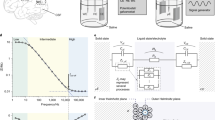
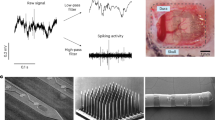
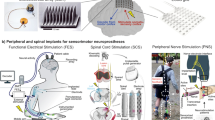
Abstract
Periodically throughout history developments from adjacent fields of science and technology reach a tipping point where together they produce unparalleled advances, such as the Allen Brain Atlas and the Human Genome Project. Today, research focused at the interface between the nervous system and electronics is not only leading to advances in fundamental neuroscience, but also unlocking the potential of implants capable of cellular-level therapeutic targeting. Ultimately, these personalized electronic therapies will provide new treatment modalities for neurodegenerative and neuropsychiatric illness;Work from our laboratory powerful control of prosthetics for restorative function in degenerative diseases, trauma and amputation; and even augmentation of human cognition. Overall, we believe that emerging advances in tissue-like electronics will enable minimally invasive devices capable of establishing a stable long-term cellular neural interface and providing long-term treatment for chronic neurological conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
24 October 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Herrington, T. M., Cheng, J. J. & Eskandar, E. N. Mechanisms of deep brain stimulation. J. Neurophysiol. 115, 19–38 (2016).
Miocinovic, S., Somayajula, S., Chitnis, S. & Vitek, J. L. History, applications, and mechanisms of deep brain stimulation. JAMA Neurol. 70, 163–171 (2013).
Kringelbach, M. L., Jenkinson, N., Owen, S. L. F. & Aziz, T. Z. Translational principles of deep brain stimulation. Nat. Rev. Neurosci. 8, 623–635 (2007).
Kook, G., Lee, S. W., Lee, H. C., Cho, I.-J. & Lee, H. J. Neural probes for chronic applications. Micromachines (Basel) 7, 179 (2016).
Wellman, S. M. et al. A materials roadmap to functional neural interface design. Adv. Funct. Mater. 28, 1701269 (2018).
Fattahi, P., Yang, G., Kim, G. & Abidian, M. R. A review of organic and inorganic biomaterials for neural interfaces. Adv. Mater. 26, 1846–1885 (2014).
Hong, G., Viveros, R. D., Zwang, T. J., Yang, X. & Lieber, C. M. Tissue-like neural probes for understanding and modulating the brain. Biochemistry 57, 3995–4004 (2018).
Hong, G., Yang, X., Zhou, T. & Lieber, C. M. Mesh electronics: a new paradigm for tissue-like brain probes. Curr. Opin. Neurobiol. 50, 33–41 (2018).
Yang, X. et al. Bioinspired neuron-like electronics. Nat. Mater. 18, 510–517, https://doi.org/10.1038/s41563-019-0292-9 (2019).
Sun, F. T. & Morrell, M. J. Closed-loop neurostimulation: the clinical experience. Neurotherapeutics 11, 553–563 (2014).
Parastarfeizabadi, M. & Kouzani, A. Z. Advances in closed-loop deep brain stimulation devices. J. Neuroeng. Rehabil. 14, 79 (2017).
Gilja, V. et al. Clinical translation of a high-performance neural prosthesis. Nat. Med. 21, 1142–1145 (2015).
Aflalo, T. et al. Decoding motor imagery from the posterior parietal cortex of a tetraplegic human. Science 348, 906–910 (2015).
Zeng, F.-G., Rebscher, S., Harrison, W., Sun, X. & Feng, H. Cochlear implants: system design, integration, and evaluation. IEEE Rev. Biomed. Eng. 1, 115–142 (2008).
Hadjinicolaou, A. E., Meffin, H., Maturana, M. I., Cloherty, S. L. & Ibbotson, M. R. Prosthetic vision: devices, patient outcomes and retinal research. Clin. Exp. Optom. 98, 395–410 (2015).
Lewis, P. M., Ackland, H. M., Lowery, A. J. & Rosenfeld, J. V. Restoration of vision in blind individuals using bionic devices: a review with a focus on cortical visual prostheses. Brain Res. 1595, 51–73 (2015).
Rosin, B. et al. Closed-loop deep brain stimulation is superior in ameliorating parkinsonism. Neuron 72, 370–384 (2011).
Lo, M.-C. & Widge, A. S. Closed-loop neuromodulation systems: next-generation treatments for psychiatric illness. Int. Rev. Psychiatry 29, 191–204 (2017).
Ezzyat, Y. et al. Closed-loop stimulation of temporal cortex rescues functional networks and improves memory. Nat. Commun. 9, 365 (2018).
Kellaway, P. The part played by electric fish in the early history of bioelectricity and electrotherapy. Bull. Hist. Med. 20, 112–137 (1946).
Ramirez-Zamora, A. et al. Evolving applications, technological challenges and future opportunities in neuromodulation: proceedings of the Fifth Annual Deep Brain Stimulation Think Tank. Front. Neurosci. 11, 734 (2018).
Cagnan, H., Denison, T., McIntyre, C. & Brown, P. Emerging technologies for improved deep brain stimulation. Nat. Biotechnol. https://doi.org/10.1038/s41587-019-0244-6 (2019).
van Dijk, K. J. et al. A novel lead design enables selective deep brain stimulation of neural populations in the subthalamic region. J. Neural Eng. 12, 046003 (2015).
Lein, E. S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
McIntyre, C. C., Chaturvedi, A., Shamir, R. R. & Lempka, S. F. Engineering the next generation of clinical deep brain stimulation technology. Brain Stimul. 8, 21–26 (2015).
Fisher, B. et al. Battery longevity comparison of two commonly available dual channel implantable pulse generators used for subthalamic nucleus stimulation in Parkinson’s disease. Stereotact. Funct. Neurosurg. 96, 151–156 (2018).
Park, K. et al. Battery life matters in deep brain stimulation. Stereotact. Funct. Neurosurg. 96, 65–66 (2018).
Helmers, A. K. et al. Comparison of the battery life of nonrechargeable generators for deep brain stimulation. Neuromodulation 21, 593–596 (2018).
Cicchetti, F. & Barker, R. A. The glial response to intracerebrally delivered therapies for neurodegenerative disorders: is this a critical issue? Front. Pharmacol. 5, 139 (2014).
Buhlmann, J., Hofmann, L., Tass, P. A. & Hauptmann, C. Modeling of a segmented electrode for desynchronizing deep brain stimulation. Front. Neuroeng. 4, 15 (2011).
Alonso, F., Latorre, M. A., Göransson, N., Zsigmond, P. & Wårdell, K. Investigation into deep brain stimulation lead designs: a patient-specific simulation study. Brain Sci. 6, 39 (2016).
Teplitzky, B. A., Zitella, L. M., Xiao, Y. & Johnson, M. D. Model-based comparison of deep brain stimulation array functionality with varying number of radial electrodes and machine learning feature sets. Front. Comput. Neurosci. 10, 58 (2016).
Little, S. et al. Bilateral adaptive deep brain stimulation is effective in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 87, 717–721 (2016).
Starr, P. A. Totally implantable bidirectional neural prostheses: a flexible platform for innovation in neuromodulation. Front. Neurosci. 12, 619 (2018).
Maling, N., Lempka, S. F., Blumenfeld, Z., Bronte-Stewart, H. & McIntyre, C. C. Biophysical basis of subthalamic local field potentials recorded from deep brain stimulation electrodes. J. Neurophysiol. 120, 1932–1944 (2018).
Swann, N. C. et al. Adaptive deep brain stimulation for Parkinson’s disease using motor cortex sensing. J. Neural Eng. 15, 046006 (2018).
Molina, R. et al. Report of a patient undergoing chronic responsive deep brain stimulation for Tourette syndrome: proof of concept. J. Neurosurg. 129, 308–314 (2018).
Shute, J. B. et al. Thalamocortical network activity enables chronic tic detection in humans with Tourette syndrome. Neuroimage Clin. 12, 165–172 (2016).
Bergey, G. K. et al. Long-term treatment with responsive brain stimulation in adults with refractory partial seizures. Neurology 84, 810–817 (2015).
Miranda, R. A. et al. DARPA-funded efforts in the development of novel brain-computer interface technologies. J. Neurosci. Methods 244, 52–67 (2015).
Neely, R. M., Piech, D. K., Santacruz, S. R., Maharbiz, M. M. & Carmena, J. M. Recent advances in neural dust: towards a neural interface platform. Curr. Opin. Neurobiol. 50, 64–71 (2018).
Heelan, C., Nurmikko, A. V. & Truccolo, W. FPGA implementation of deep-learning recurrent neural networks with sub-millisecond real-time latency for BCI-decoding of large-scale neural sensors (104 nodes). Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018, 1070–1073 (2018).
Shenoy, K. V. & Carmena, J. M. Combining decoder design and neural adaptation in brain-machine interfaces. Neuron 84, 665–680 (2014).
Wheeler, J. J. et al. An implantable 64-channel neural interface with reconfigurable recording and stimulation. in 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 7837–7840 (IEEE, 2015); https://doi.org/10.1109/EMBC.2015.7320208
Hamilton, L. et al. Neural signal processing and closed-loop control algorithm design for an implanted neural recording and stimulation system. in 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 7831–7836 (IEEE, 2015); https://doi.org/10.1109/EMBC.2015.7320207
Bjune, C. K. et al. Package architecture and component design for an implanted neural stimulator with closed loop control. in 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 7825–7830 (IEEE, 2015); https://doi.org/10.1109/EMBC.2015.7320206
Reardon, S. Worldwide brain-mapping project sparks excitement — and concern. Nature 537, 597 (2016).
Patil, A. C. & Thakor, N. V. Implantable neurotechnologies: a review of micro- and nanoelectrodes for neural recording. Med. Biol. Eng. Comput. 54, 23–44 (2016).
Hong, G. & Lieber, C. M. Novel electrode technologies for neural recordings. Nat. Rev. Neurosci. 19, 199 (2019).
Frank, J.A., Antonini, M.-J. & Anikeeva, P. Next-generation interfaces for studying neural function. Nat. Biotechnol. https://doi.org/10.1038/s41587-019-0198-8 (2019).
Hubel, D. H. & Wiesel, T. N. Receptive fields of single neurones in the cat’s striate cortex. J. Physiol. (Lond.) 148, 574–591 (1959).
O’Keefe, J. & Dostrovsky, J. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res. 34, 171–175 (1971).
Hafting, T., Fyhn, M., Molden, S., Moser, M.-B. & Moser, E. I. Microstructure of a spatial map in the entorhinal cortex. Nature 436, 801–806 (2005).
Georgopoulos, A. P., Schwartz, A. B. & Kettner, R. E. Neuronal population coding of movement direction. Science 233, 1416–1419 (1986).
Normann, R. A. & Fernández, E. Clinical applications of penetrating neural interfaces and Utah Electrode Array technologies. J. Neural Eng. 13, 061003 (2016).
Hochberg, L. R. et al. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 485, 372–375 (2012).
Jun, J. J. et al. Fully integrated silicon probes for high-density recording of neural activity. Nature 551, 232–236 (2017).
Raducanu, B. C. et al. Time multiplexed active neural probe with 1356 parallel recording sites. Sensors (Basel) 17, 2388 (2017).
Stringer, C. et al. Spontaneous behaviors drive multidimensional, brainwide activity. Science 364, 255 (2019).
Lacour, S. P., Courtine, G. & Guck, J. Materials and technologies for soft implantable neuroprostheses. Nat. Rev. Mater. 1, 16063 (2016).
Chen, R., Canales, A. & Anikeeva, P. Neural recording and modulation technologies. Nat. Rev. Mater. 2, 16093 (2017).
Polikov, V. S., Tresco, P. A. & Reichert, W. M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 148, 1–18 (2005).
Kandel, E. Principles of Neural Science 5th edn (McGraw Hill Professional, 2013).
Ghane-Motlagh, B. & Sawan, M. Design and implementation challenges of microelectrode arrays: a review. Mater. Sci. Appl. 4, 483–495 (2013).
Garcia, J. A., Pena, J. M., McHugh, S. & Jerusalem, A. A model of the spatially dependent mechanical properties of the axon during its growth. Comput. Model. Eng. Sci. 87, 411–432 (2012).
Wang, S. S. H. et al. Functional trade-offs in white matter axonal scaling. J. Neurosci. 28, 4047–4056 (2008).
Terem, I. et al. Revealing sub-voxel motions of brain tissue using phase-based amplified MRI (aMRI). Magn. Reson. Med. 80, 2549–2559 (2018).
Tyler, W. J. The mechanobiology of brain function. Nat. Rev. Neurosci. 13, 867–878 (2012).
Kasthuri, N. et al. Saturated reconstruction of a volume of neocortex. Cell 162, 648–661 (2015).
Saxena, T. & Bellamkonda, R. V. Implantable electronics: a sensor web for neurons. Nat. Mater. 14, 1190–1191 (2015).
Fu, T.-M., Hong, G., Viveros, R. D., Zhou, T. & Lieber, C. M. Highly scalable multichannel mesh electronics for stable chronic brain electrophysiology. Proc. Natl Acad. Sci. USA 114, E10046–E10055 (2017).
Hong, G. et al. Syringe injectable electronics: precise targeted delivery with quantitative input/output connectivity. Nano Lett. 15, 6979–6984 (2015).
Hong, G. et al. A method for single-neuron chronic recording from the retina in awake mice. Science 360, 1447–1451 (2018).
Zhou, T. et al. Syringe-injectable mesh electronics integrate seamlessly with minimal chronic immune response in the brain. Proc. Natl Acad. Sci. USA 114, 5894–5899 (2017).
Fu, T.-M. et al. Stable long-term chronic brain mapping at the single-neuron level. Nat. Methods 13, 875–882 (2016).
Mann, A. et al. Chronic deep brain stimulation in an Alzheimer’s disease mouse model enhances memory and reduces pathological hallmarks. Brain Stimul. 11, 435–444 (2018).
Cui, Y., Wei, Q., Park, H. & Lieber, C. M. Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species. Science 293, 1289–1292 (2001).
Gao, N. et al. General strategy for biodetection in high ionic strength solutions using transistor-based nanoelectronic sensors. Nano Lett. 15, 2143–2148 (2015).
Calabresi, P., Picconi, B., Tozzi, A., Ghiglieri, V. & Di Filippo, M. Direct and indirect pathways of basal ganglia: a critical reappraisal. Nat. Neurosci. 17, 1022–1030 (2014).
Choi, S. H. et al. Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model. Science 361, eaan8821 (2018).
Boulanger-Weill, J. et al. Functional interactions between newborn and mature neurons leading to integration into established neuronal circuits. Curr. Biol. 27, 1707–1720.e5 (2017).
Spitzer, N. C. Electrical activity in early neuronal development. Nature 444, 707–712 (2006).
Moore-Kochlacs, C. et al. Principles of high-fidelity, high-density 3-D neural recording. BMC Neurosci. 15, 122 (2014).
Guo, L. The pursuit of chronically reliable neural interfaces: a materials perspective. Front. Neurosci. 10, 599 (2016).
Patolsky, F. et al. Detection, stimulation, and inhibition of neuronal signals with high-density nanowire transistor arrays. Science 313, 1100–1104 (2006).
Tian, B. et al. Three-dimensional, flexible nanoscale field-effect transistors as localized bioprobes. Science 329, 830–834 (2010).
Qing, Q. et al. Free-standing kinked nanowire transistor probes for targeted intracellular recording in three dimensions. Nat. Nanotechnol. 9, 142–147 (2014).
Acknowledgements
S.R.P. is supported by the Cure Alzheimer’s Fund and the Henry and Allison McCance Center. C.M.L. acknowledges support of this work by the Air Force Office of Scientific Research (FA9550-14-1-0136) and a National Institutes of Health Director’s Pioneer Award (1DP1EB025835-01).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
C.M.L. is a co-inventor on patents and patent applications relating to the article that have been filed by the authors’ institution (Harvard University) as follows: ‘Scaffolds comprising nanoelectronic components, tissues, and other applications’, inventors C.M.L., J. Liu, B. Tian, T. Dvir, R. S. Langer and D. S. Kohane; US9,457,128 (issued); describes nanoscale transistors for cell recording. ‘Systems and methods for injectable devices’, inventors C.M.L., J. Liu, Z. Cheng, G. Hong, T.-M. Fu and T. Zhou; 61/975,601 (pending), PCT/US2015/024252 (pending) and 15/301,792 (pending); describes injectable mesh electronics. ‘Techniques and systems for injection and/or connection of electrical devices’, inventors C.M.L., G. Hong, T.-M. Fu and J. Huang; 62/209,255 (pending), PCT/US2016/045587 (issued) and 15/749,617 (pending); describes injection method of mesh electronics. The authors are not involved in efforts related to commercialization of this intellectual property.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patel, S.R., Lieber, C.M. Precision electronic medicine in the brain. Nat Biotechnol 37, 1007–1012 (2019). https://doi.org/10.1038/s41587-019-0234-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-019-0234-8
This article is cited by
-
3D spatiotemporally scalable in vivo neural probes based on fluorinated elastomers
Nature Nanotechnology (2024)
-
Why converging technologies need converging international regulation
Ethics and Information Technology (2024)
-
Genetically targeted chemical assembly
Nature Reviews Bioengineering (2023)
-
Flexible brain–computer interfaces
Nature Electronics (2023)
-
Bioresorbable thin-film silicon diodes for the optoelectronic excitation and inhibition of neural activities
Nature Biomedical Engineering (2022)