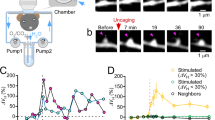
Abstract
Synaptic transmission involves cell-to-cell communication at the synaptic junction between two neurons, and chemical and electrical forms of this process have been extensively studied. In the brain, excitatory glutamatergic synapses are often made on dendritic spines that enlarge during learning1,2,3,4,5. As dendritic spines and the presynaptic terminals are tightly connected with the synaptic cleft6, the enlargement may have mechanical effects on presynaptic functions7. Here we show that fine and transient pushing of the presynaptic boutons with a glass pipette markedly promotes both the evoked release of glutamate and the assembly of SNARE (soluble N-ethylmaleimide-sensitive factor attachment protein receptor) proteins8,9,10,11,12—as measured by Förster resonance transfer (FRET) and fluorescence lifetime imaging—in rat slice culture preparations13. Both of these effects persisted for more than 20 minutes. The increased presynaptic FRET was independent of cytosolic calcium (Ca2+), but dependent on the assembly of SNARE proteins and actin polymerization in the boutons. Notably, a low hypertonic solution of sucrose (20 mM) had facilitatory effects on both the FRET and the evoked release without inducing spontaneous release, in striking contrast with a high hypertonic sucrose solution (300 mM), which induced exocytosis by itself14. Finally, spine enlargement induced by two-photon glutamate uncaging enhanced the evoked release and the FRET only when the spines pushed the boutons by their elongation. Thus, we have identified a mechanosensory and transduction mechanism15 in the presynaptic boutons, in which the evoked release of glutamate is enhanced for more than 20 min.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available from the corresponding author upon reasonable request. Source data are provided with this paper.
Code availability
The MATLAB scripts for the FRET/FLIM analysis together with other codes used in this study are available at https://github.com/sw-swatanabe/ucar-et-al-2021.
References
Matsuzaki, M., Honkura, N., Ellis-Davies, G. C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Harvey, C. D., Yasuda, R., Zhong, H. & Svoboda, K. The spread of Ras activity triggered by activation of a single dendritic spine. Science 321, 136–140 (2008).
Herring, B. E. & Nicoll, R. A. Long-term potentiation: from CaMKII to AMPA receptor trafficking. Annu. Rev. Physiol. 78, 351–365 (2016).
Noguchi, J. et al. Bidirectional in vivo structural dendritic spine plasticity revealed by two-photon glutamate uncaging in the mouse neocortex. Sci. Rep. 9, 13922 (2019).
Kasai, H., Ziv, N. E., Okazaki, H., Yagishita, S. & Toyoizumi, T. Spine dynamics in the brain, mental disorders and artificial neural networks. Nat. Rev. Neurosci. 22, 407–422 (2021).
Missler, M., Sudhof, T. C. & Biederer, T. Synaptic cell adhesion. Cold Spring Harb. Perspect. Biol. 4, a005694 (2012).
Kasai, H., Fukuda, M., Watanabe, S., Hayashi-Takagi, A. & Noguchi, J. Structural dynamics of dendritic spines in memory and cognition. Trends Neurosci. 33, 121–129 (2010).
Sudhof, T. C. & Rothman, J. E. Membrane fusion: grappling with SNARE and SM proteins. Science 323, 474–477 (2009).
Jahn, R. & Scheller, R. H. SNAREs–engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 7, 631–643 (2006).
Augustine, G. J. How does calcium trigger neurotransmitter release? Curr. Opin. Neurobiol. 11, 320–326 (2001).
Tucker, W. C., Weber, T. & Chapman, E. R. Reconstitution of Ca2+-regulated membrane fusion by synaptotagmin and SNAREs. Science 304, 435–438 (2004).
Kasai, H., Takahashi, N. & Tokumaru, H. Distinct initial SNARE configurations underlying the diversity of exocytosis. Physiol. Rev. 92, 1915–1964 (2012).
Takahashi, N. et al. Two-photon fluorescence lifetime imaging of primed SNARE complexes in presynaptic terminals and β cells. Nat. Commun. 6, 8531 (2015).
Fatt, P. & Katz, B. Spontaneous subthreshold activity at motor nerve endings. J. Physiol. 117, 109–128 (1952).
Hannezo, E. & Heisenberg, C. P. Mechanochemical feedback loops in development and disease. Cell 178, 12–25 (2019).
Imig, C. et al. The morphological and molecular nature of synaptic vesicle priming at presynaptic active zones. Neuron 84, 416–431 (2014).
Rosenmund, C. & Stevens, C. F. Definition of the readily releasable pool of vesicles at hippocampal synapses. Neuron 16, 1197–1207 (1996).
Marvin, J. S. et al. An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat. Methods 10, 162–170 (2013).
Klapoetke, N. C. et al. Independent optical excitation of distinct neural populations. Nat. Methods 11, 338–346 (2014).
Murakoshi, H., Shibata, A. C., Nakahata, Y. & Nabekura, J. A dark green fluorescent protein as an acceptor for measurement of Förster resonance energy transfer. Sci. Rep. 5, 15334 (2015).
Delmas, P. & Coste, B. Mechano-gated ion channels in sensory systems. Cell 155, 278–284 (2013).
Tapley, P., Lamballe, F. & Barbacid, M. K252a is a selective inhibitor of the tyrosine protein kinase activity of the trk family of oncogenes and neurotrophin receptors. Oncogene 7, 371–381 (1992).
Doussau, F. & Augustine, G. J. The actin cytoskeleton and neurotransmitter release: an overview. Biochimie 82, 353–363 (2000).
Cingolani, L. A. & Goda, Y. Actin in action: the interplay between the actin cytoskeleton and synaptic efficacy. Nat. Rev. Neurosci. 9, 344–356 (2008).
Tanaka, J. et al. Protein synthesis and neurotrophin-dependent structural plasticity of single dendritic spines. Science 319, 1683–1687 (2008).
Iino, M. Tension responses of chemically skinned fibre bundles of the guinea-pig taenia caeci under varied ionic environments. J. Physiol. 320, 449–467 (1981).
Harris, K. M. & Stevens, J. K. Dendritic spines of CA 1 pyramidal cells in the rat hippocampus: serial electron microscopy with reference to their biophysical characteristics. J. Neurosci. 9, 2982–2997 (1989).
de Vivo, L. et al. Ultrastructural evidence for synaptic scaling across the wake/sleep cycle. Science 355, 507–510 (2017).
Watanabe, S. et al. Ultrafast endocytosis at Caenorhabditis elegans neuromuscular junctions. Elife 2, e00723 (2013).
Honkura, N., Matsuzaki, M., Noguchi, J., Ellis-Davies, G. C. & Kasai, H. The subspine organization of actin fibers regulates the structure and plasticity of dendritic spines. Neuron 57, 719–729 (2008).
Chang, J. Y. et al. CaMKII autophosphorylation is necessary for optimal integration of Ca2+ signals during LTP induction, but not maintenance. Neuron 94, 800–808 (2017).
Kishimoto, T. et al. Vacuolar sequential exocytosis of large dense-core vesicles in adrenal medulla. EMBO J. 25, 673–682 (2006).
Ninomiya, Y., Kishimoto, T., Miyashita, Y. & Kasai, H. Ca2+-dependent exocytotic pathways in Chinese hamster ovary fibroblasts revealed by a caged-Ca2+ compound. J. Biol. Chem. 271, 17751–17754 (1996).
Iino, Y. et al. Dopamine D2 receptors in discrimination learning and spine enlargement. Nature 579, 555–560 (2020).
Shibata, A. C. E. et al. Photoactivatable CaMKII induces synaptic plasticity in single synapses. Nat. Commun. 12, 751 (2021).
Dana, H. et al. High-performance calcium sensors for imaging activity in neuronal populations and microcompartments. Nat. Methods 16, 649–657 (2019).
Grieger, J. C., Choi, V. W. & Samulski, R. J. Production and characterization of adeno-associated viral vectors. Nat. Protoc. 1, 1412–1428 (2006).
Leung, R. W., Yeh, S. C. & Fang, Q. Effects of incomplete decay in fluorescence lifetime estimation. Biomed. Opt. Express 2, 2517–2531 (2011).
Drobizhev, M., Makarov, N. S., Tillo, S. E., Hughes, T. E. & Rebane, A. Two-photon absorption properties of fluorescent proteins. Nat. Methods 8, 393–399 (2011).
Kasai, H. et al. A new quantitative (TEPIQ) analysis for diameters of exocytic vesicles and its application to mouse pancreatic islets. J. Physiol 568, 891–903 (2005).
Hayama, T. et al. GABA promotes the competitive selection of dendritic spines by controlling local Ca2+ signaling. Nat. Neurosci. 16, 1409–1416 (2013).
Acknowledgements
We thank H. Murakoshi for providing ShadowG and paCaMKII; M. Iino for discussion; and S. Fujii, C. Fujinami, Y. Sakata, R. Yonemoto, H. Ohno and A. Kurabayashi for their technical assistance. This work was supported by CREST (JPMJCR21E2 and JPMJCR1652 to H.K.) from JST, KAKENHI (JP20H05685 to H.K., JP21K15203 to H.U., JP21K07535 to S.W., JP18K06497 to J.N., JP19J10613 and JP21K20682 to Y.M., JP20K15893 to Y.I., JP21H05176 and JP21H02594 to S.Y., and JP17K08530 and JP20K07280 to N.T.) from JSPS, SRPBS (JP20dm0107120 to H.K.) and Brain/MINDS (JP21dm0207069 to S.Y.) from AMED, the World Premier International Research Center Initiative (WPI) from MEXT, Takeda Science Foundation (to S.Y.) and a grant-in-aid of The Fugaku Trust for Medical Research (to H.K.).
Author information
Authors and Affiliations
Contributions
H.U. and H.K. designed the experiments. H.U., J.N., S.W. and Y.I. performed the slice experiments. S.W. and S.Y. assisted in the programming of MATLAB and LabView. Y.M. and N.T. helped with FRET data analysis. H.U. and H.K. wrote the manuscript, and all authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Edwin Chapman, Ryohei Yasuda and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Suppression of glutamate release by the strong pushing of a presynaptic terminal.
a, Strong pushing of a presynaptic bouton labelled with iGluSnFR (green) by a glass pipette (magenta). White dashed-lines indicate the outline of the boutons. Orange lines represent the axial line of the axon. Pushing was applied over the axial line. Scale bar 0.5 µm. b, Fluorescence profiles along the yellow line (inset) before (black), during (magenta) and after (cyan) the strong pushing of the terminal shown in a. c, d, The Pr measurements before and after the strong pushing of a bouton (c) and their averaged traces (d). Orange bars (LED) indicate optogenetic stimulation (5 ms). e, The mean time courses of Pr (left) and the peak of the averaged traces (right). n = 12 boutons (8 slices, 8 rats). For the baseline fluctuations (Baseline), 3 consecutive values (5 min apart) from all boutons were obtained. For Pr, two-sided Kruskal–Wallis test (H = 22.2, **P = 1.85 × 10−4) followed by Dunn’s multiple comparison test (vs the baseline). 6.5 min, **P = 2.62 × 10−3; 11.5 min, **P = 4.2 × 10−4; 20 min, **P = 1.9 × 10−3. For the averaged trace, (H = 13.9, **P = 7.65 × 10−3) followed by post-hoc Dunn’s multiple comparison test (vs the baseline). 6.5 min, **P = 3.13 × 10−3; 11.5 min, *P = 0.015; 20 min, *P = 0.0194. f, Time courses of ΔFWHM caused by the fine pushing (black) and strong pushing (orange). For fine pushing, the trace is the replicate of Fig. 1g. For strong pushing, n =13 boutons (11 slices, 10 rats). Magenta bars in e and f indicate pushing. For strong pushing two-sided Kruskal–Wallis test (H = 28.6, **P = 6.22 × 10−7) followed by Dunn’s multiple comparison test (vs the baseline). Push (10 s), **P = 4.3×10−5; push (110 s), **P = 4.77 ×10−6. g, ΔFWHMs obtained from the same data as f. Two-tailed Mann−Whitney U test, U = 232, **P = 7.28 × 10−3. Error bars indicate s.e.m.
Extended Data Fig. 2 Pharmacological blockage of increases in FRET induced by pushing.
a, EPSCs recorded from the whole-cell clamped CA1 neurons. Optogenetic stimulations (5 ms, orange bars) were given to CA3 neurons, expressing FRET probes (Control) or FRET probes and the light chain of BoNTE (BoNTE). b, f, FRET intensities (A1%, see Methods) of the boutons that were finely pushed in the presence of BoNTE expression (b), or that were treated with K252a (2 µM, f). Scale bar indicates 0.5 µm. c, Time courses of ΔFWHM during fine pushing from HF side (black), LF side (open circle), in the absence of calcium (EGTA 100 µM, BAPTA-AM 30 µM, blue), in the presence of K252a (orange) or LatA (cyan) from the same data as Fig. 2e. The magenta bar indicates the duration of pushing. d, ΔFWHM of data in c during pushing. For the baseline fluctuations (Baseline), 2 consecutive FWHM values (20 s apart) from all boutons were plotted from the same data as Fig. 2c. Two-sided Kruskal–Wallis test (H = 127, **P = 6.41 × 10−15) followed by Dunn’s multiple comparison test (vs Baseline). HF side, n = 17, **P = 2.32 × 10−10; LF side, n = 10, **P = 2.3 × 10−8; BoNTE, n = 12, **P = 4.7 × 10−10; Ca2+-free, n = 13, **P = 2.8 × 10−10; K252a, n = 8, **P = 7.07 × 10−7; LatA, n = 10, **P = 1.02 × 10−10. Kruskal–Wallis test (H = 0.492, P = 0.992) followed by Dunn’s multiple comparison test (vs HF side). LF side, P = 0.943; BoNTE, P = 0.758; Ca2+-free, P = 0.819; K252a, P = 0.628; LatA, P = 0.563. e, FRET images of a pushed bouton and in a neighbouring bouton. Scale bar 1 µm. Error bars indicate s.e.m. g, h, Replots of the same data shown in e and f using the average lifetime, instead of A1%
Extended Data Fig. 3 The absence of the effect of pushing on the resting calcium concentrations and evoked calcium transients.
a, GCaMP7s fluorescence images of a bouton (green) before and during pushing with a glass pipette (magenta). Scale bar 0.5 µm. b, The mean time course of GCaMP7s or 6s intensities before and during pushing (magenta bar) among 9 boutons. c, Evoked calcium transients measured with GCaMP7s fluorescence from a bouton before, during and after pushing. Orange arrowheads and dashed lines indicate the timing of optogenetic stimulation with a duration of 5 ms. d, The peak values of evoked calcium responses (Exp#1–5) and their average (black). n = 5 boutons (5 slices, 3 rats). Two-sided Kruskal–Wallis test (H = 3.46, P = 0.177) followed by Dunn’s multiple comparison test (vs Before). Pushing, P = 0.752; after, P = 0.095. Error bars indicate s.e.m
Extended Data Fig. 4 Alterations of mEPSC frequencies and evoked EPSC amplitudes by varying concentrations of sucrose.
a, mEPSCs recorded from a whole-cell clamped CA1 neuron. By adding 20, 40, 60 or 150 mM sucrose to ACSF (320 mOsm), we made a series of sucrose solutions with the osmolarities of 340 mM, 360 mM, 380 mM and 470 mM. They were sequentially applied, each followed by 10 min wash periods with ACSF. b, The mean frequencies of mEPSC in the sucrose solutions normalized by the one in ACSF. n = 5 cells (5 slices, 3 rats). Two-sided Kruskal–Wallis test (H = 41.5, P = 2.11 × 10−8) followed by Dunn’s multiple comparison test (vs the baseline). 20 mM, P = 0.656; 40 mM, **P = 1.81 × 10−3; 60 mM, ** P = 7.15 × 10−4; 150 mM, **P = 3.50 × 10−8. c, Shrinkage of a bouton (left) by 20 mM sucrose quantified by the fluorescent profile (right). The profiles were aligned at the right side. n = 9 boutons (6 slices, 3 rats). Two-sided Kruskal–Wallis test (H = 17.3, **P = 1.76 × 10−4) followed by Dunn’s multiple comparison test (vs the baseline). 5 min, **P = 5.09×10−5. d, Evoked EPSCs recorded from CA1 neurons by optogenetic stimulation of the CA3 region (orange bar, 5 ms) in ACSF, isotonic sucrose (10 mM NaCl was replaced with 20 mM sucrose to yield an osmolarity the same as ACSF, 320 mOsm) and hypertonic sucrose (20 mM). We used the EPSCs from samples whose PPRs, with an interstimulus interval of 70 ms, were larger than 1.0 (see Methods). e, The mean time course for evoked EPSC amplitudes. Brown and magenta bars indicate stimulation with isotonic sucrose (n = 6) and hypertonic (n = 7) sucrose, respectively. f, Mean EPSC amplitudes during sucrose stimulations. For the baseline fluctuations (Baseline), 2 consecutive EPSC values (5 min apart) from all boutons were plotted. n = 6 cells (6 slices, 6 rats) for isotonic sucrose, n = 7 cells (7 slices, 7 rats) for hypertonic sucrose. Two-sided Kruskal–Wallis test (H = 16.1, **P = 3.28 × 10−4) followed by Dunn’s multiple comparison test (vs the baseline). Isotonic, P = 0.542, Hypertonic, **P = 6.5 × 10−5. Error bars indicate s.e.m
Extended Data Fig. 5 Effects of the three types of pressure on cell structures.
Fine pushing and the osmotic pressure distort the cellular structures, whereas the hydrostatic pressure does not.
Extended Data Fig. 6 Reduction of the paired-pulse ratio by the low-sucrose solution.
a, b, iGluSnFR fluorescence intensity traces (a) in response to optogenetic (orange bars) paired-pulse stimulation and their averaged traces (b) before, during and after application of the ACSF solution containing 20 mM sucrose. The inter-stimulus interval was 70 ms. c, d, The mean time courses for PPR (a) and ΔPPR (d) for 5 boutons, in which PPR was calculated as the ratio of the peak values of the second and first pulses from the averaged traces (b). Magenta bars indicate sucrose application. Green horizontal bars at the x-axis indicate the time windows for the Pr analysis shown in a. n = 5 boutons (5 slices, 3 rats). Two-tailed Mann−Whitney U test, ACSF−sucrose (c), U = 6, *P = 4.78 × 10−2; ACSF−sucrose (d), U = 2, **P = 7.99 × 10−3. e, ΔPPR during ACSF and sucrose solution. For the baseline fluctuations (Baseline), 2 consecutive PPR values (5 min apart) from all boutons were plotted. n = 4 boutons. Two-tailed Mann−Whitney U test, U = 2, **P = 7.99 × 10−3. Error bars indicate s.e.m
Extended Data Fig. 7 cis-SNARE formation by a high-sucrose solution but not by a low-sucrose solution.
a, The design of the FRET probe for measuring the cis-SNARE assembly. Venus (Vn) and mTurquoise (mTq1) were fused with the C-terminals of the SNAREs, syntaxin-1A and VAMP2. b, mEPSCs induced by the 300 mM sucrose solution (620 mOsm, HSS) recorded from the whole-cell clamped CA1 neuron. c, FRET images of cis-SNARE probe during application of LSS (20 mM sucrose) and after application of HSS (300 mM sucrose). Imaging was not possible during the application of 300 mM sucrose solution, because it caused an increase in the refractive index. Scale bar 0.5 µm. d, The mean time course of ΔFRET, representing cis-SNARE formation (a). Magenta and purple bars indicate 20 mM and 300 mM sucrose solutions. b1–b4 indicate time points when the images in c were obtained. n = 11 boutons (4 slices, 3 rats) for both 20 mM sucrose and 300 mM sucrose. e, ΔFRET of the cis-SNARE probe by the 20 mM and 300 mM sucrose solutions from the same dataset analysed in d. For the baseline fluctuations (Baseline), 3 consecutive FRET values (5 min apart) from all boutons were plotted. Two-sided Kruskal–Wallis test (H = 48.27, **P = 8.27 × 10−10) followed by Dunn’s multiple comparison test (vs Baseline). 20 mM sucrose, P = 0.394; 300 mM sucrose, **P = 1.33 × 10−7. Error bars indicate s.e.m
Extended Data Fig. 8 Identification of synaptic contacts.
a–c, A bouton-spine pair which showed a precise horizontal alignment and a substantial overlap. The bouton and spine were labelled with iGluSnFR and Alexa594, respectively. The X-axis was redefined according to the bouton-spine axis, and fluorescent profiles along the X-axis of XY- and XZ projections were plotted (Methods). The fluorescent profiles were normalized at their peaks. The crossing of the two profiles defines Fz and Fx, which were 0.93 and 0.9 in this example, and met the criteria of Fz > 0.8 and Fx > 0.6. d–f, An example of bouton-spine pairs which did not fulfil the criteria (Fz = 0.43 and Fx = 0.6). g, The bouton and spines were also labelled with mTq2-syntaxin-1A and Alexa594, which fulfilled the criteria with Fz = 0.95 and Fx = 0.7 in this example. Horizontal scale bars indicate 1 µm, vertical scale bars indicate 2 µm. h, Detection of functional connectivity in a bouton-spine pair by optogenetic presynaptic stimulation with CsChrimsonR and Ca2+ responses (Cal520) in postsynaptic spines. i, The Ca2+ responses were induced in 3 out of 10 stimulations in the pair shown in h. LED pulses of 5 ms were delivered every 15 s.
Extended Data Fig. 9 ΔFWHM and ΔFRET of the boutons during spine enlargement.
a, The values of FWHM of the bouton at 6 min after the onset of STDP from the data shown in Fig. 4e. n = 14 pushing spines (14 slices, 11 rats) and n = 9 non-pushing spines (9 slices, 8 rats). Two-sided Wilcoxon signed-rank test vs 0 µm, W = 4, **P = 2.58×10−3 and W = 41, *P = 3.3×10−2). b, Correlation between ΔFWHM of the bouton and ΔLength of the spine at 6 min after STDP induction from the data shown in a. Red and black circles indicate pushing and non-pushing spines, respectively. Spearman’s correlation with two tails, rho = 0.406, **P = 1.38 ×10−3. c, Correlation between ΔFRET and ΔFWHM of the boutons at 6 min after STDP induction from the data shown in a. Spearman’s correlation with two tails, rho = 0.557, **P = 6.8 ×10−5. d, Correlation between ΔFRET of the bouton and ΔV of the spine at 6 min after STDP induction from the data shown in a. Spearman’s correlation with two tails, rho = 0.002, P = 0.848. e, f, Absence of spine enlargement (e) and FRET changes (f) when uncaging was applied without postsynaptic spikes (green, n = 7 boutons, 7 slices, 5 rats). The control STDP traces are the replicates from the red traces in Fig. 4d, g. Two-sided Kruskal–Wallis test (H = 4.39, P = 0.356) for (e) and (H = 0.48, P = 0.975) for (f). Error bars indicate s.e.m
Extended Data Fig. 10 Enhancement of glutamate release by spine enlargement.
a, d, Fluorescence images (single XY- and XZ-planes) of the bouton-spine pairs, and dendritic spines before and after stimulation with STDP protocol for a pushing (a) and a non-pushing (d) spine. Horizontal scale bars indicate 1 µm, vertical scale bars indicate 2 µm. In all bouton-spine pairs shown in this figure, the criteria (Fz > 0.8 and Fx > 0.6) were met (Methods). b–f, The Pr measurements (b, e) and the averaged iGluSnFR traces (c,f) before and after induction of STDP. Orange bars indicate optogenetic stimulation (5 ms). g–j, The time courses of spine enlargement (g), spine length (h), Pr (i) and the peak of the averaged iGluSnFR traces (j) in pushing (red) and non-pushing (black) spines for the same set of data shown in k–n. k–n, The increases in spine volume (ΔV, k), spine length (ΔL, l), Pr (ΔPr, m) and average release (ΔRelease, n) in the synapses with pushing and without pushing. n = 6 (6 slices, 6 rats) and 5 (5 slices, 5 rats) for pushing and non-pushing spines at 10 min after STDP induction. Two-sided Mann–Whitney U test (U = 11, P = 0.537 [k]). Two-sided Wilcoxson signed-rank test (W = 21, *P = 0.036 [l]; W = 21, *P = 0.036 [m]; W = 21, *P = 0.036 [n]) for pushing synapses and (W = 3, P =0.281 [l]; W = 1, P = 0.104 [m]; W = 1, P = 0.106 [n]) for non-pushing synapses. o, p, Correlation for ΔPr (o) and ΔRelease (p) vs ΔV from the same dataset as k–n. Spearman’s correlation, rho = 0.257, P = 0.116 (o) and rho = 0.132, P = 0.273 (p). q, r, Correlation for ΔPr (q) and ΔRelease (r) vs ΔL with (red) and without (black) spine elongation from the same dataset as k–n. Spearman’s correlation, rho = 0.469, *P = 0.025 (q) and rho = 0.746, **P = 1.25×10−3 (r). Error bars indicate s.e.m
Supplementary information
Supplementary Video 1
Fine pushing of a bouton (iGluSnFR). An example of fine pushing of a presynaptic bouton, which expresses iGluSnFR (green), with a glass pipette (magenta). Note the change in the bouton shape and its immediate recovery after pipette retraction, as analysed in Fig. 1g. The distortion of the bouton (Fig. 1f) was very small (0.1 μm) but significant.
Supplementary Video 2
Fine pushing of a bouton (iSLIM). An example of the fine pushing of the presynaptic bouton, which expresses mTurquoise2-syntaxin-1A, as shown in Fig. 2b. The distortion of the bouton was very small but significant, as analysed in Extended Data Fig. 2c, d. The pipette was retracted 1 min after pushing.
Supplementary Video 3
Stimulation and pushing of a bouton (GCaMP7s). GCaMP7s imaging of a presynaptic bouton in the CA1 region. The bouton was optogenetically stimulated at the CA3 region. Three out of ten stimulations are displayed, and indicated as stim1, stim2 and stim3. The white dot indicates the LED stimulation. Despite responding to the optogenetic stimulation (Extended Data Fig. 3c), the bouton showed no increase in fluorescence intensity by pushing (Extended Data Fig. 3b).
Source data
Rights and permissions
About this article
Cite this article
Ucar, H., Watanabe, S., Noguchi, J. et al. Mechanical actions of dendritic-spine enlargement on presynaptic exocytosis. Nature 600, 686–689 (2021). https://doi.org/10.1038/s41586-021-04125-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-04125-7
This article is cited by
-
Mechanisms of SNARE proteins in membrane fusion
Nature Reviews Molecular Cell Biology (2024)
-
Mechanical stimulation and electrophysiological monitoring at subcellular resolution reveals differential mechanosensation of neurons within networks
Nature Nanotechnology (2024)
-
Loss of spines in the prelimbic cortex is detrimental to working memory in mice with early-life adversity
Molecular Psychiatry (2023)
-
Single-neuron mechanical perturbation evokes calcium plateaus that excite and modulate the network
Scientific Reports (2023)
-
Drosophila motor neuron boutons remodel through membrane blebbing coupled with muscle contraction
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.