Abstract
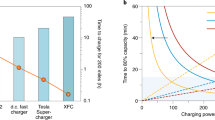
Lithium-ion batteries with nickel-rich layered oxide cathodes and graphite anodes have reached specific energies of 250–300 Wh kg−1 (refs. 1,2), and it is now possible to build a 90 kWh electric vehicle (EV) pack with a 300-mile cruise range. Unfortunately, using such massive batteries to alleviate range anxiety is ineffective for mainstream EV adoption owing to the limited raw resource supply and prohibitively high cost. Ten-minute fast charging enables downsizing of EV batteries for both affordability and sustainability, without causing range anxiety. However, fast charging of energy-dense batteries (more than 250 Wh kg−1 or higher than 4 mAh cm−2) remains a great challenge3,4. Here we combine a material-agnostic approach based on asymmetric temperature modulation with a thermally stable dual-salt electrolyte to achieve charging of a 265 Wh kg−1 battery to 75% (or 70%) state of charge in 12 (or 11) minutes for more than 900 (or 2,000) cycles. This is equivalent to a half million mile range in which every charge is a fast charge. Further, we build a digital twin of such a battery pack to assess its cooling and safety and demonstrate that thermally modulated 4C charging only requires air convection. This offers a compact and intrinsically safe route to cell-to-pack development. The rapid thermal modulation method to yield highly active electrochemical interfaces only during fast charging has important potential to realize both stability and fast charging of next-generation materials, including anodes like silicon and lithium metal.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article, the Extended Data and the Supplementary Information.
References
Wu, Y. et al. An empirical model for the design of batteries. ACS Energy Lett. 5, 807–816 (2020).
Deng, J., Bae, C., Denlinger, A. & Miller, T. Electric vehicles batteries: requirements and challenges. Joule 4, 511–515 (2020).
Cheeseman, H. Fast-charging Li-metal batteries. ARPA-E https://arpa-e.energy.gov/open-2021/webinars (2021).
Howell, D. et al. Enabling fast charging: a technology gap assessment. US Department of Energy https://www.energy.gov/sites/default/files/2017/10/f38/XFC%20Technology%20Gap%20Assessment%20Report_FINAL_10202017.pdf (2017).
Zaghib, K. et al. Safe and fast-charging Li-ion battery with long shelf life for power applications. J. Power Sources 196, 3949–3954 (2011).
Colclasure, A. M. et al. Electrode scale and electrolyte transport effects on extreme fast charging of lithium-ion cells. Electrochim. Acta 337, 135854 (2020).
Logan, E. R. et al. Ester-based electrolytes for fast charging of energy dense lithium-ion batteries. J. Phys. Chem. C 124, 12269–12280 (2020).
Liu, T., Yang, X., Ge, S., Leng, Y. & Wang, C. Y. Ultrafast charging of energy-dense lithium-ion batteries for urban air mobility. eTransportation 7, 100103 (2021).
Du, Z., Wood Iii, D. L. & Belharouak, I. Enabling fast charging of high energy density Li-ion cells with high lithium ion transport electrolytes. Electrochem. Comm. 103, 109–113 (2019).
Chen, K. et al. Efficient fast-charging of lithium-ion batteries enabled by laser-patterned three-dimensional graphite anode architectures. J. Power Sources 471, 228475 (2020).
Yang, X.-G. et al. Asymmetric temperature modulation for extreme fast charging of lithium-ion batteries. Joule 3, 3002–3019 (2019).
Han, J.-G. et al. An electrolyte additive capable of scavenging HF and PF5 enables fast charging of lithium-ion batteries in LiPF6-based electrolytes. J. Power Sources 446, 227366 (2020).
Gonzalez, A. F., Yang, N.-H. & Liu, R.-S. Silicon anode design for lithium-ion batteries: progress and perspectives. J. Phys. Chem. C 121, 27775–27787 (2017).
Lee, S. K., McDowell, M. T., Choi, J. W. & Cui, Y. Anomalous shape changes of silicon nanopillars by electrochemical lithiation. Nano Lett. 11, 3034–3039 (2011).
Son, I. H. et al. Graphene balls for lithium rechargeable batteries with fast charging and high volumetric energy densities. Nat. Commun. 8, 1561 (2017).
Wang, B. et al. Ultrafast-charging silicon-based coral-like network anodes for lithium-ion batteries with high energy and power densities. ACS Nano 13, 2307–2315 (2019).
Kim, N., Chae, S., Ma, J., Ko, M. & Cho, J. Fast-charging high-energy lithium-ion batteries via implantation of amorphous silicon nanolayer in edge-plane activated graphite anodes. Nat. Commun. 8, 812 (2017).
McBrayer, J. D. et al. Calendar aging of silicon-containing batteries. Nat. Energy 6, 866–872 (2021).
Masias, A., Marcicki, J. & Paxton, W. A. Opportunities and challenges of batteries in automotive applications. ACS Energy Lett. 6, 621–630 (2021).
Zheng, J. et al. Electrolyte additive enabled fast charging and stable cycling lithium metal batteries. Nat. Energy 2, 17012 (2017).
Peng, Z. et al. High-power lithium metal batteries enabled by high-concentration acetonitrile-based electrolytes with vinylene carbonate additive. Adv. Funct. Mater. 30, 2001285 (2020).
Lee, Y.-G. et al. High-energy long-cycling all-solid-state lithium metal batteries enabled by silver–carbon composite anodes. Nat. Energy 5, 299–308 (2020).
Niu, C. et al. High-energy lithium metal pouch cells with limited anode swelling and long stable cycles. Nat. Energy 4, 551–559 (2019).
Ren, X. et al. Enabling high-voltage lithium-metal batteries under practical conditions. Joule 3, 1662–1676 (2019).
Leng, Y. et al. Fast charging of energy-dense lithium metal batteries in localized ether-based highly concentrated electrolytes. J. Electrochem. Soc. 168, 060548 (2021).
Liu, T., Ge, S., Yang, X.-G. & Wang, C.-Y. Effect of thermal environments on fast charging Li-ion batteries. J. Power Sources 511, 230466 (2021).
Keil, J. et al. Linear and nonlinear aging of lithium-ion cells investigated by electrochemical analysis and in-situ neutron diffraction linear and nonlinear aging of lithium-ion cells investigated by electrochemical analysis and in-situ neutron diffraction. J. Electrochem. Soc. 166, A3908 (2019).
Schuster, S. F. et al. Nonlinear aging characteristics of lithium-ion cells under different operational conditions. J. Energy Storage 1, 44–53 (2015).
Yang, X.-G., Leng, Y., Zhang, G., Ge, S. & Wang, C.-Y. Modeling of lithium plating induced aging of lithium-ion batteries: transition from linear to nonlinear aging. J. Power Sources 360, 28–40 (2017).
Ma, X. et al. Hindering Rollover Failure of Li[Ni0.5Mn0.3Co0.2]O2/Graphite Pouch Cells during Long-Term Cycling. J. Electrochem. Soc. 166, A711 (2019).
Yang, X.-G., Ge, S., Liu, T., Leng, Y. & Wang, C.-Y. A look into the voltage plateau signal for detection and quantification of lithium plating in lithium-ion cells. J. Power Sources 395, 251–261 (2018).
Ogihara, N. et al. Theoretical and experimental analysis of porous electrodes for lithium-ion batteries by electrochemical impedance spectroscopy using a symmetric cell. J. Electrochem. Soc. 159, A1034–A1039 (2012).
Newman, J. S. & Tobias, C. W. Theoretical analysis of current distribution in porous electrodes. J. Electrochem. Soc. 109, 1183 (1962).
Li, L. et al. Transport and electrochemical properties and spectral features of non-aqueous electrolytes containing LiFSI in linear carbonate solvents. J. Electrochem. Soc. 158, A74 (2011).
Han, H.-B. et al. Lithium bis(fluorosulfonyl)imide (LiFSI) as conducting salt for nonaqueous liquid electrolytes for lithium-ion batteries: physicochemical and electrochemical properties. J. Power Sources 196, 3623–3632 (2011).
Landesfeind, J., Hattendorff, J., Ehrl, A., Wall, W. A. & Gasteiger, H. A. Tortuosity determination of battery electrodes and separators by impedance spectroscopy. J. Electrochem. Soc. 163, A1373–A1387 (2016).
Bi, Y. et al. Reversible planar gliding and microcracking in a single-crystalline Ni-rich cathode. Science 370, 1313–1317 (2020).
Li, Y. et al. Growth of conformal graphene cages on micrometre-sized silicon particles as stable battery anodes. Nat. Energy 1, 15029 (2016).
Yang, X.-G., Zhang, G. & Wang, C. Y. Computational design and refinement of self-heating lithium ion batteries. J. Power Sources 328, 203–211 (2016).
Zhang, J., Wu, B., Li, Z. & Huang, J. Simultaneous estimation of thermal parameters for large-format laminated lithium-ion batteries. J. Power Sources 259, 106–116 (2014).
Ye, Y., Saw, L. H., Shi, Y., Somasundaram, K. & Tay, A. A. O. Effect of thermal contact resistances on fast charging of large format lithium ion batteries. Electrochim. Acta 134, 327–337 (2014).
Aiken, C. P. et al. Li[Ni0.5Mn0.3Co0.2]O2 as a superior alternative to LiFePO4 for long-lived low voltage li-ion cells. J. Electrochem. Soc. 169, 050512 (2022).
Longchamps, R. S., Yang, X. G. & Wang, C. Y. Fundamental insights into battery thermal management and safety. ACS Energy Lett. 7, 1103–1111 (2022).
Lima, P. Samsung SDI 94 Ah battery cell full specifications. PushEVs https://pushevs.com/2018/04/05/samsung-sdi-94-ah-battery-cell-full-specifications/ (2021).
Wang, C. Y. et al. Lithium-ion battery structure that self-heats at low temperatures. Nature 529, 515–518 (2016).
Yang, X. G., Liu, T. & Wang, C. Y. Innovative heating of large-size automotive Li-ion cells. J. Power Sources 342, 598–604 (2017).
Zhang, G. et al. Rapid self-heating and internal temperature sensing of lithium-ion batteries at low temperatures. Electrochim. Acta 218, 149–155 (2016).
Leng, Y. et al. Electrochemical cycle-life characterization of high energy lithium-ion cells with thick Li(Ni0.6Mn0.2Co 0.2)O2 and graphite electrodes. J. Electrochem. Soc. 164, A1037–A1049 (2017).
Acknowledgements
Partial support from the US Department of Energy’s Office of Energy Efficiency and Renewable Energy (EERE) under award no. DE-EE0008355, the William E. Diefenderfer Endowment and Air Force STTR under contract FA864921P1620 is gratefully acknowledged. We are also grateful to Gamma Technologies for offering GT-AutoLion software.
Author information
Authors and Affiliations
Contributions
C.Y.W., T.L., B.D.M. and E.S.R. wrote the manuscript. S.G. designed and built the cells. T.L. and X.G.Y. designed the experiments. T.L. built the test stand and carried out the experiments. Y.L. optimized electrolytes in coin cells. C.Y.W., N.V.S. and B.D.M. designed and performed 3D numerical simulations. All authors contributed to development of the manuscript and to discussions as the project developed.
Corresponding authors
Ethics declarations
Competing interests
B.D.M., E.S.R., N.V.S. and C.Y.W. have a financial interest in EC Power.
Peer review
Peer review information
Nature thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 LiB design for ATM cycling.
a, LiB with an embedded nickel foil for internal heating. Before each charging, current goes through the internal heating structure and heats up the cell to 65 °C in less than one minute. After reaching the target temperature, the charging channel starts to take in energy and maintains thermal balance throughout the charging process. b, Cell and heating-foil temperatures versus heating time. A heating rate of 0.75 °C s−1 was achieved when applying 3.3 V on the heating channel.
Extended Data Fig. 2 Rate performance tests and model validation.
a, Cell with baseline electrolyte. b, Cell with dual-salt electrolyte; the simulated transference number was 0.48.
Extended Data Fig. 3 Ageing mechanisms under ATM cycling.
a, b, Lithium plating detection with voltage relaxation method for 4.2 mAh cm−2 LiBs and 3.4 mAh cm−2 LiBs. c, d, Coulombic efficiency during cycling. e, f, Change of resistance attained by EIS tests. Baseline electrolyte was used in these tests.
Extended Data Fig. 4 Numerical prediction of lithium plating at various charge rates with T=60°C.
a, Experimental and simulated voltage profiles for baseline cells. b, Simulated lithium deposition potential for baseline cells. c, Experimental and simulated voltage profiles for cells with dual-salt electrolyte. d, Simulated lithium deposition potential for cells with dual-salt electrolyte.
Extended Data Fig. 5 Ageing mechanisms under fast charging of batteries with enhanced ion transport.
a, Lithium plating detection with voltage relaxation method for 3.4 mAh cm−2 LiBs with enhanced ion transport. b, Coulombic efficiency during cycling. c, Change of resistance attained by EIS tests.
Extended Data Fig. 6 Fast charging cycling with different combinations of electrodes and electrolytes.
Use of either the mixed dual-salt electrolyte or higher porosity anodes failed to give long cycle lifetime; both were required for long cycle lifetime.
Extended Data Fig. 7 Parameter map of lithium plating-free (LPF) charging (3.4 mAh cm−2).
a, Simulated LPF region given by relative ion transport resistance and charge transfer resistance when 3C charging the battery (*denotes unitless variables, referenced with the properties of a fresh baseline cell charging at 60 °C). b, Simulated LPF region given by relative ion transport resistance and charge transfer resistance when 4C charging the battery, in which the open symbol labelled as 65 °C denotes the high porosity anode. c, Change of charge transfer resistance during aging attained from EIS tests and analysis. d, Change of ion transport resistance during ageing attained from EIS tests and analysis.
Extended Data Fig. 8 Effects of heat transfer coefficient.
Effects during C/3 discharge on 4C charge - C/3 discharge cycling under otherwise the cooling condition of 140 W per m2K, indicative of dramatic shortening in time for the battery to cool down to below 40 °C. Note that a heat transfer coefficient of 300 W per m2K is still attainable by strong aspirated air convection. a, Cell temperature curves. b, Cell voltage curves. c, Spatial temperature nonuniformity, Tmax – Tmin, as a function of time.
Extended Data Fig. 9 EIS tests and equivalent circuit for fitting.
a, Typical shape and interpretation for cell impedance in the complex plane. b, Equivalent circuit and the mathematical expression to fit the experimental results. c, EIS results and fitting curve for baseline cell under ATM cycling with 3C charging to 80% SOC. d, EIS results and fitting curve for cell with enhanced ion-transport under ATM cycling with 4C charging to 70% SOC. e, EIS results and fitting curve for cell with enhanced ion-transport under ATM cycling with 4C charging to 75% SOC.
Supplementary information
Supplementary Information
Cell design details, Tables 1–4, Figures 1–5, the electrochemical–thermal coupled model and equivalent circuit for EIS.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, CY., Liu, T., Yang, XG. et al. Fast charging of energy-dense lithium-ion batteries. Nature 611, 485–490 (2022). https://doi.org/10.1038/s41586-022-05281-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05281-0
This article is cited by
-
Advances in 3D silicon-based lithium-ion microbatteries
Communications Materials (2024)
-
Safe electrolyte for long-cycling alkali-ion batteries
Nature Sustainability (2024)
-
Sustainable plug-in electric vehicle integration into power systems
Nature Reviews Electrical Engineering (2024)
-
Origin of fast charging in hard carbon anodes
Nature Energy (2024)
-
Advances in All-Solid-State Lithium–Sulfur Batteries for Commercialization
Nano-Micro Letters (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.