Abstract
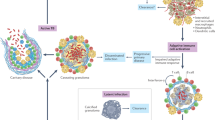
An effective vaccine is needed for the prevention and elimination of malaria. The only immunogens that have been shown to have a protective efficacy of more than 90% against human malaria are Plasmodium falciparum (Pf) sporozoites (PfSPZ) manufactured in mosquitoes (mPfSPZ)1,2,3,4,5,6,7. The ability to produce PfSPZ in vitro (iPfSPZ) without mosquitoes would substantially enhance the production of PfSPZ vaccines and mosquito-stage malaria research, but this ability is lacking. Here we report the production of hundreds of millions of iPfSPZ. iPfSPZ invaded human hepatocytes in culture and developed to mature liver-stage schizonts expressing P. falciparum merozoite surface protein 1 (PfMSP1) in numbers comparable to mPfSPZ. When injected into FRGhuHep mice containing humanized livers, iPfSPZ invaded the human hepatocytes and developed to PfMSP1-expressing late liver stage parasites at 45% the quantity of cryopreserved mPfSPZ. Human blood from FRGhuHep mice infected with iPfSPZ produced asexual and sexual erythrocytic-stage parasites in culture, and gametocytes developed to PfSPZ when fed to mosquitoes, completing the P. falciparum life cycle from infectious gametocyte to infectious gametocyte without mosquitoes or primates.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The NCBI BioProject accession number for the RNA-seq fastq files generated in this study is PRJNA753927. Details on how to use the files are provided in Supplementary Table 4. Genomes were obtained from PlasmoDB v.32 (https://plasmodb.org/plasmo/app/downloads/release-32/Pfalciparum3D7/); BDGP6.32, https://nov2020.archive.ensembl.org/Drosophila_melanogaster/Info/Index); and UC Irvine for A. stephensi (v.1.0, https://www.ncbi.nlm.nih.gov/genome/2653?genome_assembly_id=985930).
References
Hoffman, S. L. et al. Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum. Vaccin. 6, 97–106 (2010).
Seder, R. A. et al. Protection against malaria by intravenous immunization with a nonreplicating sporozoite vaccine. Science 341, 1359–1365 (2013).
Epstein, J. E. et al. Protection against Plasmodium falciparum malaria by PfSPZ vaccine. JCI Insight 2, e89154 (2017).
Mordmuller, B. et al. Sterile protection against human malaria by chemoattenuated PfSPZ vaccine. Nature 542, 445–449 (2017).
Jongo, S. A. et al. Increase of dose associated with decrease in protection against controlled human malaria infection by PfSPZ vaccine in Tanzanian adults. Clin. Infect. Dis. 71, 2849–2857 (2020).
Mwakingwe-Omari, A. et al. Two chemoattenuated PfSPZ malaria vaccines induce sterile hepatic immunity. Nature 595, 289–294 (2021).
Sissoko, M. S. et al. Safety and efficacy of a three-dose regimen of Plasmodium falciparum sporozoite vaccine in adults during an intense malaria transmission season in Mali: a randomised, controlled phase 1 trial. Lancet Infect. Dis. 22, 377–389 (2022).
Trager, W. & Jensen, J. B. Human malaria parasites in continuous culture. Science 193, 673–675 (1976).
Haynes, J. D., Diggs, C. L., Hines, F. A. & Desjardins, R. E. Culture of human malaria parasites Plasmodium falciparum. Nature 263, 767–769 (1976).
Ifediba, T. & Vanderberg, J. P. Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294, 364–366 (1981).
Campbell, C. C., Collins, W. E., Nguyen Dinh, P., Barber, A. & Broderson, J. R. Plasmodium falciparum gametocytes from culture in vitro develop to sporozoites that are infectious to primates. Science 217, 1048–1050 (1982).
Warburg, A. & Schneider, I. In vitro culture of the mosquito stages of Plasmodium falciparum. Exp. Parasitol. 76, 121–126 (1993).
World Malaria Report 2021 (WHO, 2021).
Epstein, J. E. et al. Live attenuated malaria vaccine designed to protect through hepatic CD8+ T cell immunity. Science 334, 475–480 (2011).
Ishizuka, A. S. et al. Protection against malaria at 1 year and immune correlates following PfSPZ vaccination. Nat. Med. 22, 614–623 (2016).
Sissoko, M. S. et al. Safety and efficacy of PfSPZ Vaccine against Plasmodium falciparum via direct venous inoculation in healthy malaria-exposed adults in Mali: a randomised, double-blind phase 1 trial. Lancet Infect. Dis. 17, 498–509 (2017).
Lyke, K. E. et al. Attenuated PfSPZ Vaccine induces strain-transcending T cells and durable protection against heterologous controlled human malaria infection. Proc. Natl Acad. Sci. USA 114, 2711–2716 (2017).
Goswami, D. et al. A replication-competent late liver stage–attenuated human malaria parasite. JCI Insight 5, e135589 (2020).
Warburg, A. & Miller, L. H. Sporogonic development of a malaria parasite in vitro. Science 255, 448–450 (1992).
Al-Olayan, E. M., Beetsma, A. L., Butcher, G. A., Sinden, R. E. & Hurd, H. Complete development of mosquito phases of the malaria parasite in vitro. Science 295, 677–679 (2002).
Porter-Kelley, J. M. et al. Plasmodium yoelii: axenic development of the parasite mosquito stages. Exp. Parasitol. 112, 99–108 (2006).
Barr, P. J. et al. Recombinant Pfs25 protein of Plasmodium falciparum elicits malaria transmission-blocking immunity in experimental animals. J. Exp. Med. 174, 1203–1208 (1991).
Posthuma, G. et al. Immunogold localization of circumsporozoite protein of the malaria parasite Plasmodium falciparum during sporogony in Anopheles stephensi midguts. J. Cell Biol. 46, 18–24 (1988).
Benton, G., Arnaoutova, I., George, J., Kleinman, H. K. & Koblinski, J. Matrigel: from discovery and ECM mimicry to assays and models for cancer research. Adv. Drug Deliv. Rev. 79-80, 3–18 (2014).
Hoffman, S. L. et al. Sporozoite vaccine induces genetically restricted T cell elimination of malaria from hepatocytes. Science 244, 1078–1081 (1989).
Doolan, D. L. & Hoffman, S. L. The complexity of protective immunity against liver-stage malaria. J. Immunol. 165, 1453–1462 (2000).
Hoffman, S. L. & Doolan, D. L. Malaria vaccines-targeting infected hepatocytes. Nat. Med. 6, 1218–1219 (2000).
Weiss, W. R. & Jiang, C. G. Protective CD8+ T lymphocytes in primates immunized with malaria sporozoites. PLoS ONE 7, e31247 (2012).
Camponovo, F. et al. Proteome-wide analysis of a malaria vaccine study reveals personalized humoral immune profiles in Tanzanian adults. eLife 9, e53080 (2020).
Aly, A. S., Vaughan, A. M. & Kappe, S. H. Malaria parasite development in the mosquito and infection of the mammalian host. Annu. Rev. Microbiol. 63, 195–221 (2009).
Longley, R. J. et al. Comparative assessment of vaccine vectors encoding ten malaria antigens identifies two protective liver-stage candidates. Sci. Rep. 5, 11820 (2015).
Atella, G. C., Silva-Neto, M. A., Golodne, D. M., Arefin, S. & Shahabuddin, M. Anopheles gambiae lipophorin: characterization and role in lipid transport to developing oocyte. Insect Biochem. Mol. Biol. 36, 375–386 (2006).
Costa, G. et al. Non-competitive resource exploitation within mosquito shapes within-host malaria infectivity and virulence. Nat. Commun. 9, 3474 (2018).
Gare, D. C., Piertney, S. B. & Billingsley, P. F. Anopheles gambiae collagen IV genes: cloning, phylogeny and midgut expression associated with blood feeding and Plasmodium infection. Int. J. Parasitol. 33, 681–690 (2003).
Nacer, A., Walker, K. & Hurd, H. Localisation of laminin within Plasmodium berghei oocysts and the midgut epithelial cells of Anopheles stephensi. Parasit. Vectors 1, 33 (2008).
Ponnudurai, T., Meuwissen, J. H., Leeuwenberg, A. D., Verhave, J. P. & Lensen, A. H. The production of mature gametocytes of Plasmodium falciparum in continuous cultures of different isolates infective to mosquitoes. Trans. R. Soc. Trop. Med. Hyg. 76, 242–250 (1982).
Li, T. et al. Robust, reproducible, industrialized, standard membrane feeding assay for assessing the transmission blocking activity of vaccines and drugs against Plasmodium falciparum. Malar. J. 14, 150 (2015).
Feldmann, A. M. & Ponnudurai, T. Selection of Anopheles stephensi for refractoriness and susceptibility to Plasmodium falciparum. Med. Vet. Entomol. 3, 41–52 (1989).
Bounkeua, V., Li, F. & Vinetz, J. M. In vitro generation of Plasmodium falciparum ookinetes. Am. J. Trop. Med. Hyg. 83, 1187–1194 (2010).
FiberCell Systems Hollow Fiber Cell Culture: An Overview (FibreCellSystems); https://www.fibercellsystems.com/instructional-video-fibercell-systems-hollow-fiber-cell-culture-an-overview/ (2012).
Operation of a FiberCell Systems Duet Pump (FibreCellSystems); https://www.fibercellsystems.com/instructional-video-operation-of-a-fibercell-systems-duet-pump/ (2013).
Zavala, F., Gwadz, R. W., Collins, F. H., Nussenzweig, R. S. & Nussenzweig, V. Monoclonal antibodies to circumsporozoite proteins identify the species of malaria parasites in infected mosquitoes. Nature 299, 737–738 (1982).
Roestenberg, M. et al. Controlled human malaria infections by intradermal injection of cryopreserved Plasmodium falciparum sporozoites. Am. J. Trop. Med. Hyg. 88, 5–13 (2013).
Sattabongkot, J. et al. Establishment of a human hepatocyte line that supports in vitro development of the exo-erythrocytic stages of the malaria parasites Plasmodium falciparum and P. vivax. Am. J. Trop. Med. Hyg. 74, 708–715 (2006).
Holder, A. A. The carboxy-terminus of merozoite surface protein 1: structure, specific antibodies and immunity to malaria. Parasitology 136, 1445–1456 (2009).
Tsuji, M., Mattei, D., Nussenzweig, R. S., Eichinger, D. & Zavala, F. Demonstration of heat-shock protein 70 in the sporozoite stage of malaria parasites. Parasitol. Res. 80, 16–21 (1994).
Sanchez, G. I., Rogers, W. O., Mellouk, S. & Hoffman, S. L. Plasmodium falciparum: exported protein-1, a blood stage antigen, is expressed in liver stage parasites. Exp. Parasitol. 79, 59–62 (1994).
Guerin-Marchand, C. et al. A liver-stage-specific antigen of Plasmodium falciparum characterized by gene cloning. Nature 329, 164–167 (1987).
Vaughan, A. M. et al. Complete Plasmodium falciparum liver-stage development in liver-chimeric mice. J. Clin. Invest. 122, 3618–3628 (2012).
Kefi, M. et al. New rapid one-step PCR diagnostic assay for Plasmodium falciparum infective mosquitoes. Sci. Rep. 8, 1462 (2018).
Mensah, V. A. et al. Safety, immunogenicity and efficacy of prime-boost vaccination with ChAd63 and MVA encoding ME-TRAP against Plasmodium falciparum infection in adults in Senegal. PLoS ONE 11, e0167951 (2016).
Zanghì, G. et al. A specific PfEMP1 is expressed in P. falciparum sporozoites and plays a role in hepatocyte infection. Cell Rep. 22, 2951–2963 (2018).
Gardner, M. J. et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 419, 498–511 (2002).
Celniker, S. E. et al. Finishing a whole-genome shotgun: release 3 of the Drosophila melanogaster euchromatic genome sequence. Genome Biol. 3, research0079.1 (2002).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-seq. Nat. Methods 5, 621–628 (2008).
Wagner, G. P., Kin, K. & Lynch, V. J. Measurement of mRNA abundance using RNA-seq data: RPKM measure is inconsistent among samples. Theory Biosci. 131, 281–285 (2012).
Breitling, R., Armengaud, P., Amtmann, A. & Herzyk, P. Rank products: a simple, yet powerful, new method to detect differentially regulated genes in replicated microarray experiments. FEBS Lett. 573, 83–92 (2004).
Tusher, V. G., Tibshirani, R. & Chu, G. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl Acad. Sci. USA 98, 5116–5121 (2001).
Robinson, M. D. & Smyth, G. K. Small-sample estimation of negative binomial dispersion, with applications to SAGE data. Biostatistics 9, 321–332 (2008).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome Biol. 11, R106 (2010).
Diehl, K. H. et al. A good practice guide to the administration of substances and removal of blood, including routes and volumes. J. Appl. Toxicol. 21, 15–23 (2001).
Acknowledgements
We thank A. Warburg and E. H. Hurd for advice regarding in vitro culture; T. L. Richie for reading the manuscript; A. Vaughan for advice regarding infection of FRG-knockout FRGhuHep mice; A. J. Radtke for confocal imaging of oocysts; E. James for cryopreservation of PfSPZ; R. Shepherd and M. Thambi for iPfSPZ production; A. Belmonte for assistance with T cell studies; and the members of Sanaria’s gametocyte and mosquito production teams and experimental animal study teams for their support. This work was supported by funds from the NIAID, NIH under SBIR grants 1R43AI085740-01, 2R44AI085740-03A1 and 2R44AI085740-06A1, and the CDMRP Program, USA Medical Research Acquisition Activity under contract W81XWH-16-2-0025. RNA-seq studies were funded by seed funds from Seattle Children’s Research Institute to S.H.I.K. The views expressed in this article reflect the results of research conducted by the author and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, nor the US Government.
Author information
Authors and Affiliations
Contributions
S.L.H. led the entire project. A.G.E., S.C., P.F.B., B.K.L.S. and S.L.H. designed the studies. A.G.E., M.M., H.H., I.M. and A.A.Y. performed in vitro culture experiments and data collection. P.D.L.V., B.U.H. and A.R. performed in vitro infectivity assays using HC-04 cells and data collection. P.D.L.V., H.H., H.K. and A.G.E. performed in vitro infectivity assays using PHHs and data collection. S.C., N.K., T.L., C.T. and A.G.E. performed FRG mouse infection studies and data collection. T.L., A.P., Y.A., C.T. and B.K.L.S. produced stage V gametocytes. T.L., A.G.E., I.M., H.H., C.T., B.U.H. and S.C. performed P. falciparum life cycle without mosquitoes and data collection. G.Z., R.D.M. and S.H.I.K. performed transcriptome analysis and data collection. E.I. performed RT–qPCR. M.B. and M.S. performed T cell studies and data collection. S.C. and N.K. immunized mice. J.J.C. performed proteome assays. A.S.I.A., M.L. and T.W. performed immunoblot analysis. A.G.E., P.F.B., B.K.L.S. and S.L.H. wrote the manuscript. B.K.L.S. and S.L.H. supervised the project. All of the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
A.G.E., S.C., N.K, A.P., Y.A., A.A.Y., E.I., A.S.I.A., T.W., M.L., P.F.B., B.K.L.S. and S.L.H. are employees of Sanaria. B.K.L.S. and S.L.H. own stock in Sanaria. US patents on in vitro PfSPZ, 9878026B2 (2018) and 10441646B2 (2019) have been issued (inventors A.G.E. and S.L.H.). T.L., M.M., H.H., C.T., I.M., A.R. and P.D.V. were employees of Sanaria at the time the study was conducted.
Peer review
Peer review information
Nature thanks Laurent Renia and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Ookinetes and oocysts in vitro.
a, b. In vitro transformed Pf zygotes and S2 feeder cells were added to Matrigel matrix coated wells of 8-well chamber slides and 24 h (a) or 36 (b) later an anti-Pfs25 mAb was added to the wells and the parasites were assessed by immunofluorescence assay. a. At 24 h, Pfs25 staining of an ookinete and the trail it had shed moving across the well are shown. b. At 36 h, Pfs25 staining of an early oocyst and the trail it had shed indicating the likely path the ookinete took before transforming to an oocyst are shown. Scale bars, 10 µm. Ookinetes expressing Pfs25 in 8-well chamber slide Matrigel cultures were identified in 3 different experiments. c. At 3 days (upper panels) an anti-Pfs25 mAb or at 8 days (lower panels) an anti-PfCSP mAb was added to the cultures and oocysts were assessed by immunofluorescence using confocal microscopy. Punctate localization in 8-day oocysts indicates budding PfSPZ. DAPI signals not localized to oocysts indicate feeder cell nuclei. In vitro produced 3-day oocysts expressing Pf25 and 8-day oocysts expressing PfCSP were detected in numerous experiments in 8-well chamber slides with Matrigel. d. A. stephensi mosquitoes were fed a blood meal containing stage V Pf gametocytes. At 3 days (upper panels) and 8 days (lower panels) the midguts were dissected and stained with anti-Pfs25 mAb (upper panels) and anti-PfCSP mAb (lower panels) and assessed by immunofluorescence using confocal microscopy. e. Comparison of conversion rates of gametocytes to iPfSPZ in different culture conditions. Center lines show the medians; box limits indicate the 25th and 75th percentiles as determined by R software; whiskers extend 1.5 times the interquartile range from the 25th and 75th percentiles, outliers are represented by dots. There were 7, 23, 24, 11, and 37 different independent experiments respectively for 8-well chamber slides with Sf21 cells, 8-well chamber slides with Matrigel, 12-well plates with S2 cells and collagen I, 12-well plates with S2 cells and GMP collagen, and 12-well plates with S2 cells and no matrix. All data were compared using Kruskal-Wallis test of median values (<0.0001), and post-hoc pairwise comparisons were made using Dunn’s multiple comparisons test. Significant differences and their respective p values are shown on the figure. f-g. In vitro transformed Pf zygotes (the product of 2x103 stage V gametocytes) with (f) and without (g) S2 feeder cells were added to Matrigel matrix coated wells of 8-well chamber slides. On day 15 post initiation of the culture, oocysts were assessed by IFA using an anti-PfCSP mAb. The culture including S2 cells (f) had many oocysts and iPfSPZ, indicated by arrows. The culture without S2 cells (g) had significantly lower numbers of oocysts (Supplementary Table 2). The oocysts in the cultures without S2 cells (g) had many budding PfSPZ expressing PfCSP (dots), but no fully developed iPfSPZ, suggesting an arrest in development. This experiment was performed three times and photomicrographs taken once.
Extended Data Fig. 2 Pf life cycle without mosquitoes or humans.
iPfSPZ were injected into mice containing humanized livers (FRGhuHep mice). Six and 7 days later the mice were transfused with human erythrocytes, and 6 h after the day 7 transfusion, blood was removed from the mice and cultured in vitro using standard methods. a, Parasites developed in these cultures to form Pf rings, trophozoites and schizonts; b. After induction, Pf gametocytes were produced in the cultures.
Extended Data Fig. 3 Gene and protein expression of iPfSPZ.
a. Scatter plot showing correlation of transcriptomes; x axis mPfSPZ, y axis iPfSPZ, right upper corner in red print, expressed liver stage genes. b. Heatmap of genes expressed in PfSPZ detected by RNASeq. 3,656 genes with ≥5 TPM in at least one sample are plotted. The raw gene expression reads from both iPfSPZ and mPfSPZ samples are in Supplementary Table 4. c. Comparative transcriptomics of mPfSPZ and iPfSPZ. Volcano plot of differentially expressed genes are plotted based upon average fold change and unadjusted average p-value from the 5 DE tools (significance of differential expression, see methods). Extreme low p-values are cropped at 1e-08 for plotting. Notable, differentially expressed genes are highlighted in red. d. PfCSP expression by mPfSPZ and iPfSPZ by immunoblot analysis. Lanes 1-3 are 2.0x105, 5.0x104 and 1.25x104 mPfSPZ, and lanes 5-7 are 1.1x106, 2.0x105, 5.0x104 iPfSPZ respectively. No sample was loaded in lane 4. Molecular size markers are indicated on the left.
Extended Data Fig. 4 T-cell responses in fresh and cryopreserved splenocytes.
Data show spot forming cells (SFC) expressing interferon gamma (IFNγ)/106 splenocytes from 6 mice immunized with iPfSPZ and 6 mice immunized with mPfSPZ at 2 weeks after the fourth dose of iPfSPZ or mPfSPZ. Splenocytes from 2 mice were pooled so that there were 3 samples from each group of immunized mice (a, b, c, d, e, f and h). Each pooled splenocyte sample was assayed in duplicate and each data point represents the mean of the duplicates. Results are median and interquartile ranges of SFC/106 splenocytes after incubation with stimulating cells at different concentrations. P values were calculated using the Mann-Whitney U test. The same symbol was used for each pool sample at the 3 different concentrations of stimulating cells. T-cell responses against Plasmodium falciparum-infected red blood cells (PfRBC) in fresh (a) and cryopreserved (b) splenocytes from immunized mice. One pool (●) had no detectable signal at 1x105 PfRBC but had a signal at both 2x105 PfRBC and 5x104 PfRBC. The negative response at 5x104 was considered a technical error and removed from the analysis. c, d. T-cell responses against mPfSPZ in fresh (c) and cryopreserved (d) splenocytes from immunized mice. Splenocytes were stimulated with 2.5x104 mPfSPZ. The responses to incubation with vaccine diluent alone are also shown. e, f. T-cell responses against uninfected red blood cell (uRBC) in fresh (E) and cryopreserved (F) splenocytes from immunized mice. The same symbol was used for each pool at the 3 different concentrations of uRBC (2x105, 1x105, and 5x104). g. T-cell responses against mPfSPZ in fresh splenocytes from naïve mice. Splenocytes of 2 naïve mice were pooled and frozen before assaying in duplicates and splenocytes of 2 naïve mice were pooled. and assayed fresh in duplicate. Splenocytes from naïve mice were stimulated with 2.5x104 mPfSPZ. h. T-cell responses against PfRBC in fresh and cryopreserved splenocytes from naïve mice. Fresh and cryopreserved splenocytes from naïve mice were stimulated with 2x105, 1x105, and 5x104 PfRBCs.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Supplementary Table 4
Raw gene expression reads from both iPfSPZ and mPfSPZ samples.
Supplementary Table 5
Relative abundance of the 50 most highly expressed mPfSPZ gene transcripts in iPfSPZ as determined by RNA-seq.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eappen, A.G., Li, T., Marquette, M. et al. In vitro production of infectious Plasmodium falciparum sporozoites. Nature 612, 534–539 (2022). https://doi.org/10.1038/s41586-022-05466-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05466-7
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.