Abstract
The global emergence of many severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants jeopardizes the protective antiviral immunity induced after infection or vaccination. To address the public health threat caused by the increasing SARS-CoV-2 genomic diversity, the National Institute of Allergy and Infectious Diseases within the National Institutes of Health established the SARS-CoV-2 Assessment of Viral Evolution (SAVE) programme. This effort was designed to provide a real-time risk assessment of SARS-CoV-2 variants that could potentially affect the transmission, virulence, and resistance to infection- and vaccine-induced immunity. The SAVE programme is a critical data-generating component of the US Government SARS-CoV-2 Interagency Group to assess implications of SARS-CoV-2 variants on diagnostics, vaccines and therapeutics, and for communicating public health risk. Here we describe the coordinated approach used to identify and curate data about emerging variants, their impact on immunity and effects on vaccine protection using animal models. We report the development of reagents, methodologies, models and notable findings facilitated by this collaborative approach and identify future challenges. This programme is a template for the response to rapidly evolving pathogens with pandemic potential by monitoring viral evolution in the human population to identify variants that could reduce the effectiveness of countermeasures.
Similar content being viewed by others
Main
SARS-CoV-2, the aetiological agent of coronavirus disease 2019 (COVID-19), has caused a devastating pandemic resulting in more than 6 million deaths worldwide (https://covid19.who.int). With continuous transmission cycles occurring around the world, SARS-CoV-2 variants have arisen with mutations throughout its genome, including in the spike protein gene, the principal antigenic target of all SARS-CoV-2 vaccines currently in use1,2. The rapid emergence of variants—the latest being Omicron in November 2021—has raised concerns about how new mutations affect virus replication, infectivity, transmission and infection, and vaccine-induced immunity. This rapid genetic evolution of SARS-CoV-2 created an immediate need to monitor and characterize variants for potential resistance to medical countermeasures.
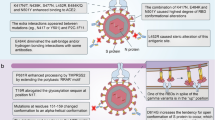
The US Department of Health and Human Services established the SARS-CoV-2 Interagency Group (SIG) to maximize coordination between the Centers for Disease Control and Prevention, the National Institutes of Health (NIH), the Food and Drug Administration, the Biomedical Advanced Research and Development Authority and Department of Defense for the US public health response to the COVID-19 pandemic3. The National Institute of Allergy and Infectious Diseases (NIAID) formed the SAVE consortium in January 2021 as a critical data-generating component for the SIG and to facilitate rapid data sharing with global partners and the scientific community (Fig. 1). The SAVE programme provides a comprehensive real-time risk assessment of emerging mutations in SARS-CoV-2 strains that could affect transmissibility, virulence, and infection- or vaccine-induced immunity. SAVE was constructed as a rational, structured and iterative risk-assessment pipeline with a goal of providing critical data to support SIG actions and ensure the effectiveness of countermeasures against emerging variants.
The SAVE programme is divided into three working groups to provide real-time risk assessments of SARS-CoV-2 variants on infection and vaccine-induced immunity. The early-detection and analysis group curates and prioritizes emerging SARS-CoV-2 variants. The in vitro group evaluates the effect of SARS-CoV-2 variants on humoral and cell-mediated immune responses. The in vivo group uses animal models to test vaccine efficacy, transmission, and define immune mechanisms and correlates of protection. These data are fed into the SIG, which coordinates between different US government agencies to assess the impact of variants on critical SARS-CoV-2 countermeasures, including vaccines, therapeutics and diagnostics. This iterative approach allows for information flow between the SAVE programme and the SIG to continue prioritizing and testing SARS-CoV-2 variants.
The SAVE programme is composed of an international team of scientists with expertise in virology, immunology, vaccinology, structural biology, bioinformatics, viral genetics and evolution. Each team member is responsible for key contributions ranging from curation of viral mutations, bioinformatics analysis, development of new reagents, assay development and testing, in vitro characterization, and in vivo model development and countermeasure testing. The SAVE programme is divided into three working groups: (1) the early-detection and analysis group; (2) the in vitro group; and (3) the in vivo group. The early-detection group uses public databases and analysis tools to curate and prioritize emerging SARS-CoV-2 variants. The in vitro group evaluates the impact of SARS-CoV-2 variants on humoral and cell-mediated immune responses using in vitro assays. The in vivo group uses small and large animal models to test vaccine efficacy, transmission, and define immune mechanisms and correlates of protection. A common theme across these subgroups is the integration of orthogonal experimental and computational approaches to validate findings and strengthen the evidence for recommendations. Collaborative efforts between the early-detection geneticists and evolutionary biologists, and the in vitro group virologists/immunologists enable the rapid determination of relationships between viral evolution and neutralization sensitivity. In turn, these results enable the in vivo team to assess and evaluate vaccine protection in animal studies. The SAVE programme has regularly scheduled (usually weekly) meetings that include individual subgroup meetings and an all-hands meeting, which serves as an opportunity to share key information across groups and align priorities for the most urgent experimental questions. NIAID programme staff and intramural and extramural scientists share leadership responsibilities. Collaboration within and across these groups has accelerated research and discovery due to the immediate and open sharing of ideas, reagents, protocols and data4,5,6,7,8,9,10,11,12. The SAVE group routinely invites scientists from international sites to present a real-time assessment of SARS-CoV-2 variants and infections within their region. The SAVE group coordinates with the Biodefense and Emerging Infections (BEI) Research Resources Repository, the World Reference Center for Emerging Viruses and Arboviruses (WRCEVA) and the World Health Organization (WHO) to distribute SARS-CoV-2 isolates, proteins and plasmids. The SAVE group also has an open-face sharing policy in which findings are quickly disseminated through preprint servers while manuscripts undergo formal peer review. The head-to-head comparison, review and discussion of unpublished data has yielded real-time peer review that would otherwise take months to achieve.
The early-detection and analysis group
SARS-CoV-2 genome sequencing data have been shared in public databases. As of December 2021, GISAID—the most widely used database for SARS-CoV-2—has more than 6.5 million sequences deposited with more than 150,000 sequences added weekly. This depth and rate of growth of genetic information for an emerging virus is unprecedented, providing a unique resource to track virus evolution. From late 2020, the emergence of variants of concern (VOCs) with an increased risk to global public health prompted scientists to establish variant detection and tracking pipelines (such as Outbreak.info13, CDC SARS-CoV-2 Variant Classifications and Definitions14 and the BV-BRC SARS-CoV-2 Real-time Tracking and Early Warning System for Variants and Lineages of Concern (https://www.bv-brc.org)). The early-detection and analysis group was assembled to establish a systematic approach to identify and predict SARS-CoV-2 variants that might increase virus replication, transmission and/or escape immunity. The team’s main goal is to select and prioritize variants for development of key experimental reagents (for example, spike proteins for binding assays and pseudoviruses (PSVs) for neutralization assays) and viruses for challenge studies, as well as to inform the in vitro and in vivo groups about predicted variant properties to guide their experiments. The initial and primary focus has been on variants with mutations in the spike protein that might lead to antibody escape, with subsequent analyses considering T cell escape, infectivity and transmission. Other important characteristics—such as replication fitness and virulence—and genomic regions outside of the spike gene are also evaluated. The process is collaborative and iterative, with seven teams using independent models and methodologies to prioritize mutations and lineages as well as rank importance for downstream testing. Although the focus is on human infections, the early-detection group also monitors variants circulating in animal populations, such as mink and deer, as they represent a potential reservoir source.
Methodology
Genomic surveillance consists of weekly downloads of SARS-CoV-2 genomes from GISAID/GENBANK, quality filtering, alignment, and the identification of variant or co-variant substitutions. The main focus has been on potential antibody escape to identify mutations in key epitopes in the receptor-binding domain (RBD) and the N-terminal domain (NTD) supersite, but regions proximal to the furin-cleavage site or experiencing convergent/parallel evolution are also considered. The dynamics of these spike substitutions, as a function of time and geographical spread, are evaluated considering sequence prevalence and viral population growth rate, including comparative analyses to other variants co-circulating in a given geographical location (Fig. 2a). One example of recurrent substitutions with phenotypic relevance are those near to the furin-cleavage site, which result in enhanced spike cleavage and infectivity15,16. These mutations have been identified in different variants and in newly expanding lineages. Some teams take into account vaccine coverage when prioritizing an emerging lineage for analysis.
a, The trajectory of SARS-CoV-2 variant sequence prevalence over a one-year period, 1 January 2021 to 31 December 31 2021, tracking frequencies of weekly counts based on PANGO lineage designations. The data in the graphs are based on the 4.8 million SARS-CoV-2 sequences sampled in 2021 and made available through the GISAID Initiative. Updated graphs can be found online (https://cov.lanl.gov; the tracking tool is called Embers). Global summary and status of five continents. Europe and North America remain the most highly sampled regions of the world, biasing the global sampling. b, Tangle plots for comparative prioritization of circulating variants across subgroups. The list of variants to prioritize was built collectively by the whole group and prioritized by individual teams to arrive at a consensus list. Each column graph refers to the prioritization order made by each subteam for circulating variants in December 2021 (top, highest priority; bottom, lowest priority): A, Cambridge University; B, LANL; C, ISMMS; D, JCVI/BV-BRC; E, UCR SOM; F, Broad Institute; G, WRAIR. The final consensus ranking of the 43 variants was produced by ordering the lineages by their mean rank across the different teams, who also have the option to defer from ranking a lineage or to assign multiple lineages a tied ranking and, after discussion with the group, determine priority categories. The dashed arrow indicates the order of priority. The colours refer to each PANGO lineage tracked, but blocks of the same colour can also refer to different variants within a PANGO lineage. For example, in addition to the coloured Delta AY.* sublineages indicated, Delta has 26 subvariants (purple) with different combinations of mutations that are being prioritized for analysis.
The rankings are split into two broadly distinct methodologies, each with slight variations: one is based on convergent evolution as the main signal for selection and functional impact of mutations (that is, the Cambridge and Walter Reed Army Institute of Research (WRAIR) teams); whereas the other is anchored on prevalence and growth patterns of mutations and defined lineages (that is, the Los Alamos National Lab (LANL), Icahn School of Medicine at Mount Sinai (ISMMS), J. Craig Venter Institute/Bacterial Viral Bioinformatic Resource Center (JCVI/BV-BRC), UC Riverside and Broad Institute teams) (Fig. 2b).
The functional impact of mutations
Cambridge prioritizes substitutions that are likely to cause immune escape by looking at both experimentally determined escape from polyclonal sera and the effect of mutations on spike protein structure. Substitutions are given higher priority if they appear to be emerging and if they are in a different Barnes class17 from previously observed substitutions, and lower priority if they have already been tested experimentally. The WRAIR team tracks the prevalence of substitutions at a set of sites selected based on the strength of the interaction with known SARS-CoV-2 antibodies (using complex structures in the Protein Data Bank; https://www.rcsb.org) as well as structural information or knowledge from deep mutational scanning or mutagenesis studies. Weight scores for ranking are also given for various characteristics, such as the fold increase in detection over time and geographical spread or population growth in the context of high vaccination coverage.
Prevalence and growth patterns
The ISMMS team has a similar approach, whereby variants are ranked on the basis of an aggregate score for sequence prevalence increase and genetic changes of concern in sites of importance associated with functional changes (such as ACE2 binding, antibody escape) but also assigns weight to mutations in the active sites of viral enzymes. Moreover, data from surveillance cohorts in the New York City metropolitan area are used to assess lineages associated with local outbreaks and breakthrough infections after vaccination. LANL identifies emergent mutational patterns within the spike, RBD and NTD supersite to determine global and regional sampling frequencies. Variant dynamics and global spread are tracked at multiple geographical levels using a suite of tools5 (https://cov.lanl.gov/). The JCVI/BV-BRC team uses an algorithm combining sequence prevalence dynamics with functional impact predictions to rank emerging variants. Each mutation is given a sequence-prevalence score, reflecting geographically localized prevalence changes, and a functional impact score, on the basis of the location of the mutation within important spike protein regions and whether studies have demonstrated significant changes in either antibody- or ACE2-receptor binding18,19,20,21. UC Riverside uses relative growth in the prevalence of specific substitutions and deletions/insertions to identify the fastest growing variants and mutation combinations (https://coronavirus3d.org). For the final variant and subvariant ranking, additional criteria are included, such as their potential impact on protein structure (by modelling) and the re-emergence of individual mutations in previously undescribed combinations in new variants. Finally, the team from the Broad Institute, similar to the UC Riverside team, examines the accelerated growth of a variant relative to its peers, across multiple geographical regions, but fits a binomial logistic regression to each lineage’s proportion over time. Moreover, they fit hierarchical multinomial logistic regression models across geographical regions22.
Challenges for the early-detection and analysis group
The early-detection and analysis group has faced six main challenges in identifying emerging variants for functional testing: (1) the newest data are the most subject to bias and the least representative because of small numbers. The longer that one waits, the more accurate the data, but the greater the delay in identifying newly emergent variants for evaluation. (2) Disentanglement of epidemiological from evolutionary effects. A variant might show increased sequence prevalence within a geographical region due to founder effects, or increased incidence could be conferred by epidemiological factors rather than an evolutionary fitness advantage. An example of a founder effect is Delta AY.25, which is very common in North America but not increasing in frequency over time (Fig. 2a), versus AY.4.2, which was first sampled well after Delta was increasing in the UK and was constantly increasing in frequency in 27 countries where it was found and, furthermore, it never significantly decreased relative to other Delta variants once it emerged, suggesting positive selection. (3) Selective pressures on the virus are in flux, and mutations may be transient due to a balance with requirements for retention of fitness. Pressures are exerted by the host at the level of transmission, epidemiological interventions and immune evasion. (4) Under-representation of variant spread and evolution in countries with limited sampling and sequencing capacity. Although some parts of the world have an abundance of sequencing data(such as the UK and USA), others are under-represented (such as the African continent and China). There is an urgent need to increase sampling and sequencing capacity in resource-poor countries. (5) Variability in data quality. The submission of consensus assemblies without underlying raw read-level data means that quality cannot be independently evaluated. Erroneous genome sequences due to technical artifacts, low coverage or bioinformatic strategies that default to ancestral bases in regions without sequence coverage can affect the accuracy of variant amino acid calls23. (6) The database curation quality-control steps can filter on the basis of criteria that do not apply uniformly across lineages. The B.1.621/Mu lineage had an unexpected stop codon in ORF3a that caused B.1.621 sequences to be flagged during automated uploads to the GISAID database, which initially led Mu to be undercounted. This can lead to a false understanding of the dynamics of a given variant lineage globally. Despite these challenges, our prioritization methods continue to evolve as more information becomes available. These efforts have allowed for the rapid generation of reagents for multiple variants before they have spread extensively in the USA and have been critical for guiding the in vitro and in vivo groups. A list of regularly updated prioritized variants is available online (https://docs.google.com/spreadsheets/d/167uJP9LfJN07410sWaMSKU1Se-4XX687j8IgVX4MV_w/edit?usp=sharing).
The in vitro group
The in vitro group performs antibody binding, neutralization, Fc effector and T cell stimulation assays to understand how SARS-CoV-2 variants affect vaccine- and infection-induced immunity. The in vitro group serves as a critical intermediary between the early detection and analysis and in vivo groups by providing valuable data to confirm variant lineage prioritization, and ranking viruses for prioritized in vivo challenge studies. The in vitro group was initially tasked with developing key reagents (for example, spike and RBD antigens, and plasmids for generating PSVs) and procuring biospecimens (such as authentic viruses and sera/plasma from infected and vaccinated individuals). At the beginning of 2021, reagents for generating data—including variant virus isolates, recombinant infectious clones, recombinant variant spike proteins for antibody binding assays, variant-specific expression plasmids for PSV particle entry inhibition assays and variant-specific sera—were not widely available (Fig. 3a). A key lesson from this process is that the streamlining of administrative procedures for reagent sharing facilitates data generation that directly informs urgent policy- and decision-making. A substantial and ongoing challenge requiring numerous administrative steps is to obtain authentic virus isolates from domestic and international sources. To expedite this process, we developed a pipeline between SAVE investigators to isolate, propagate and sequence emerging viruses. This effort led to cataloguing and isolating hundreds of SARS-CoV-2 variants representing over 40 lineages. For more difficult to obtain SARS-CoV-2, additional efforts have been made to generate infectious clones24,25,26. Furthermore, the early-detection group prioritized viral variants and curated sequences to accelerate the production of recombinant variant spike proteins and expression plasmids.
a, Live-virus nasal swabs in viral transport medium or seed stocks are obtained followed by plaque purification and deep-sequencing. Pseudotyped virus plasmids encoding the variant spike sequence are synthesized to generate pseudotyped lentivirus stocks. The vesicular stomatitis virus (VSV) chimeric virus glycoprotein gene (G) is replaced with the spike protein of SARS-CoV-2 (VSV-eGFP-SARS-CoV-2) and a GFP reporter gene33. b, The in vitro group conducted performance testing between 12 neutralization assays involving live authentic virus consisting of focus-reduction neutralization test (FRNT-1), recombinant SARS-CoV-2 reporter virus FRNT (FRNT-2), plaque-reduction neutralization test (PRNT), recombinant SARS-CoV-2 expressing nano-luc (Nano-luc), cytopathic effect assay (CPE), microneutralization assay (MNA), focus-reduction neutralization assay (FRNA), lentivirus and VSV pseudotyped neutralization assays, and VSV chimeric assays. An example comparison between the wild-type (WT) and Beta virus is presented. c, Antigenic cartography; 50% infectious dose (ID50) neutralization titres in a lentivirus-based PSV assay were determined against a panel of SARS-CoV-2 variants and serum from individuals who were vaccinated with the Moderna vaccine, or individuals with SARS-CoV-2 infection. The distance between serum to an antigen corresponds to the titre of that serum for the antigen37. The grid lines represent twofold dilution of antiserum. The y and x axes represent antigenic distance. Circles, antigens; squares, sera. d, T cell responses to SARS-CoV-2 variants. Sequencing data are curated for coding mutations (pink boxes). Curated mutations are tested on convalescent T cell responses using functional assays (activation-induced marker (AIM) assays; green boxes). Immune Epitope Database (IEDB) and the immunocode multiplex identification of T cell receptor antigen specificity dataset (MIRA) are analysed to generate curated peptide sets of immunodominant epitopes (blue boxes). Data are integrated to produce a ranked score list of variant epitope changes weighted by their likelihood to disrupt epitope binding and the relative size of the affected population (grey boxes). MPs, megapools. Partially created using BioRender.
At the start of the pandemic, the correlates of immune protection were unknown for COVID-19. Multiple teams within the in vitro group conducted assessments of vaccine-induced serum neutralization using parallel but independent methods across laboratories. Studies with clinical samples show neutralizing antibody titres are a strong predictor of protection against severe disease27. As such, a major undertaking of the in vitro group has been to use neutralization assays to assess the effect of spike mutations on the inhibitory activity of clinically approved monoclonal antibodies and serum/plasma from vaccinated or infected individuals. One of the strengths of the in vitro group is the use of orthogonal SARS-CoV-2 neutralization assays based on authentic live viruses, PSVs and chimeric viruses. An initial task of this group was to compare neutralization assay platforms across 12 independent laboratories using a defined serum panel from individuals vaccinated with the Pfizer and Moderna vaccines. Using either the ancestral wild-type virus (Wuhan-1) or more recent variants (for example, the Beta variant shown in Fig. 3b), team members performed neutralization assays that varied on the basis of live virus assay readouts (foci, plaques, cytopathic effect, luciferase and fluorescence), target cells, and expression of ACE2 and/or TMPRSS2 on target cells28,29,30,31,32,33,34,35. This type of performance testing has highlighted differences between assay platforms, cell targets and readouts that can impact neutralization potency. Nonetheless, in most cases, there was considerable congruence across platforms. Another area of emphasis is using variant-infected serum/plasma samples to visualize the antigenic evolution of spike through a process called antigenic cartography36,37 (Fig. 3c). This two-dimensional map provides a landscape of how spike mutations drive loss in neutralizing activity.
For many viruses, the affinity and magnitude of antibody binding to viral glycoproteins associates with virus-neutralizing activity, and a strong correlation has been shown for SARS-CoV-238,39,40,41. Investigating the correlation between the neutralizing and binding activity of vaccine-induced antibodies showed that spike mutations alter this slope, and virus neutralization is often more affected than antibody binding42,43. This has been confirmed through different platforms measuring changes in binding to either native spike proteins or the RBD, including ELISA44 and multiplexed spike antigen detection platforms7. One potential explanation for this is that many more binding than neutralizing epitopes exist on the spike protein. Some antibodies that have neutralizing activity against the wild-type virus may lose activity to variants, yet overall binding is still maintained—a phenomenon observed for other viruses (such as influenza virus45).
Binding antibodies can still have a considerable protective effect, irrespective of neutralizing activity due to Fc effector functions, as seen with influenza virus or Ebola virus46,47,48. The humoral immune response restricts microbes through the coordinated effort of the Fab (antigen-binding) and Fc (constant) domains49. After infection or vaccination, polyclonal antibodies are induced that target pathogens at multiple sites through their Fab domains. Fab domains that directly or indirectly hinder virus entry are neutralizing; however, the remaining ‘non-neutralizing’ antibodies can bind to and opsonize the pathogen to form immune complexes, or bind to spike proteins on the surface of infected cells. Once complexed, the Fc domains act as molecular beacons that draw in immune cells through Fc-gamma receptors (FcγRs), providing instructions on how the immune system should destroy the antibody-opsonized material. Fc-effector functions of antibodies are linked to natural resolution of COVID-1950,51,52,53, correlate with vaccine-mediated protection from infection in animal models54,55,56 and are associated with protection after the transfer of passive convalescent serum or monoclonal antibodies57,58,59,60. Although emerging variants of SARS-CoV-2 can escape neutralizing antibodies, their substitutions alter a limited fraction of the overall humoral immune response to the SARS-CoV-2 spike56,61. Thus, Fc-effector functions have more resilience in the face of variation across spike, for both mRNA and the adenoviral 26 (Ad26) vaccines, offering mechanisms through which antibodies may continue to confer protection despite escaping neutralization.
Growing evidence from animal models and human studies indicates that CD4+ and CD8+ T cells have protective roles in preventing severe disease and death from SARS-CoV-2 infection6,62,63,64. T cells are an attractive target for intervention as they are less susceptible to viral escape than antibodies6,65. This is largely for two reasons: (1) in convalescent individuals, T cells can target peptides derived from the entire proteome, not just surface-exposed epitopes; and (2) HLA-restriction and diversity creates interpersonal variation in the repertoire of targets, limiting the immunological pressure on any one epitope. Given the presumed role of T cells in limiting severe disease and their potential for sustaining protection against variant mutation, the SAVE in vitro group included assessment of T cell responses. The goal was to determine empirical drift from vaccination and infection-induced immunity, and to develop tools to predict the impact of variant-associated mutations on immunodominant T cell responses.
The T cell investigations follow two parallel approaches to assess the impact of variant mutations on T cell reactivity and a broad range of different variants (Fig. 3d). The first involves measuring the overall reactivity against the entire spike protein (in the case of vaccination) or the entire proteome (in the case of infection) and expressing the results as the fold difference relative to the ancestral sequences. A parallel approach characterizes the mutational impact on specific single epitopes, and monitors whether individuals with decreased T cell reactivity have responses that selectively recognize certain epitopes in the context of particular HLA types. Regarding the first approach, at the general population level, the results to date have detected a limited impact of mutations within spike after natural infection or mRNA vaccination6 against the most concerning variants at the time the study was performed (B.1.1.7, B.1.351, P.1 and B.1.427/429). These findings were corroborated66 and expanded to adenoviral-vector-based vaccination67. However, in a minority of individuals, two- to threefold decreases in the CD8+ T cell responses against the B.1.351/Beta and B.1.427/429/Epsilon variants were noted6. These findings suggest that a more in-depth characterization at the single-epitope level is required to understand the mechanisms behind the reduced CD8+ T cell response in specific individuals. Moreover, it is critical to monitor and predict the effect of emerging circulating variants on T cell reactivity, particularly regarding the most concerning (to date) B.1.617.2/Delta variant (including the AY.* sublineages) and B.1.1.529/Omicron variant. The experimental data will be used to confirm and improve the bioinformatics analysis and infer the impact of current and upcoming variants on SARS-CoV-2 specific T cell responses.
Advanced computational tools for assessing SARS-CoV-2 genome mutations on HLA binding have enabled prediction of the effect of mutations within a VOC on T cell reactivity. Owing to the broad diversity of HLA genotypes, T cell escape at the population level is not likely, as demonstrated for multiple VOCs6. However, previous work on HIV and influenza virus has identified associations between specific HLA class I alleles, disease severity68,69 and vaccine efficacy70. We anticipate that, as SARS-CoV-2 continues to spread globally, T cell immunity will eventually drive viral evolution. In these situations, specific HLA alleles may become associated with a reduced ability to mount responses against dominant T cell epitopes, which may affect clinical outcomes. The T cell subgroup has developed a computational pipeline to assess the effects of specific mutations on HLA binding by also ranking all individual mutations on a T cell escape score, based on experimentally verified and predicted T cell responses (Fig. 3d). This ranking will provide early identification of specific mutations associated with T cell escape, particularly CD8+ T cells, and testable hypotheses for T cell experiments. In our preliminary analyses of VOCs, the B.1.617.2 variant was identified as the first in which mutations were associated with reduced HLA binding at the population level. These data suggest that T cell cross-reactivity to B.1.617.2 may be reduced in some individuals. Owing to the extensive number of SARS-CoV-2 viral genomes, and large-scale clinical cohorts that are being studied, the T cell SAVE group plans to assemble a database linking HLA genotypes with clinical outcome and viral genomes, which may provide a unique opportunity to study HLA associations with clinical disease and viral evolution at a resolution that has not previously been attempted.
Challenges for the in vitro group
Work by the in vitro group has focused mostly on characterizing neutralizing antibody responses to the spike protein with some analysis of the impact of variants on T cell responses as well. With the recent increase in Omicron infections in vaccinated and unvaccinated individuals, a challenge moving forward will be to disentangle vaccine- and infection-induced immunity, breakthrough infections, waning immunity and other covariates associated with increased risk of symptomatic infection (immunocompromised, age, obesity, diabetes). Many other key aspects of SARS-CoV-2 and its variants remain uninvestigated. Although neutralizing antibodies correlate with protection from SARS-CoV-2, neutralization is not the only function of antibodies. In fact, non-neutralizing antibodies can afford substantial protection against influenza virus46,71,72,73 and similar mechanisms remain to be examined for SARS-CoV-2. Furthermore, differences between wild-type and variant viruses in ACE2 binding, fusion, impact of mutations on spike processing by proteases, and potentially fusion at the cell membrane and cell-to-cell fusion, remain poorly understood15. Furthermore, the spike protein is just one of many SARS-CoV-2 proteins. The effect of mutations in non-spike proteins on immunity and viral fitness, including transmission, virus–host interaction and polymerase fidelity has not yet been assessed. The use of reverse genetics systems and PSVs can be leveraged to understand the contribution of individual mutations to viral fitness and evasion of antibody responses24,25,26,74. We acknowledge that differences between the ancestral and variant viruses may also impact neutralization assays in different cell lines. Although we have seen some of these cell-line-specific effects in in vitro neutralization, we do not yet understand their underlying mechanisms. Furthermore, we need to increase the use of reference standards in binding and neutralization assays, such as the WHO International Standard and International Reference Panel for anti-SARS-CoV-2 immunoglobulin75, to calibrate assays and provide a means to compare serological findings. Rare and volume-limited variant-specific sera/plasma are difficult to obtain and share across borders, and/or between academic institutions, and the process is often slowed by administrative hurdles. On occasion, SARS-CoV-2 variant sample sharing has not been possible within the needed timeframe, impeding the research response to this public health emergency. Although access to virus isolates outside the USA and variant-specific human sera remains limited, the in vitro group has created an extensive network of collaborations to overcome these hurdles. Finally, much remains to be examined for both antibody and T cell responses about emerging variants such as B.1.617.2, B.1.617.2 subvariants, B.1.1.529 and other new variants of interest and concern.
The in vivo group
SARS-CoV-2 animal models have been critical for the development and testing of vaccines and antiviral therapeutics76,77,78,79,80,81,82,83,84. Initial countermeasures targeted the spike protein from the SARS-CoV-2 strain circulating during the early phase of the pandemic in 2020 and focused on efficacy testing against homologous strains. However, the emergence of variants and their possible effects on transmission, pathogenesis, and infection- or vaccine-mediated immunity required rapid adaptation of animal models to confirm vaccine efficacy against VOCs. The in vivo group was assembled to develop animal models, standardize reagents and assays, and examine the effect of SARS-CoV-2 variants on protection elicited by vaccine- or infection-induced immunity and transmission. The variants identified for investigation by the early-detection and analysis group that are validated in the in vitro group are then forwarded to the in vivo group. The in vivo group studies protection against SARS-CoV-2 variants using an array of animal models, including mice, hamsters and non-human primates (NHPs). This has led to a collaborative process in which transmission, pathogenesis and protection data are shared to develop a consensus on the effect of emerging variants on protective immunity.
Development of animal models
One of the first tasks of the in vivo group was to standardize viral challenge stocks, routes and doses of infection, and vaccination strategies across each of the animal models. To minimize variability and adventitious mutations associated with virus propagation in different cell types, Vero-TMPRSS2 cells were used to generate challenge stocks and distributed among team members85. As new variants emerge, viruses are tested in small-animal models to determine infectivity and pathogenicity. For vaccination studies, the group focused on evaluating both approved (including Emergency Use Authorization (EUA)) vaccines and those undergoing advanced clinical testing in humans, including mRNA vaccines (Pfizer BNT162b and Moderna mRNA-1273), protein-based vaccines (Novavax NVX-CoV2373) and virus-vectored vaccines (J&J Ad26.COV2.S). For these studies, vaccine doses were optimized to model magnitude and durability of vaccine-induced immunity across the animal models. For each vaccination experiment, many parameters are studied, including neutralizing antibody potency and kinetics, pathogenesis of ancestral and variant viruses, as well as the levels of virus in various respiratory tract tissues.
Mouse models
The ancestral SARS-CoV-2 strain does not replicate in conventional laboratory mice as the spike protein inefficiently binds to mouse ACE286,87. To overcome this obstacle, several mouse models were developed, including human ACE2 (hACE2) transgenic mice (for example, K18-hACE288, originally developed for studies of SARS-CoV88) and mice that express hACE2 transiently after transduction with viral vectors (such as adenovirus)89,90. The K18-hACE2 transgenic mice are highly permissive for most SARS-CoV-2 strains and variants, and infection typically results in weight loss, nasal turbinate and lung infection, pneumonia and death60,91,92. Lungs from SARS-CoV-2-infected mice show denuding bronchiolitis, mixed inflammatory infiltrate, alveolar oedema and alveolitis92,93,94. Some mice, especially young K18-hACE2 mice, develop infection in the brain and encephalitis, which may confound interpretation of clinical disease95. The spike mutation at position N501Y, which is found in mouse-adapted strains and several emerging variants (Alpha, Beta, Gamma, Mu and Omicron), increases the affinity of the SARS-CoV-2 spike protein for the murine ACE2 receptor and enables direct infection of inbred mice86. Thus, in addition to the K18-hACE2 mouse model, challenge of conventional laboratory mice with mouse-adapted virus or SARS-CoV-2 variants containing an N501Y substitution within the spike protein cause pneumonia in BALB/c, 129S2 and C57BL/6 mice in an age-dependent manner89,96,97,98. The initial characterization of each variant in a variety of mouse strains at different doses allowed for vaccination studies to be conducted with validated stocks and consistent phenotypes. The in vivo group has used both inbred mouse strains (such as BALB/c, C57BL/6, 129S1, 129S2) and K18-hACE2 transgenic mice for iterative infection and vaccination studies.
As part of the experimental design, the in vivo team uses a high and low vaccine dose strategy with Ad26.COV2.S, NVX-CoV2373, BNT162b and mRNA-1273 to study the effect of variant mutations on protection. Mice inoculated with a high vaccine dose are useful for evaluating antibody-mediated protection of the upper and lower respiratory tract after challenge. Mice inoculated with the lower vaccine dose serve as a model for suboptimal immunity (as might be seen in the elderly or immunocompromised) and breakthrough infection. Through this effort, the group has demonstrated that low doses of mRNA vaccines (BNT162b and mRNA-1273) or protein vaccine (NVX-CoV2373) show reduced protection compared with high vaccine doses (Fig. 4) against the B.1.351 and B.1.617.2 variants99. These experiments are being extended to study additional variants and a range of vaccination doses.
a, Animal model development. After selecting variants for analysis by the early-detection and in vitro analysis group, isolates are grown, validated using next-generation sequencing, and analysed in each animal model at different doses to determine the pathogenicity, viral kinetics and transmission (in hamsters). Weight loss, lung titre and lung pathology was assessed to generate benchmarks for vaccine studies. b, Vaccine challenge. Each animal model is immunized with selected vaccines. Animal serum is examined after vaccination for neutralizing antibody levels and across a systems serology analysis before viral challenge with the chosen variants. Protection against infection and disease in each model is analysed to determine the protective ability of each vaccine and variant. Data on the protection of each animal model with each vaccine platform and challenged with variant viruses are shared with the SAVE consortium and SIG. Created using BioRender.
Another goal of the SAVE team is to assess the ability of previous infection to protect against secondary challenge with SARS-CoV-2. To model this, middle-aged C57BL/6 mice are inoculated with SARS-CoV-2 (B.1.1.7/Alpha, B.1.351/Beta or P.1/Gamma variants) followed by a homologous or heterologous challenge up to 120 days later with either variants or mouse-adapted SARS-CoV-2. Mice infected with any of the three variants remain asymptomatic but develop a neutralizing antibody response that is measurable at 21 days after infection. However, neutralizing antibodies cannot be detected in most mice at 3 months after infection, yet mice are still partly protected from variant virus challenge or from a dose of mouse-adapted SARS-CoV-2 that is lethal to mice without pre-existing immunity. Part of the goal of this project is to measure T and B cell memory responses to understand why the mice remain protected.
Hamster models
Syrian golden hamsters are highly susceptible to SARS-CoV-2 infection and disease without any species-specific adaptation of the virus, and have a disease phenotype that resembles mild disease observed in human COVID-19 cases. Loss of 10–20% of the initial body weight is seen at 6–7 days after infection depending on the age and sex of the animal and the variant and dose of the virus100,101,102. Virus replication is confined to the upper and lower respiratory tract, which peaks at 3 days after infection and then wanes to undetectable amounts by 10 days after infection. Imaging of the lungs of infected hamsters shows abnormalities during the course of infection that do not directly resolve even after virus clearance100.
Pathological changes in the lungs of the hamsters are comparable to those in some humans and are characterized by widespread, moderate to severe bronchointerstitial pneumonia100,103. Lung lesions comprise focal extensive areas of pulmonary oedema and consolidation with evidence of interstitial pneumonia. Histopathological lesions include fibrin deposits and oedema in alveolar spaces, influx of neutrophils and macrophages into alveolar spaces, the presence of syncytial cells and prominent type II pneumocyte hyperplasia. Secondary bacterial infections are often detected in the lungs. Despite a robust infection, SARS-CoV-2 is not lethal in healthy hamsters and infected animals recover.
As the use of the hamster model for SARS-CoV-2 infection was established using an early isolate of SARS-CoV-2, infection studies have been performed with several variants. Hamsters are largely agnostic to the variants, demonstrating little differences in viral replication and shedding kinetics between different variants including D614G, B.1.1.7 and B.1.351104. Although competition infections are more sensitive to revealing small effects of SARS-CoV-2 mutations on fitness for airway infection and transmission25,105, recent studies indicate that B.1.1.529/Omicron is attenuated in hamsters with less infection in the lungs106.
Given their general susceptibility to SARS-CoV-2, hamsters are an excellent model to study vaccine-induced immunity against variants. Although immunological reagents are less widely available for hamsters, antibody responses induced by vaccination can be measured in neutralization assays and ELISAs by using hamster-specific IgG, IgA and IgM secondary antibodies. Cohorts of animals receive two immunizations with either the Pfizer BNT162b2 and Moderna mRNA-1273 vaccine given three and four weeks apart, respectively. The vaccine dose in these studies has generally been one-third of the dose given to humans (that is, 10 µg of the Pfizer vaccine or 35 µg of the Moderna vaccine), and an additional freeze–thaw of the Pfizer and Janssen vaccines does not decrease immunogenicity. IgG antibody titres against the SARS-CoV-2 spike can be detected after the first vaccination but are more robust three weeks after the second vaccination, similar to data in human studies. As long-term vaccine immunity is a key question in SARS-CoV-2 research, vaccinated animals are held for extended periods of time before challenge with new emerging variants.
Transmission studies are well established in hamsters, as SARS-CoV-2 can transmit efficiently through the aerosol route from an infected to naive hamster. Increases in the transmission potential of the D614G and the B.1.1.7 variants have been observed compared with other isolates of SARS-CoV-2. Using a direct-contact transmission model, intramuscular or intranasal vaccinated ChadOx1/AZD1222 hamsters were protected from disease but not upper respiratory tract infection107,108. This suggests the hamster transmission model is a useful tool to study vaccine efficacy in the context of natural exposure.
NHP model
Vaccinated NHPs are an important experimental model for demonstrating immunogenicity and protective efficacy against SARS-CoV-230,109,110,111,112. NHPs have several advantages for clinical translation. First, NHPs are outbred, and their innate immune responses and B and T cell repertoires have greater similarity to humans than those of rodents. Owing to the diversity in class I and II MHC, NHPs also support the study of the breadth of T cell responses induced by vaccines. Second, NHPs enable the use of clinically relevant vaccine doses and are an excellent model to study the durability of immune responses. Third, after intranasal and intratracheal administration, viral replication occurs rapidly in the upper and lower airways, respectively, with similar kinetics to humans109,110. For most SARS-CoV-2 strains, infection is cleared by 7–10 days after challenge. Inflammation and pathology in the lungs are consistent with mild infection as described in humans. The NHP model has been used to show immunogenicity and protection after vaccination with mRNA77, ChAdOx-178, Ad26-spike84, protein/adjuvant79 or inactivated whole virus113,114, which have all been approved for use in humans. The NHP model has also been used to understand immune correlates and mechanisms of protection. The SAVE investigators are currently studying homologous and heterologous prime–boost vaccinations using EUA or approved vaccines for their ability to induce humoral and cellular immunity, longevity of immune responses, and the mechanisms associated with induction of long-lived immunity and protection in the upper and lower airway.
Challenges for the in vivo group
SARS-CoV-2 animal models provide an opportunity to understand mechanisms of infection, inflammation, pathogenesis and transmission across species and against different vaccine platforms. Similar to the concerns raised with the in vitro group, an initial challenge for the in vivo group was obtaining authentic viruses without cell-culture-adaptive mutations for challenge studies. To overcome this hurdle, a parallel pipeline for propagating and sequencing challenge stocks was developed to ensure not only the presence of lineage-defining mutations but also the absence of mutations associated with propagation in tissue culture, and that included standardizing virus dose and routes of inoculation and distributing the same stocks to all of the team members. SARS-CoV-2 strains are constantly evolving, challenging the decision as to which strains are most relevant for in vivo study. Each animal model has unique opportunities and limitations that are considered when evaluating protective immunity against a variant. Mice are a tractable system with an array of immunological tools, assays and genetic knockout strains that allow for experimental rigour and mechanistic analysis. However, inbred mouse strains are limited to SARS-CoV-2 variants that possess an N501Y mutation in the spike protein, and the genetic background (such as BALB/c, 129S2, C57BL/6) can impact viral replication and pulmonary pathology. Most variants appear to infect hamsters at similar levels with comparable lung inflammation and pathology, with the apparent exception of B.1.1.529 (ref. 106). However, infection in hamsters causes a mild to moderate disease and there are limited immunological reagents to probe the response to infection and vaccination. The transmission models are exquisitely sensitive and further studies are needed to understand experimental parameters (such as airflow, contact time, relative humidity, temperature) that modulate the transmission efficiency and standardize experimental systems. The NHP model has been used in conjunction with the small-animal models using the same viral stocks to provide a more comprehensive analysis across species for how the vaccines are mediating protection. As NHPs are more limited in their availability, the rodent models can inform the best use of this model. As the SARS-CoV-2 pandemic continues, animal models will need to be adapted to reflect the immune status of the population (for example, natural infection, vaccination and booster shots). These large- and small-animal models will be essential for further testing of next-generation vaccines, boosters formulated with variant spikes and immunological imprinting.
Summary
Collaborative science and open sharing of results in near real time have defined the SAVE programme. This cross-fertilization has enabled the efficient and rapid analysis of the effect of emerging variants on infection- and vaccine-induced immunity. The emergence of the B.1.1.529/Omicron variant, which contains more than 30 mutations in the spike protein, threatens clinically approved monoclonal antibodies and infection- and vaccine-induced immunity. The SAVE group rapidly responded by generating plasmids and spike protein, isolating, propagating and distributing authentic Omicron viral stocks, submitting reagents to public repositories, performing binding and neutralization assays and evaluating virus infection across different animal models106,115,116,117,118,119,120,121,122. The data from these studies were rapidly shared with government agencies and submitted as manuscripts on preprint servers.
Over the past two decades, we have witnessed the emergence and re-emergence of several RNA viruses, including West Nile virus, H1N1 influenza virus, chikungunya virus, Zika virus, SARS-CoV-1, MERS-CoV and Ebola virus that have threatened global public health. Developing collaborative programmes between academic, industry and commercial partners is essential to respond to rapidly evolving viruses. This progressive approach combined with open communication and coordination by NIAID/NIH has facilitated rapid prioritization, reagent development, testing and assessment of SARS-CoV-2 variants. The mutual relationship between the SAVE group and the SIG has provided feedforward and feedback loops to aid with key decision matters involving risk assessment, SARS-CoV-2 countermeasures, diagnostics and public health policy. In addition to the SAVE group, other national and international networks have been developed for assessing the risk of SARS-CoV-2 mutations on immunity. This includes the Genotype to Phenotype–UK National Virology Consortium, the Genotype to Phenotype–Japan Consortium, the NIH Accelerating COVID-19 therapeutic interventions and vaccines (ACTIV) tracking resistance and coronavirus evolution (Trace) consortium, the WHO R&D Blueprint and the National Cancer Institute Serological Sciences Network (SeroNet). These partnerships must continue to increasingly include scientists across the world to ensure that variants are rapidly identified and characterized to determine their impact on transmission, infection, replication and immune evasion. This SAVE programme is a template to develop reagents, models, assays and diagnostics, and test therapeutics and vaccines in preclinical models against rapidly evolving pathogens.
References
Krammer, F. SARS-CoV-2 vaccines in development. Nature 586, 516–527 (2020).
Plante, J. A. et al. The variant gambit: COVID-19’s next move. Cell Host Microbe 29, 508–515 (2021).
Walensky, R. P., Walke, H. T. & Fauci, A. S. SARS-CoV-2 variants of concern in the United States—challenges and opportunities. JAMA 325, 1037–1038 (2021).
Fischer, W. et al. HIV-1 and SARS-CoV-2: patterns in the evolution of two pandemic pathogens. Cell Host Microbe 29, 1093–1110 (2021).
Korber, B. et al. Tracking changes in SARS-CoV-2 spike: evidence that D614G increases infectivity of the COVID-19 virus. Cell 182, 812–827 (2020). This was the first study showing that a newly emerging mutation in the spike protein could come to rapidly dominate across the globe, indicating a fitness advantage.
Tarke, A. et al. Impact of SARS-CoV-2 variants on the total CD4+ and CD8+ T cell reactivity in infected or vaccinated individuals. Cell Rep. Med. 2, 100355 (2021).
Pegu, A. et al. Durability of mRNA-1273 vaccine-induced antibodies against SARS-CoV-2 variants. Science 373, 1372–1377 (2021). This study describes the impact of several SARS-CoV-2 variants on antibody binding and neutralization on mRNA-1273 vaccine sera between 1 and 6 months after the second dose.
Carreno, J. M. et al. Evidence for retained spike-binding and neutralizing activity against emerging SARS-CoV-2 variants in serum of COVID-19 mRNA vaccine recipients. EBioMedicine 73, 103626 (2021).
Jansen, J. E. & Retpen, J. A. Acute spontaneous streptococcal myositis. Case report. Acta Chir. Scand. 154, 323–324 (1988).
Lusvarghi, S. et al. Key substitutions in the spike protein of SARS-CoV-2 variants can predict resistance to monoclonal antibodies, but other substitutions can modify the effects. J. Virol. 96, JVI0111021 (2021).
Neerukonda, S. N. et al. Establishment of a well-characterized SARS-CoV-2 lentiviral pseudovirus neutralization assay using 293T cells with stable expression of ACE2 and TMPRSS2. PLoS ONE 16, e0248348 (2021).
Pino, M. et al. A yeast expressed RBD-based SARS-CoV-2 vaccine formulated with 3M-052-alum adjuvant promotes protective efficacy in non-human primates. Sci. Immunol. 6, eabh3634 (2021).
Mullen, J. L. et al. Outbreak.info https://outbreak.info/ (2020).
SARS-CoV-2 Variant Classifications and Definitions (CDC, 2021).
Escalera, A. et al. Mutations in SARS-CoV-2 variants of concern link to increased spike cleavage and virus transmission. Cell Host Microbe 30, 373–387 (2022).
Liu, Y. et al. Delta spike P681R mutation enhances SARS-CoV-2 fitness over Alpha variant. Cell Reports https://doi.org/10.1016/j.celrep.2022.110829 (2022).
Barnes, C. O. et al. SARS-CoV-2 neutralizing antibody structures inform therapeutic strategies. Nature 588, 682–687 (2020).
Starr, T. N., Greaney, A. J., Dingens, A. S. & Bloom, J. D. Complete map of SARS-CoV-2 RBD mutations that escape the monoclonal antibody LY-CoV555 and its cocktail with LY-CoV016. Cell Rep. Med. 2, 100255 (2021).
Greaney, A. J. et al. Comprehensive mapping of mutations in the SARS-CoV-2 receptor-binding domain that affect recognition by polyclonal human plasma antibodies. Cell Host Microbe 29, 463–476 (2021).
Starr, T. N. et al. Prospective mapping of viral mutations that escape antibodies used to treat COVID-19. Science 371, 850–854 (2021).
Starr, T. N. et al. Deep mutational scanning of SARS-CoV-2 receptor binding domain reveals constraints on folding and ACE2 binding. Cell 182, 1295–1310 (2020).
Obermeyer, F. et al. Analysis of 6.4 million SARS-CoV-2 genomes identifies mutations associated with transmissibility. Preprint at medRxiv https://doi.org/10.1101/2021.09.07.21263228 (2022).
Davis, J. J. et al. Analysis of the ARTIC version 3 and version 4 SARS-CoV-2 primers and their impact on the detection of the G142D amino acid substitution in the spike protein. Microbiol. Spectr. 9, e0180321 (2021).
Xie, X. et al. An infectious cDNA clone of SARS-CoV-2. Cell Host Microbe 27, 841–848 (2020).
Liu, Y. et al. The N501Y spike substitution enhances SARS-CoV-2 infection and transmission. Nature 602, 294–299 (2021). This study tested all of the spike mutations identified in the first variant of concern, B.1.1.7/Alpha, and identified N501Y as being important for enhanced transmission and an adaptive mutation of concern.
Xie, X. et al. Neutralization of SARS-CoV-2 spike 69/70 deletion, E484K and N501Y variants by BNT162b2 vaccine-elicited sera. Nat. Med. 27, 620–621 (2021).
Khoury, D. S. et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 27, 1205–1211 (2021).
Vanderheiden, A. et al. Development of a rapid focus reduction neutralization test assay for measuring SARS-CoV-2 neutralizing antibodies. Curr. Protoc. Immunol. 131, e116 (2020).
Wang, P. et al. Increased resistance of SARS-CoV-2 variant P.1 to antibody neutralization. Cell Host Microbe 29, 747–751 (2021).
Corbett, K. S. et al. Protection against SARS-CoV-2 beta variant in mRNA-1273 vaccine-boosted nonhuman primates. Science 374, 1343–1353 (2021).
Chen, R. E. et al. Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies. Nat. Med. 27, 717–726 (2021).
Hou, Y. J. et al. SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract. Cell 182, 429–446 (2020).
Case, J. B. et al. Neutralizing antibody and soluble ACE2 inhibition of a replication-competent VSV-SARS-CoV-2 and a clinical isolate of SARS-CoV-2. Cell Host Microbe 28, 475–485 (2020).
Saadat, S. et al. Binding and neutralization antibody titers after a single vaccine dose in health care workers previously infected with SARS-CoV-2. JAMA 325, 1467–1469 (2021).
Muruato, A. E. et al. A high-throughput neutralizing antibody assay for COVID-19 diagnosis and vaccine evaluation. Nat. Commun. 11, 4059 (2020).
Fonville, J. M. et al. Antibody landscapes after influenza virus infection or vaccination. Science 346, 996–1000 (2014).
Smith, D. J. et al. Mapping the antigenic and genetic evolution of influenza virus. Science 305, 371–376 (2004).
Wajnberg, A. et al. Robust neutralizing antibodies to SARS-CoV-2 infection persist for months. Science 370, 1227–1230 (2020).
Amanat, F. et al. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat. Med. 26, 1033–1036 (2020).
Suthar, M. S. et al. Rapid generation of neutralizing antibody responses in COVID-19 patients. Cell Rep. Med. 1, 100040 (2020).
Cohen, K. W. et al. Longitudinal analysis shows durable and broad immune memory after SARS-CoV-2 infection with persisting antibody responses and memory B and T cells. Cell Rep. Med. 2, 100354 (2021). This study describes the durability of humoral and T-cell-mediated immunity after SARS-CoV-2 infection.
Carreño, J. M. et al. Evidence for retained spike-binding and neutralizing activity against emerging SARS-CoV-2 variants in serum of COVID-19 mRNA vaccine recipients. eBioMedicine 73, 103626 (2021).
Amanat, F. et al. SARS-CoV-2 mRNA vaccination induces functionally diverse antibodies to NTD, RBD, and S2. Cell 184, 3936–3948 (2021).
Stadlbauer, D. et al. SARS-CoV-2 seroconversion in humans: a detailed protocol for a serological assay, antigen production, and test setup. Curr. Protoc. Microbiol. 57, e100 (2020).
Roubidoux, E. K. et al. Mutations in the hemagglutinin stalk domain do not permit escape from a protective, stalk-based vaccine-induced immune response in the mouse model. mBio 12, e03617–20 (2021).
Asthagiri Arunkumar, G. et al. Broadly cross-reactive, nonneutralizing antibodies against influenza b virus hemagglutinin demonstrate effector function-dependent protection against lethal viral challenge in mice. J. Virol. 93, e01696–18 (2019).
Saphire, E. O. et al. Systematic analysis of monoclonal antibodies against ebola virus GP defines features that contribute to protection. Cell 174, 938–952 (2018).
DiLillo, D. J., Tan, G. S., Palese, P. & Ravetch, J. V. Broadly neutralizing hemagglutinin stalk-specific antibodies require FcγR interactions for protection against influenza virus in vivo. Nat. Med. 20, 143–151 (2014).
Lu, L. L., Suscovich, T. J., Fortune, S. M. & Alter, G. Beyond binding: antibody effector functions in infectious diseases. Nat. Rev. Immunol. 18, 46–61 (2018).
Zohar, T. et al. Compromised humoral functional evolution tracks with SARS-CoV-2 mortality. Cell 183, 1508–1519 (2020).
Atyeo, C. et al. Distinct early serological signatures track with SARS-CoV-2 survival. Immunity 53, 524–532 (2020).
Anand, S. P. et al. Longitudinal analysis of humoral immunity against SARS-CoV-2 spike in convalescent individuals up to 8 months post-symptom onset. Cell Rep. Med. 2, 100290 (2021).
Tso, F. Y. et al. Presence of antibody-dependent cellular cytotoxicity (ADCC) against SARS-CoV-2 in COVID-19 plasma. PLoS ONE 16, e0247640 (2021).
McMahan, K. et al. Correlates of protection against SARS-CoV-2 in rhesus macaques. Nature 590, 630–634 (2021).
Mercado, N. B. et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 586, 583–588 (2020).
Gorman, M. J. et al. Fab and Fc contribute to maximal protection against SARS-CoV-2 following NVX-CoV2373 subunit vaccine with Matrix-M vaccination. Cell Rep. Med. 2, 100405 (2021).
Begin, P. et al. Convalescent plasma for hospitalized patients with COVID-19: an open-label, randomized controlled trial. Nat. Med. 27, 2012–2024 (2021).
Winkler, E. S. et al. Human neutralizing antibodies against SARS-CoV-2 require intact Fc effector functions for optimal therapeutic protection. Cell 184, 1804–1820 (2021).
Schafer, A. et al. Antibody potency, effector function, and combinations in protection and therapy for SARS-CoV-2 infection in vivo. J. Exp. Med. 218, e20201993 (2021).
Yamin, R. et al. Fc-engineered antibody therapeutics with improved anti-SARS-CoV-2 efficacy. Nature 599, 465–470 (2021).
Kaplonek, P. et al. mRNA-1273 and BNT162b2 COVID-19 vaccines elicit antibodies with differences in Fc-mediated effector functions. Sci Transl Med https://doi.org/10.1126/scitranslmed.abm2311 (2022).
Mateus, J. et al. Selective and cross-reactive SARS-CoV-2 T cell epitopes in unexposed humans. Science 370, 89–94 (2020).
Grifoni, A. et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 181, 1489–1501 (2020). This study described the SARS-CoV-2-specific CD4+ and CD8+ T cell responses in patients with COVID-19 and in unexposed individuals.
Dan, J. M. et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 371, eabf4063 (2021). This study assessed the durability of immune responses against SARS-CoV-2 variants in individuals who received the mRNA vaccine.
Goel, R. R. et al. mRNA vaccines induce durable immune memory to SARS-CoV-2 and variants of concern. Science 374, eabm0829 (2021).
Geers, D. et al. SARS-CoV-2 variants of concern partially escape humoral but not T-cell responses in COVID-19 convalescent donors and vaccinees. Sci. Immunol. 6, eabj1750 (2021).
Alter, G. et al. Immunogenicity of Ad26.COV2.S vaccine against SARS-CoV-2 variants in humans. Nature 596, 268–272 (2021).
Moore, C. B. et al. Evidence of HIV-1 adaptation to HLA-restricted immune responses at a population level. Science 296, 1439–1443 (2002).
Hertz, T. et al. HLA targeting efficiency correlates with human T-cell response magnitude and with mortality from influenza A infection. Proc. Natl Acad. Sci. USA 110, 13492–13497 (2013).
Gartland, A. J. et al. Analysis of HLA A*02 association with vaccine efficacy in the RV144 HIV-1 vaccine trial. J. Virol. 88, 8242–8255 (2014).
Henry Dunand, C. J. et al. Both neutralizing and non-neutralizing human H7N9 influenza vaccine-induced monoclonal antibodies confer protection. Cell Host Microbe 19, 800–813 (2016).
Tan, G. S. et al. Broadly-reactive neutralizing and non-neutralizing antibodies directed against the H7 influenza virus hemagglutinin reveal divergent mechanisms of protection. PLoS Pathog. 12, e1005578 (2016).
Jegaskanda, S., Weinfurter, J. T., Friedrich, T. C. & Kent, S. J. Antibody-dependent cellular cytotoxicity is associated with control of pandemic H1N1 influenza virus infection of macaques. J. Virol. 87, 5512–5522 (2013).
Plante, J. A. et al. Spike mutation D614G alters SARS-CoV-2 fitness. Nature 592, 116–121 (2020).
Knezevic, I. et al. WHO international standard for evaluation of the antibody response to COVID-19 vaccines: call for urgent action by the scientific community. Lancet Microbe 3, e235–e240 (2021).
Corbett, K. S. et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 586, 567–571 (2020).
Vogel, A. B. et al. BNT162b vaccines protect rhesus macaques from SARS-CoV-2. Nature 592, 283–289 (2021).
van Doremalen, N. et al. ChAdOx1 nCoV-19 vaccine prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature 586, 578–582 (2020).
Guebre-Xabier, M. et al. NVX-CoV2373 vaccine protects cynomolgus macaque upper and lower airways against SARS-CoV-2 challenge. Vaccine 38, 7892–7896 (2020).
Tian, J. H. et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373 immunogenicity in baboons and protection in mice. Nat. Commun. 12, 372 (2021).
Pruijssers, A. J. et al. Remdesivir inhibits SARS-CoV-2 in human lung cells and chimeric SARS-CoV expressing the SARS-CoV-2 RNA polymerase in mice. Cell Rep. 32, 107940 (2020).
Hansen, J. et al. Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail. Science 369, 1010–1014 (2020).
Boras, B. et al. Preclinical characterization of an intravenous coronavirus 3CL protease inhibitor for the potential treatment of COVID19. Nat. Commun. 12, 6055 (2021).
He, X. et al. Low-dose Ad26.COV2.S protection against SARS-CoV-2 challenge in rhesus macaques. Cell 184, 3467–3473 (2021).
Hirose, R. et al. A cytopathic effect-based tissue culture method for HCoV-OC43 titration using TMPRSS2-expressing VeroE6 cells. mSphere 6, e00159–21 (2021).
Munoz-Fontela, C. et al. Animal models for COVID-19. Nature 586, 509–515 (2020).
Ozono, S. et al. SARS-CoV-2 D614G spike mutation increases entry efficiency with enhanced ACE2-binding affinity. Nat. Commun. 12, 848 (2021).
McCray, P. B. Jr et al. Lethal infection of K18-hACE2 mice infected with severe acute respiratory syndrome coronavirus. J. Virol. 81, 813–821 (2007).
Sun, J. et al. Generation of a broadly useful model for COVID-19 pathogenesis, vaccination, and treatment. Cell 182, 734–743 (2020).
Sun, C. P. et al. Rapid generation of mouse model for emerging infectious disease with the case of severe COVID-19. PLoS Pathog. 17, e1009758 (2021).
Oladunni, F. S. et al. Lethality of SARS-CoV-2 infection in K18 human angiotensin-converting enzyme 2 transgenic mice. Nat. Commun. 11, 6122 (2020).
Winkler, E. S. et al. SARS-CoV-2 infection of human ACE2-transgenic mice causes severe lung inflammation and impaired function. Nat. Immunol. 21, 1327–1335 (2020).
Golden, J. W. et al. Human angiotensin-converting enzyme 2 transgenic mice infected with SARS-CoV-2 develop severe and fatal respiratory disease. JCI Insight 5, e142032 (2020).
Moreau, G. B. et al. Evaluation of K18-hACE2 mice as a model of SARS-CoV-2 infection. Am. J. Trop. Med. Hyg. 103, 1215–1219 (2020).
Zheng, J. et al. COVID-19 treatments and pathogenesis including anosmia in K18-hACE2 mice. Nature 589, 603–607 (2021).
Vanderheiden, A. et al. CCR2 signaling restricts SARS-CoV-2 infection. mBio 12, e0274921 (2021).
Muruato, A. et al. Mouse-adapted SARS-CoV-2 protects animals from lethal SARS-CoV challenge. PLoS Biol. 19, e3001284 (2021).
Rathnasinghe, R. et al. The N501Y mutation in SARS-CoV-2 spike leads to morbidity in obese and aged mice and is neutralized by convalescent and post-vaccination human sera. Preprint at medRxiv https://doi.org/10.1101/2021.01.19.21249592 (2021).
Ying, B. et al. Protective activity of mRNA vaccines against ancestral and variant SARS-CoV-2 strains. Sci. Transl. Med. 14, eabm3302 (2021).
Imai, M. et al. Syrian hamsters as a small animal model for SARS-CoV-2 infection and countermeasure development. Proc. Natl Acad. Sci. USA 117, 16587–16595 (2020).
Bricker, T. L. et al. A single intranasal or intramuscular immunization with chimpanzee adenovirus-vectored SARS-CoV-2 vaccine protects against pneumonia in hamsters. Cell Rep. 36, 109400 (2021).
Sia, S. F. et al. Pathogenesis and transmission of SARS-CoV-2 in golden hamsters. Nature 583, 834–838 (2020).
Imai, M. et al. Characterization of a new SARS-CoV-2 variant that emerged in Brazil. Proc. Natl Acad. Sci. USA 118, e2106535118 (2021).
Schmitz, A. J. et al. A vaccine-induced public antibody protects against SARS-CoV-2 and emerging variants. Immunity 54, 2159–2166 (2021).
Plante, J. A. et al. Spike mutation D614G alters SARS-CoV-2 fitness. Nature 592, 116–121 (2021).
Halfmann, P. J. et al. SARS-CoV-2 Omicron virus causes attenuated disease in mice and hamsters. Nature 603, 687–692 (2022).
van Doremalen, N. et al. Intranasal ChAdOx1 nCoV-19/AZD1222 vaccination reduces viral shedding after SARS-CoV-2 D614G challenge in preclinical models. Sci. Transl. Med. 13, eabh0755 (2021).
Fischer, R. J. et al. ChAdOx1 nCoV-19 (AZD1222) protects Syrian hamsters against SARS-CoV-2 B.1.351 and B.1.1.7. Nat. Commun. 12, 5868 (2021).
Francica, J. R. et al. Protective antibodies elicited by SARS-CoV-2 spike protein vaccination are boosted in the lung after challenge in nonhuman primates. Sci. Transl. Med. 13, eabi4547 (2021).
Corbett, K. S. et al. Immune correlates of protection by mRNA-1273 vaccine against SARS-CoV-2 in nonhuman primates. Science 373, eabj0299 (2021).
Corbett, K. S. et al. mRNA-1273 protects against SARS-CoV-2 beta infection in nonhuman primates. Nat. Immunol. 22, 1306–1315 (2021).
Gagne, M. et al. Protection from SARS-CoV-2 Delta one year after mRNA-1273 vaccination in rhesus macaques coincides with anamnestic antibody response in the lung. Cell 185, 113–130 (2022).
Bewley, K. R. et al. Immunological and pathological outcomes of SARS-CoV-2 challenge following formalin-inactivated vaccine in ferrets and rhesus macaques. Sci. Adv. 7, eabg7996 (2021).
Zhou, J. et al. Immunogenicity, safety, and protective efficacy of an inactivated SARS-associated coronavirus vaccine in rhesus monkeys. Vaccine 23, 3202–3209 (2005).
Cameroni, E. et al. Broadly neutralizing antibodies overcome SARS-CoV-2 Omicron antigenic shift. Nature 602, 664–670 (2022).
Pajon, R. et al. SARS-CoV-2 Omicron variant neutralization after mRNA-1273 booster vaccination. N. Engl. J. Med. 386, 1088–1091 (2022).
Edara, V.-V. et al. mRNA-1273 and BNT162b2 mRNA vaccines have reduced neutralizing activity against the SARS-CoV-2 Omicron variant. Cell Rep. Med. 3, 100529 (2022).
Liu, L. et al. Striking antibody evasion manifested by the Omicron variant of SARS-CoV-2. Nature 602, 676–681 (2022).
Tarke, A. et al. SARS-CoV-2 vaccination induces immunological T cell memory able to cross-recognize variants from Alpha to Omicron. Cell 185, 847–859 (2022).
Valanparambil, R. M. et al. Antibody response to SARS-CoV-2 mRNA vaccine in lung cancer patients: reactivity to vaccine antigen and variants of concern. Preprint at medRxiv https://doi.org/10.1101/2022.01.03.22268599 (2022).
Bartsch, Y. et al. Omicron variant Spike-specific antibody binding and Fc activity are preserved in recipients of mRNA or inactivated COVID-19 vaccines. Sci Transl Med https://doi.org/10.1126/scitranslmed.abn9243 (2022).
Carreno, J. M. et al. Activity of convalescent and vaccine serum against SARS-CoV-2 Omicron. Nature 602, 682–688 (2022).
Acknowledgements
A.A. is funded by HHSN272201700060C; G.A. is funded by the Ragon Institute of MGH, MIT and Harvard, the Massachusetts Consortium on Pathogen Readiness (MassCPR), the National Institutes of Health (NIH) (3R37AI080289-11S1, R01AI146785, U19AI42790-01, U19AI135995-02, U19AI42790-01, 1U01CA260476-01), the Gates Foundation Global Health Vaccine Accelerator Platform funding (OPP1146996 and INV-001650) and the Musk Foundation; D.C.M. and X.S. are supported in part by the National Institute of Allergy and Infectious Diseases (NIAID)/NIH Collaborative Influenza Vaccine Innovation Centers (CIVIC) under contract 75N93019C00050, Duke University; A.E., A. Gordon, J.M.C., F.K. and V.S. are funded in part by NIH/NIAID CIVIC under contract 75N93019C00051, Icahn School of Medicine at Mount Sinai; G.A., B.L., V.A.M., S.S.-C. and P.G.T. are funded in part by the NIH/NIAID Center for Influenza Vaccine Research for High-Risk Populations (CIVR-HRP) CIVIC under contract 75N93019C0052, University of Georgia; M.S.D., V.S., L.B.T., H.v.B., L.A.V., R.A.M.F., A.G.-S., M.B.F., R.M.J., J.P.L., S.M.W., B.L.H., D.D.H., Y.H., Y.L., M.S.N., P.W., M.W., D.J.S., A.N., E.B.L., S.L.J., S. Türeli, S.M.W., S.A.T., J.M.C., F.K., B.M.W., B.R. and B.Y. are supported by the NIH/NIAID Centers of Excellence for Influenza Research and Response (CEIRR) under contract 75N93021C00014, Icahn School of Medicine at Mount Sinai; T.P.F., G.F., J.F., T.J., L.K., B.L., V.A.M., S.S.-C., A.S., S.T., P.G.T., E.K., L.C.-L., T.H., S. Sacharen and R.J.W. are funded in part by the NIH\NIAID CEIRR under contract 75N9302100016, St Jude Children’s Research Hospital; R.R.A., V.-V.E., M.E.D.-G. and M.S.S. are funded in part by the NIH/NIAID CEIRR under contract 75N93021C00017, Emory University; A.E., T.P.F., J.F., T.J., L.K., B.L., V.A.M., S.S.-C., S.T. S.P.J.W., Z.L., L.-M.B., P.W.R., M.C.P., S. Stumpf, A.G., A.S. and R.J.W. are funded in part by the NIH/NIAID Centers of Excellence for Influenza Research and Surveillance (CEIRS) under contract HHSN272201400006C, St Jude Children’s Research Hospital; A.P. is supported in part by the NIH/NIAID CEIRS under contract HHS N2772201400007C, Johns Hopkins University; A.E., M.B.F., R.M.J., A.G.-S., J.P.L., S.M.W., P.J.H., Y.K., J.M.C., F.K., M. Schotsaert and V.S. are funded in part by the NIH/NIAID CEIRS under contract HHSN272201400008C, Icahn School of Medicine at Mount Sinai; R.R.A. is funded by 3R01AI148378-01S1; J.D.B. is supported in part by R01AI141707 and is an Investigator of the Howard Hughes Medical Institute; A.C.M.B. is funded by U01AI151810; E.A.B. is supported by an Intramural NIH Research Program project ZIA AI005156-02; D.L.B. is supported by NIH/NIAID under contract 75N93019D0025 to CAMRIS International; E.G. and V.J.M. are supported by the Intramural Research Program of the NIAID, NIH; B.L.D. and M.R. are supported by a cooperative agreement between The Henry M. Jackson Foundation for the Advancement of Military Medicine and the US Department of the Army (W81XWH-18-2-0040); M.S.D. is supported in part by R01AI157155; M.D. is supported by 1R01AI143639; N.A.D.-R., D.C.D., A.B.M., N.J.S., R.A.K., S.O., S.D.S. and R.A.S. are supported by the Intramural Research Program of the Vaccine Research Center, NIAID and NIH; C.D., B.A.M. and T.C.J. are funded in part by the German Ministry of research under project codes DZIF, MolTrax and PREPARED; A.E. is supported by NIAID grants U01AI141990 and 1U01AI150747; V.-V.E., M.E.D.-G. and M.S.S. are supported in part by the NIH\NIAID (P51OD011132 and 3U19AI057266-17S1), Emory Executive Vice President for Health Affairs Synergy Fund award, and Woodruff Health Sciences Center 2020 COVID-19 CURE Award, Agod; L.J. and M. Sedova are supported by HHSN272201700060C; A. Gordon is supported by R01AI20997; M.R.H. is supported by NIAID contract HHSN272201800013C; M.P.G.K. is supported by VEO, versatile emerging infectious disease observatory: forecasting, nowcasting and tracking in a changing world. VEO has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement no. 874735, COVID-19 vaccination in kidney patients (RECOVAC), ZonMw 10430072010002 monitoring the evolution, spread and transmission of SARS-CoV-2 through whole-genome sequencing to enable fast genotype to phenotype prediction’ funded by ZonMw (project 10150062010005, APW 202645827); to support the structured evaluation of SARS-CoV-2 evolution funded by the WHO (APW 202605269); to gain in-depth understanding of the evolution, spread and transmission of SARS-CoV-2 during the coming phase of the pandemic funded by the WHO. B.K., J.T. and H.Y. are funded by the Los Alamos National Laboratory and the Gates Foundation OPP1169339; R.A.K. is funded by Intramural NIH funding; M.J.M. is supported by UM1AI068618; V.D.M. is supported by R01AI153602; S.P. is supported by P01AI060699 and R01AI129269; P.-Y.S. is supported by NIH grants AI134907 and awards from the Sealy Smith Foundation, the Kleberg Foundation, the John S. Dunn Foundation, the Amon G. Carter Foundation, the Gillson Longenbaugh Foundation and the Summerfield Robert Foundation; V.S. is supported by the NIH/NCI: Serological Sciences Network (SeroNet) 75N91019D00024; P.G.T. is supported by U01AI144616, U01AI150747, R01AI136514, R01AI154470; Z.S.W., R.H.S., A.M.N. and M. Shukla are supported by NIH, NIAID Contract 75N93019C00076; W.W. and C.D.W. are supported by US Food and Drug Administration institutional research funds; and S.C.W. is supported by NIH grant R24 AI120942 and by the Sealy and Smith Foundation. The findings and conclusions in this Review are those of the authors and do not necessarily represent the official position of the US Centers for Disease Control and Prevention. Use of trade names is for identification only and does not imply endorsement by the US Centers for Disease Control and Prevention or the US Department of Health and Human Services.
Author information
Authors and Affiliations
Contributions
M.M.D., E.G., M.B.F., F.K., A.G., N.J.S., A.S., P.G.T., D.J.S., M.S.D., D.J.P. and M.S.S. wrote the initial draft. A.A., G.A., R.R.A., R.S.B., D.H.B., J.D.B., L.B., G.B., A.C.M.B., E.A.B., D.L.B., T.L.B., L.-M.B., W.J.B., J.M.C., L.C.-L., T.L.D., M.E.D.-G., B.L.D., H.D., M.D., N.A.D.-R., D.C.D., C.D., V.-V.E., A.E., T.P.F., G.F., W.C.F., R.A.M.F., J.F., A.G.-S., A. Godzik, A.S.G.-R., A. Gordon., B.L.H., P.J.H., D.D.H., M.R.H., Y.H., S.L.J., L.J., T.J., R.M.J., T.C.J., A.J., Y.K., L.K., M.P.G.K., B.K., E.K., R.A.K., E.B.L., J.E.L., M.J.L., Z.L., B.L., J.P.L., Y.L., A.B.M., M.J.M., V.A.M., V.D.M., D.C.M., B.M., V.J.M., J.E.M., M.S.N., A.N., A.M.N., S.O., A.P., S.P., M.C.P., B.R., M.R., P.W.R., S. Sacharen, R.H.S., S.D.S., M. Schotsaert, S.S.-C., R.A.S., M. Sedova, A.S., R.S.S., X.S., P.-Y.S., M. Shukla., V.S., S. Stumpf, L.B.T., J.T., S. Trifkovic, S. Tureli, S.A.T., M.A.V., H.v.B., L.A.V., L.R.V., Z.S.W., L.W., M.W., P.W., W.W., S.C.W., R.J.W., C.D.W., D.E.W., S.M.W., S.P.J.W., B.M.W., S.H.W., X.X., B.Y., H.Y., B.Z., T.H., M.M.D., E.G., M.B.F., F.K., A.G., N.J.S., P.G.T., D.J.S., M.S.D., D.J.P. and M.S.S. provided input, concepts, editing and revisions.
Corresponding authors
Ethics declarations
Competing interests
D.H.B. is listed as a co-inventor on provisional vaccine patents (63/121,482; 63/133,969; 63/135,182). J.D.B. consults for Moderna and Flagship Labs 77 on topics related to viral evolution, and is an inventor on Fred Hutch licensed patents related to viral deep mutational scanning (62/692,398; PCT/US2019/039952; 17/281,540; 19824586.2; 62/935,954; 17/097,853; 62/812,804; PCT/US2020/020429). The Boon laboratory has received unrelated funding support in sponsored research agreements from AI Therapeutics, GreenLight Biosciences, and Nano targeting & Therapy Biopharma. The Boon laboratory has received funding support from AbbVie, for the commercial development of SARS-CoV-2 monoclonal antibodies. A.C.M.B. was a recipient of a licensing agreement with Abbvie for the commercial development of SARS-CoV-2 monoclonal antibodies. M.S.D. is a consultant for Inbios, Vir Biotechnology, Senda Biosciences and Carnival Corporation, and on the scientific advisory boards of Moderna and Immunome. The Diamond laboratory has received unrelated funding support in sponsored research agreements from Vir Biotechnology, Moderna and Emergent BioSolutions. The Ellebedy laboratory received funding under sponsored research agreements that are unrelated to the data presented in the current study from Emergent BioSolutions and from AbbVie. A.E. has received consulting fees from InBios International, Fimbrion Therapeutics, Mubadala Investment Company and Goldman Sachs and is the founder of ImmuneBio Consulting. M.B.F. has funding from Novavax, which is outside the scope of this research. They had no role in the funded research from the SAVES consortium. The Garcia-Sastre laboratory has received research support from Pfizer, Senhwa Biosciences, Kenall Manufacturing, Avimex, Johnson & Johnson, Dynavax, 7Hills Pharma, Pharmamar, ImmunityBio, Accurius, Nanocomposix, Hexamer, N-fold, Model Medicines and Merck, outside of the reported work. A.G.-S. has consulting agreements for the following companies involving cash and/or stock: Vivaldi Biosciences, Contrafect, 7Hills Pharma, Avimex, Vaxalto, Pagoda, Accurius, Esperovax, Farmak, Applied Biological Laboratories, Pharmamar and Pfizer, outside of the reported work. A.G.-S. is listed as an inventor on patents and patent applications on the use of antivirals and vaccines for the treatment and prevention of virus infections and cancer, owned by the Icahn School of Medicine at Mount Sinai, New York, outside of the reported work (5,820,871; 5,854,037; 6,001,634; 6,146,642; 6,451,323; 6,468,544; 6,544,785; 6,573,079; 6,635,416; 6,649,372; 6,669,943; 6,740,519; 6,852,522; 6,866,853; 6,884,414; 6,887,699; 7,060,430; 7,384,774; 7,442,379; 7,494, 808; 7,588,768; 7,833,774; 8,012,490; 8,057,803; 8,124,101; 8,137,676; 8,591,881; 8,629,283; 8,673,314; 8,709,442; 8,709,730; 8,765,139; 8,828,406; 8,999,352; 9,051,359; 9,096,585; 9,175,069; 9,217,136; 9,217,157; 9,238,851; 9,352,033; 9,371,366; 9,387,240; 9,387,242; . 9,549,975; 9,701,723; 9,708,373; 9,849,172; 9,908,930; 9,968,670; 10,035,984; .10,098,945; 10,131,695; 10,137,189; 10,179,806; 10,251,922; 10,308,913; 10,543,268; 10,544,207; 10,583,188; 10,736,956; 11,254,733; 11,266,734). A. Gordon serves on a scientific advisory board for Janssen, Erasmus MC has a proprietary IP on MERS. B.K. is part of provisional patent applications for strategies for next-generation SARS-CoV-2 vaccines that address diversity (63/256,848, 17/234,590; S133955). The Icahn School of Medicine at Mount Sinai has filed patent applications relating to SARS-CoV-2 serological assays that list V.S. as a co-inventor (62/994,252; 63/018,457; 63/020,503; 63/024,436) and NDV-based SARS-CoV-2 vaccines that list F.K. as a co-inventor (63/251,020). Mount Sinai has spun out a company, Kantaro, to market serological tests for SARS-CoV-2. F.K. has consulted for Merck and Pfizer (before 2020), and is currently consulting for Pfizer, Seqirus, 3rd Rock Ventures and Avimex. The Krammer laboratory is also collaborating with Pfizer on animal models of SARS-CoV-2. V.D.M. has filed a patent on the reverse genetic system and reporter SARS-CoV-2 (63/000,713; 63/041,667). D.C.M. receives funding from Moderna to perform blinded assessments of vaccine-elicited neutralizing antibody responses in clinical studies of their COVID-19 vaccines. A.S. is a consultant for Gritstone Bio, Flow Pharma, Arcturus Therapeutics, ImmunoScape, CellCarta, Avalia, Moderna, Fortress and Repertoire. P.-Y.S. laboratory has received funding support in sponsored research agreements from GSK, Pfizer, Gilead, Novartis, Merck, IGM Biosciences and Atea Pharmaceuticals. P.-Y.S. is a member of the scientific advisory boards of AbImmune and is Founder of FlaviTech. M.S.S. serves on the advisory board for Moderna and Ocugen. P.G.T. serves on the scientific advisory board for Immunoscape and Cytoagents and has consulted for Johnson and Johnson. P.G.T. has received travel support and honoraria from Illumina and 10x Genomics. P.G.T. has patents related to viral infection treatment and T cell receptor biology (11,083,725; 2021/0299118; US-2019-0040381; 17/616,279; WO2021/214637). S.P.J.W. has filed a patent with Washington University for VSV- SARS-CoV-2 mutants to characterize antibody panels (PCT/US2021/027275). S.P.J.W. has received unrelated funding support in sponsored research agreements with Vir Biotechnology, AbbVie, and sAB therapeutics.
Peer review
Peer review information
Nature thanks Ravindra Gupta, Volker Thiel and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
DeGrace, M.M., Ghedin, E., Frieman, M.B. et al. Defining the risk of SARS-CoV-2 variants on immune protection. Nature 605, 640–652 (2022). https://doi.org/10.1038/s41586-022-04690-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-04690-5
This article is cited by
-
The spike-specific TCRβ repertoire shows distinct features in unvaccinated or vaccinated patients with SARS-CoV-2 infection
Journal of Translational Medicine (2024)
-
Vaccine design via antigen reorientation
Nature Chemical Biology (2024)
-
Mucosal vaccine-induced cross-reactive CD8+ T cells protect against SARS-CoV-2 XBB.1.5 respiratory tract infection
Nature Immunology (2024)
-
Bioengineered amyloid peptide for rapid screening of inhibitors against main protease of SARS-CoV-2
Nature Communications (2024)
-
Cepharanthine analogs mining and genomes of Stephania accelerate anti-coronavirus drug discovery
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.