Abstract
Mammaliamorpha comprises the last common ancestor of Tritylodontidae and Mammalia plus all its descendants1. Tritylodontids are nonmammaliaform herbivorous cynodonts that originated in the Late Triassic epoch, diversified in the Jurassic period2,3,4,5 and survived into the Early Cretaceous epoch6,7. Eutriconodontans have generally been considered to be an extinct mammalian group, although different views exist8. Here we report a newly discovered tritylodontid and eutriconodontan from the Early Cretaceous Jehol Biota of China. Eutriconodontans are common in this biota9, but it was not previously known to contain tritylodontids. The two distantly related species show convergent features that are adapted for fossorial life, and are the first ‘scratch-diggers’ known from this biota. Both species also show an increased number of presacral vertebrae, relative to the ancestral state in synapsids or mammals10,11, that display meristic and homeotic changes. These fossils shed light on the evolutionary development of the axial skeleton in mammaliamorphs, which has been the focus of numerous studies in vertebrate evolution12,13,14,15,16,17 and developmental biology18,19,20,21,22,23,24,25,26,27,28. The phenotypes recorded by these fossils indicate that developmental plasticity in somitogenesis and HOX gene expression in the axial skeleton—similar to that observed in extant mammals—was already in place in stem mammaliamorphs. The interaction of these developmental mechanisms with natural selection may have underpinned the diverse phenotypes of body plan that evolved independently in various clades of mammaliamorph.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
In addition to the material presented in the Article and the accessioned specimens, supporting images, graphics and essential phylogenetic data are provided in the Extended Data figures and Supplementary Information. Computed tomography and computed laminography scanning data are available on the Archives of Digital Morphology, Key Laboratory of Vertebrate Evolutionary Systems Science of the Chinese Academy of Sciences (http://www.admorph.org), at https://doi.org/10.12112/M.101 (F. sinensis) and https://doi.org/10.12112/M.102 (J. cheni). Life Science Identifiers for the new genera and species have been registered at Zoobank as: Fossiomanus Mao, Zhang, Liu & Meng, LSIDurn:lsid:zoobank.org:act:C579314D-6330-4D7F-9C71-AF7E8DC9A1DD; Fossiomanus sinensis Mao, Zhang, Liu & Meng, LSIDurn:lsid:zoobank.org:act:3D66E77B-F3A1-4A0D-923B-11121CB934EC; Jueconodon Mao, Zhang, Liu & Meng, LSIDurn:lsid:zoobank.org:act:580A4D81-BFFE-4BE7-BDE9-267ED929CA3A; and Jueconodon cheni Mao, Zhang, Liu & Meng, LSIDurn:lsid:zoobank.org:act:C97D7EF4-68C4-4B09-9E78-BA6F48ADCA09. The data matrix for the phylogenetic analysis is in the Supplementary Information, and has been as deposited in MorphoBank (http://www.morphobank.org; project number 3943).
Code availability
The MrBayes commands for Bayesian analyses are provided in the Supplementary Information.
References
Rowe, T. Definition, diagnosis, and origin of Mammalia. J. Vertebr. Paleontol. 8, 241–264 (1988).
Sues, H.-D. Skull and dentition of two tritylodontid synapsids from the Lower Jurassic of western North America. Bull. Mus. Comp. Zool. 151, 217–268 (1986).
Sues, H.-D. & Jenkins, F. A. Jr. in Amniote Paleobiology: Perspectives on the Evolution of Mammals, Birds, and Reptiles (eds Carrano, M. T. et al.) 114–152 (Univ. Chicago Press, 2006).
Averianov, A. O. et al. A tritylodontid synapsid from the Middle Jurassic of Siberia and the taxonomy of derived tritylodontids. J. Vertebr. Paleontol. 37, e1363767 (2017).
Velazco, P. M., Buczek, A. J. & Novacek, M. J. Two new tritylodontids (Synapsida, Cynodontia, Mammaliamorpha) from the Upper Jurassic, southwestern Mongolia. Am. Mus. Novit. 3874, 1–35 (2017).
Lopatin, A. V. & Agadjanian, A. K. A tritylodont (Tritylodontidae, Synapsida) from the Mesozoic of Yakutia. Dokl. Biol. Sci. 419, 107–110 (2008).
Matsuoka, H., Kusuhashi, N. & Corfe, I. J. A new Early Cretaceous tritylodontid (Synapsida, Cynodontia, Mammaliamorpha) from the Kuwajima Formation (Tetori Group) of central Japan. J. Vertebr. Paleontol. 36, e1112289 (2016).
Krause, D. W. et al. Skeleton of a Cretaceous mammal from Madagascar reflects long-term insularity. Nature 581, 421–427 (2020).
Meng, J. Mesozoic mammals of China: implications for phylogeny and early evolution of mammals. Natl. Sci. Rev. 1, 521–542 (2014).
Narita, Y. & Kuratani, S. Evolution of the vertebral formulae in mammals: a perspective on developmental constraints. J. Exp. Zool. 304B, 91–106 (2005).
Müller, J. et al. Homeotic effects, somitogenesis and the evolution of vertebral numbers in recent and fossil amniotes. Proc. Natl Acad. Sci USA 107, 2118–2123 (2010).
Sánchez‐Villagra, M. R., Narita, Y. & Kuratani, S. Thoracolumbar vertebral number: the first skeletal synapomorphy for afrotherian mammals. Syst. Biodivers. 5, 1–7 (2007).
Asher, R. J., Lin, K. H., Kardjilov, N. & Hautier, L. Variability and constraint in the mammalian vertebral column. J. Evol. Biol. 24, 1080–1090 (2011).
Varela-Lasheras, I. et al. Breaking evolutionary and pleiotropic constraints in mammals: on sloths, manatees and homeotic mutations. Evodevo 2, 11 (2011).
Galis, F. et al. Fast running restricts evolutionary change of the vertebral column in mammals. Proc. Natl Acad. Sci. USA 111, 11401−11406 (2014).
Jones, K. E. et al. Fossils reveal the complex evolutionary history of the mammalian regionalized spine. Science 361, 1249–1252 (2018).
Williams, S. A. et al. Increased variation in numbers of presacral vertebrae in suspensory mammals. Nat. Ecol. Evol. 3, 949–956 (2019).
Burke, A. C., Nelson, C. E., Morgan, B. A. & Tabin, C. Hox genes and the evolution of vertebrate axial morphology. Development 121, 333–346 (1995).
Richardson, M. K., Allen, S. P., Wright, G. M., Raynaud, A. & Hanken, J. Somite number and vertebrate evolution. Development 125, 151–160 (1998).
Wellik, D. M. & Capecchi, M. R. Hox10 and Hox11 genes are required to globally pattern the mammalian skeleton. Science 301, 363–367 (2003).
Dequéant, M.-L. & Pourquié, O. Segmental patterning of the vertebrate embryonic axis. Nat. Rev. Genet. 9, 370–382 (2008).
Gomez, C. et al. Control of segment number in vertebrate embryos. Nature 454, 335–339 (2008).
Hautier, L., Weisbecker, V., Sánchez-Villagra, M. R., Goswami, A. & Asher, R. J. Skeletal development in sloths and the evolution of mammalian vertebral patterning. Proc. Natl Acad. Sci. USA 107, 18903–18908 (2010).
Vinagre, T. et al. Evidence for a myotomal Hox/Myf cascade governing nonautonomous control of rib specification within global vertebral domains. Dev. Cell 18, 655–661 (2010).
Guerreiro, I. et al. Role of a polymorphism in a Hox/Pax-responsive enhancer in the evolution of the vertebrate spine. Proc. Natl Acad. Sci. USA 110, 10682–10686 (2013).
Jurberg, A. D., Aires, R., Varela-Lasheras, I., Nóvoa, A. & Mallo, M. Switching axial progenitors from producing trunk to tail tissues in vertebrate embryos. Dev. Cell 25, 451–462 (2013).
Aires, R. et al. Oct4 is a key regulator of vertebrate trunk length diversity. Dev. Cell 38, 262–274 (2016).
Head, J. J. & Polly, P. D. Evolution of the snake body form reveals homoplasy in amniote Hox gene function. Nature 520, 86–89 (2015).
Lewis, G. E. in The Ecology and Biology of Mammal-like Reptiles (eds Hotton, N. et al.) 295−303 (Smithsonian Institution, 1986).
Chen, M. & Wilson, G. P. A multivariate approach to infer locomotor modes in Mesozoic mammals. Paleobiology 41, 280–312 (2015).
Luo, Z.-X., Chen, P., Li, G. & Chen, M. A new eutriconodont mammal and evolutionary development in early mammals. Nature 446, 288–293 (2007).
Meng, J., Wang, Y. & Li, C. Transitional mammalian middle ear from a new Cretaceous Jehol eutriconodont. Nature 472, 181–185 (2011).
Ji, Q., Luo, Z.-X. & Ji, S.-A. A Chinese triconodont mammal and mosaic evolution of the mammalian skeleton. Nature 398, 326–330 (1999).
Hou, S.-L. & Meng, J. A new eutriconodont mammal from the Early Cretaceous Jehol biota of Liaoning, China. Chin. Sci. Bull. 59, 546–553 (2014).
King, B. & Beck, R. M. D. Tip dating supports novel resolutions of controversial relationships among early mammals. Proc. R. Soc. B 287, 20200943 (2020).
Mao, F. et al. Integrated hearing and chewing modules decoupled in a Cretaceous stem therian mammal. Science 367, 305–308 (2020).
Shimer, H. Adaptations to aquatic, arboreal, fossorial and cursorial habits in mammals. III. Fossorial adaptations. Am. Nat. 37, 819–825 (1903).
Hildebrand, M. in Functional Vertebrate Morphology Vol. 6 (eds Hildebrand, M. et al.) 89–109 (Belknap, 1985).
Salton, J. A. & Sargis, E. J. in Mammalian Evolutionary Morphology: A Tribute to Frederick S. Szalay (eds Sargis, E. J. & Dagosto, M.) 51–71 (Springer, 2008).
Wake, M. H. in The Skull Vol. 3 (eds Hanken, J. & Hall, B. K.) 197–240 (Univ. Chicago Press, 1993).
Kümmell, S., Abdala, F., Sassoon, J. & Abdala, V. Evolution and identity of synapsid carpal bones. Acta Palaeontol. Pol. 65, 649–678 (2020).
Arlegi, M., Veschambre-Couture, C. & Gómez-Olivencia, A. Evolutionary selection and morphological integration in the vertebral column of modern humans. Am. J. Phys. Anthropol. 171, 17–36 (2020).
Oates, A. C., Morelli, L. G. & Ares, S. Patterning embryos with oscillations: structure, function and dynamics of the vertebrate segmentation clock. Development 139, 625–639 (2012).
Wellik, D. M. Hox patterning of the vertebrate axial skeleton. Dev. Dyn. 236, 2454–2463 (2007).
Carapuço, M., Nóvoa, A., Bobola, N. & Mallo, M. Hox genes specify vertebral types in the presomitic mesoderm. Genes Dev. 19, 2116–2121 (2005).
McIntyre, D. C. et al. Hox patterning of the vertebrate rib cage. Development 134, 2981–2989 (2007).
Mallo, M., Wellik, D. M. & Deschamps, J. Hox genes and regional patterning of the vertebrate body plan. Dev. Biol. 344, 7–15 (2010).
Böhmer, C. Correlation between Hox code and vertebral morphology in the mouse: towards a universal model for Synapsida. Zool. Lett. 3, 8 (2017).
Buchholtz, E. A. et al. Fixed cervical count and the origin of the mammalian diaphragm. Evol. Dev. 14, 399–411 (2012).
Luo, Z.-X. et al. Evolutionary development in basal mammaliaforms as revealed by a docodontan. Science 347, 760–764 (2015).
Buchholtz, E. A. Crossing the frontier: a hypothesis for the origins of meristic constraint in mammalian axial patterning. Zoology 117, 64–69 (2014).
Jones, K. E., Benitez, L., Angielczyk, K. D. & Pierce, S. E. Adaptation and constraint in the evolution of the mammalian backbone. BMC Evol. Biol. 18, 172 (2018).
Liu, J. & Olsen, P. The phylogenetic relationships of Eucynodontia (Amniota: Synapsida). J. Mamm. Evol. 17, 151–176 (2010).
McKenna, M. C. & Bell, S. K. Classification of Mammals: Above the Species Level (Columbia Univ. Press, 1997).
O’Leary, M. A. et al. The placental mammal ancestor and the post-K–Pg radiation of placentals. Science 339, 662–667 (2013).
Wallace, R. V. S., Martínez, R. & Rowe, T. First record of a basal mammaliamorph from the early Late Triassic Ischigualasto Formation of Argentina. PLoS ONE 14, e0218791 (2019).
Swofford, D. L. Phylogenetic Analysis Using Parsimony, version 4.0b10 (Sinauer Associates, 2002).
Ronquist, F. & Huelsenbeck, J. P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19, 1572–1574 (2003).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Lewis, P. O. A likelihood approach to estimating phylogeny from discrete morphological character data. Syst. Biol. 50, 913–925 (2001).
Yang, Z. Maximum likelihood phylogenetic estimation from DNA sequences with variable rates over sites: approximate methods. J. Mol. Evol. 39, 306–314 (1994).
Rannala, B., Zhu, T. & Yang, Z. Tail paradox, partial identifiability, and influential priors in Bayesian branch length inference. Mol. Biol. Evol. 29, 325–335 (2012).
Zhang, C., Rannala, B. & Yang, Z. Robustness of compound Dirichlet priors for Bayesian inference of branch lengths. Syst. Biol. 61, 779–784 (2012).
Geyer, C. J. Practical Markov chain Monte Carlo. Stat. Sci. 7, 473–483 (1992).
Ronquist, F. et al. A total-evidence approach to dating with fossils, applied to the early radiation of the hymenoptera. Syst. Biol. 61, 973–999 (2012).
Gavryushkina, A., Welch, D., Stadler, T. & Drummond, A. J. Bayesian inference of sampled ancestor trees for epidemiology and fossil calibration. PLoS Comput. Biol. 10, e1003919 (2014).
Zhang, C., Stadler, T., Klopfstein, S., Heath, T. A. & Ronquist, F. Total-evidence dating under the fossilized birth–death process. Syst. Biol. 65, 228–249 (2016).
Stadler, T. Sampling-through-time in birth-death trees. J. Theor. Biol. 267, 396–404 (2010).
Heath, T. A., Huelsenbeck, J. P. & Stadler, T. The fossilized birth-death process for coherent calibration of divergence-time estimates. Proc. Natl Acad. Sci. USA 111, E2957–E2966 (2014).
Drummond, A. J., Ho, S. Y., Phillips, M. J. & Rambaut, A. Relaxed phylogenetics and dating with confidence. PLoS Biol. 4, e88 (2006).
Chen, M., Luo, Z.-X. & Wilson, G. P. The postcranial skeleton of Yanoconodon allini from the Early Cretaceous of Hebei, China, and its implications for locomotor adaptation in eutriconodontan mammals. J. Vertebr. Paleontol. 37, e1315425 (2017).
Liao, H.-Y., Shen, Y.-B. & Huang, D.-Y. Serrated microstructures on carapaces of Eosestheria (Branchiopoda: Diplostraca) in the Early Cretaceous Jehol biota and discussion on the taxonomic value of these structures. Cretac. Res. 95, 310–317 (2019).
Acknowledgements
We thank the Jinzhou Paleontological Museum for providing one of the studied specimens housed in their collections; J. Hooker, P. Brewer and M. Day (Natural History Museum), B. Lindow (Natural History Museum of Denmark), G. Billet and M. Godinot (Museum National d’Histoire Naturelle) for access to specimens under their curatorial care; S.-H. Xie and S.-J. Li (IVPP) for specimen preparation; D. W. Krause (Denver Museum of Nature and Science), S. Hoffmann (New York Institute of Technology) and M. Ruta (University of Lincoln) for data sharing; Y.-M. Hou, P.-F. Yin and P.-H. Wang (IVPP), S.-Y. Hou, M.-J. Han, Y.-Y. Zhang (Yinghua Inspection and Testing) for computed tomography and computed laminography scanning of the specimens; N. Wong (American Museum of Natural History (AMNH)) for drawing the figures; Z.-H. Zhou, X.-L. Wang, J.-Y. Zhang and F. Jin (IVPP) for discussions of localities and the identification of fossil fishes; H.-Y. Liao (Yunnan University) for identification of Eosestheria; and C.-Y. Yu (AMNH) for data transformation. This work was supported by the National Natural Science Foundation of China (41688103 and 42072002); the Strategic Priority Research Program (B) of the CAS (XDB26000000); the Youth Innovation Promotion Association, CAS (2019076); the Kalbfleisch Fellowship, Richard Gilder Graduate School, AMNH; the 100 Young Talents Program of the CAS.
Author information
Authors and Affiliations
Contributions
F.M. and J.M. conceived the study and wrote the paper; F.M. conducted the computed tomography scan and rendering work; C.L. collected and curated specimens; C.Z. ran the Bayesian analyses; and all authors edited and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Fernando Abdala, P. David Polly, Hans-Dieter Sues and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Holotype of F. sinensis (JZMP-2107500093).
a, Holotype of F. sinensis, primarily in ventral view. b, Composed computed laminographic image of the holotype. The black box in a and the inset at the bottom left of b indicate fish specimens of Jinanichthys, an index vertebrate fossil for the Jiufotang Formation. The close-up views of caudal vertebrae in a, b correspond to the area outlined in a red-dashed line in a.
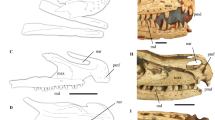
Extended Data Fig. 2 Optical and computed tomography-rendered skulls of F. sinensis.
a, c, f, Ventral view of the skull of F. sinensis. b, d, Dorsal view of the skull of F. sinensis. e, One slice of the computed tomography scanning images of the skull of F. sinensis, showing the contrast quality of the image. f, Preserved positions of upper teeth (white) and lower teeth (yellow). There are six upper postcanines that are visible (indicated as ‘upc’ in a) in the specimen, of which the mesial five are fully erupted and functional; PC6 is still erupting and the rest of the tooth crowns are not fully developed within the maxilla (Extended Data Fig. 3a–d). cev, cervical vertebrae; lin, lower incisor; lpc, lower postcanine(s); na, nasal; occ, occipital condyle; pdu, postdentary unit; syd, symphysis (suture) of the dentary; uin, upper incisor; zya, zygomatic arch.
Extended Data Fig. 3 Tooth morphologies of F. sinensis.
a–d, Occlusal (a), buccal (b), lingual (c) and dorsal (root) (d) views of the right upper postcanine dentition. e, Occlusal view of a left upper postcanine. f, g, Occlusal (f) and dorsal (root) (g) views of two left upper postcanines. Teeth shown in e and f are probably PC1 and PC3, respectively, as judged by their wear. h, i, Buccal (h) and lingual (i) views of two left upper postcanines. j–l, Crown (j), distal (k) and lingual (l) views of a left lower postcanine. m, n, Crown (m) and buccal (n) views for two left lower postcanines. o, Buccal view of a loose left lower postcanine. p–r, Lingual (p), buccal (q) and lateral (r) views of the left upper incisor. s, t, Lateral (s) and lingual (t) views of the right upper incisor. u, v, Lateral (u) and lingual (v) views of the left lower incisor. B, buccal cusp (with number) of upper tooth; L, lingual cusp (with number) of upper tooth; M, medial cusp (with number) of upper tooth; b, buccal cusp (with number) of lower tooth; l, lingual cusp (with number) of lower tooth. PC1 to PC5 are functional postcanine teeth with roots developed, and PC6 is erupting and lacks roots.
Extended Data Fig. 4 Limb structures of F. sinensis.
a, b, Optical (a) and computed laminography (b) images of the forelimb. c, d, Optical (c) and computed laminography (d) images of the hindlimb. as, astragalus; ca, calcaneus; cu, cuboid; dc1, distal carpal 1 (trapezium); dc2, distal carpal 2 (trapezoid); dc3, distal carpal 3 (capitate, magnum); ectc, ectocuneiform; entc, entocuneiform; itl, intermedium (lunate); lcl, lateral centrale (unciform, hamate); mc, metacarpal; mcl, medial centrale (centrale); mec, mesocuneiform; mt, metatarsal; na, navicular; phd, distal phalanx (number referring to digit); phi, intermediate phalanx; php, proximal phalanx; ps, pisiform; rad, radiale (scaphoid); uln, ulnare (triquetrum, cuneiform). The tritylodontid terminology for the carpal bones (labelled in the figure) is based on refs. 3,71. The lateral centrale may be interpreted as a fused distal carpal IV and V41. Intermediate phalanx 5 was broken anteriorly.
Extended Data Fig. 5 Holotype of J. cheni (ZGY0052).
a, Holotype of J. cheni in dorsal view (optical photograph). b, Composed computed laminography image of the holotype. The black boxes in a highlight carapaces of the conchostracan Eosestheria, an index invertebrate fossil in the Yixian Formation72. The inset in the bottom right of a shows the structure of Eosestheria. The computed laminography image of the right arm was digitally placed to closer to its original position in b. -l, left side; -r, right side; ac, acetabulum; cp, carpus; mp, metacarpus; na, navicular; nap, nasal anterior process; ph, phalanx; sm, sternum.
Extended Data Fig. 6 Optical and computed tomography-rendered skulls of J. cheni.
a, b, Ventral view of the skull of J. cheni. c, d, Dorsal view of the skull of J. cheni. cop, coronoid process; glf, glenoid fossa; mac, mandibular condyle; nap, nasal projections; ocp, occiput; omc, ossified Meckelian cartilage; ors, orbital socket; pm, promontorium; pop, postorbital process.
Extended Data Fig. 7 Computed tomography-rendered dentition of J. cheni.
a, b, Original preservation of the dentition. c–f, Digitally restored dentition. Arrows point to tooth germs in the dentition.
Extended Data Fig. 8 The forelimbs and hindlimbs of J. cheni.
a, b, Optic (a) and computed laminography (b) images of the left forelimb. c, d, Optic (c) and computed laminography (d) images of the right hindlimb. e, f, Optic (e) and computed laminography (f) images of the right forearm. na, navicular; ph, phalanges; phi, intermediate phalanx (number referring to digit); php, proximal phalanx (number referring to digit). Images in a–d are shown on the same scale.
Extended Data Fig. 9 The consensus tree using Bayesian phylogenetic tip-dating analysis.
In the consensus tree (50% majority rule), the node ages (divergence times) are the median estimates (numbers shown in small font) and node bars (blue) represent the 95% highest posterior density intervals. Fossoriality and meristic or homeotic changes in various taxa and clades are mapped on the tree. Red dots indicate major clades of mammaliamorph. Red bar highlights the paraphyletic eutriconodontans. Extant taxa are not shown (indicated by green arrows); see Extended Data Fig. 10b for trees including extant species.
Extended Data Fig. 10 Strict consensus of most parsimonious trees and result of non-clock Bayesian analysis.
a, Strict consensus of 319 most parsimonious trees obtained in this study. The data matrix consists of 121 generic-level taxa and 551 characters. All characters are unordered and have equal weight. Two characters are parsimony-uninformative (informative for Bayesian analyses). Tree length = 2,864; consistency index = 0.321; retention index = 0.790; rescaled consistency index = 0.254; homoplasy index = 0.679. The two species described in this Article are denoted by black dots. b, Result of nonclock Bayesian analysis using Lewis Mkv model with gamma rate variation across characters (Mkv+Γ). Numbers at selected nodes are posterior clade probabilities. Black bar indicates the position of the taxa described in this Article. Arrow indicates the branch connection. More details are provided in Methods and Supplementary Information.
Supplementary information
Supplementary Information
This file contains additional supplementary material, including: definition of vertebral region, sources for vertebral formula, sources for fossoriality, meristic-homeotic changes, chronological constraints, character list, references, and results of phylogenetic analyses.
Rights and permissions
About this article
Cite this article
Mao, F., Zhang, C., Liu, C. et al. Fossoriality and evolutionary development in two Cretaceous mammaliamorphs. Nature 592, 577–582 (2021). https://doi.org/10.1038/s41586-021-03433-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-03433-2
This article is cited by
-
Jurassic shuotheriids show earliest dental diversification of mammaliaforms
Nature (2024)
-
Divergent vertebral formulae shape the evolution of axial complexity in mammals
Nature Ecology & Evolution (2023)
-
Middle ear innovation in Early Cretaceous eutherian mammals
Nature Communications (2023)
-
Petrosal morphology of the Early Cretaceous triconodontid Astroconodon from the Cloverly Formation (Montana, USA)
Journal of Mammalian Evolution (2023)
-
A long-tailed marine reptile from China provides new insights into the Middle Triassic pachypleurosaur radiation
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.