Abstract
Tumours often contain B cells and plasma cells but the antigen specificity of these intratumoral B cells is not well understood1,2,3,4,5,6,7,8. Here we show that human papillomavirus (HPV)-specific B cell responses are detectable in samples from patients with HPV-positive head and neck cancers, with active production of HPV-specific IgG antibodies in situ. HPV-specific antibody secreting cells (ASCs) were present in the tumour microenvironment, with minimal bystander recruitment of influenza-specific cells, suggesting a localized and antigen-specific ASC response. HPV-specific ASC responses correlated with titres of plasma IgG and were directed against the HPV proteins E2, E6 and E7, with the most dominant response against E2. Using intratumoral B cells and plasma cells, we generated several HPV-specific human monoclonal antibodies, which exhibited a high degree of somatic hypermutation, consistent with chronic antigen exposure. Single-cell RNA sequencing analyses detected activated B cells, germinal centre B cells and ASCs within the tumour microenvironment. Compared with the tumour parenchyma, B cells and ASCs were preferentially localized in the tumour stroma, with well-formed clusters of activated B cells indicating ongoing germinal centre reactions. Overall, we show that antigen-specific activated and germinal centre B cells as well as plasma cells can be found in the tumour microenvironment. Our findings provide a better understanding of humoral immune responses in human cancer and suggest that tumour-infiltrating B cells could be harnessed for the development of therapeutic agents.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The following protein sequences were used for generating recombinant HPV proteins: E2 (Uniprot P03120), E6 (Uniprot03126) and E7 (Uniprot P03129). RNA-seq and scRNA-seq data are available in the NCBI Gene Expression Omnibus (GEO) database under the SuperSeries accession numbers GSE149327 and GSE153559, respectively. Normalized gene expression counts of sorted B cell subsets are available in Supplementary Table 1. HPV-specific monoclonal antibodies are available with a completed Material Transfer Agreement. Other relevant data are available from the corresponding authors upon reasonable request.
Code availability
Custom code for RNA-seq and scRNA-seq is available from the corresponding authors upon reasonable request.
References
Cabrita, R. et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 577, 561–565 (2020).
Helmink, B. A. et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature 577, 549–555 (2020).
Petitprez, F. et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 577, 556–560 (2020).
Flynn, N. J., Somasundaram, R., Arnold, K. M. & Sims-Mourtada, J. The multifaceted roles of B cells in solid tumors: emerging treatment opportunities. Target. Oncol. 12, 139–152 (2017).
Yuen, G. J., Demissie, E. & Pillai, S. B lymphocytes and cancer: a love-hate relationship. Trends Cancer 2, 747–757 (2016).
Shalapour, S. et al. Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy. Nature 521, 94–98 (2015).
Kroeger, D. R., Milne, K. & Nelson, B. H. Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. Clin. Cancer Res. 22, 3005–3015 (2016).
Lechner, A. et al. Tumor-associated B cells and humoral immune response in head and neck squamous cell carcinoma. OncoImmunology 8, 1535293 (2019).
Ang, K. K. et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 363, 24–35 (2010).
Nulton, T. J., Olex, A. L., Dozmorov, M., Morgan, I. M. & Windle, B. Analysis of The Cancer Genome Atlas sequencing data reveals novel properties of the human papillomavirus 16 genome in head and neck squamous cell carcinoma. Oncotarget 8, 17684–17699 (2017).
Marur, S., D’Souza, G., Westra, W. H. & Forastiere, A. A. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 11, 781–789 (2010).
Mei, H. E. et al. Blood-borne human plasma cells in steady state are derived from mucosal immune responses. Blood 113, 2461–2469 (2009).
Ward, M. J. et al. Tumour-infiltrating lymphocytes predict for outcome in HPV-positive oropharyngeal cancer. Br. J. Cancer 110, 489–500 (2014).
Lang Kuhs, K. A. et al. Characterization of human papillomavirus antibodies in individuals with head and neck cancer. Cancer Epidemiol. 42, 46–52 (2016).
Lang Kuhs, K. A. et al. Human papillomavirus 16 E6 antibodies are sensitive for human papillomavirus-driven oropharyngeal cancer and are associated with recurrence. Cancer 123, 4382–4390 (2017).
Dahlstrom, K. R. et al. Diagnostic accuracy of serum antibodies to human papillomavirus type 16 early antigens in the detection of human papillomavirus-related oropharyngeal cancer. Cancer 123, 4886–4894 (2017).
Ellebedy, A. H. et al. Defining antigen-specific plasmablast and memory B cell subsets in human blood after viral infection or vaccination. Nat. Immunol. 17, 1226–1234 (2016).
Davis, C. W. et al. Longitudinal analysis of the human B cell response to Ebola virus infection. Cell 177, 1566–1582 (2019).
Sullivan, N. L. et al. Breadth and functionality of Varicella-Zoster virus glycoprotein-specific antibodies identified after Zostavax vaccination in humans. J. Virol. 92, e00269-18 (2018).
Li, G. M. et al. Pandemic H1N1 influenza vaccine induces a recall response in humans that favors broadly cross-reactive memory B cells. Proc. Natl Acad. Sci. USA 109, 9047–9052 (2012).
Scheid, J. F. et al. Broad diversity of neutralizing antibodies isolated from memory B cells in HIV-infected individuals. Nature 458, 636–640 (2009).
Pereira, J. P., Kelly, L. M., Xu, Y. & Cyster, J. G. EBI2 mediates B cell segregation between the outer and centre follicle. Nature 460, 1122–1126 (2009).
Barrena, S. et al. Aberrant expression of tetraspanin molecules in B-cell chronic lymphoproliferative disorders and its correlation with normal B-cell maturation. Leukemia 19, 1376–1383 (2005).
Arce, S. et al. CD38 low IgG-secreting cells are precursors of various CD38 high-expressing plasma cell populations. J. Leukoc. Biol. 75, 1022–1028 (2004).
Green, J. A. et al. The sphingosine 1-phosphate receptor S1P2 maintains the homeostasis of germinal center B cells and promotes niche confinement. Nat. Immunol. 12, 672–680 (2011).
Sharonov, G. V., Serebrovskaya, E. O., Yuzhakova, D. V., Britanova, O. V. & Chudakov, D. M. B cells, plasma cells and antibody repertoires in the tumour microenvironment. Nat. Rev. Immunol. 20, 294–307 (2020).
Sautès-Fridman, C., Petitprez, F., Calderaro, J. & Fridman, W. H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 19, 307–325 (2019).
Skeate, J. G., Woodham, A. W., Einstein, M. H., Da Silva, D. M. & Kast, W. M. Current therapeutic vaccination and immunotherapy strategies for HPV-related diseases. Hum. Vaccin. Immunother. 12, 1418–1429 (2016).
Chaturvedi, A. K. et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J. Clin. Oncol. 29, 4294–4301 (2011).
Krishna, S. et al. Human papilloma virus specific immunogenicity and dysfunction of CD8+ T cells in head and neck cancer. Cancer Res. 78, 6159–6170 (2018).
Dahlstrom, K. R. et al. HPV serum antibodies as predictors of survival and disease progression in patients with HPV-positive squamous cell carcinoma of the oropharynx. Clin. Cancer Res. 21, 2861–2869 (2015).
Wieland, A. & Ahmed, R. Fc receptors in antimicrobial protection. Curr. Top. Microbiol. Immunol. 423, 119–150 (2019).
Kamphorst, A. O. et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science 355, 1423–1427 (2017).
de León, D. C. et al. Human papillomavirus (HPV) in breast tumors: prevalence in a group of Mexican patients. BMC Cancer 9, 26 (2009).
Smith, K. et al. Rapid generation of fully human monoclonal antibodies specific to a vaccinating antigen. Nat. Protocols 4, 372–384 (2009).
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F. & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
DeTomaso, D. & Yosef, N. FastProject: a tool for low-dimensional analysis of single-cell RNA-Seq data. BMC Bioinformatics 17, 315 (2016).
Milpied, P. et al. Human germinal center transcriptional programs are de-synchronized in B cell lymphoma. Nat. Immunol. 19, 1013–1024 (2018).
Hudson, W. H. et al. Expression of novel long noncoding RNAs defines virus-specific effector and memory CD8+ T cells. Nat. Commun. 10, 196 (2019).
Zerbino, D. R. et al. Ensembl 2018. Nucleic Acids Res. 46, D754–D761 (2018).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Lê, S., Josse, J. & Husson, F. FactoMineR: AnRPackage for Multivariate Analysis. J. Stat. Softw. 25, 1–18 (2008).
Wilkerson, M. D. & Hayes, D. N. ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics 26, 1572–1573 (2010).
Acknowledgements
This work was supported by funding from the Ambrose Monell Foundation (R.A.), a Winship Invest$ Pilot grant (to R.A., Z.G.C. and N.F.S.), and NCI grant 1-R00-CA197891 (H.T.K.). W.H.H. is a Cancer Research Institute Irvington Fellow supported by the Cancer Research Institute. We would like to acknowledge the pathology personnel involved in sample handling, the Emory Flow Cytometry Core supported by the National Center for Georgia Clinical and Translational Science Alliance of the National Institutes of Health (NIH) under award number UL1TR002378, and the Yerkes NHP Genomics Core, which is supported in part by NIH P51 OD011132. We would also like to thank C. W. Davis and L. J. Sudmeier for feedback on the manuscript.
Author information
Authors and Affiliations
Contributions
A.W. conceived and designed the project. A.W. and R.A. designed experiments and wrote the manuscript. A.W. performed most of the experiments (including cell isolation, ELISPOT, serology, flow cytometry and monoclonal antibody generation) and analysed the generated data. C.S.E. performed MBC assays and helped with flow cytometry experiments. M.A.C. and H.T.K. analysed scRNA-seq data. W.H.H. analysed bulk RNA-seq data. R.C.O. performed and analysed multiplex immunohistochemistry experiments. M.R.P. collected and provided human specimens, and analysed patient data. C.C.G. and X.W. handled human specimens. N.F.S. and Z.G.C. initiated the clinical specimen protocol. All authors contributed to the revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A.W. and R.A. are inventors on a patent (US patent application no. 16/971,627) filed by Emory University relating to HPV-specific monoclonal antibodies and HPV E2 as potential immunological target in HPV-positive cancers. All other authors declare no competing interests.
Additional information
Peer review information Nature thanks Shiv Pillai and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 ASC responses in patients with HPV-positive HNSCC.
a, Sequencing-based HPV genotyping of p16+ HNSCC cases (n = 32). b, Representative ELISPOT showing total ASCs among lymphocytes from metLNs, primary tumours (TILs) and PBMCs of a patient with p16+ HNSCC. c–e, Summary graphs showing the frequency of ASCs producing IgG, IgA and IgM among lymphocytes from metLNs (n = 37) (c), TILs (n = 22) (d) or PBMCs (n = 39) of patients with p16+ HNSCC (e). Data are mean ± s.e.m. ***P = 0.0001, ****P < 0.0001 (c); **P = 0.0021, ****P < 0.0001 (d); ****P < 0.0001 (e), Friedman test with two-sided Dunn’s multiple comparisons test. Not significant (ns) = 0.1720. f, Representative ELISPOT of E2/6/7-specific IgG-secreting ASCs in PBMCs, metLNs and TILs of a patient with p16+ HNSCC. MBP indicates negative control. g, Correlation (Spearman) of antigen-specific IgG-secreting ASCs in metLNs and TILs (n = 18 patients) with r = 0.7536 and P < 0.0001.
Extended Data Fig. 2 Patients with HPV-negative HNSCC exhibit reduced lymphocyte infiltration into the tumour and lack HPV-specific ASCs.
a, Number of isolated lymphocytes per gram primary tumour in patients with p16+ (n = 35) and p16− (n = 9) HNSCC. ***P = 0.0007, two-tailed Mann–Whitney test. b, Frequency of ASCs producing IgG, IgA and IgM among lymphocytes from metLNs of patients with p16+ (n = 37) and p16− (n = 6 for IgG, n = 5 for IgA and IgM) HNSCC. c, Frequency of E2/6/7-specific IgG-secreting ASCs among total IgG-secreting ASCs in metLNs (n = 6) and TILs (n = 1) of patients with p16− HNSCC. d, Frequency of E2/6/7- and influenza-specific IgG+ MBCs among total IgG+ MBCs in the peripheral blood of patients with p16+ (n = 27) and p16− (n = 9) HNSCC. Numbers indicate detected responses among tested samples. Data are mean ± s.e.m.
Extended Data Fig. 3 Serological analyses.
a–c, IgG titres against E2 (a), E6 (b) and E7 (c) in plasma of healthy individuals (n = 50) and patients with p16+ HNSCC (n = 39). Data are median and quartiles. ****P < 0.0001, two-tailed Mann–Whitney test. d, e, E2/6/7-specific IgA (d) and IgM (e) titres in plasma of patients with p16+ HNSCC (n = 39). f, E2/6/7-specific IgG titres in plasma of p16+ patients and graph demonstrating an IgG response against at least two HPV proteins in the vast majority of patients (n = 39). Data are median and quartiles. ***P = 0.0006, Friedman test with two-sided Dunn’s multiple comparisons test. ns, 0.1852. g, Heat map showing E2/6/7-specific IgG antibody titres in patients with p16+ HNSCC (n = 39) with each column representing a patient. h, Correlation (Spearman) between E2/6/7-specific IgG+ ASCs in primary tumour and E2/6/7-specific IgG titres in plasma (n = 18 patients) with r = 0.7343 and P < 0.0001.
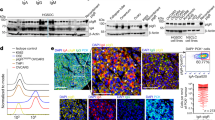
Extended Data Fig. 4 Human monoclonal antibodies against HPV E antigens.
a, Clustered binding pattern of E2-specific monoclonal antibodies performed by competition ELISA. Recognition of linear epitopes was determined by western blot. b, ELISA of E6-specific antibodies (21E2, 21E11, 21H3) generated from single-cell sorted ASCs from metLNs of a patient with HPV+ HNSCC. An E2-specific monoclonal antibody 22B10) is shown as negative control. a.u., arbitrary units. c, Number of SHMs in the Vh and Vl chain of E6-specific monoclonal antibodies (n = 3) with indicated mean.
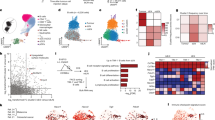
Extended Data Fig. 5 Activated cells of the B cell lineage from the TME are present in distinct clusters.
a, UMAP plots showing enrichment for ABC, ASC, GCB and proliferation gene sets. b, Violin plots showing gene set enrichment scores among the four clusters identified by scRNA-seq. P values determined by two-sided Pearson’s Chi-squared test for binary variables with Yates continuity correction. c, UMAP plots showing expression of selected genes. d, UMAP plots showing distribution of cells of the indicated patient and tissue origin (in red) among the identified clusters. Bar graphs quantifying the composition of the respective sample in terms of frequency among the identified clusters: ASCs, ABCs, GCBs and transitory cells.
Extended Data Fig. 6 Gene expression of cytokines and other immunomodulators by B cells and plasma cells in the TME.
a, Flow plots showing the presence of ASCs and ABCs but absence of germinal centre (GC) B cells in the peripheral blood of a healthy volunteer 7 days after vaccination with Fluarix. b, ASC ELISPOT showing total IgG/A/M-secreting cells (top) and influenza-specific IgG/A/M-secreting cells (bottom) in PBMCs 7 days after vaccination with Fluarix. c, Representative histogram of ASCs from peripheral blood (red) or metLNs (blue) of patients with p16+ HNSCC showing Ki67 expression. Numbers indicate frequency of Ki67+ cells among total ASCs. Summary graph showing paired frequencies of Ki67-expressing ASCs in PBMCs, metLNs and TILs (n = 14). ****P < 0.0001, paired two-tailed t-test. d, Heat map showing gene expression (normalized reads) of selected cytokines and immunomodulators as well as CD19 and CXCR5 as reference. Immunomodulators related to B cells and previously described as negative regulators in the TME are highlighted in red. An expression threshold was set to 50 normalized reads, with reads less than 50 displayed in white.
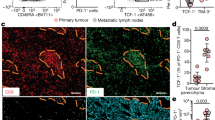
Extended Data Fig. 7 Multiplex immunohistochemistry analysis of B cells and ASCs in the TME.
a, Representative multiplex immunohistochemistry (mIHC) section of HPV+ HNSCC tumour (n = 7) with B cell infiltrates and associated germinal centres (white arrows) (see also Fig. 5e). Seven-colour composite mIHC images of CD19, CD20, Ki67, IRF4, CD138, P16 and DAPI (left), individual images of CD20, CD19, Ki67, and IRF4 (middle), and high magnification (right) of a region of interest (white box). b, Frequency of Ki67+ and CD138+ ASCs (CD19+CD20−IRF4+) in mIHC sections of seven HPV+ HNSCC tumours. Data are mean ± s.e.m. c, Quantification of B cells (CD19+CD20+), ABCs (CD19+CD20+Ki67+), and ASCs (CD19+CD20−IRF4+) in the stroma and tumour parenchyma of three patients with HPV− HNSCC.
Extended Data Fig. 8 Gating strategy for isolation and analysis of B cell subsets.
Gating strategy for B cell subsets used for flow cytometric analyses, bulk RNA-seq analyses, scRNA-seq analyses or the generation of E2-specific monoclonal antibodies. B cell subsets used for bulk RNA-seq analyses are highlighted in red: ASCs, ABCs and GCBs.
Supplementary information
Supplementary Table 1
RNA-seq data of B cell subsets. Normalized gene expression is shown.
Rights and permissions
About this article
Cite this article
Wieland, A., Patel, M.R., Cardenas, M.A. et al. Defining HPV-specific B cell responses in patients with head and neck cancer. Nature 597, 274–278 (2021). https://doi.org/10.1038/s41586-020-2931-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2931-3
This article is cited by
-
B cells in head and neck squamous cell carcinoma: current opinion and novel therapy
Cancer Cell International (2024)
-
The effect of physical exercise on anticancer immunity
Nature Reviews Immunology (2024)
-
The tumor ecosystem in head and neck squamous cell carcinoma and advances in ecotherapy
Molecular Cancer (2023)
-
Germinal center-dependent and -independent immune responses of tumor-infiltrating B cells in human cancers
Cellular & Molecular Immunology (2023)
-
Improving head and neck cancer therapies by immunomodulation of the tumour microenvironment
Nature Reviews Cancer (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.