Abstract
T follicular helper (TFH) cells are crucial for B cell-mediated humoral immunity1. Although transcription factors such as BCL6 drive the differentiation of TFH cells2,3, it is unclear whether and how post-transcriptional and metabolic programs enforce TFH cell programming. Here we show that the cytidine diphosphate (CDP)–ethanolamine pathway co-ordinates the expression and localization of CXCR5 with the responses of TFH cells and humoral immunity. Using in vivo CRISPR–Cas9 screening and functional validation in mice, we identify ETNK1, PCYT2, and SELENOI—enzymes in the CDP–ethanolamine pathway for de novo synthesis of phosphatidylethanolamine (PE)—as selective post-transcriptional regulators of TFH cell differentiation that act by promoting the surface expression and functional effects of CXCR5. TFH cells exhibit unique lipid metabolic programs and PE is distributed to the outer layer of the plasma membrane, where it colocalizes with CXCR5. De novo synthesis of PE through the CDP–ethanolamine pathway co-ordinates these events to prevent the internalization and degradation of CXCR5. Genetic deletion of Pcyt2, but not of Pcyt1a (which mediates the CDP–choline pathway), in activated T cells impairs the differentiation of TFH cells, and this is associated with reduced humoral immune responses. Surface levels of PE and CXCR5 expression on B cells also depend on Pcyt2. Our results reveal that phospholipid metabolism orchestrates post-transcriptional mechanisms for TFH cell differentiation and humoral immunity, highlighting the metabolic control of context-dependent immune signalling and effector programs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information files. Microarray data of wild-type and Pcyt2-deficient cells at day 3 after LCMV infection have been deposited in the NCBI Gene Expression Omnibus (GEO) database and are accessible through the GEO SuperSeries accession number: GSE147190. For GSEA using manually curated gene signatures from public datasets, the datasets GSE21380 (ref. 9), GSE72188 (ref. 17) and GSE74854 (ref. 37) from the NCBI GEO database were used to generate the relevant gene signatures (<5% FDR). Source data are provided with this paper.
Code availability
The code used for the colocalization index analysis of TIRF–STORM or other co-ordinate-based data has been applied successfully42 and the accessible link of the code is https://rdrr.io/cran/colocalization/.
References
Crotty, S. T follicular helper cell biology: a decade of discovery and diseases. Immunity 50, 1132–1148 (2019).
Johnston, R. J. et al. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science 325, 1006–1010 (2009).
Nurieva, R. I. et al. Bcl6 mediates the development of T follicular helper cells. Science 325, 1001–1005 (2009).
Zeng, H. et al. mTORC1 and mTORC2 kinase signaling and glucose metabolism drive follicular helper T cell differentiation. Immunity 45, 540–554 (2016).
Chapman, N. M., Boothby, M. R. & Chi, H. Metabolic coordination of T cell quiescence and activation. Nat. Rev. Immunol. 20, 55–70 (2020).
Wei, J. et al. Targeting REGNASE-1 programs long-lived effector T cells for cancer therapy. Nature 576, 471–476 (2019).
Huang, H. et al. In vivo CRISPR screening reveals nutrient signaling processes underpinning CD8+ T cell fate decisions. Cell 184, 1245–1261 (2021).
Xu, L. et al. The transcription factor TCF-1 initiates the differentiation of TFH cells during acute viral infection. Nat. Immunol. 16, 991–999 (2015).
Choi, Y. S. et al. LEF-1 and TCF-1 orchestrate TFH differentiation by regulating differentiation circuits upstream of the transcriptional repressor Bcl6. Nat. Immunol. 16, 980–990 (2015).
Boularan, C. et al. B lymphocyte-specific loss of Ric-8A results in a Gα protein deficit and severe humoral immunodeficiency. J. Immunol. 195, 2090–2102 (2015).
Preite, S. et al. Hyperactivated PI3Kδ promotes self and commensal reactivity at the expense of optimal humoral immunity. Nat. Immunol. 19, 986–1000 (2018).
Lemonnier, F. et al. Recurrent TET2 mutations in peripheral T-cell lymphomas correlate with TFH-like features and adverse clinical parameters. Blood 120, 1466–1469 (2012).
Johnston, R. J., Choi, Y. S., Diamond, J. A., Yang, J. A. & Crotty, S. STAT5 is a potent negative regulator of TFH cell differentiation. J. Exp. Med. 209, 243–250 (2012).
Calzada, E., Onguka, O. & Claypool, S. M. Phosphatidylethanolamine metabolism in health and disease. Int. Rev. Cell Mol. Biol. 321, 29–88 (2016).
Haynes, N. M. et al. Role of CXCR5 and CCR7 in follicular Th cell positioning and appearance of a programmed cell death gene-1high germinal center-associated subpopulation. J. Immunol. 179, 5099–5108 (2007).
Wang, L., Magdaleno, S., Tabas, I. & Jackowski, S. Early embryonic lethality in mice with targeted deletion of the CTP:phosphocholine cytidylyltransferase alpha gene (Pcyt1a). Mol. Cell. Biol. 25, 3357–3363 (2005).
Liu, X. et al. Genome-wide analysis identifies Bcl6-controlled regulatory networks during T follicular helper cell differentiation. Cell Rep. 14, 1735–1747 (2016).
Shi, J. et al. PD-1 controls follicular T helper cell positioning and function. Immunity 49, 264–274 (2018).
Zhao, M. Lantibiotics as probes for phosphatidylethanolamine. Amino Acids 41, 1071–1079 (2011).
Irie, A., Yamamoto, K., Miki, Y. & Murakami, M. Phosphatidylethanolamine dynamics are required for osteoclast fusion. Sci. Rep. 7, 46715 (2017).
Burr, M. L. et al. CMTM6 maintains the expression of PD-L1 and regulates anti-tumour immunity. Nature 549, 101–105 (2017).
Zeng, H. et al. Discrete roles and bifurcation of PTEN signaling and mTORC1-mediated anabolic metabolism underlie IL-7-driven B lymphopoiesis. Sci. Adv. 4, eaar5701 (2018).
Platt, R. J. et al. CRISPR–Cas9 knockin mice for genome editing and cancer modeling. Cell 159, 440–455 (2014).
Oxenius, A., Bachmann, M. F., Zinkernagel, R. M. H. & Hengartner, H. Virus-specific MHC-class II-restricted TCR-transgenic mice: effects on humoral and cellular immune responses after viral infection. Eur. J. Immunol. 28, 390–400 (1998).
Leonardi, R., Frank, M. W., Jackson, P. D., Rock, C. O. & Jackowski, S. Elimination of the CDP-ethanolamine pathway disrupts hepatic lipid homeostasis. J. Biol. Chem. 284, 27077–27089 (2009).
Zhang, D. et al. Macrophages deficient in CTP:phosphocholine cytidylyltransferase-α are viable under normal culture conditions but are highly susceptible to free cholesterol-induced death. J. Biol. Chem. 275, 35368–35376 (2000).
Bettini, M. L., Bettini, M., Nakayama, M., Guy, C. S. & Vignali, D. A. Generation of T cell receptor-retrogenic mice: improved retroviral-mediated stem cell gene transfer. Nat. Protocols 8, 1837–1840 (2013).
Locci, M. et al. Human circulating PD-1+CXCR3−CXCR5+ memory Tfh cells are highly functional and correlate with broadly neutralizing HIV antibody responses. Immunity 39, 758–769 (2013).
Chen, R. et al. In vivo RNA interference screens identify regulators of antiviral CD4+ and CD8+ T cell differentiation. Immunity 41, 325–338 (2014).
Birsoy, K. et al. An essential role of the mitochondrial electron transport chain in cell proliferation is to enable aspartate synthesis. Cell 162, 540–551 (2015).
Sanson, K. R. et al. Optimized libraries for CRISPR–Cas9 genetic screens with multiple modalities. Nat. Commun. 9, 5416 (2018).
Hanayama, R. et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science 304, 1147–1150 (2004).
Todo, T. et al. Anti-CD3ε induces splenic B220lo B-cell expansion following anti-CD20 treatment in a mouse model of allosensitization. Int. Immunol. 24, 529–538 (2012).
Zeng, H. et al. mTORC1 couples immune signals and metabolic programming to establish Treg-cell function. Nature 499, 485–490 (2013).
Shrestha, S. et al. Treg cells require the phosphatase PTEN to restrain TH1 and TFH cell responses. Nat. Immunol. 16, 178–187 (2015).
Yu., D. et al. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity 31, 457–468 (2009).
Shaw, L. A. et al. Id2 reinforces TH1 differentiation and inhibits E2A to repress TFH differentiation. Nat. Immunol. 17, 834–843 (2016).
Li, W. et al. Chimeric antigen receptor designed to prevent ubiquitination and downregulation showed durable antitumor efficacy. Immunity 53, 456–470 (2020).
Bates, M., Huang, B., Dempsey, G. T. & Zhuang, X. Multicolor super-resolution imaging with photo-switchable fluorescent probes. Science 317, 1749–1753 (2007).
Guy, C. S. et al. Distinct TCR signaling pathways drive proliferation and cytokine production in T cells. Nat. Immunol. 14, 262–270 (2013).
Liedmann, S. et al. Viral suppressors of the RIG-I-mediated interferon response are pre-packaged in influenza virions. Nat. Commun. 5, 5645 (2014).
Samir, P. et al. DDX3X acts as a live-or-die checkpoint in stressed cells by regulating NLRP3 inflammasome. Nature 573, 590–594 (2019).
Malkusch, S. et al. Coordinate-based colocalization analysis of single-molecule localization microscopy data. Histochem. Cell Biol. 137, 1–10 (2012).
Azuma, T. & Kei, T. Super-resolution spinning-disk confocal microscopy using optical photon reassignment. Opt. Express 23, 15003–15011 (2015).
Guagliardi, L. E. et al. Co-localization of molecules involved in antigen processing and presentation in an early endocytic compartment. Nature 343, 133–139 (1990).
Folch, J., Lees, M. & Sloane Stanley, G. H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226, 497–509 (1957).
Pasternak, C. A. & Bergeron, J. J. M. Turnover of mammalian phospholipids. Biochem. J. 119, 473–480 (1970).
Keckesova, Z. et al. LACTB is a tumour suppressor that modulates lipid metabolism and cell state. Nature 543, 681–686 (2017).
Acknowledgements
The authors acknowledge J. Saravia for human samples; Y. Wang and J. Raynor for editing of the manuscript; M. Hendren for animal colony management; the St. Jude Immunology FACS core facility for cell sorting; the Center for Advanced Genome Engineering (CAGE) for testing sgRNA deletion efficiency; and the Tetramer Facility of the NIH for gp66. Electron microscopy support was provided by the Cell and Tissue Imaging Center at St. Jude Children’s Research Hospital and by NCI P30 CA021765. This work was supported by NIH AI105887, AI131703, AI140761, AI150241, AI150514 and CA221290, by the Lupus Research Alliance, and by ALSAC. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
G.F. designed and performed in vitro and in vivo experiments, analysed data and wrote the manuscript; C.S.G. performed imaging experiments and analysed the data with the help of S.R.; N.M.C. analysed human samples; G.P. performed lipidomic experiments; J.W. and L.L. developed the lentiviral sgRNA metabolic library; P.Z., H.H. and A.K. helped with cellular and molecular experiments; Y.-D.W., C.Q. and J.Y. analysed CRISPR–Cas9 screening data; Y.D., H.S. and X.L. performed bioinformatic analyses; S.A.B. helped with serum immunoglobulin analysis; A.J., R.W. and C.G.R. helped with electron microscopy experiments; A.S. provided human tonsil samples; S.J. provided the Pcyt2fl and Pcyt1afl mice and contributed scientific insight and guidance on PE metabolism; H.C. designed experiments, co-wrote the manuscript and provided overall direction.
Corresponding author
Ethics declarations
Competing interests
H. Chi is a consultant for Kumquat Biosciences. The remaining authors declare no competing interests.
Additional information
Peer review information Nature thanks Michael Dustin, Alessio Lanna and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
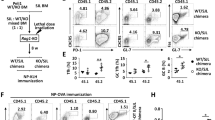
Extended Data Fig. 1 Pooled in vivo CRISPR–Cas9 screening and an in vivo dual transfer system to identify and validate potential regulators of TFH cells.
a, Diagram of the screening system. Naive Cas9-expressing SMARTA cells were transduced with lentiviral-derived sgRNA metabolic library, expanded in vitro and transferred into C57BL/6 mice, which were then infected with LCMV 24 h later. At day 7 after infection, splenic TFH (CXCR5+SLAM−) and TH1 (CXCR5−SLAM+) cells were purified and those gRNAs that were upregulated (corresponding to negative regulators of TFH responses) or downregulated (corresponding to positive regulators of TFH responses) in TFH versus TH1 cells (|log2(TFH/TH1)| > 0.5; adjusted P < 0.05) were determined for metabolism-related genes that establish TFH over TH1 cell differentiation. b, Diagram of in vivo dual transfer system. SMARTA cells transduced with sgRNA viral vectors expressing distinct fluorescent proteins were mixed and transferred into the same C57BL/6 mice, followed by LCMV infection and experimental analyses. c, SMARTA cells transduced with the indicated sgRNA viral vectors (Ametrine+) were mixed at a 2:1 ratio with sgNTC (mCherry+)-transduced SMARTA cells and transferred into C57BL/6 mice, followed by LCMV infection. Analyses of the proportion of donor-derived TFH (CXCR5+SLAM− or PSGL-1−Ly6C−) and TH1 (CXCR5−SLAM+ or PSGL-1+Ly6C+) cells and quantification of relative TFH cell percentage and number (lower right) in the spleen at day 7 (n = 4 mice). d, Insertion and deletion (indel) mutations after CRISPR–Cas9 targeted disruption in SMARTA cells transduced with sgNTC or sgPcyt2, through deep sequencing analysis of indels generated at the exonic target site of the Pcyt2 gene, including 74% of indel events in sgPcyt2-transduced cells compared to 0.6% in sgNTC-transduced cells. e, Immunoblot analyses of ETNK1 and PCYT2 in splenic SMARTA cells at day 3 after infection. Asterisk (*), non-specific band; arrow, the target band. Data are representative of two (d, e), or at least three (c) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (c). ***P < 0.001. Numbers in quadrants or gates indicate percentage of cells.
Extended Data Fig. 2 Validation of the effects of CDP–ethanolamine pathway genes on TFH generation during both viral infection and protein immunization.
a, b, Quantification of relative proportions and numbers of CXCR5+PD-1+ TFH cells (a) and CXCR5−SLAM+ TH1 cells (b) in donor-derived cells from the spleen of mice receiving the indicated sgRNA-transduced SMARTA cells at day 7 after LCMV infection (n = 4 mice). c, Indel mutations after CRISPR–Cas9 targeted disruption in SMARTA cells transduced with the indicated sgRNAs through deep sequencing analysis of indels generated at the exonic target sites of the indicated genes. d, Immunoblot analyses of the indicated proteins in the indicated sgRNA-transduced SMARTA cells isolated from the spleen at 3–5 days after adoptive transfer and LCMV infection. Asterisk (*), non-specific band; arrow, the target band. e, SMARTA cells transduced with the indicated sgRNA-expressing vectors (Ametrine+) were mixed at a 2:1 ratio with sgNTC (mCherry+)-transduced SMARTA cells and transferred into C57BL/6 mice, followed by LCMV infection. Quantification of donor-derived TFH and TH1 cells from the host spleen at day 7 after infection (n = 4 mice). f, OT-II cells transduced with the indicated sgRNAs were transferred into C57BL/6 mice, followed by immunization with NP-OVA and LPS in alum. Quantification of TFH cell percentage and number in the spleen at day 7 (n = 4 mice). Data are representative of two (c–e), or at least three (a, b, f) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (a, b, e, f). NS, not significant; *P < 0.05, **P < 0.01 and ***P < 0.001.
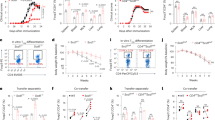
Extended Data Fig. 3 Dynamic regulation of PE metabolic programs in TFH cells.
a, Lipidomic analysis of the lipid content (PC and PS) in naive (CD4+) T cells, wild-type TFH (transduced with sgNTC), and Pcyt2-deficient TFH cells (n = 3 samples, each pooled from multiple mice). b, c, The dependence on Pcyt2 for PE alterations during TFH cell differentiation from naive CD4+ T cells. b, Heat map showing the 39 significantly downregulated PE molecules in Pcyt2-deficient TFH cells as compared with wild-type TFH cells (log2 ratio ≥ 0.5, P < 0.05). c, Venn diagram showing the overlap of upregulated (left) or downregulated (right) PE molecules in wild-type TFH versus naive CD4+ T cells compared to wild-type versus Pcyt2-deficient TFH cells (n = 3 samples, each pooled from multiple mice). d, Lipidomic analysis of the lipid content (PE and PC) in wild-type and Selenoi-deficient TFH cells (n = 3 samples, each pooled from multiple mice). e, Primary TFH and TH1 cells were sorted from LCMV-infected mice and incubated with [3H]-Etn (2 μCi ml−1) for 3 h. To assess PE synthesis, lipids were extracted and [3H]-Etn incorporation into TFH cells was assessed with a scintillation counter (see synthesis column, left graph). To assess PE turnover, after 3 h of [3H]-Etn incubation, cells were washed and chased with unlabelled ethanolamine for another 3 h (see turnover column, left graph). The presence of [3H] radioactivity in cells and culture medium (right graph) was measured with a scintillation counter (n = 4 samples, each pooled from multiple mice). CPM, counts per minute. Data are representative of at least two independent experiments (a–e). Data are mean ± s.e.m. P values are determined one-way ANOVA (a) or by two-tailed unpaired Student’s t-test (d, e). NS, not significant; *P < 0.05, **P < 0.01 and ***P < 0.001.
Extended Data Fig. 4 The CDP–ethanolamine pathway but not the CDP–choline pathway is required for TFH cell differentiation.
a, Diagram summary of the effects of single or combined deficiency of genes in the CDP–ethanolamine and CDP–choline pathways on TFH cell differentiation after LCMV infection. b, Immunoblot analyses of the indicated proteins in the indicated sgRNA-transduced SMARTA cells isolated from the spleen at 3–5 days after adoptive transfer and LCMV infection. Asterisk (*), non-specific band; arrow, the target band. c, Summary of the relative proportions of TFH (CXCR5+SLAM−) and TH1 (CXCR5−SLAM+) cells in donor-derived cells in spleen of mice receiving the indicated sgRNA-transduced SMARTA cells at day 7 after LCMV infection (n = 4 mice). d, Lipidomic analysis of the lipid content (PC and PE) in wild-type TFH (transduced with sgNTC), Pcyt1a-deficient TFH and Pcyt1a and Pcyt1b doubly deficient TFH cells (n = 3 samples, each pooled from multiple mice). e, Flow cytometry analysis (left) and summary of the proportion of wild-type and Pcyt2-deficient CXCR5+ SMARTA cells (middle) and their dilution of CellTrace Violet (CTV; right) at day 2 after LCMV infection (n = 3 mice). f, Summary of apoptotic wild-type and Pcyt2-deficient SMARTA cells as analysed by Annexin V and 7-AAD staining (or unstained) in freshly isolated splenocytes at day 2 after LCMV infection (n = 3 mice). g, Distribution of SMARTA cells in the follicle in the spleen at day 3 after infection. Scale bar, 50 μm (n = 50 sections). h, Diagram of TFH cell effector functional assay. sgNTC (mCherry+) and sgPcyt2 (Ametrine+)-transduced SMARTA cells (CD45.1+) were transferred (first transfer) into C57BL/6 mice (CD45.2+), followed by LCMV infection. Seven days later, the fully differentiated wild-type or Pcyt2-deficient CXCR5+SLAM− TFH cells (CD45.1+) were sorted and equal numbers of these cells were transferred (second transfer) into LCMV-infected mice (CD45.2+; this transfer occurred at day 1 after LCMV infection). GC B cell and plasma cell formation was analysed at day 5 after the second adoptive transfer. Data are representative of one (d) or at least two (b, c, e–g) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (c, d) or by two-tailed unpaired Student’s t-test (e, f). NS, not significant; **P < 0.01 and ***P < 0.001. Numbers in gates indicate percentage of cells.
Extended Data Fig. 5 The CDP–ethanolamine pathway regulates TFH cell differentiation independently of the BCL6–T-bet axis.
a, sgNTC or sgPcyt2-transduced SMARTA cells (CD45.1+) were transferred into C57BL/6 recipients (CD45.2+) that were subsequently infected with LCMV. CD45.1+ cells were isolated at day 3 for transcriptional profiling by microarray. GSEA of the TFH signature in Pcyt2-deficient compared to wild-type T cells (n = 3 samples, each pooled from multiple mice). b, c, sgNTC, sgEtnk1, sgPcyt2 or sgSelenoi-transduced SMARTA cells were transferred into C57BL/6 recipients, followed by infection with LCMV. Quantification of the percentages of BCL6+T-bet– and BCL6−T-bet+ cells among donor-derived T cells in the spleen at day 3 (b) and day 7 (c) after infection (n = 4 mice). d, GSEA of BCL6 target genes (identified by ChIP–seq17) in Pcyt2-deficient compared to wild-type T cells (samples are the same as Extended Data Fig. 5a; n = 3 samples, each pooled from multiple mice). e, GSEA of the TFH-specific genes that are directly downregulated by BCL6 (in total 48 genes, identified by combining ChIP–seq, RNA-seq and microarray datasets17) in Pcyt2-deficient compared to wild-type T cells (samples are the same as Extended Data Fig. 5a; n = 3 samples, each pooled from multiple mice). f, Analysis and quantification of PSGL-1−Ly6C− cells, CXCR5+ cells and CXCR5 MFI among PSGL-1−Ly6C− TFH cells at day 3 after infection (n = 4 mice). g, sgNTC, sgPcyt2, sgBcl6, or dual sgPcyt2 and sgBcl6-transduced SMARTA cells were transferred into C57BL/6 recipients that were subsequently infected with LCMV. Quantification of the proportion of TFH cells (CXCR5+PD-1+ and CXCR5+SLAM−) in donor-derived cells from the spleen at day 7 after infection (n = 4 mice). h, sgNTC, sgEtnk1, sgPcyt2 or sgSelenoi-transduced SMARTA cells (CD45.1+) were transferred into C57BL/6 recipients (CD45.2+) that were subsequently infected with LCMV. Quantification of the proportion of TFH cells (CXCR5+PD-1+) in donor-derived T cells from the spleen at day 3 after infection (n = 4 mice). i–o, SMARTA cells transduced with the indicated sgRNAs were transferred into C57BL/6 recipients, followed by infection with LCMV. i, Quantification of the frequency and MFI of CXCR5 on TFH cells (PSGL-1−Ly6C−) in the spleen at day 2 after infection (n = 3 mice). j, Quantification of the frequency and MFI of PD-1 on TFH cells (PSGL-1−Ly6C−) in the spleen at day 2 after infection (n = 3 mice). k, SMARTA cells were transduced with sgNTC or sgPcyt2 alone or in combination with a retrovirus overexpressing empty vector (Empty-RV) or CXCR5 (CXCR5-RV), followed by adoptive transfer into C57BL/6 mice that were then infected with LCMV. Analysis (top) and quantification (bottom) of the proportion of TFH cells (CXCR5+PD-1+ and PSGL-1−Ly6C−) in donor-derived cells from the spleen at day 7 after infection. Numbers above the graphs indicate fold change (n = 4 mice in CXCR5-RV group of CXCR5+PD-1+ TFH data, n = 3 mice in other groups). l, MFI of PD-1, ICOS, Ly6C, PSGL-1, CD44 and CD62L on CD45.1+ SMARTA cells at day 3 after infection (n = 4 mice). m, Quantification of the frequency and MFI of CXCR3 on CD45.1+ SMARTA cells at day 3 after infection (n = 3 mice). n, MFI of CCR7 expression on donor-derived T cells from the host spleen at day 3 after infection (n = 3 mice). o, Quantification of phosphorylated AKT (pAKT(S473)) levels in purified wild-type or Pcyt2-deficient TFH cells (CXCR5+SLAM−) that were stimulated with CXCL13 for 3 h (n = 4 mice). Data are representative of one (a, d, e), at least two (b, c, g, h), or at least three (f, i–o) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (b, c, f–h, k, l), or by two-tailed unpaired Student’s t-test (i, j, m–o). NS, not significant; *P < 0.05, **P < 0.01 and ***P < 0.001. Numbers in quadrants or gates indicate percentage of cells.
Extended Data Fig. 6 PE is selectively distributed on the outer layer of the plasma membrane of TFH cells but not TH1 cells.
a, Analysis and quantification of surface levels of PE on TFH (PSGL-1−Ly6C−) and TH1 (PSGL-1+Ly6C+) membrane at day 5 after infection (n = 4 mice). b, Quantification of the colocalization index of PE and CCR7 or CXCR5 on PSGL-1−Ly6C− TFH cells in Fig. 3b using the CBC algorithm (PE and CCR7, n = 33 cells; PE and CXCR5, n = 25 cells). A colocalization index value of 1 indicates complete colocalization, a value of 0 represents spatial randomness and a value of −1 indicates intermolecular exclusion. c, Three-dimensional super resolution confocal image of the entire TFH cell on poly-l-lysine-coated coverslips. Surface staining was reconstructed using Imaris software. Scale bars, 2 μm. Layers are translucent to readily visualize both colours. Right, quantification of colocalization index of PE and CCR7 or PE and CXCR5 on PSGL-1−Ly6C− TFH cells (n = 4 mice and 20 cells were quantified). d, Confocal microscopy imaging of PE distribution on TFH (PSGL-1−Ly6C−) and TH1 (PSGL-1+Ly6C+) cells at the indicated time points after LCMV infection. Scale bar, 10 μm (n = 3 mice). e, f, Lipids associated with Flag–CXCR5 or Flag–CCR7 expressed in SMARTA cells isolated from LCMV-infected mice were immunoprecipitated by anti-Flag M2 magnetic beads and quantified using LC–MS/MS. Analysis of lipid content (PE, PC and PS) (e) and heat map showing significantly changed (P < 0.05) PE molecules (f) in the indicated groups (n = 2 samples, each pooled from multiple mice). Asterisks indicate PE molecules with the exact mass. g, SMARTA cells were transduced with the indicated sgRNA and the retrovirus overexpressing empty vector (Empty-RV) or CXCR5 (CXCR5-RV), followed by adoptive transfer into C57BL/6 mice and LCMV infection. Quantification of the proportion of PE+ cells in PSGL-1−Ly6C− TFH cells in donor-derived cells from the spleen at day 7. Numbers above the graphs indicate fold change (n = 4 mice). h, SMARTA cells were transduced with the indicated sgRNAs, followed by adoptive transfer into C57BL/6 mice and LCMV infection. Quantification of the proportion of PE+ cells in donor-derived PSGL-1−Ly6C− TFH cells from the spleen at day 7 after infection (n = 4 mice). i, Analysis and quantification of PE outer-layer membrane distribution on freshly isolated wild-type and Pcyt2-deficient TFH (PSGL-1−Ly6C−) and TH1 (PSGL-1+Ly6C+) cells (TFH, n = 5 mice; TH1, n = 4 mice). j, sgNTC or sgPcyt2-transduced SMARTA cells were transferred into C57BL/6 recipients that were subsequently infected with LCMV. PS exposure in intact TFH (PSGL-1−Ly6C−) and TH1 (PSGL-1+Ly6C+) membrane was assessed by Annexin V and 7-AAD (left) or MFG-E8 and 7-AAD (right) staining analysis at day 5 after infection (n = 4 mice). Data are representative of two (e–h, j), or at least three (a–d, i) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (h, j), or two-tailed unpaired Student’s t-test (a–c, g, i). NS, not significant; ***P < 0.001. Numbers in quadrants or gates indicate percentage of cells.
Extended Data Fig. 7 PE regulates CDP–ethanolamine pathway-mediated TFH responses.
a, Wild-type and Pcyt2-deficient TFH cells were sorted from LCMV-infected mice and incubated with [3H]-Etn (2 μCi ml−1) for 3 h. Lipids were extracted and [3H]-Etn incorporated into TFH cells was assessed with a scintillation counter (n = 4 samples). b–e, Lipid add-back–rescue assay. The sgNTC and sgPcyt2-transduced SMARTA cells were supplemented with the indicated lipids, followed by transfer of these cells to C57BL/6 mice and LCMV infection. b, Diagram showing the time points of lipid treatment. PE was supplemented at days −5, −3 and −1 before adoptive transfer, and LCMV-infected mice were analysed at day 3 after infection (to capture the effects induced by in vitro PE supplementation). c, Lipidomic analysis showing the PE content in the indicated groups (left), and Venn diagram showing the rescued PE molecules by diacyl-type and ether-type PE supplementation (right) at day 3 after infection. d, Analysis and quantification of the proportion of PE+ cells in donor-derived TFH cells (PSGL-1−Ly6C−) from the spleen (n = 5 mice). e, Analysis and quantification of the proportion of donor-derived CXCR5+SLAM− TFH cells from the spleen (n = 5 mice). f, GSEA plot for KEGG signature of ABC transporters in TFH versus TH1 cells (from a public dataset GSE74854 (ref. 37)). g, Heat map showing the top 22 leading-edge genes from the enrichment plot in f. Data are representative of two (a, c) or at least three (d, e) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (d, e) or two-tailed unpaired Student’s t-test (a). *P < 0.05, **P < 0.01 and ***P < 0.001. Numbers in gates indicate percentage of cells.
Extended Data Fig. 8 PE stabilizes surface CXCR5 and prevents it from being targeted for lysosome-mediated degradation.
a, The internalization of surface CXCR5 over time was traced by confocal imaging. b, Wild-type or Pcyt2-deficient SMARTA cells were isolated from LCMV-infected mice and labelled with unconjugated PD-1-specific antibody before incubation at 37 °C. The amount of surface PD-1 remaining over time was detected by fluorophore-conjugated secondary antibody staining (n = 3 mice). c, Flow cytometry analysis of CXCR5 surface expression showed that proteinase K treatment (0.1 mg ml−1) efficiently removed the non-internalized CXCR5 on the cell surface. d, CXCR5 recycling assay. Cell-surface CXCR5 was labelled with unconjugated anti-CXCR5 antibody and incubated at 30 °C to allow internalization of antibody-labelled CXCR5. After washing, the remaining surface-bound antibody was stripped by resuspension in proteinase K, followed by washing and incubation at different time points to allow CXCR5 recycling. The amount of surface CXCR5 recycled over time was detected by fluorophore-conjugated secondary antibody staining and analysed by flow cytometry (left). Right, quantification of percentage of recycled CXCR5 (relative ratio to untreated cells) (n = 4 mice). e, Validation of anti-CXCR5 antibody (clone number, EPR23463-30) by immunoblot analysis of CXCR5 expression in wild-type and CXCR5-deficient SMARTA cells that were isolated from LCMV-infected mice. f, Immunoblot analysis of CXCR5 and PCYT2 expression in wild-type and Pcyt2-deficient SMARTA cells that were isolated from LCMV-infected mice and treated with the indicated concentrations of BafA1 for 8 h. g, Immunoblot analysis of CXCR5 expression in wild-type and Pcyt2-deficient SMARTA cells isolated from LCMV-infected mice after treatment with the proteasome inhibitor MG-132. h, Flow cytometry analysis of CXCR5 surface expression on wild-type and Pcyt2-deficient SMARTA cells that were isolated from LCMV-infected mice and treated with the indicated concentrations of BafA1 for 8 h. i, Quantification of surface PE levels on human CXCR5+ memory TFH, CXCR5+ central memory (TCM) and CD45RA+ naive-like T cells in peripheral blood (n = 4 donors). j, Analysis and quantification of surface PE levels on human tonsil CXCR5+PD-1hi TFH cells, transitional CXCR5+PD-1+ TFH cells, CXCR5− non-TFH and CD45RA+ naive-like T cells (n = 8 donors). Data are representative of two (b, e–h) or at least three (a, c, d, i, j) independent experiments. P values are determined by one-way ANOVA (i, j) or two-tailed unpaired Student’s t-test (b, d). NS, not significant; *P < 0.05 and ***P < 0.001. Numbers in gates indicate percentage of cells.
Extended Data Fig. 9 Loss of Pcyt2 in activated T cells reveals selective impairments of TFH accumulation that are associated with reduced GC responses.
a, Pcyt2 mRNA expression in freshly isolated (day 0) naive CD4+ T cells from wild-type and OX40CrePcyt2fl/fl mice, or after in vitro anti-CD3/CD28 antibody stimulation for the indicated times (n = 4 samples). b, Analysis and quantification of TFH cells (CXCR5+BCL6+ or CXCR5+PD-1+) among B220−CD4+TCRβ+ cells in Peyer’s patches (PPs) and mesenteric lymph nodes (mLNs) from wild-type and OX40CrePcyt2fl/fl mice (8 weeks old; CXCR5+BCL6+ TFH cells, n = 4 mice; CXCR5+PD-1+ TFH cells, n = 3 mice). c, Analysis and quantification of GC B cells (Fas+GL7+) among B220+CD19+ cells in PPs and mLNs from wild-type and OX40CrePcyt2fl/fl mice (n = 3 mice). d, Immunohistochemistry of GCs in the mLNs of wild-type and OX40CrePcyt2fl/fl mice. Scale bar, 50 μm (n = 3 mice). e, Pcyt1a mRNA expression in freshly isolated (day 0) naive CD4+ T cells from wild-type and OX40CrePcyt1afl/fl mice, or after in vitro stimulation with anti-CD3/CD28 antibodies for indicated times (n = 3 samples). f, Analysis and quantification of TFH cells (CXCR5+BCL6+ or CXCR5+PD-1+) among B220−CD4+TCRβ+ cells in PPs and mLNs from wild-type and OX40CrePcyt1afl/fl mice (8 weeks old; n = 3 mice). g, Analysis and quantification of GC B cells (Fas+GL7+) among B220+CD19+ cells in PPs and mLNs from wild-type and OX40CrePcyt1afl/fl mice (n = 3 mice). h, Mixed bone marrow chimaeras were constructed by mixing bone marrow cells from wild-type or OX40CrePcyt2fl/fl mice and CD45.1+ ‘spike’ mice followed by injection into sub-lethally irradiated Rag1−/− recipient mice. Quantification of TFH cells (CXCR5+BCL6+) among CD45.1+B220–CD4+TCRβ+ or CD45.2+B220−CD4+TCRβ+cells in the PPs and mLNs under steady state (n = 3 mice). i, Analysis and quantification of gp66-tetramer-positive CXCR5+PD-1+ TFH cells in the spleen of wild-type and OX40CrePcyt2fl/fl mice at day 7 after infection (n = 4 mice). j, Retrogenic mouse-derived naive CD4+ T cells deficient for Etnk1 or Selenoi were transferred into C57BL/6 mice, followed by LCMV infection. Analyses of the proportion of donor-derived TFH (CXCR5+SLAM−, CXCR5+PD-1+ or PSGL-1−Ly6C−) cells (left) and quantification of TFH cell percentage and number (right) in the spleen at day 7 after infection (n = 4 mice). k, Quantification of numbers of TFH cells (CXCR5+PD-1+, CXCR5+ICOS+ or CXCR5+Ly6C−) or GC B cells in the spleen from wild-type and OX40CrePcyt2fl/fl mice at day 7 after intraperitoneal immunization with NP-OVA and LPS in alum (n = 4 mice). l, Measurements of anti-NP immunoglobulins in the serum from wild-type and OX40CrePcyt2fl/fl mice at day 7 after immunization with NP-OVA and LPS (n = 16, collected from 8 mice). Data are representative of two (a, e–l) or at least three (b–d) independent experiments. Data are mean ± s.e.m. P values are determined by one-way ANOVA (j) or by two-tailed unpaired Student’s t-test (b, c, f–i, k, l). NS, not significant; *P < 0.05, **P < 0.01 and ***P < 0.001. Numbers in gates indicate percentage of cells.
Extended Data Fig. 10 Surface PE distribution and CXCR5 expression on B cells are dependent on the CDP–ethanolamine pathway.
a, Analysis and quantification of the proportions of CXCR5+PE+ cells on mature conventional B cells (B220hiIgDhiIgMlo) or total B220+IgM+ B cells from the spleen, immature B cells (B220+IgM+ cells from the bone marrow (BM)), as well as splenic naive CD4+ T cells (n = 3 mice). b, Quantification of CXCR5 and PE MFI among B220+ cells in spleen, peripheral lymph nodes (pLNs), mesenteric lymph nodes (mLNs) and Peyer’s patches (PPs) of wild-type and Cd19CrePcyt2fl/fl mice (n = 3 mice). c, Proportion of migrated B cells from the spleen of wild-type and Cd19CrePcyt2fl/fl mice was assessed by flow cytometry after treatment with CXCL13 for 3 h (n = 3 mice). d, Two-step model of TFH cell differentiation. Expression of BCL6 and other transcription factors in activated CD4+ T cells orchestrates the initiation and commitment to the TFH program, leading to the induction of chemokine receptor CXCR5 (left cell). Right cell: In BCL6- and CXCR5-expressing TFH cells, the CDP–ethanolamine pathway (composed of ETNK1, PCYT2 and SELENOI), which mediates de novo PE synthesis, acts as a critical post-transcriptional program for the functional maintenance of the TFH program. Maintenance of the TFH program is essential for GC responses, plasma cell formation and antigen-specific immunoglobulin secretion. By contrast, the CDP–choline pathway that promotes PC synthesis and the PISD-dependent decarboxylation of PS are dispensable for TFH accumulation, as indicated by grey shading. e, Mechanistically, the CDP–ethanolamine pathway controls TFH responses by stabilizing CXCR5 surface expression, CXCL13–CXCR5-mediated signalling events and cellular trafficking to B cell follicles. De novo synthesis of PE downstream of the CDP–ethanolamine pathway enables PE to interact with CXCR5 and prevents its targeting for lysosome-mediated degradation. Depletion of the CDP–ethanolamine pathway reduces the stability, accelerates the internalization rate and decreases the surface recycling of CXCR5. Data are representative of at least two (a–c) independent experiments. Data are mean ± s.e.m. P values are determined by two-tailed unpaired Student’s t-test (b, c). NS, not significant; *P < 0.05 and **P < 0.01. Numbers in gates indicate percentage of cells.
Supplementary information
Supplementary Figure 1
This file contains the uncropped Western blot images with size marker indications.
Supplementary Table 1
Analysis of mouse sgRNA metabolic library CRISPR screening data. This file contains the output of the analysis of mouse metabolic library CRISPR screening data at the gene (Tab a) and sgRNA (Tab b) levels, with the first row showing the label identifying each column. For data analysis, FastQ files obtained after sequencing were demultiplexed using the HiSeq Analysis software (Illumina). Single-end reads were trimmed and quality-filtered using the CLC Genomics Workbench v11 (Qiagen) and matched against sgRNA sequences from the sgRNA metabolic library. Read counts for sgRNAs were normalized against total read counts across all samples. For each sgRNA, the fold change (log2-transformed ratio) for enrichment was calculated between each of the biological replicates and the input experiment. After merging the quantification results from two sub-libraries, candidate genes were ranked based on the average enrichment of their six gene-specific sgRNAs in Tfh (CXCR5+SLAM-) relative to Th1 (CXCR5-SLAM+) cells (log2 ratio (Tfh/Th1); adjusted P < 0.05), Tfh cells relative to input (log2 ratio (Tfh/input); adjusted P < 0.05), and Th1 cells relative to input (log2 ratio (Th1/input); adjusted P < 0.05). The gene level false discovery rate-adjusted P value was calculated among multiple sgRNAs (n = 6) for each gene, using a two-tailed paired Student’s t-test between log2-transformed average normalized read counts of Tfh, Th1 or input cells, and the P value was further adjusted using Bonferroni correction with gene size.
Supplementary Table 2
Analysis of lipidomic data in naïve T cells and sgNTC or sgPcyt2-transduced Tfh cells. This file contains the output of the analysis of the lipidomic data from wild-type Tfh, Pcyt2-deficient Tfh and naïve CD4+ T cells, with the first row showing the label for each column. For data analysis, the Thermo Fisher Scientific LipidSearch software (version 4.1) was used for identification and relative quantification of lipids with the following parameters: precursor and product ion mass tolerance of ± 5 ppm; main adducts search (M+H, M-H, M+NH4, M+CH3COO, M+2H, M-2H, M+Na) for mass. All lipid sub-classes were searched within major lipid classes (phospholipids, sphingolipids, glycerolipids and neutral lipids). All individual data files were searched for product ion MS/MS spectra of lipid precursor ions, and the data were merged for each annotated lipid. Compound Discoverer 3.1 (CD31; Thermo Fisher Scientific) software was used for multivariate statistical and differential expression analyses of individual molecules using a pre-defined workflow from CD31: untargeted metabolomics with statistics; detect ID of unknowns using online databases; map compounds to biological pathways using Metabolika. The mass-to-charge (m/z) data from positive or negative ion modes processed from CD31 was combined and then formatted to comma-separated value (CSV) file and imported into MetaboAnalyst 4.0. The peak areas were normalized using the parameters for sample normalization sum, data transformation log10 and range data scaling. After normalization, the fold change (log2-transformed ratio) of lipids between each group was calculated.
Supplementary Table 3
PE molecular composition in Selenoi-deficient Tfh cells and in Pcyt2-deficient Tfh cells upon ether-type or diacyl-type PE supplementation. This file contains two tabs (a, b). Tab a shows the 79 PE molecules that were significantly downregulated (P < 0.05) upon Selenoi deletion, with 47 and 32 PE molecules being ether-type and diacyl-type PE, respectively. Tab b shows that Pcyt2-deficient SMARTA cells supplemented with ether-type (left) or diacyl-type (right) PE resulted in the generation of both species of PE molecules. PE molecules that were restored by either ether-type or diacyl-type PE supplementation are underlined.
Supplementary Table 4
Expression levels of 48 lipid translocases in Tfh and Th1 cells, and CRISPR-based in vivo screening of the 48 lipid translocases. This file contains two tabs (a, b). Tab a shows the average gene expression levels in Tfh and Th1 cells (log2 values, from a public dataset GSE74854) of the 48 translocase genes that we examined by CRISPR-based in vivo functional testing (see Tab b). Tab b shows the data summary of CRISPR-based in vivo functional testing of 48 candidate lipid translocases. SMARTA cells transduced with sgNTC (mCherry+) and the indicated gene-specific sgRNA (Ametrine+) were mixed at a 1:2 ratio and transferred into C57BL/6 hosts followed by LCMV infection. Positive regulators of cell proliferation (middle columns) or Tfh differentiation (right columns) were defined by quantification of the relative proportions of donor-derived cells (normalized to ‘spike’ cells) or Tfh cells (CXCR5+SLAM–, CXCR5+PD-1+, PSGL-1−Ly6C−), respectively (as comparisons, 49.4% reduction and 53.3% increase in Tfh differentiation were observed for deletion of Bcl6 and Blimp1, respectively; data not shown).
Source data
Rights and permissions
About this article
Cite this article
Fu, G., Guy, C.S., Chapman, N.M. et al. Metabolic control of TFH cells and humoral immunity by phosphatidylethanolamine. Nature 595, 724–729 (2021). https://doi.org/10.1038/s41586-021-03692-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-03692-z
This article is cited by
-
Functional CRISPR screens in T cells reveal new opportunities for cancer immunotherapies
Molecular Cancer (2024)
-
Inhibition of choline metabolism in an angioimmunoblastic T-cell lymphoma preclinical model reveals a new metabolic vulnerability as possible target for treatment
Journal of Experimental & Clinical Cancer Research (2024)
-
Metabolic waypoints during T cell differentiation
Nature Immunology (2024)
-
CRISPR screening in hematology research: from bulk to single-cell level
Journal of Hematology & Oncology (2023)
-
CRISPR/Cas9 system: recent applications in immuno-oncology and cancer immunotherapy
Experimental Hematology & Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.