmid. For all plots, dots represent individual biological replicates and bars represent mean±s.d. of three independent biological replicates.
Extended Data Fig. 4 TadA-8e V106W analysis for SaCas9 and LbCas12a.
a, DNA editing comparing SaABE7.10, SaABE8e, and SaABE8e(TadA-8e V106W) at four genomic sites in HEK293T cells. b, DNA editing comparing LbABE7.10, LbABE8e, and LbABE8e(TadA-8e V106W) at six genomic sites in HEK293T cells. For all plots, dots represent individual biological replicates and bars represent mean±s.d. of three independent biological replicates.
Supplementary information
Supplementary Information
Supplementary Figs. 1–17, Tables 1–5, Note 1 and Sequences 1
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Richter, M.F., Zhao, K.T., Eton, E. et al. Phage-assisted evolution of an adenine base editor with improved Cas domain compatibility and activity. Nat Biotechnol 38, 883–891 (2020). https://doi.org/10.1038/s41587-020-0453-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-020-0453-z
This article is cited by
-
CRISPR base editing-mediated correction of a tau mutation rescues cognitive decline in a mouse model of tauopathy
Translational Neurodegeneration (2024)
-
Precise fine-turning of GhTFL1 by base editing tools defines ideal cotton plant architecture
Genome Biology (2024)
-
CRISPR technologies for genome, epigenome and transcriptome editing
Nature Reviews Molecular Cell Biology (2024)
-
Domain-inlaid Nme2Cas9 adenine base editors with improved activity and targeting scope
Nature Communications (2024)
-
Glycosylase-based base editors for efficient T-to-G and C-to-G editing in mammalian cells
Nature Biotechnology (2024)