Abstract
Multiple sclerosis (MS) lesions that do not resolve in the months after they form harbour ongoing demyelination and axon degeneration, and are identifiable in vivo by their paramagnetic rims on MRI scans1,2,3. Here, to define mechanisms underlying this disabling, progressive neurodegenerative state4,5,6 and foster development of new therapeutic agents, we used MRI-informed single-nucleus RNA sequencing to profile the edge of demyelinated white matter lesions at various stages of inflammation. We uncovered notable glial and immune cell diversity, especially at the chronically inflamed lesion edge. We define ‘microglia inflamed in MS’ (MIMS) and ‘astrocytes inflamed in MS’, glial phenotypes that demonstrate neurodegenerative programming. The MIMS transcriptional profile overlaps with that of microglia in other neurodegenerative diseases, suggesting that primary and secondary neurodegeneration share common mechanisms and could benefit from similar therapeutic approaches. We identify complement component 1q (C1q) as a critical mediator of MIMS activation, validated immunohistochemically in MS tissue, genetically by microglia-specific C1q ablation in mice with experimental autoimmune encephalomyelitis, and therapeutically by treating chronic experimental autoimmune encephalomyelitis with C1q blockade. C1q inhibition is a potential therapeutic avenue to address chronic white matter inflammation, which could be monitored by longitudinal assessment of its dynamic biomarker, paramagnetic rim lesions, using advanced MRI methods.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All raw snRNA-seq data (fastq files) were deposited to GEO (https://www.ncbi.nlm.nih.gov/geo/ under accession number GSE180759). Source data are provided with this paper.
Code availability
Codes used to analyse the snRNA-seq data in the current study are available from the corresponding authors upon request.
References
Bagnato, F., et al. Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 tesla. Brain 134, 3602–3615 (2011).
Absinta, M., et al. Persistent 7-tesla phase rim predicts poor outcome in new multiple sclerosis patient lesions. J. Clin. Invest. 126, 2597–2609 (2016).
Dal-Bianco, A., et al. Slow expansion of multiple sclerosis iron rim lesions: pathology and 7 T magnetic resonance imaging. Acta Neuropathol. 133, 25–42 (2017).
Absinta, M., et al. Association of chronic active multiple sclerosis lesions with disability in vivo. JAMA Neurol. 76, 1474–1483 (2019).
Elliott, C., et al. Chronic white matter lesion activity predicts clinical progression in primary progressive multiple sclerosis. Brain 142, 2787–2799 (2019).
Maggi, P., et al. Chronic white matter inflammation and neurofilament levels in patients with multiple sclerosis. Neurology 97, e543–e553 (2021).
Kuhlmann, T., et al. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 133, 13–24 (2017).
Reich, D. S., Lucchinetti, C. F. & Calabresi, P. A. Multiple sclerosis. N. Engl. J. Med. 378, 169–180 (2018).
Krishnaswami, S. R., et al. Using single nuclei for RNA-seq to capture the transcriptome of postmortem neurons. Nat. Protoc. 11, 499–524 (2016).
Hendrickx, D. A. E., et al. Gene expression profiling of multiple sclerosis pathology identifies early patterns of demyelination surrounding chronic active lesions. Front. Immunol. 8, 1810 (2017).
Elkjaer, M. L., et al. Molecular signature of different lesion types in the brain white matter of patients with progressive multiple sclerosis. Acta Neuropathol. Commun. 7, 205 (2019).
Jackle, K., et al. Molecular signature of slowly expanding lesions in progressive multiple sclerosis. Brain 143, 2073–2088 (2020).
Schirmer, L., et al. Neuronal vulnerability and multilineage diversity in multiple sclerosis. Nature 573, 75–82 (2019).
Maric, D., et al. Whole-brain tissue mapping toolkit using large-scale highly multiplexed immunofluorescence imaging and deep neural networks. Nat. Commun. 12, 1550 (2021).
Comabella, M., Montalban, X., Munz, C. & Lunemann, J. D. Targeting dendritic cells to treat multiple sclerosis. Nat. Rev. Neurol. 6, 499–507 (2010).
Clayton, B. L. L. & Popko, B. Endoplasmic reticulum stress and the unfolded protein response in disorders of myelinating glia. Brain Res. 1648, 594–602 (2016).
Krasemann, S., et al. The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47, 566–581.e569 (2017).
Keren-Shaul, H., et al. A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169, 1276–1290.e1217 (2017).
Masuda, T., et al. Spatial and temporal heterogeneity of mouse and human microglia at single-cell resolution. Nature 566, 388–392 (2019).
Sankowski, R., et al. Mapping microglia states in the human brain through the integration of high-dimensional techniques. Nat. Neurosci. 22, 2098–2110 (2019).
Jakel, S., et al. Altered human oligodendrocyte heterogeneity in multiple sclerosis. Nature 566, 543–547 (2019).
Miron, V. E., et al. M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat. Neurosci. 16, 1211–1218 (2013).
Cignarella, F., et al. TREM2 activation on microglia promotes myelin debris clearance and remyelination in a model of multiple sclerosis. Acta Neuropathol. 140, 513–534 (2020).
Breij, E. C., et al. Homogeneity of active demyelinating lesions in established multiple sclerosis. Ann. Neurol. 63, 16–25 (2008).
van der Poel, M., Hoepel, W., Hamann, J., Huitinga, I. & Dunnen, J. D. IgG immune complexes break immune tolerance of human microglia. J. Immunol. 205, 2511–2518 (2020).
Browaeys, R., Saelens, W. & Saeys, Y. NicheNet: modeling intercellular communication by linking ligands to target genes. Nat. Methods 17, 159–162 (2020).
Rusinova, I., et al. Interferome v2.0: an updated database of annotated interferon-regulated genes. Nucleic Acids Res. 41, D1040–D1046 (2013).
Kirby, L., et al. Oligodendrocyte precursor cells present antigen and are cytotoxic targets in inflammatory demyelination. Nat. Commun. 10, 3887 (2019).
Falcao, A. M., et al. Disease-specific oligodendrocyte lineage cells arise in multiple sclerosis. Nat. Med. 24, 1837–1844 (2018).
Liddelow, S. A., et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541, 481–487 (2017).
Zhang, B. & Horvath, S. A general framework for weighted gene co-expression network analysis. Stat. Appl. Genet. Mol. Biol. 4, 17 (2005).
Dal-Bianco, A., et al. Long-term evolution of multiple sclerosis iron rim lesions in 7 T MRI. Brain 144, 833–847 (2021).
International Multiple Sclerosis Genetics Consortium. Multiple sclerosis genomic map implicates peripheral immune cells and microglia in susceptibility. Science 365, eaav7188 (2019).
Fitzgerald, K. C., et al. Early complement genes are associated with visual system degeneration in multiple sclerosis. Brain 142, 2722–2736 (2019).
Kaiser, T. & Feng, G. Tmem119-EGFP and Tmem119-CreERT2 transgenic mice for labeling and manipulating microglia. eNeuro 6, 0448–18.2019 (2019).
Werneburg, S., et al. Targeted complement inhibition at synapses prevents microglial synaptic engulfment and synapse loss in demyelinating disease. Immunity 52, 167–182.e167 (2020).
Fonseca, M. I., et al. Cell-specific deletion of C1qa identifies microglia as the dominant source of C1q in mouse brain. J. Neuroinflammation 14, 48 (2017).
Stephan, A. H., et al. A dramatic increase of C1q protein in the CNS during normal aging. J. Neurosci. 33, 13460–13474 (2013).
Michailidou, I., et al. Complement C1q–C3-associated synaptic changes in multiple sclerosis hippocampus. Ann. Neurol. 77, 1007–1026 (2015).
van der Valk, P. & De Groot, C. J. Staging of multiple sclerosis (MS) lesions: pathology of the time frame of MS. Neuropathol. Appl. Neurobiol. 26, 2–10 (2000).
Matson, K. J. E., et al. Isolation of adult spinal cord nuclei for massively parallel single-nucleus RNA sequencing. J. Vis. Exp. 140, 58413 (2018).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Stuart, T., et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902.e1821 (2019).
Korsunsky, I., et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Fabregat, A., et al. The Reactome Pathway Knowledgebase. Nucleic Acids Res. 46, D649–D655 (2018).
Raudvere, U., et al. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 47, W191–W198 (2019).
Zhou, Y., et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 1523 (2019).
Bonnardel, J., et al. Stellate cells, hepatocytes, and endothelial cells imprint the Kupffer cell identity on monocytes colonizing the liver macrophage niche. Immunity 51, 638–654.e639 (2019).
Krzywinski, M., et al. Circos: an information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Schindelin, J., et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Absinta, M., et al. Postmortem magnetic resonance imaging to guide the pathologic cut: individualized, 3-dimensionally printed cutting boxes for fixed brains. J. Neuropathol. Exp. Neurol. 73, 780-788 (2014).
Polman, C. H., et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 69, 292–302 (2011).
Absinta, M., et al. Identification of chronic active multiple sclerosis lesions on 3T MRI. Am. J. Neuroradiol. 39, 1233–1238 (2018).
Yao, B., et al. Chronic multiple sclerosis lesions: characterization with high-field-strength MR imaging. Radiology 262, 206–215 (2012).
Sati, P., et al. Rapid, high-resolution, whole-brain, susceptibility-based MRI of multiple sclerosis. Mult. Scler. 20, 1464–1470 (2014).
Acknowledgements
This study was supported by the Intramural Research Program of NINDS, the Adelson Medical Research Foundation (to D.S.R. and D.P.S.), the Conrad N. Hilton Foundation (grant no. 17313 to M.A.) and the Cariplo Foundation (grant no. 2019-1677 to M.A.). We thank Y. Li and Y. Luo from the DNA Sequencing and Genomics Core (NHLBI/NIH); G. Trivellin (NICHD/NIH), who helped with some technical aspects of single nucleus RNA-seq processing; C. Covino from the Advanced Light and Electron Microscopy BioImaging Center (ALEMBIC, Vita-Salute San Raffaele University, Milan), who helped with visualization of multiplex immunofluorescence; T. Yednock and V. Mehta at Annexon Biosciences for providing the anti-C1q monoclonal antibody (ANX-M1.21); G. Nair, P. Sati, E. S. Beck, M. Donadieu, the NINDS Translational Neuroradiology Section and the NIMH Functional MRI Facility for assistance with MRI scanning; I. Cortese, J. Ohayon and the NINDS Neuroimmunology Clinic for outstanding patient care; and D. Bergles, R. Franklin, J. Kipnis and N. Mihelson for fruitful discussion of the results.
Author information
Authors and Affiliations
Contributions
M.A., J.-P.L. and P.L. performed the nucleic extraction and snRNA-seq experiments; M.A. and K.R.J. performed the bioinformatic analysis; M.A. and D.M. performed the immunostainings and multiplex immunofluorescence; A.S., M.G., T.G., M.D.S., J.J., D.P.S. and P.A.C. performed the animal studies; M.A., D.S.R. and T.W. performed the MRI studies; P.A.C. and K.C.F. performed the genotyping study; M.A., D.S.R., P.A.C., D.P.S. and D.B.M. discussed and interpreted findings; M.A. and D.S.R. designed the study and wrote the manuscript. All co-authors read, revised and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests. M.A. received consulting fees from Sanofi, unrelated to this study. P.A.C. received grant support from Annexon Biosciences for testing the anti-C1q-blocking antibody in EAE. The funders of the study had no role in the collection, analysis, or interpretation of data, in the writing of the manuscript, or in the decision to submit the paper for publication.
Additional information
Peer review information Nature thanks Oleg Butovsky and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
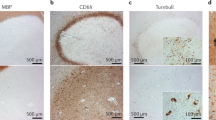
Extended Data Fig. 1 Chronic active/slowly expanding rim lesions can be visualized with MRI in vivo.
a, Susceptibility-based axial MRI at three levels showing the chronic active MS lesions with paramagnetic rims (magnified view in the insets, arrows) of a 38-year-old man with progressive MS, who requires assistance to walk 100 meters without resting. Scale bar = 10 mm. b, MRI-pathology of a frontal periventricular chronic active/slowly expanding lesion in a man with progressive MS who died at age 59. On the serial in vivo T1-weighted coronal images (clinical MR images sensitive to both demyelination and axonal loss), the rim lesion clearly expanded over a period of 7 years. The lesion was classified as chronic active by histological analysis. Accumulation of iron-laden phagocytes (CD68 and Turnbull iron staining) was seen at the lesion edge. Smouldering demyelination can also be inferred by the co-presence of early (LFB+, blue) and late (PAS+, purple) myelin degradation products within phagocytes at the lesion edge. Most reactive GFAP+ astrocytes do not contain iron. Magnified views are shown in the insets. Scale bar = 20 μm (myelin PLP); 10 μm (LFB-PAS); 50 μm (CD68, Turnbull iron/CD68, Turnbull iron/GFAP). c, In vivo long-term evolution of PRLs: representative examples of persistent/stable, faded, and disappeared rims. d, Bar graph showing the evolution of PRLs in each MS case followed over time. All cases had >5 years of yearly MRI follow up, except for cases 2–4, where follow-up was between 3.5 and 5 years. e, Survival analysis showed that the median PRL survival time is about 7 years.
Extended Data Fig. 2 Cell cluster proportions vary across tissue types.
a, Bar graphs showing the distribution of nuclei percentages by cell type (mean, standard error). See Fig. 1 for corresponding UMAP plots and distribution of nuclei count per sample. b, Bar graph showing the percentage of MIMS-foamy and MIMS-iron nuclei for each sample and location. WM, white matter; CA, chronic active; CI, chronic inactive; MIMS, microglia inflamed in multiple sclerosis; OPC, oligodendrocyte progenitor cells; mono/moDC, monocytes and monocyte-derived dendritic cells; oligo, oligodendrocytes; ox, oxidative stress.
Extended Data Fig. 3 Subclustering markers for astrocytes, oligodendrocytes and OPC.
a–c, Subclustering UMAP plots for astrocytes, oligodendrocytes and OPC, and associated stacked violin plots depicting selected differentially expressed genes for each subcluster. d, Bar graph showing A1-marker30 expression (Z-scores relative to all astrocytes) for each astrocyte cluster. Most A1 markers were highly expressed in AIMS. UMAP, uniform manifold approximation and projection; OPC, oligodendrocyte progenitor cells; oligo, oligodendrocytes; AIMS, astrocytes inflamed in MS.
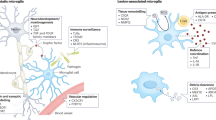
Extended Data Fig. 4 Lymphocytes, monocyte-derived dendritic cells, MIMS and AIMS are identified at the chronic active lesion edge.
Chronic active lesion overview. Multiplex immunostaining of a brain tissue block from a 48-year-old woman with progressive MS (case MS4 in Supplementary Table 1), including a chronic active demyelinated lesion (devoid of myelin proteolipid protein (PLP) staining). Numbers indicate areas magnified in subsequent panels for validation of immune cell and inflamed astrocytes and represent the chronic active lesion edge, lesion centre and periplaque white matter. Lymphocytes and microglia at the lesion edge (panels 1 and 2). At the chronic active lesion edge, there are groups of CD8 T cells within the perivascular space and sparsely within the parenchyma (arrows). CD20 B cells are fewer than CD8 T cells and are located prevalently within the perivascular space (dashed arrows). Transition from myelination to demyelination is shown with staining for CNPase, an oligodendrocyte and myelin marker. Residual myelin fragments can be seen at the edge (arrowheads), presumably not yet removed by phagocytes. IBA1-microglia/macrophages are frequent, and they have an activated morphology (round shape without ramifications). Bar graphs (second row) show the gene expression Z-scores of markers implemented for the identification in tissue of the most relevant glial cell populations at the chronic active lesion edge. Homeostatic microglia vs. MIMS: different spatial locations (insets 3–5). In the periplaque white matter, most microglia are P2RY12+ (a homeostatic marker) with short and thick processes, whereas at the chronic active lesion edge, most are CD68+ (indicating upregulation of antigen and lipid processing) with round, activated morphology. At the lesion core, fewer microglia can be identified, and these show a round morphology consistent with activation. Interestingly, some of them are P2RY12+, potentially suggesting the return of some homeostatic markers. MIMS-iron (inset 6). Accumulation of iron-laden phagocytes (CD68 and ferritin light chain, FTL) is seen at the lesion edge. FTL is within the first 100 top differentially expressed genes in MIMS, especially in MIMS-iron (Fig. 2c). Iron retention in phagocytes can be seen by MRI at the chronic active lesion edge as a paramagnetic rim (MRI biomarker; Fig. 1a). MIMS-foamy (inset 7). Colocalization of PPARG and CD68. PPARG and CD68 double positive (white, arrows) microglia are especially seen at the lesion edge, suggesting their involvement in energy metabolism and modulation of inflammation as well as clearance of myelin debris. Monocytes/monocyte-derived dendritic cells and MIMS (inset 8). CD68 microglia outnumber CD83 monocyte-derived mature dendritic cells (arrows) at the chronic active lesion edge. AIMS and MIMS (insets 9, 10a, and 10b). In addition to IBA1+/CD68+ microglia, the lesion edge is enriched for inflamed astrocytes (positive for VIM and APOE but negative for IBA1 and CD68), sometimes in close proximity (dashed white box). Compared to activated microglia, inflamed astrocytes are bigger and show radial processes. moDC, monocyte-derived dendritic cells; MIMS, microglia inflamed in MS; AIMS, astrocytes inflamed in MS. Scale bar: 20 μm.
Extended Data Fig. 5 Pathway analysis of the two MIMS populations and comparison with DAM.
a, b, Gene enrichment and pathway analysis of the top 100 differentially expressed genes are shown for MIMS-foamy and MIMS-iron populations, separately. Using the gProfileR package, different sources were compared (GO:BP, GO:CC, GO:MF, KEGG). Only significant pathway terms are plotted in the graphs (p < 0.05, correction methods "g_SCS"). See the text for interpretation. c, Homeostatic microglia vs. MIMS vs. DAM: direct comparison of two single-cell RNA-seq datasets (MS [current work] and a mouse AD model [5XFAD]). Data for the AD model are derived from supplementary table 2 of ref. 18. In each dataset, only significant differentially expressed genes in the comparison between homeostatic vs. MIMS and homeostatic vs. DAM were retained (p < 0.001). Volcano plots report overlapping genes for each microglia phenotype. MS, multiple sclerosis; MIMS, microglia inflamed in MS; AD, Alzheimer disease; DAM, degenerative disease-associated microglia.
Extended Data Fig. 6 Two MIMS populations were identified through re-analysis of two published snRNA-seq MS datasets.
a, snRNA-seq initial mapping (UMAP plot) and annotations based on the top differentially expressed genes in each cluster after re-analysis of raw data from Schirmer et al. 13 and Jäkel et al. 21. b, Immune cell nuclei were mapped onto our immune subclustering map. Most of the immune cell populations identified were seen also in the other datasets, including the two MIMS populations. UMAP, uniform manifold approximation and projection; MIMS, microglia inflamed in MS.
Extended Data Fig. 7 Multiplex immunostaining of human chronic active lesions.
a, Overview of the multiplex immunostaining method (see the text for details). b, A representative example of the chronic active edge and myeloid subpopulations. In the magnified view, identification of MIMS-foamy, MIMS-iron, and macrophages using 7 primary antibodies (combined or separate channels). c, Within the lesion core, most cells (both myeloid cells and astrocytes) are positive for the senescence marker p16INK4a. Separate channels are shown to facilitate the visualization of different markers. d, Quantification of the proportion of p16-positive nuclei at different locations. The lesion core showed the higher percentage of p16-positive nuclei (ANOVA p < 0.0001, Tukey’s multiple comparison post-hoc analysis *p < 0.05, **p < 0.01, ***p < 0.001). Scale bar: 20 μm. Blue: DAPI (nuclei).
Extended Data Fig. 8 MIMS-AIMS gene modules and correlation.
a, b, Hierarchical clustering dendrograms of genes (a) and module colours (b) based on weighted correlation network analysis (WGCNA) of 918 variable genes from immune cells and astrocyte clusters 5, 9, 10. We identified 2 MIMS gene modules (pink: C1QB, CD74, CEBPD, HLA-DRA, ITM2B, RPS19; black: ACTB, APOE, CD81, EEF1A1, FTH1, FTL, PSAP) and an AIMS gene module (magenta: CALM1, CLU, CRYAB, CST3, GAPDH, GFAP). The complete list of modules is shown in Supplementary Table 9. c, Heatmap showing the eigengene adjacency matrix that represents the relationships among the identified gene modules. MIMS gene modules (pink and black) were highly correlated with other myeloid gene modules and with the AIMS module, but not with the nonreactive astrocyte module. AST, astrocytes; IMM, immune cells; MIMS, microglia inflamed in MS; AIMS, astrocytes inflamed in MS; ME, module eigengene; MT, mitochondrial genes.
Extended Data Fig. 9 Mapping complement and MS susceptibility genes onto the snRNA-seq dataset.
a, Heatmap showing the expression of genes involved in the classical complement cascade, including complement genes, receptors, and regulators. Z-scores are relative to all cells. Most of these genes map onto immune cell, astrocyte, and vascular cell populations. Cluster identifiers correspond to Fig. 1c, d. b, For each cell population, the number of complement cascade-related genes with Z-score>1.5 is plotted on the UMAP. c, Heatmaps showing the expression of genes involved in the classical complement cascade for the immune cell (left, pink) and astrocyte (right, green) subclusters. Z-scores are relative to immune cells and astrocytes, respectively. Among microglia, C1Q, C3 and CFD (C3 activator) were expressed mainly by MIMS-iron, whereas negative regulators of complement activation (LAIR1, LAIR2, CR1) were expressed mainly by homeostatic microglia and perivascular macrophages. Among astrocytes, C1 complex (C1Q, C1R, C1S, C1QBP), C3, C4 and CALR were expressed mainly by AIMS, whereas C2 and C6 were expressed by some other reactive astrocytes. d, Mapping MS-susceptibility risk genes onto the snRNA-seq dataset: the list of 558 prioritized MS susceptibility genes was obtained from a recent genome-wide association study (GWAS)33 and mapped onto all three snRNA-seq datasets (the current dataset as well as that of Schirmer et al. 13 and Jäkel et al. 21). Low-expressed genes (within the 25th percentile average expression) were excluded. MS susceptibility genes were then assigned to clusters if the z-scored average gene expression was >2. Clusters were classified based on the number of MS susceptibility genes (>80, 50–80, 30–50, 1–30 and < 10 genes). Results were colour-mapped onto each snRNA-seq UMAP. Most MS susceptibility genes mapped onto the immune and vascular cell clusters. Interestingly, an excitatory neuronal population (“py”) expressed some MS susceptibility genes as well. oligo, oligodendrocytes; imm, immune cells; OPC, oligodendrocyte progenitor cells; lym, lymphocytes; ast, astrocytes; endo, endothelial cells; peri, pericytes; neu, neurons; MIMS, microglia inflamed in MS; mono/moDC, monocytes and monocyte-derived dendritic cells; AIMS, astrocytes inflamed in MS; exc, excitatory; in, inhibitory.
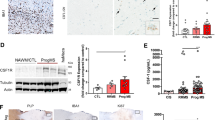
Extended Data Fig. 10 C1q mediates microglia activation in mouse EAE.
Iba1+ cells in microglia-specific C1q cKO with EAE appear less reactive. a, Visual thalamus was immunostained at PID12–14 for Iba1 (microglia/macrophages) and Clec7a (disease-associated microglia). Yellow arrows denote Iba1+Clec7a+ cells, white arrows Iba1+Clec7a- cells. Scale bar = 100μm. b, c, Clec7a decreased in cKO mice with EAE compared to Ctrl-EAE littermates. The density of Iba1+ cells and Iba1 MFI was attenuated to control values in cKO-EAE mice. *p < 0.05, by one-way ANOVA and Tukey’s posthoc test (b) or Kruskal-Wallis test and Dunn posthoc test (c). d, Iba1+ cells were morphologically characterized into 3 categories (representative images in the panel below the quantification, scale bar = 20 μm). Iba1+ cells in cKO-EAE mice were indistinguishable from CFA controls. cKO-EAE showed more ramified Iba1+ cells and fewer amoeboid cells compared to Ctrl-EAE littermates. **p < 0.01, ***p < 0.001, ****p < 0.0001 by two-way ANOVA and Tukey’s posthoc test. Error bars: SEM. Anti-C1q treatment reduces expression of FTL and Iba1 in chronic EAE. e, Experimental paradigm (twice weekly treatment with isotype control or C1q-blocking antibody (M1.21) from EAE onset until PID42). f, Representative images of FTL and Iba1 immunostaining. g, h, Higher magnifications of f. White arrows: Iba1+ cells. i, EAE scores for each treatment arm. j, k, Quantification of the expression of C1q, FTL and Iba1 in hippocampal white matter (outlined with the dashed line in f) and count of Iba1+FTL+ cells. Student t-test, p* ≤ 0.05, *** ≤ 0.001. Error bars: SEM. TAM, tamoxifen; FTL, ferritin light chain; MFI, mean fluorescent intensity; PID, post-immunization day; SEM,standard error of the mean.
Supplementary information
Supplementary Information
This file contains supplementary text and supplementary references
Supplementary Tables 1–13
This file contains Supplementary Tables 1–13 and their accompanying legends.
Source data
Rights and permissions
About this article
Cite this article
Absinta, M., Maric, D., Gharagozloo, M. et al. A lymphocyte–microglia–astrocyte axis in chronic active multiple sclerosis. Nature 597, 709–714 (2021). https://doi.org/10.1038/s41586-021-03892-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-03892-7
This article is cited by
-
Bone-derived PDGF-BB enhances hippocampal non-specific transcytosis through microglia-endothelial crosstalk in HFD-induced metabolic syndrome
Journal of Neuroinflammation (2024)
-
Microglia regulation of central nervous system myelin health and regeneration
Nature Reviews Immunology (2024)
-
Profiling of microglia nodules in multiple sclerosis reveals propensity for lesion formation
Nature Communications (2024)
-
Myeloid cell replacement is neuroprotective in chronic experimental autoimmune encephalomyelitis
Nature Neuroscience (2024)
-
The aging mouse CNS is protected by an autophagy-dependent microglia population promoted by IL-34
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.