Abstract
Southeast Asia has emerged as an important region for understanding hominin and mammalian migrations and extinctions. High-profile discoveries have shown that Southeast Asia has been home to at least five members of the genus Homo1,2,3. Considerable turnover in Pleistocene megafauna has previously been linked with these hominins or with climate change4, although the region is often left out of discussions of megafauna extinctions. In the traditional hominin evolutionary core of Africa, attempts to establish the environmental context of hominin evolution and its association with faunal changes have long been informed by stable isotope methodologies5,6. However, such studies have largely been neglected in Southeast Asia. Here we present a large-scale dataset of stable isotope data for Southeast Asian mammals that spans the Quaternary period. Our results demonstrate that the forests of the Early Pleistocene had given way to savannahs by the Middle Pleistocene, which led to the spread of grazers and extinction of browsers—although geochronological limitations mean that not all samples can be resolved to glacial or interglacial periods. Savannahs retreated by the Late Pleistocene and had completely disappeared by the Holocene epoch, when they were replaced by highly stratified closed-canopy rainforest. This resulted in the ascendency of rainforest-adapted species as well as Homo sapiens—which has a unique adaptive plasticity among hominins—at the expense of savannah and woodland specialists, including Homo erectus. At present, megafauna are restricted to rainforests and are severely threatened by anthropogenic deforestation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All accession numbers, and data generated and used during this study, are included in the Article and its Supplementary Information.
References
Détroit, F. et al. A new species of Homo from the Late Pleistocene of the Philippines. Nature 568, 181–186 (2019).
Kaifu, Y. Archaic hominin populations in Asia before the arrival of modern humans: their phylogeny and implications for the “Southern Denisovans”. Curr. Anthropol. 58, S418–S433 (2017).
Reich, D. et al. Denisova admixture and the first modern human dispersals into Southeast Asia and Oceania. Am. J. Hum. Genet. 89, 516–528 (2011).
Louys, J., Curnoe, D. & Tong, H. Characteristics of Pleistocene megafauna extinctions in Southeast Asia. Palaeogeogr. Palaeoclimatol. Palaeoecol. 243, 152–173 (2007).
Klein, R. G. Stable carbon isotopes and human evolution. Proc. Natl Acad. Sci. USA 110, 10470–10472 (2013).
Cerling, T. E. et al. Woody cover and hominin environments in the past 6 million years. Nature 476, 51–56 (2011).
Heaney, L. R. in Tropical Forests and Climate (ed. Myers, N.) 53–61 (Springer, 1991).
Bird, M. I., Taylor, D. & Hunt, C. Palaeoenvironments of insular Southeast Asia during the Last Glacial Period: a savanna corridor in Sundaland? Quat. Sci. Rev. 24, 2228–2242 (2005).
Louys, J. & Turner, A. Environment, preferred habitats and potential refugia for Pleistocene Homo in Southeast Asia. C. R. Palevol 11, 203–211 (2012).
Dennell, R. & Roebroeks, W. An Asian perspective on early human dispersal from Africa. Nature 438, 1099–1104 (2005).
van den Bergh, G. D., de Vos, J. & Sondaar, P. Y. The Late Quaternary palaeogeography of mammal evolution in the Indonesian Archipelago. Palaeogeogr. Palaeoclimatol. Palaeoecol. 171, 385–408 (2001).
Steiper, M. E. Population history, biogeography, and taxonomy of orangutans (Genus: Pongo) based on a population genetic meta-analysis of multiple loci. J. Hum. Evol. 50, 509–522 (2006).
Patel, R. P. et al. Two species of Southeast Asian cats in the genus Catopuma with diverging histories: an island endemic forest specialist and a widespread habitat generalist. R. Soc. Open Sci. 3, 160350 (2016).
Cannon, C. H., Morley, R. J. & Bush, A. B. G. The current refugial rainforests of Sundaland are unrepresentative of their biogeographic past and highly vulnerable to disturbance. Proc. Natl Acad. Sci. USA 106, 11188–11193 (2009).
Sun, X., Li, X., Luo, Y. & Chen, X. The vegetation and climate at the last glaciation on the emerged continental shelf of the South China Sea. Palaeogeogr. Palaeoclimatol. Palaeoecol. 160, 301–316 (2000).
Louys, J. & Meijaard, E. Palaeoecology of Southeast Asian megafauna-bearing sites from the Pleistocene and a review of environmental changes in the region. J. Biogeogr. 37, 1432–1449 (2010).
Raes, N. et al. Historical distribution of Sundaland’s dipterocarp rainforests at Quaternary glacial maxima. Proc. Natl Acad. Sci. USA 111, 16790–16795 (2014).
Handiani, D. et al. Tropical vegetation response to Heinrich Event 1 as simulated with the UVic ESCM and CCSM3. Clim. Past Discuss. 8, 5359–5387 (2012).
Chabangborn, A., Brandefelt, J. & Wohlfarth, B. Asian monsoon climate during the Last Glacial Maximum: palaeo-data–model comparisons: LGM Asian monsoon climate. Boreas 43, 220–242 (2014).
Levin, N. E. et al. Herbivore enamel carbon isotopic composition and the environmental context of Ardipithecus at Gona, Ethiopia. Geol. S. Am. S. 446, 215–235 (2008).
Cerling, T. E., Hart, J. A. & Hart, T. B. Stable isotope ecology in the Ituri Forest. Oecologia 138, 5–12 (2004).
Secord, R., Wing, S. L. & Chew, A. Stable isotopes in early Eocene mammals as indicators of forest canopy structure and resource partitioning. Paleobiology 34, 282–300 (2008).
Fannin, L. D. & McGraw, W. S. Does oxygen stable isotope composition in primates vary as a function of vertical stratification or folivorous behaviour? Folia Primatol. 91, 219–227 (2020).
Clark, P. U. et al. The middle Pleistocene transition: characteristics, mechanisms, and implications for long-term changes in atmospheric pCO2. Quat. Sci. Rev. 25, 3150–3184 (2006).
Sarr, A. C. et al. Subsiding Sundaland. Geology 47, 119–122 (2019).
Di Nezio, P. N. et al. The climate response of the Indo-Pacific warm pool to glacial sea level. Paleoceanogr 31, 866–894 (2016).
Roberts, P. et al. Isotopic evidence for initial coastal colonization and subsequent diversification in the human occupation of Wallacea. Nat. Commun. 11, 2068 (2020).
Barker, G. & Farr, L. E. Archaeological Investigations in the Niah Caves, Sarawak, The Archaeology of Niah Caves, Sarawak (McDonald Institute Monographs, 2016).
Piper, P. J. & Rabett, R. J. Hunting in a tropical rainforest: evidence from the Terminal Pleistocene at Lobang Hangus, Niah Caves, Sarawak. Int. J. Osteoarchaeol. 19, 551–565 (2009).
Steiner, C. C., Houck, M. L. & Ryder, O. A. Genetic variation of complete mitochondrial genome sequences of the Sumatran rhinoceros (Dicerorhinus sumatrensis). Conserv. Genet. 19, 397–408 (2018).
Sodhi, N. S., Koh, L. P., Brook, B. W. & Ng, P. K. Southeast Asian biodiversity: an impending disaster. Trends Ecol. Evol. 19, 654–660 (2004).
Spehar, S. N. et al. Orangutans venture out of the rainforest and into the Anthropocene. Sci. Adv. 4, e1701422 (2018).
Craig, H. The geochemistry of the stable carbon isotope. Geochim. Cosmochim. Acta 3, 53–92 (1953).
Smith, B. N. & Epstein, S. Two categories of 13C/12C ratios for higher plants. Plant Physiol. 47, 380–384 (1971).
Tieszen, L. L. Natural variations in the carbon isotope values of plants: implications for archaeology, ecology, and paleoecology. J. Archaeol. Sci. 18, 227–248 (1991).
Sponheimer, M. et al. Do “savanna” chimpanzees consume C4 resources? J. Hum. Evol. 51, 128–133 (2006).
Sponheimer, M. et al. Isotopic evidence of early hominin diets. Proc. Natl Acad. Sci. USA 110, 10513–10518 (2013).
Codron, J. et al. Stable isotope series from elephant ivory reveal lifetime histories of a true dietary generalist. Proc. R. Soc. Lond. B 279, 2433–2441 (2012).
Crowley, B. E. et al. Extinction and ecology retreat in a community of primates. Proc. R. Soc. Lond. B 279, 3597–3605 (2012).
Farquhar, G. D., Ehleringer, J. R. & Hubick, K. T. Carbon isotope discrimination and photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 40, 503–537 (1989).
van der Merwe, N. J. & Medina, E. The canopy effect, carbon isotope ratios and foodwebs in Amazonia. J. Archaeol. Sci. 18, 249–259 (1991).
Pearcy, R. W. & Pfitsch, W. A. Influence of sunflecks on the δ13C of Adenocaulon bicolor plants occurring in contrasting forest understory microsites. Oecologia 86, 457–462 (1991).
Bonafini, M., Pellegrini, M., Ditchfield, P. & Pollard A. M. Investigation of the ‘canopy effect’ in the isotope ecology of temperate woodlands. J. Archaeol. Sci. 40, 3926–3935 (2013).
Ehleringer, J. R., Rundel, P. W. & Nagy, K. A. Stable isotopes in physiological ecology and food web research. Trends Ecol. Evol. 1, 42–45 (1986).
van der Merwe, N. J. & Medina, E. Photosynthesis and 13C/12C ratios in Amazonian rainforests. Geochim. Cosmochim. Acta 53, 1091–1094 (1989).
Ometto, J. P. H. B. et al. The stable carbon and nitrogen isotopic composition of vegetation in tropical forests of the Amazon Basin, Brazil. Biogeochemistry 79, 251–274 (2006).
Gonfiantini, R., Gratziu, S. & Tongiorgi, E. in Isotopes and Radiation in Soil Plant Nutrition Studies (Technical Report Series No. 206) (ed. Joint FAO/IAEA Division of Atomic Energy in Agriculture) 405–410 (Isotope Atomic Energy Commission, 1965).
Flanagan, L. B., Comstock, J. P. & Ehleringer, J. R. Comparison of modelled and observed environmental influences on the stable oxygen and hydrogen isotope composition of leaf water in Phaseolus vulgaris L. Plant Physiol. 96, 588–596 (1991).
Yakir, D., Berry, J. A., Giles, L. & Osmond, C. B. Isotopic heterogeneity of water in transpiring leaves: Identification of the component that controls the δ18O of atmospheric O2 and CO2. Plant Cell Environ. 17, 73–80 (1994).
Sheshshayee, M. S. et al. Oxygen isotope enrichment (Δ18O) as a measure of time-averaged transpiration rate. J. Exp. Bot. 56, 3033–3039 (2005).
Buchmann, N. & Ehleringer, J. R. CO2 concentration profiles, and carbon and oxygen isotopes in C3 and C4 crop canopies. Agric. For. Meteorol. 89, 45–58 (1998).
Buchmann, N., Guehl, J. M., Barigah, T. S. & Ehleringer, J. R. Interseasonal comparison of CO2 concentrations, isotopic composition, and carbon dynamics in an Amazonian rainforest (French Guiana). Oecologia 110, 120–131 (1997).
da Silveira, L., Sternberg, L., Mulkey, S. S. & Joseph Wright, S. Oxygen isotope ratio stratification in a tropical moist forest. Oecologia 81, 51–56 (1989).
McCarroll, D. & Loader, N. J. in Isotopes in Palaeonvironmental Research (ed. Leng, M. J.) 67–116 (Springer, 2006).
Carter, M. L. & Bradbury, M. W. Oxygen isotope ratios in primate bone carbonate reflect amount of leaves and vertical stratification in the diet. Am. J. Primatol. 78, 1086–1097 (2016).
Kohn, M. J., Schoeninger, M. J. & Valley, J. W. Herbivore tooth oxygen isotope compositions: effects of diet and physiology. Geochim. Cosmochim. Acta 60, 3889–3896 (1996).
Levin, N. E., Cerling, T. E., Passey, B. H., Harris, J. M. & Ehleringer, J. R. A stable isotope aridity index for terrestrial environments. Proc. Natl Acad. Sci. USA 103, 11201–11205 (2006).
Lee-Thorp, J. et al. Isotopic evidence for an early shift to C4 resources by Pliocene hominins in Chad. Proc. Natl Acad. Sci. USA 109, 20369–20372 (2012).
Roberts, P. et al. Fruits of the forest: Human stable isotope ecology and rainforest adaptations in Late Pleistocene and Holocene (<36 to 3 ka) Sri Lanka. J. Hum. Evol. 106, 102–118 (2017).
Snoeck, C. & Pellegrini, M. Comparing bioapatite carbonate pre-treatments for isotopic measurements: part 1 – impact on structure and chemical composition. Chem. Geol. 417, 394–403 (2015).
Pellegrini, M. & Snoeck, C. Comparing bioapatite carbonate pre-treatments for isotopic measurements: part 2 – impact on carbon and oxygen isotope compositions. Chem. Geol. 420, 88–96 (2016).
Jiang, Q. Y., Zhao, L. X. & Hu, Y. W. Variations of fossil enamel bioapatite caused by different preparation and measurement protocols: a case study of Gigantopithecus fauna. Vertebrata PalAsiatica 58, 159–168 (2020).
Pushkina, D., Bocherens, H., Chaimanee, Y. & Jaeger, J. J. Stable carbon isotope reconstructions of diet and paleoenvironment from the late Middle Pleistocene Snake Cave in Northeastern Thailand. Naturwissenschaften 97, 299–309 (2010).
Ma, J. et al. Isotopic evidence of foraging ecology of Asian elephant (Elephas maximus) in South China during the Late Pleistocene. Quat. Int. 443, 160–167 (2017).
Ma, J. et al. Ecological flexibility and differential survival of Pleistocene Stegodon orientalis and Elephas maximus in mainland southeast Asia revealed by stable isotope (C, O) analysis. Quat. Sci. Rev. 212, 33–44 (2019).
Bacon, A. M. et al. Nam Lot (MIS 5) and Duoi U’Oi (MIS 4) Southeast Asian sites revisited: zooarchaeological and isotopic evidences. Palaeogeogr. Palaeoclimatol. Palaeoecol. 512, 132–144 (2018).
Bacon, A. M. et al. Testing the savannah corridor hypothesis during MIS2: the Boh Dambang hyena site in southern Cambodia. Quat. Int. 464, 417–439 (2018).
Suraprasit, K. et al. Late Middle Pleistocene ecology and climate in northeastern Thailand inferred from the stable isotope analysis of Khok Sung herbivore tooth enamel and the land mammal cenogram. Quat. Sci. Rev. 193, 24–42 (2018).
Suraprasit, K. et al. New fossil and isotope evidence for the Pleistocene zoogeographic transition and hypothesized savanna corridor in peninsular Thailand. Quat. Sci. Rev. 221, 105861 (2019).
Bocherens, H. et al. Flexibility of diet and habitat in Pleistocene South Asian mammals: implications for the fate of the giant fossil ape Gigantopithecus. Quat. Int. 434, 148–155 (2017).
Puspaningrum, M. R. et al. Isotopic reconstruction of proboscidean habitats and diets on Java since the Early Pleistocene: implications for adaptation and extinction. Quat. Sci. Rev. 228, 106007 (2020).
Janssen, R. et al. Tooth enamel stable isotopes of Holocene and Pleistocene fossil fauna reveal glacial and interglacial paleoenvironments of hominins in Indonesia. Quatern. Sci. Rev. (Singap.) 144, 145–154 (2016).
Wang, W. et al. Sequence of mammalian fossils, including hominoid teeth, from the Bubing Basin caves, South China. J. Hum. Evol. 52, 370–379 (2007).
Nelson, S. V. The paleoecology of early Pleistocene Gigantopithecus blacki inferred from isotopic analyses. Am. J. Phys. Anthropol. 155, 571–578 (2014).
Qu, Y. et al. Preservation assessments and carbon and oxygen isotopes analysis of tooth enamel of Gigantopithecus blacki and contemporary animals from Sahne Cave, Chongzuo, South China during the Early Pleistocene. Quat. Int. 354, 52–58 (2014).
Uno, K. T. et al. Late Miocene to Pliocene carbon isotope record of differential diet change among East African herbivores. Proc. Natl Acad. Sci. USA 108, 6509–6514 (2011).
LeGeros, R. Z. Calcium Phosphates in Oral Biology and Medicine (Monographs in Oral Science 15) (1991).
Lee-Thorp, J. A. On isotopes and old bones. Archaeometry 50, 925–950 (2008).
Friedli, H. et al. Ice core record of the 13C/12C ratio of atmospheric CO2 in the past two centuries. Nature 324, 237–238 (1986).
Graven, H. et al. Compiled records of carbon isotopes in atmospheric CO2 for historical simulations in CMIP6. Geosci. Model Dev. 10, 4405–4417 (2017).
Ambrose, S. H. & Norr, L. in Prehistoric Human Bone 1–37 (Springer, Berlin, Heidelberg, 1993).
Cerling, T. E. & Harris, J. M. Carbon isotope fractionation between diet and bioapatite in ungulate mammals and implications for ecological and paleoecological studies. Oecologia 120, 347–363 (1999).
Crowley, B. E. et al. Stable carbon and nitrogen isotope enrichment in primate tissues. Oecologia 164, 611–626 (2010).
Lee-Thorp, J. A., Sealy, J. C. & van der Merwe, N. J. Stable carbon isotope ratio differences between bone collagen and bone apatite, and their relationship to diet. J. Archaeol. Sci. 16, 585–599 (1989).
Kellner, C. M. & Schoeninger, M. J. A simple carbon isotope model for reconstructing prehistoric human diet. Am. J. Phys. Anthropol. 133, 1112–1127 (2007).
Karasov, W. H. & Douglas, A. E. Comparative digestive physiology. Compr. Physiol. 3, 741–783 (2013).
Ley, R. E. et al. Evolution of mammals and their gut microbes. Science 320, 1647–1651 (2008).
Furness, J. B., Cottrell, J. J. & Bravo, D. M. Comparative gut physiology symposium: comparative physiology of digestion. J. Anim. Sci. 93, 485–491 (2015).
Hammer, Ø., Harper, D. A. & Ryan, P. D. PAST: paleontological statistics software package for education and data analysis. Palaeontol. Electronica 4, 9 (2001).
Cleveland, W. S. Robust locally weighted fitting and smoothing scatterplots. J. Am. Stat. Assoc. 74, 829–836 (1979).
Cleveland, W. S. A program for smoothing scatterplots by robust locally weighted fitting. Am. Stat. 35, 54 (1981).
Lisiecki, L. E., & Raymo M. E. A. Pliocene–Pleistocene stack of 57 globally distributed benthic δ18O records. Paleoceanogr. 20, PA1003 (2005).
Pickering, R. et al. U–Pb-dated flowstones restrict South African early hominin record to dry climate phases. Nature 565, 226–229 (2019).
Chuan, G. K. in The Physical Geography of Southeast Asia (ed. Gupta, A.) 80–93 (Oxford Univ. Press, 2005).
Candy, I. et al. Pronounced warmth during early Middle Pleistocene interglacials: investigating the Mid-Brunhes Event in the British terrestrial sequence. Earth Sci. Rev. 103, 183–196 (2010).
Meckler, A. N., Clarkson, M. O., Cobb, K. M., Sodemann, H. & Adkins, J. F. Interglacial hydroclimate in the tropical west Pacific through the Late Pleistocene. Science 336, 1301–1304 (2012).
Cheng, H. et al. The Asian monsoon over the past 640,000 years and ice age terminations. Nature 534, 640–646 (2016).
Maloney, B. K. & McCormac, F. G. Palaeoenvironments of north Sumatra: a 30,000 year old pollen record from Pea Bullok. Bull. Indo-Pacific Prehist. Ass. 14, 73–82 (1996).
van der Kaars, W. A. & Dam, M. A. C. A. 135,000-year record of vegetational and climatic change from the Bandung area, West-Java, Indonesia. Palaeogeogr. Palaeoclimatol. Palaeoecol. 117, 55–72 (1995).
van der Kaars, W. A. & Dam, M. A. C. Vegetation and climate change in West-Java, Indonesia during the last 135,000 years. Quat. Int. 37, 67–71 (1997).
Wurster, C. M. et al. Forest contraction in north equatorial Southeast Asia during the last glacial period. Proc. Natl Acad. Sci. USA 107, 15508–15511 (2010).
Wurster, C. M., Rifai, H., Zhou, B., Haig, J. & Bird, M. I. Savanna in equatorial Borneo during the Late Pleistocene. Sci. Rep. 9, 6392 (2019).
Dubois, N. et al. Indonesian vegetation response to changes in rainfall seasonality over the past 25,000 years. Nat. Geosci. 7, 513–517 (2014).
Sun, X. et al. Deep-sea pollen from the South China Sea: Pleistocene indicators of East Asian monsoon. Mar. Geol. 201, 97–118 (2003).
Yu, S. et al. Pollen record in the northwestern continental shelf of the South China Sea in the past 82 ka: paleoenvironmental changes in the last glacial period. J. Asian Earth Sci. 199, 104457 (2020).
IUCN. The IUCN Red List of Threatened Species. Version 2019-3 http://www.iucnredlist.org (accessed 6 November 2019).
Yang, D. et al. Researches of Ailuropoda–Stegodon Fauna from Gulin China (in Chinese with English abstract) (Chongqing, 1995).
Turvey, S. T. et al. Holocene survival of Late Pleistocene megafauna in China: a critical review of the evidence. Quat. Sci. Rev. 76, 156–166 (2013).
Jin, C. et al. Chronological sequence of the early Pleistocene Gigantopithecus faunas from cave sites in the Chongzuo, Zuojiang River area, South China. Quat. Int. 354, 4–14 (2014).
Rizal, Y. et al. Last appearance of Homo erectus at Ngandong, Java, 117,000–108,000 years ago. Nature 577, 381–385 (2020).
Joordens, J. C. et al. Homo erectus at Trinil on Java used shells for tool production and engraving. Nature 518, 228–231 (2015).
Zhang, Y. et al. New 400–320 ka Gigantopithecus blacki remains from Hejiang Cave, Chongzuo City, Guangxi, South China. Quat. Int. 354, 35–45 (2014).
Han, D. & Xu, C. in Palaeoanthropology and Palaeolithic Archaeology in the People’s Republic of China (eds Rukang, W. & Olsen, J. W.) 267–289 (Academic, 1985).
Lu, C., Xu, X. & Sun, X. Re-dating Changyang Cave in Hubei Province, southern China. Quat. Int. 537, 1–8 (2020).
van den Bergh, G. D. et al. The Early Pleistocene terrestrial vertebrate faunal sequence of Java, Indonesia. J. Vert. Paleol. Abstract 210 (2019).
Dong, W. et al. New materials of Early Pleistocene Sus from Sanhe Cave, Chongzuo, Guangxi, South China. Acta Anthropol. Sin. 32, 63–76 (2013).
Shao, Q. et al. Coupled ESR and U-series dating of early Pleistocene Gigantopithecus faunas at Mohui and Sanhe Caves, Guangxi, southern China. Quat. Geochronol. 30, 524–528 (2015).
Rink, W. J., Wei, W., Bekken, D. & Jones, H. L. Geochronology of Ailuropoda–Stegodon fauna and Gigantopithecus in Guangxi Province, southern China. Quat. Res. 69, 377–387 (2008).
Wang, Y., Jin, C. Z. & Mead, J. I. New remains of Sinomastodon yangziensis (Proboscidea, Gomphotheriidae) from Sanhe karst cave, with discussion on the evolution of Pleistocene Sinomastodon in South China. Quat. Int. 339–340, 90–96 (2014).
Duval, M. et al. Direct ESR dating of the Pleistocene vertebrate assemblage from Khok Sung locality, Nakhon Ratchasima Province, Northeast Thailand. Pal. Electr. 22, 1–25 (2019).
Li, H., Li, C. & Kuman, K. Longgudong, an Early Pleistocene site in Jianshi, South China, with stratigraphic association of human teeth and lithics. Sci. China Earth Sci. 60, 452–462 (2017).
Bacon, A. M. et al. Late Pleistocene mammalian assemblages of Southeast Asia: new dating, mortality profiles and evolution of the predator–prey relationships in an environmental context. Palaeogeogr. Palaeoclimatol. Palaeoecol. 422, 101–127 (2015).
Westaway, K. E. et al. Age and biostratigraphic significance of the Punung rainforest fauna, East Java, Indonesia, and implications for Pongo and Homo. J. Hum. Evol. 53, 709–717 (2007).
Matsu’ura, S. et al. Age control of the first appearance datum for Javanese Homo erectus in the Sangiran area. Science 367, 210–214 (2020).
Sun, L. et al. Magnetochronological sequence of the early Pleistocene Gigantopithecus faunas in Chongzuo, Guangxi, southern China. Quat. Int. 354, 15–23 (2014).
Esposito, M., Reyss, J. L., Chaimanee, Y. & Jaeger, J. J. U-series dating of fossil teeth and carbonates from Snake Cave, Thailand. J. Archaeol. Sci. 29, 341–349 (2002).
Storm, P. et al. U-series and radiocarbon analyses of human and faunal remains from Wajak, Indonesia. J. Hum. Evol. 64, 356–365 (2013).
Acknowledgements
We thank E. Hoeger, R. Voss, L. Kok Peng, A. van Heteren, J. Cuisin, V. Nicolas, G. Véron, J. Lesur and C. Lefèvre for allowing access to specimens under their care, N. Boivin and the Max Planck Society for support and J. Ilgner, M. Lucas, E. Perruchini and S. Marzo for their assistance with analysis of the samples. The map in Fig. 1 was provided by CartoGIS Services, ANU College of Asia and the Pacific, The Australian National University; we thank S. Potter and K. Pelling for providing the map. This research was supported by an Australian Research Council Future Fellowship to J.L. (FT160100450). P.R. was funded by the Max Planck Society and the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement no. 850709).
Author information
Authors and Affiliations
Contributions
J.L. conceived this research and conducted the statistical analyses. P.R. performed the stable isotope analyses. Both authors contributed equally to study design, data acquisition, interpretation of data and the writing of the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Thure Cerling and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Distribution of δ13C values across the Quaternary.
Distribution is shown with a jitter plot and corresponding kernel density for Indochina (blue) and Sundaland (red). Kernel densities are exaggerated vertically, such that the peaks for both provinces are equal. Shaded boxes represent the division between δ13C values associated with forests (left) and grasslands (right).
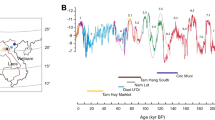
Extended Data Fig. 2 Temporal trends of δ13C and δ18O values under different geochronological scenarios.
a, δ13C values assuming minimum age for each site. b, δ18O values assuming minimum age for each site. c, δ13C values assuming median age for each site. d, δ18O values assuming median age for each site. e, δ13C values assuming maximum age for each site. f, δ18O values assuming maximum age for each site. Each panel is shown relative to the Lisiecki Raymo benthic oxygen-isotope stack. The 95% confidence interval for each curve was based on 999 random replicates using resampling of residuals.
Extended Data Fig. 3 Distribution of δ13C values for browsers across fossil sites through Southeast Asia.
Indochina, dark green; Sundaland, light green. Horizontal line represents the −29‰ zone that indicates the beginning of subcanopy and closed-canopy environments. The long lower whiskers in the box and whisker plot, which indicate a very negatively skewed distribution, are most closely associated with highly stratified forests. The boxes show the median and the lower (25%) and upper (75%) quartiles; the whiskers encompass the minimum and maximum values. Independent sample sizes: Juyuandong, n = 4; Longudong, n = 26; Mohui, n = 5; Sanhe, n = 25; Semedo, n = 6; Sangiran, n = 4; Upper Pubu, n = 4; Khok Sung, n = 5; Pha Bong, n = 15; Tham Wiman Nakin, n = 10; Baxian, n = 32; Boh Damban, n = 18; Nam Lot, n = 39; Quzai, n = 32; Sibrambang, n = 6; Wajak, n = 4; Cipeundeuy, n = 2; Indochina, n = 74; and Sundaland, n = 158.
Extended Data Fig. 4 Changes in mean δ13C and δ18O values for mammals classified at the ordinal level.
Continuities (non-significant differences in mean) of δ13C values within orders between epochs are illustrated with arrows at the top of each plot. Continuities between orders in a single epoch are illustrated with circles bounding similar δ13C means. Variation within orders and epochs is indicated at 1 s.d.
Supplementary information
Supplementary Table
This excel spreadsheet lists all the isotope data considered in the manuscript, including all published data, raw original data, and fractionation adjustments. Worksheet 1 lists the combined dataset. Worksheet 2 list data from Zoologische Staatssammlung München (ZSM), Germany; Worksheet 3 the Lee Kong Chian Natural History Museum (LKCNHM); Worksheet 4 the Muséum National d’Histoire Naturelle (MNHN), Paris, France; and Worksheet 5 the American Museum of Natural History (AMNH), New York, United States of America.
Rights and permissions
About this article
Cite this article
Louys, J., Roberts, P. Environmental drivers of megafauna and hominin extinction in Southeast Asia. Nature 586, 402–406 (2020). https://doi.org/10.1038/s41586-020-2810-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2810-y
This article is cited by
-
The demise of the giant ape Gigantopithecus blacki
Nature (2024)
-
Tooth enamel nitrogen isotope composition records trophic position: a tool for reconstructing food webs
Communications Biology (2023)
-
Palaeoenvironments and hominin evolutionary dynamics in southeast Asia
Scientific Reports (2023)
-
Evolutionary ecology of Miocene hominoid primates in Southeast Asia
Scientific Reports (2022)
-
Javanese Homo erectus on the move in SE Asia circa 1.8 Ma
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.