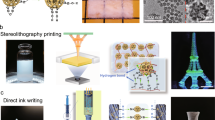
Abstract
Owing to their ultralow thermal conductivity and open pore structure1,2,3, silica aerogels are widely used in thermal insulation4,5, catalysis6, physics7,8, environmental remediation6,9, optical devices10 and hypervelocity particle capture11. Thermal insulation is by far the largest market for silica aerogels, which are ideal materials when space is limited. One drawback of silica aerogels is their brittleness. Fibre reinforcement and binders can be used to overcome this for large-volume applications in building and industrial insulation5,12, but their poor machinability, combined with the difficulty of precisely casting small objects, limits the miniaturization potential of silica aerogels. Additive manufacturing provides an alternative route to miniaturization, but was “considered not feasible for silica aerogel”13. Here we present a direct ink writing protocol to create miniaturized silica aerogel objects from a slurry of silica aerogel powder in a dilute silica nanoparticle suspension (sol). The inks exhibit shear-thinning behaviour, owing to the high volume fraction of gel particles. As a result, they flow easily through the nozzle during printing, but their viscosity increases rapidly after printing, ensuring that the printed objects retain their shape. After printing, the silica sol is gelled in an ammonia atmosphere to enable subsequent processing into aerogels. The printed aerogel objects are pure silica and retain the high specific surface area (751 square metres per gram) and ultralow thermal conductivity (15.9 milliwatts per metre per kelvin) typical of silica aerogels. Furthermore, we demonstrate the ease with which functional nanoparticles can be incorporated. The printed silica aerogel objects can be used for thermal management, as miniaturized gas pumps and to degrade volatile organic compounds, illustrating the potential of our protocol.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The raw data on particle size distribution (Fig. 1f), rheological measurements (Fig. 1g, h), nitrogen sorption (Fig. 2m, n), thermogravimetric analysis (Fig. 2o), pumping flow rate and toluene degradation (Fig. 4f, g), and thermal conductivity measurements and reconstructed X-ray tomography used for the image analysis (Fig. 2j–l), are available at https://doi.org/10.5281/zenodo.3794969. All other data (raw data used for Extended Data Figs. 1–9 and Extended Data Table 1) are available from the corresponding authors on request.
Code availability
The codes for 3D printing and tomographic analysis are available at https://doi.org/10.5281/zenodo.3794969.
References
Kistler, S. S. Coherent expanded aerogels and jellies. Nature 127, 741 (1931).
Hüsing, N. & Schubert, U. Aerogels—airy materials: chemistry, structure, and properties. Angew. Chem. Int. Ed. 37, 22–45 (1998).
Prakash, S. S., Brinker, J. C., Hurd, A. J. & Rao, S. M. Silica aerogel films prepared at ambient pressure by using surface derivatization to induce reversible drying shrinkage. Nature 374, 439–443 (1995); erratum 375, 431 (1995).
Wordsworth, R., Kerber, L. & Cockell, C. Enabling Martian habitability with silica aerogel via the solid-state greenhouse effect. Nat Astron. 3, 898–903 (2019).
Koebel, M. M., Rigacci, A. & Achard, P. in Aerogels Handbook (eds Aegerter, M. et al.) 607–633 (Springer, 2011).
Morris, C. A., Anderson, M. L., Stroud, R. M., Merzbacher, C. I. & Rolison, D. R. Silica sol as a nanoglue: flexible synthesis of composite aerogels. Science 284, 622–624 (1999).
Kim, C. et al. Large-scale nanoporous metal-coated silica aerogels for high SERS effect improvement. Sci. Rep. 8, 15144 (2018).
Pollanen, J. et al. New chiral phases of superfluid 3He stabilized by anisotropic silica aerogel. Nat. Phys. 8, 317–320 (2012).
Dumée, L. F. et al. Silver metal nano-matrixes as high efficiency and versatile catalytic reactors for environmental remediation. Sci. Rep. 7, 45112 (2017).
Shin, D. et al. Scalable variable-index elasto-optic metamaterials for macroscopic optical components and devices. Nat. Commun. 8, 16090 (2017).
Sandford, S. A. et al. Organics captured from Comet 81P/Wild 2 by the Stardust spacecraft. Science 314, 1720–1724 (2006).
Lee, K. P., Gould, G. L., Gronemeyer, W. & Stepanian, C. J. Methods to produce gel sheets. US patent 7,399,439 B2 (2008).
Bertino, M. F. Rapid fabrication of hybrid aerogels and 3D printed porous materials. J. Sol-Gel Sci. Technol. 86, 239–254 (2018).
Collins, R. Aerogels 2019–2029: technologies, markets and players. IDTechX, https://www.idtechex.com/en/research-report/aerogels-2019–2029-technologies-markets-and-players/644 (2018).
Zhang, Q. et al. 3D printing of graphene aerogels. Small 12, 1702–1708 (2016).
Zhu, C. et al. Highly compressible 3D periodic graphene aerogel microlattices. Nat. Commun. 6, 6962 (2015).
Jiang, Y. et al. Direct 3D printing of ultralight graphene oxide aerogel microlattices. Adv. Funct. Mater. 28, 1707024 (2018).
Guo, F. et al. Highly stretchable carbon aerogels. Nat. Commun. 9, 881 (2018).
He, P. et al. Patterned carbon nitride-based hybrid aerogel membranes via 3D printing for broadband solar wastewater remediation. Adv. Funct. Mater. 28, 1801121 (2018).
Zhu, C. et al. Toward digitally controlled catalyst architectures: hierarchical nanoporous gold via 3D printing. Sci. Adv. 4, eaas9459 (2018).
Chandrasekaran, S. et al. Direct ink writing of organic and carbon aerogels. Mater. Horiz. 5, 1166–1175 (2018).
Hausmann, M. K. et al. Dynamics of cellulose nanocrystal alignment during 3D printing. ACS Nano 12, 6926–6937 (2018).
Truby, R. L. & Lewis, J. A. Printing soft matter in three dimensions. Nature 540, 371–378 (2016).
Gladman, A. S., Matsumoto, E. A., Nuzzo, R. G., Mahadevan, L. & Lewis, J. A. Biomimetic 4D printing. Nat. Mater. 15, 413–418 (2016).
Kim, Y., Yuk, H., Zhao, R., Chester, S. A. & Zhao, X. Printing ferromagnetic domains for untethered fast-transforming soft materials. Nature 558, 274–279 (2018).
Maleki, H., Montes, S., Hayati-Roodbari, N., Putz, F. & Huesing, N. Compressible, thermally insulating, and fire retardant aerogels through self-assembling silk fibroin biopolymers inside a silica structure—an approach towards 3D printing of aerogels. ACS Appl. Mater. Interfaces 10, 22718–22730 (2018).
Hyun, K., Kim, S. H., Ahn, K. H. & Lee, S. J. Large amplitude oscillatory shear as a way to classify the complex fluids. J. Non-Newt. Fluid Mech. 107, 51–65 (2002).
Yang, Y., Li, Y., Mao, M., Zeng, M. & Zhao, X. UV–visible–infrared light-driven thermocatalysis for environmental purification on ramsdellite MnO2 hollow spheres considerably promoted by a novel photoactivation. ACS Appl. Mater. Interfaces 9, 2350–2357 (2017).
He, X., Bahk, Y. K. & Wang, J. Organic dye removal by MnO2 and Ag micromotors under various ambient conditions: the comparison between two abatement mechanisms. Chemosphere 184, 601–608 (2017).
Wei, G., Liu, Y., Zhang, X., Yu, F. & Du, X. Thermal conductivities study on silica aerogel and its composite insulation materials. Int. J. Heat Mass Transf. 54, 2355–2366 (2011).
Iswar, S. et al. Reinforced and superinsulating silica aerogel through in situ cross-linking with silane terminated prepolymers. Acta Mater. 147, 322–328 (2018).
Muntz, E., Sone, Y., Aoki, K., Vargo, S. & Young, M. Performance analysis and optimization considerations for a Knudsen compressor in transitional flow. J. Vac. Sci. Technol. A 20, 214–224 (2002).
Zhao, S. et al. Dimensional and structural control of silica aerogel membranes for miniaturized motionless gas pumps. ACS Appl. Mater. Interfaces 7, 18803–18814 (2015).
Pajonk, G. M. et al. Physical properties of silica gels and aerogels prepared with new polymeric precursors. J. Non-Cryst. Solids 186, 1–8 (1995).
Stahl, T., Brunner, S., Zimmermann, M. & Ghazi Wakili, K. Thermo-hygric properties of a newly developed aerogel based insulation rendering for both exterior and interior applications. Energy Build. 44, 114–117 (2012).
Brunauer, S., Emmett, P. H. & Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309–319 (1938).
Marone, F. & Stampanoni, M. Regridding reconstruction algorithm for real-time tomographic imaging. J. Synchrotron Radiat. 19, 1029–1037 (2012).
Fox, P., Hall, A. & Schryer, N. L. The PORT mathematical subroutine library. ACM Trans. Math. Softw. 4, 104–126 (1978).
R: a language and environment for statistical computing (R Core Team, 2013).
Maxwell, J. C. VII. On stresses in rarified gases arising from inequalities of temperature. Philos. Trans. R. Soc. Lond. 170, 231–256 (1879).
Loeb, L. B. The Kinetic Theory of Gases 3rd edn, Ch. VII, 290–300 (Dover, 2004).
Sone, Y. Molecular Gas Dynamics Theory, Techniques, and Applications Ch. 1 (Birkhäuser, 2007).
Malfait, W. J., Verel, R. & Koebel, M. M. Hydrophobization of silica aerogels: insights from quantitative solid-state NMR spectroscopy. J. Phys. Chem. C 118, 25545–25554 (2014).
Malfait, W. J. et al. Surface chemistry of hydrophobic silica aerogels. Chem. Mater. 27, 6737–6745 (2015).
Acknowledgements
We thank M. Giannakou and D. Sivaraman for help in the laboratory, D. Rentsch for support with the NMR spectrometer, funded in part by grant 150638 from the Swiss National Science Foundation, F. Winnefeld for support with the rheometer, A. Braun for support with the photocatalytic setup, S. Carl for support with the infrared thermal imaging system, R. Pauer for support with the STEM and SAED pattern analysis, N. Guerrero for video editing and B. Fischer for TGA analysis. M.G. was supported by MSCA grant 746992.
Author information
Authors and Affiliations
Contributions
S.Z., G.S., M.M.K. and W.J.M. designed and coordinated the study. S.Z., G.S., D.N. and C.U. optimized the ink formulation. G.S. and S.Z. carried out the printing experiments, supervised by G.N. J.W. supervised the MnO2 synthesis and light-driven pump test. S.D. and S.Z. conducted the pumping/degradation experiments. S.Z. carried out the SEM and BET analysis. S.B. and S.Z. conducted the thermal conductivity and infrared imaging measurements. M.G. prepared STL files and photographic artwork. W.J.M. did the NMR analysis. A.B. collected, and S.G. and Z.P. processed, the X-ray tomography data. S.Z. and W.J.M. wrote the manuscript with input from all coauthors. All authors reviewed and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A patent for the additive manufacturing of silica aerogels (EP19208179.2) has been filed on behalf of Empa. S.Z., G.S., W.J.M., M.M.K., C.U. and D.N. are listed as inventors.
Additional information
Peer review information Nature thanks L. Zhao and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Photographs and hydrophobicity of 3D-printed silica aerogel objects.
a–c, Photographs of the 3D-printed silica aerogel lotus flower (c) show it is light-weight (a) and superhydrophobic (b). d, Water and 1-pentanol contact-angle measurements.
Extended Data Fig. 2 Rheology of silica aerogel inks.
a–d, Apparent viscosity as a function of applied shear rate (a, c), and storage (G′) and loss (G″) modulus as a function of applied shear stress (b, d), for the silica aerogel ink with various loadings of silica aerogel particles (as labelled). e, f, Shelf life of ink SP1.6: apparent viscosity as a function of applied shear rate (e), and storage (G′) and loss (G″) modulus as a function of applied shear stress (f), for the fresh silica aerogel ink and after 30 days of storage at room temperature (25 °C, 50% relative humidity).
Extended Data Fig. 3 Solidification and printing performance.
a, The evolution of solidification of the silica aerogel ink with and without PPGNH. The gels are more transparent when PPGNH has been added to the sol, indicative of a more homogenous pore structure. b, Filaments printed from 100-μm, 250-μm and 410-μm nozzles. c, Demonstrations of overhang and bridging with ink SP2.5. d, Schematic designs of the 2D and 3D structures in Fig. 1e: a lotus flower (31–50 layers) and its leaf (24 layers). Models modified from a design by J. Watkins, https://www.thingiverse.com/thing:415314.
Extended Data Fig. 4 X-ray tomography image analysis.
a, Histogram of grey values with deconvoluted peaks of aerogel particles (blue), MnO2-loaded aerogel matrix (green) and MnO2-enriched areas (orange), and the resulting fit (red). b, Line profile across a particle (see inset), before (grey dotted line) and after (black line) Gaussian blurring. The chosen binder cutoff (red dashed line) and the final phase separation after erosion (green) are also shown. c–f, Different orientations of the 3D volume rendering of the filament (c, d), and y–z (e) and x–y (f) cross-sections. The segmented image consists of 57.9% SiO2 particles, 40.5% binder phase and 1.6% MnO2-enriched areas.
Extended Data Fig. 5 Spectroscopic data.
a, b, 1H–29Si (a) and 1H–13C (b) cross-polarization magic-angle spinning NMR spectra of a 3D-printed silica aerogel. The spectra have the typical peaks for hydrophobized silica aerogel43,44. The 1H–29Si spectrum has peaks Q4, Q3 and Q2 from silica particles, corresponding to silicon atoms coordinated by 4, 3 and 2 bridging oxygen atoms and 0, 1 and 2 non-bridging oxygen atoms, respectively, and a peak from trimethylsilyl (TMS) groups grafted onto the silica surfaces. The 1H–13C spectrum has an intense peak from grafted TMS groups (63.0% of total spectral intensity) and two strong peaks from ethoxy groups grafted onto the silica surfaces (Et, CH2 and Et, CH3 with 34.5% of total spectral intensity), from the TEOS precursor of the matrix aerogel and/or from alcoholysis during solvent exchanges. An additional five weak peaks (P1–P5), corresponding to either pentanol adsorbed on the surface or petanoxy groups grafted onto the silica, together account for approximately 2.5% of the total spectral intensity. The peaks labelled ‘ssb’ correspond to spinning sidebands. No other peaks were detected. c, FTIR spectra of the silica aerogel particles (SAPs; red), the PPGNH (blue) and the printed object (dashed black).
Extended Data Fig. 6 Mechanical properties of silica-ink-based aerogels.
a, b, Stress–strain curves during uniaxial compression (a) and Brazilian test (b) of reference sol–gel aerogels and silica-ink-based aerogels. c, The corresponding mechanical properties of the various aerogels. σCmax, maxium compressive strength; ECmodulus, elastic modulus derived from the compression test; σTmax, maxium Brazilian tensile strength; ETmodulus, elastic modulus derived from the Brazilian tensile strength. d, e, Subtractive manufacture of the silica-ink-based gel.
Extended Data Fig. 7 Protection of a temperature-sensitive capacitor from a heat source.
a–c, Without protection (T/C, T-type thermocouple; a), with a polystyrene foam cap (XPS; b) and with a printed silica aerogel cap (c). d, STL file. e, 3D-printed silica aerogel cap. f, Temperature evolution after contact with the cartridge heater.
Extended Data Fig. 8 Properties of MnO2 and MnO2-doped silica aerogels.
a–g, X-ray diffraction (a), SEM image (b), HRTEM image and lattice spacing (c), GC/FID spectra of the toluene degradation shown in Fig. 4 on a MnO2–SiO2 bilayer aerogel (d), SAED of the MnO2 microspheres (e), and STEM image (f; g, right-most image) and elemental analysis (g, left three images) of the MnO2 distribution in the silica aerogel. h, Energy-dispersive X-ray spectroscopy (right-most image) of a cross-section of the interface between the silica and MnO2-loaded silica aerogel within the thermal transpiration membrane, and the element distribution maps (Mn, Si, O; left three images).
Extended Data Fig. 9 Light-driven gas pump and VOC degradation system.
a, b, Photograph (a) and working scheme (b) of the setup.
Supplementary information
Video 1
Additive manufacturing of a SiO2 aerogel lotus flower (high-speed video).
Video 2
Additive manufacturing of a SiO2-MnO2 aerogel lattice cube.
Video 3
Additive manufacturing of various 3D patterns.
Video 4
Additive manufacturing of gel array for thermal management.
Video 5
Additive manufacturing of 50×50×10 cm3 aerogel plate for thermal conductivity measurement.
Rights and permissions
About this article
Cite this article
Zhao, S., Siqueira, G., Drdova, S. et al. Additive manufacturing of silica aerogels. Nature 584, 387–392 (2020). https://doi.org/10.1038/s41586-020-2594-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2594-0
This article is cited by
-
Gradient matters via filament diameter-adjustable 3D printing
Nature Communications (2024)
-
Stretchable and negative-Poisson-ratio porous metamaterials
Nature Communications (2024)
-
Improving Thermal-oxidative Aging Resistance of Styrene-butadiene Rubber by Antioxidant Loaded Silica Aerogel
Chinese Journal of Polymer Science (2024)
-
Dual-filler reinforced PVDF-HFP based polymer electrolyte enabling high-safety design of lithium metal batteries
Nano Research (2024)
-
Study on preparation of SiO2/PTFE aerogel-like materials via atmospheric drying and their thermal insulation performance
Journal of Sol-Gel Science and Technology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.