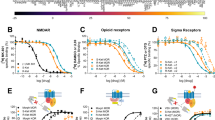
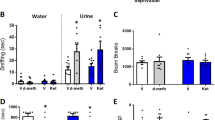
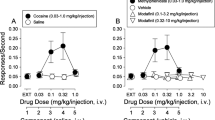
Abstract
Ketamine is used clinically as an anaesthetic and a fast-acting antidepressant, and recreationally for its dissociative properties, raising concerns of addiction as a possible side effect. Addictive drugs such as cocaine increase the levels of dopamine in the nucleus accumbens. This facilitates synaptic plasticity in the mesolimbic system, which causes behavioural adaptations and eventually drives the transition to compulsion1,2,3,4. The addiction liability of ketamine is a matter of much debate, in part because of its complex pharmacology that among several targets includes N-methyl-d-aspartic acid (NMDA) receptor (NMDAR) antagonism5,6. Here we show that ketamine does not induce the synaptic plasticity that is typically observed with addictive drugs in mice, despite eliciting robust dopamine transients in the nucleus accumbens. Ketamine nevertheless supported reinforcement through the disinhibition of dopamine neurons in the ventral tegmental area (VTA). This effect was mediated by NMDAR antagonism in GABA (γ-aminobutyric acid) neurons of the VTA, but was quickly terminated by type-2 dopamine receptors on dopamine neurons. The rapid off-kinetics of the dopamine transients along with the NMDAR antagonism precluded the induction of synaptic plasticity in the VTA and the nucleus accumbens, and did not elicit locomotor sensitization or uncontrolled self-administration. In summary, the dual action of ketamine leads to a unique constellation of dopamine-driven positive reinforcement, but low addiction liability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available in the Zenodo repository (https://doi.org/10.5281/zenodo.5772449)51.
Code availability
The MATLAB code used for analysing the fibre photometry raw data is available in the Zenodo repository (https://doi.org/10.5281/zenodo.5772449).
References
Bellone, C., Loureiro, M. & Luscher, C. Drug-evoked synaptic plasticity of excitatory transmission in the ventral tegmental area. Cold Spring Harb. Perspect. Med. 11, a039701 (2021).
Di Chiara, G. et al. Dopamine and drug addiction: the nucleus accumbens shell connection. Neuropharmacology 47, 227–241 (2004).
Lüscher, C., Robbins, T. W. & Everitt, B. J. The transition to compulsion in addiction. Nat. Rev. Neurosci. 21, 247–263 (2020).
Lüscher, C. & Ungless, M. A. The mechanistic classification of addictive drugs. PLoS Med. 3, e437 (2006).
Franks, N. P. & Lieb, W. R. Molecular and cellular mechanisms of general anaesthesia. Nature 367, 607–614 (1994).
Zanos, P. et al. Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacol. Rev. 70, 621–660 (2018).
Masuzawa, M. et al. Pentobarbital inhibits ketamine-induced dopamine release in the rat nucleus accumbens: a microdialysis study. Anesth. Analg. 96, 148–152 (2003).
Witkin, J. M. et al. The rapidly acting antidepressant ketamine and the mGlu2/3 receptor antagonist LY341495 rapidly engage dopaminergic mood circuits. J. Pharmacol. Exp. Ther. 358, 71–82 (2016).
Littlewood, C. L. et al. Mapping the central effects of ketamine in the rat using pharmacological MRI. Psychopharmacology 186, 64–81 (2006).
Rocha, B. A., Ward, A. S., Egilmez, Y., Lytle, D. A. & Emmett-Oglesby, M. W. Tolerance to the discriminative stimulus and reinforcing effects of ketamine. Behav. Pharmacol. 7, 160–168 (1996).
De Luca, M. T. & Badiani, A. Ketamine self-administration in the rat: evidence for a critical role of setting. Psychopharmacology 214, 549–556 (2011).
Zanos, P. et al. A negative allosteric modulator for alpha5 subunit-containing GABA receptors exerts a rapid and persistent antidepressant-like action without the side effects of the NMDA receptor antagonist ketamine in mice. eNeuro 4, ENEURO.0285-16.2017 (2017).
Suzuki, T. et al. Effects of the non-competitive NMDA receptor antagonist ketamine on morphine-induced place preference in mice. Life Sci. 67, 383–389 (2000).
Luscher, C. & Malenka, R. C. Drug-evoked synaptic plasticity in addiction: from molecular changes to circuit remodeling. Neuron 69, 650–663 (2011).
Luscher, C. The emergence of a circuit model for addiction. Annu. Rev. Neurosci. 39, 257–276 (2016).
Ungless, M. A., Whistler, J. L., Malenka, R. C. & Bonci, A. Single cocaine exposure in vivo induces long-term potentiation in dopamine neurons. Nature 411, 583–587 (2001).
Bellone, C. & Luscher, C. Cocaine triggered AMPA receptor redistribution is reversed in vivo by mGluR-dependent long-term depression. Nat. Neurosci. 9, 636–641 (2006).
Brown, M. T. et al. Drug-driven AMPA receptor redistribution mimicked by selective dopamine neuron stimulation. PLoS One 5, e15870 (2010).
Conrad, K. L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121 (2008).
Kourrich, S., Rothwell, P. E., Klug, J. R. & Thomas, M. J. Cocaine experience controls bidirectional synaptic plasticity in the nucleus accumbens. J. Neurosci. 27, 7921–7928 (2007).
Mameli, M. et al. Cocaine-evoked synaptic plasticity: persistence in the VTA triggers adaptations in the NAc. Nat. Neurosci. 12, 1036–1041 (2009).
Pascoli, V. et al. Stochastic synaptic plasticity underlying compulsion in a model of addiction. Nature 564, 366–371 (2018).
Brodie, M. S. & Dunwiddie, T. V. Cocaine effects in the ventral tegmental area: evidence for an indirect dopaminergic mechanism of action. Naunyn Schmiedebergs Arch. Pharmacol. 342, 660–665 (1990).
Hunker, A. C. et al. Conditional single vector CRISPR/SaCas9 viruses for efficient mutagenesis in the adult mouse nervous system. Cell Rep. 30, 4303–4316 (2020).
Corre, J. et al. Dopamine neurons projecting to medial shell of the nucleus accumbens drive heroin reinforcement. eLife 7, e39945 (2018).
Luscher, C. & Malenka, R. C. NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb. Perspect. Biol. 4, a005710 (2012).
Zanos, P. et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 533, 481–486 (2016).
Ganguly, S., Panetta, J. C., Roberts, J. K. & Schuetz, E. G. Ketamine pharmacokinetics and pharmacodynamics are altered by P-glycoprotein and breast cancer resistance protein efflux transporters in mice. Drug Metab. Dispos. 46, 1014–1022 (2018).
Saland, S. K. & Kabbaj, M. Sex differences in the pharmacokinetics of low-dose ketamine in plasma and brain of male and female rats. J. Pharmacol. Exp. Ther. 367, 393–404 (2018).
Valjent, E. et al. Involvement of the extracellular signal-regulated kinase cascade for cocaine-rewarding properties. J. Neurosci. 20, 8701–8709 (2000).
Valjent, E. et al. Regulation of a protein phosphatase cascade allows convergent dopamine and glutamate signals to activate ERK in the striatum. Proc. Natl Acad. Sci. USA 102, 491–496 (2005).
Bertran-Gonzalez, J. et al. Opposing patterns of signaling activation in dopamine D1 and D2 receptor-expressing striatal neurons in response to cocaine and haloperidol. J. Neurosci. 28, 5671–5685 (2008).
Pascoli, V. et al. Contrasting forms of cocaine-evoked plasticity control components of relapse. Nature 509, 459–464 (2014).
Pascoli, V., Turiault, M. & Luscher, C. Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature 481, 71–75 (2012).
Deroche-Gamonet, V., Belin, D. & Piazza, P. V. Evidence for addiction-like behavior in the rat. Science 305, 1014–1017 (2004).
Tan, K. R. et al. Neural bases for addictive properties of benzodiazepines. Nature 463, 769–774 (2010).
Cruz, H. G. et al. Bi-directional effects of GABAB receptor agonists on the mesolimbic dopamine system. Nat. Neurosci. 7, 153–159 (2004).
Melis, M. et al. Endocannabinoids mediate presynaptic inhibition of glutamatergic transmission in rat ventral tegmental area dopamine neurons through activation of CB1 receptors. J. Neurosci. 24, 53–62 (2004).
Paoletti, P., Bellone, C. & Zhou, Q. NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease. Nat. Rev. Neurosci. 14, 383–400 (2013).
Moghaddam, B., Adams, B., Verma, A. & Daly, D. Activation of glutamatergic neurotransmission by ketamine: a novel step in the pathway from NMDA receptor blockade to dopaminergic and cognitive disruptions associated with the prefrontal cortex. J. Neurosci. 17, 2921–2927 (1997).
Ali, F. et al. Ketamine disinhibits dendrites and enhances calcium signals in prefrontal dendritic spines. Nat. Commun. 11, 72 (2020).
Kapur, S. & Seeman, P. NMDA receptor antagonists ketamine and PCP have direct effects on the dopamine D2 and serotonin 5-HT2 receptors—implications for models of schizophrenia. Mol. Psychiatry 7, 837–844 (2002).
Luscher, C. & Slesinger, P. A. Emerging roles for G protein-gated inwardly rectifying potassium (GIRK) channels in health and disease. Nat. Rev. Neurosci. 11, 301–315 (2010).
Can, A. et al. Effects of ketamine and ketamine metabolites on evoked striatal dopamine release, dopamine receptors, and monoamine transporters. J. Pharmacol. Exp. Ther. 359, 159–170 (2016).
Engblom, D. et al. Glutamate receptors on dopamine neurons control the persistence of cocaine seeking. Neuron 59, 497–508 (2008).
Uchihashi, Y., Kuribara, H., Morita, T. & Fujita, T. The repeated administration of ketamine induces an enhancement of its stimulant action in mice. Jpn. J. Pharmacol. 61, 149–151 (1993).
Wiley, J. L., Evans, R. L., Grainger, D. B. & Nicholson, K. L. Age-dependent differences in sensitivity and sensitization to cannabinoids and 'club drugs' in male adolescent and adult rats. Addict. Biol. 13, 277–286 (2008).
Berman, R. M. et al. Antidepressant effects of ketamine in depressed patients. Biol. Psychiatry 47, 351–354 (2000).
Bariselli, S. et al. Role of VTA dopamine neurons and neuroligin 3 in sociability traits related to nonfamiliar conspecific interaction. Nat. Commun. 9, 3173 (2018).
Zweifel, L. S., Argilli, E., Bonci, A. & Palmiter, R. D. Role of NMDA receptors in dopamine neurons for plasticity and addictive behaviors. Neuron 59, 486–496 (2008).
Simmler, L. D. et al. Dual-action of ketamine confines addiction liability. Zenodo https://doi.org/10.5281/zenodo.5772449 (2022).
Acknowledgements
We thank J. Cand for laboratory assistance and L. Zweifel for providing the conditional NR1-KO and control viruses. We thank A. Kwan, C. Bellone and M. Mameli for their comments on an earlier version of the manuscript. This study was supported by the Swiss National Science Foundation (L.D.S., grant number PZ00P3_174178; C.L., 310030_189188) and the European Research Council (AdG F-Addict).
Author information
Authors and Affiliations
Contributions
L.D.S. conceived the experiments and performed patch recordings, fibre photometry and behavioural experiments. Y.L. performed fibre photometry, patch recordings and behavioural experiments. L.C.H. performed immunohistochemistry, fibre photometry and behavioural experiments. A.H. performed mice surgeries and behavioural experiments. R.v.Z. performed fibre photometry experiments. L.D.S., Y.L., L.C.H. and R.v.Z. performed analyses. C.L., L.D.S. and Y.L. wrote the manuscript with the help of all authors. C.L. supervised the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Jean-Antoine Girault and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Lever presses and infusion intervals of self-administration.
a, Lever presses on active and inactive lever during self-administration for each session. n (mice) = 8 (saline), 10 (ketamine), 8 (cocaine). b, Median inter-infusion interval per mouse in session 6 to estimate rate of infusion during non-contingent self-administration (Fig. 3k). n (mice) = 10 (ketamine), 7 (cocaine). Data are represented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001.
Extended Data Fig. 2 Dose-dependence of accumbal dopamine transients.
a, Schematic of virus injection and fibre implantation targeting the medial shell of the nucleus accumbens. b, Mean dLight response to i.p. injections of different doses of ketamine. n = 5 mice. c, AUC from 0–10 min after i.p. injection of different doses of ketamine. n = 5 mice. Data are represented as mean ± SEM.
Extended Data Fig. 3 Validation of NR1 ablation.
a, Schematic of experimental details with virus injections and location of patch-clamp recordings. b, Representative traces of NMDA/AMPA recordings of NR1-KO (KO; sgGrin1) and control (CT; sgRosa26). Scale bar, 10 ms, 200 pA. c, NMDA/AMPA ratios (amplitudes measured for NMDA component 20 ms after the peak at +40 mV and AMPA component at −70 mV) of VTA GABA neurons from KO and CT mice. n (cells) = 6 (KO) and 6 (CT). Data are represented as mean ± SEM. *P < 0.05.
Extended Data Fig. 4 Fentanyl- and ketamine-induced GABA inhibition and dopamine transients.
a,c,e, Schematic of virus injection and fibre implantation. b, Mean Ca2+ signal of VTA GABA neurons from NR1-control (CT) and KO mice with i.p. injections of saline or fentanyl. n (mice) = 3 (CT) and 3 (KO). d, Average dopamine transients induced by ketamine (30 mg/kg) and fentanyl (0.3 mg/kg). n = 8 mice. f, Average dopamine transients induced by ketamine (30 mg/kg) and cocaine (15 mg/kg) in NR1-CT mice. n = 11 mice. g, Average dopamine transients induced by ketamine and cocaine in NR1-KO mice. n = 11 mice. Data are represented as mean ± SEM.
Extended Data Fig. 5 In vitro NMDAR inhibition in acute brain slices of the NAc.
a, Example traces of NMDAR EPSCs induced by electrical stimulation in the NAc, recorded in Mg2+-free aCSF. Top: before bath-application of ketamine; bottom: with 50 µM ketamine. Stimulation artefact was removed. Scale bar is 50 ms, 50 pA. b, NMDAR EPSCs before and with ketamine. n = 5 cells. Data are presented as mean ± SEM. **P < 0.01.
Extended Data Fig. 6 Compulsive ketamine self-administration and pain perception affected by ketamine infusions.
a. Number of ketamine infusions in baseline and punishment sessions. n = 17 mice. b. Latency to hotplate before and after mocked saline and ketamine i.v. infusions (1 mg/kg/infusion, 30 infusions). n (mice) = 16 (saline) and 17 (ketamine). Data are presented as mean ± SEM. ***P < 0.001.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Simmler, L.D., Li, Y., Hadjas, L.C. et al. Dual action of ketamine confines addiction liability. Nature 608, 368–373 (2022). https://doi.org/10.1038/s41586-022-04993-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-04993-7
This article is cited by
-
Ketamine in neuropsychiatric disorders: an update
Neuropsychopharmacology (2024)
-
Sex dependence of opioid-mediated responses to subanesthetic ketamine in rats
Nature Communications (2024)
-
Dynamic 3D imaging of cerebral blood flow in awake mice using self-supervised-learning-enhanced optical coherence Doppler tomography
Communications Biology (2023)
-
Mechanisms and molecular targets surrounding the potential therapeutic effects of psychedelics
Molecular Psychiatry (2023)
-
Decoupling SERT-nNOS Interaction to Generate Fast-Onset Antidepressants
Neuroscience Bulletin (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.