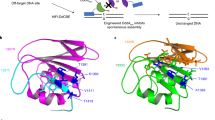
Abstract
Bacterial toxins represent a vast reservoir of biochemical diversity that can be repurposed for biomedical applications. Such proteins include a group of predicted interbacterial toxins of the deaminase superfamily, members of which have found application in gene-editing techniques1,2. Because previously described cytidine deaminases operate on single-stranded nucleic acids3, their use in base editing requires the unwinding of double-stranded DNA (dsDNA)—for example by a CRISPR–Cas9 system. Base editing within mitochondrial DNA (mtDNA), however, has thus far been hindered by challenges associated with the delivery of guide RNA into the mitochondria4. As a consequence, manipulation of mtDNA to date has been limited to the targeted destruction of the mitochondrial genome by designer nucleases9,10.Here we describe an interbacterial toxin, which we name DddA, that catalyses the deamination of cytidines within dsDNA. We engineered split-DddA halves that are non-toxic and inactive until brought together on target DNA by adjacently bound programmable DNA-binding proteins. Fusions of the split-DddA halves, transcription activator-like effector array proteins, and a uracil glycosylase inhibitor resulted in RNA-free DddA-derived cytosine base editors (DdCBEs) that catalyse C•G-to-T•A conversions in human mtDNA with high target specificity and product purity. We used DdCBEs to model a disease-associated mtDNA mutation in human cells, resulting in changes in respiration rates and oxidative phosphorylation. CRISPR-free DdCBEs enable the precise manipulation of mtDNA, rather than the elimination of mtDNA copies that results from its cleavage by targeted nucleases, with broad implications for the study and potential treatment of mitochondrial disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Coordinates and structure factors for DddA have been deposited in the PDB under accession code 6U08. High-throughput sequencing and whole-mitochondria sequencing data have been deposited in the NCBI Sequence Read Archive under accession code PRJNA603010. Amino acids sequences of all base editors in this study are provided in the Supplementary Information, sequences 1–4.
References
Rees, H. A. & Liu, D. R. Base editing: precision chemistry on the genome and transcriptome of living cells. Nat. Rev. Genet. 19, 770–788 (2018).
Komor, A. C., Badran, A. H. & Liu, D. R. CRISPR-based technologies for the manipulation of eukaryotic genomes. Cell 169, 559 (2017).
Salter, J. D. & Smith, H. C. Modeling the embrace of a mutator: APOBEC selection of nucleic acid ligands. Trends Biochem. Sci. 43, 606–622 (2018).
Gammage, P. A., Moraes, C. T. & Minczuk, M. Mitochondrial genome engineering: the revolution may not be CRISPR-ized. Trends Genet. 34, 101–110 (2018).
Vafai, S. B. & Mootha, V. K. Mitochondrial disorders as windows into an ancient organelle. Nature 491, 374–383 (2012).
Gopal, R. K. et al. Early loss of mitochondrial complex I and rewiring of glutathione metabolism in renal oncocytoma. Proc. Natl Acad. Sci. USA 115, E6283–E6290 (2018).
Stewart, J. B. & Chinnery, P. F. The dynamics of mitochondrial DNA heteroplasmy: implications for human health and disease. Nat. Rev. Genet. 16, 530–542 (2015).
Bacman, S. R. et al. MitoTALEN reduces mutant mtDNA load and restores tRNAAla levels in a mouse model of heteroplasmic mtDNA mutation. Nat. Med. 24, 1696–1700 (2018).
Bacman, S. R., Williams, S. L., Pinto, M., Peralta, S. & Moraes, C. T. Specific elimination of mutant mitochondrial genomes in patient-derived cells by mitoTALENs. Nat. Med. 19, 1111–1113 (2013).
Gammage, P. A., Rorbach, J., Vincent, A. I., Rebar, E. J. & Minczuk, M. Mitochondrially targeted ZFNs for selective degradation of pathogenic mitochondrial genomes bearing large-scale deletions or point mutations. EMBO Mol. Med. 6, 458–466 (2014).
Nissanka, N., Bacman, S. R., Plastini, M. J. & Moraes, C. T. The mitochondrial DNA polymerase gamma degrades linear DNA fragments precluding the formation of deletions. Nat. Commun. 9, 2491 (2018).
Peeva, V. et al. Linear mitochondrial DNA is rapidly degraded by components of the replication machinery. Nat. Commun. 9, 1727 (2018).
Gaudelli, N. M. et al. Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Iyer, L. M., Zhang, D., Rogozin, I. B. & Aravind, L. Evolution of the deaminase fold and multiple origins of eukaryotic editing and mutagenic nucleic acid deaminases from bacterial toxin systems. Nucleic Acids Res. 39, 9473–9497 (2011).
Coulthurst, S. The type VI secretion system: a versatile bacterial weapon. Microbiology 165, 503–515 (2019).
Hood, R. D. et al. A type VI secretion system of Pseudomonas aeruginosa targets a toxin to bacteria. Cell Host Microbe 7, 25–37 (2010).
Richardson, S. R., Narvaiza, I., Planegger, R. A., Weitzman, M. D. & Moran, J. V. APOBEC3A deaminates transiently exposed single-strand DNA during LINE-1 retrotransposition. eLife 3, e02008 (2014).
Krokan, H. E. & Bjørås, M. Base excision repair. Cold Spring Harb. Perspect. Biol. 5, a012583 (2013).
Urnov, F. D., Rebar, E. J., Holmes, M. C., Zhang, H. S. & Gregory, P. D. Genome editing with engineered zinc finger nucleases. Nat. Rev. Genet. 11, 636–646 (2010).
Joung, J. K. & Sander, J. D. TALENs: a widely applicable technology for targeted genome editing. Nat. Rev. Mol. Cell Biol. 14, 49–55 (2013).
Guilinger, J. P., Thompson, D. B. & Liu, D. R. Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat. Biotechnol. 32, 577–582 (2014).
Qi, L. S. et al. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152, 1173–1183 (2013).
Kleinstiver, B. P. et al. Broadening the targeting range of Staphylococcus aureus CRISPR–Cas9 by modifying PAM recognition. Nat. Biotechnol. 33, 1293–1298 (2015).
Nishimasu, H. et al. Crystal structure of Staphylococcus aureus Cas9. Cell 162, 1113–1126 (2015).
Anzalone, A. V. et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157 (2019).
Nishida, K. et al. Targeted nucleotide editing using hybrid prokaryotic and vertebrate adaptive immune systems. Science 353, aaf8729 (2016).
Komor, A. C. et al. Improved base excision repair inhibition and bacteriophage Mu Gam protein yields C:G-to-T:A base editors with higher efficiency and product purity. Sci. Adv. 3, eaao4774 (2017).
Nilsen, H. et al. Nuclear and mitochondrial uracil-DNA glycosylases are generated by alternative splicing and transcription from different positions in the UNG gene. Nucleic Acids Res. 25, 750–755 (1997).
Koblan, L. W. et al. Improving cytidine and adenine base editors by expression optimization and ancestral reconstruction. Nat. Biotechnol. 36, 843–846 (2018).
Bao, X. R. et al. Mitochondrial dysfunction remodels one-carbon metabolism in human cells. eLife 5, e10575 (2016).
Magnusson, J., Orth, M., Lestienne, P. & Taanman, J.-W. Replication of mitochondrial DNA occurs throughout the mitochondria of cultured human cells. Exp. Cell Res. 289, 133–142 (2003).
Gopal, R. K. et al. Widespread chromosomal losses and mitochondrial DNA alterations as genetic drivers in Hürthle cell carcinoma. Cancer Cell 34, 242–255.e5 (2018).
Rinaldi, F. C., Doyle, L. A., Stoddard, B. L. & Bogdanove, A. J. The effect of increasing numbers of repeats on TAL effector DNA binding specificity. Nucleic Acids Res. 45, 6960–6970 (2017).
Yang, L. et al. Engineering and optimising deaminase fusions for genome editing. Nat. Commun. 7, 13330 (2016).
Finn, R. D., Clements, J. & Eddy, S. R. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 39, W29–W37 (2011).
Fazli, M., Harrison, J. J., Gambino, M., Givskov, M. & Tolker-Nielsen, T. In-frame and unmarked gene deletions in Burkholderia cenocepacia via an allelic exchange system compatible with gateway technology. Appl. Environ. Microbiol. 81, 3623–3630 (2015).
Spiewak, H. L. et al. Burkholderia cenocepacia utilizes a type VI secretion system for bacterial competition. MicrobiologyOpen 8, e774 (2019).
Choi, K. H., DeShazer, D. & Schweizer, H. P. mini-Tn7 insertion in bacteria with multiple glmS-linked attTn7 sites: example Burkholderia mallei ATCC 23344. Nat. Protoc. 1, 162–169 (2006).
Rees, H. A., Wilson, C., Doman, J. L. & Liu, D. R. Analysis and minimization of cellular RNA editing by DNA adenine base editors. Sci. Adv. 5, eaax5717 (2019).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Painter, J. & Merritt, E. A. Optimal description of a protein structure in terms of multiple groups undergoing TLS motion. Acta Crystallogr. D 62, 439–450 (2006).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Bhagwat, A. S. et al. Strand-biased cytosine deamination at the replication fork causes cytosine to thymine mutations in Escherichia coli. Proc. Natl Acad. Sci. USA 113, 2176–2181 (2016).
Kotrys, A. V. et al. Quantitative proteomics revealed C6orf203/MTRES1 as a factor preventing stress-induced transcription deficiency in human mitochondria. Nucleic Acids Res. 47, 7502–7517 (2019).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Koboldt, D. C. et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res. 22, 568–576 (2012).
Miller, S. M. et al. Continuous evolution of SpCas9 variants compatible with non-G PAMs. Nat. Biotechnol. 38, 471–481 (2020).
Rees, H. A. et al. Improving the DNA specificity and applicability of base editing through protein engineering and protein delivery. Nat. Commun. 8, 15790 (2017).
Clement, K. et al. CRISPResso2 provides accurate and rapid genome editing sequence analysis. Nat. Biotechnol. 37, 224–226 (2019).
Ludwig, L. S. et al. Lineage tracing in humans enabled by mitochondrial mutations and single-cell genomics. Cell 176, 1325–1339.e22 (2019).
Diroma, M. A., Ciaccia, L., Pesole, G. & Picardi, E. Elucidating the editome: bioinformatics approaches for RNA editing detection. Brief. Bioinform. 20, 436–447 (2019).
Doman, J. L., Raguram, A., Newby, G. A. & Liu, D. R. Evaluation and minimization of Cas9-independent off-target DNA editing by cytosine base editors. Nat. Biotechnol. 38, 620–628 (2020).
Vaser, R., Adusumalli, S., Leng, S. N., Sikic, M. & Ng, P. C. SIFT missense predictions for genomes. Nat. Protoc. 11, 1–9 (2016).
Acknowledgements
We thank C. Wilson, L. Ludwig, T. Wang, K. Zhao, W. H. Yeh, S. Miller, A. Sousa, H. Hayden and A. Vo for materials and technical advice. R. Harris provided APOBEC3A and L. Eberl provided B. cen H111. This work was supported by the Merkin Institute of Transformative Technologies in Healthcare; US National Institutes of Health (NIH) grants R01AI080609, U01AI142756, RM1HG009490, R35GM122455, R35GM118062 and P30DK089507; US Defense Threat Reduction Agency (DTRA) grant 1-13-1-0014; and the University of Washington Cystic Fibrosis Foundation Research Development Program (RDP) SINGH15R0. B.Y.M. was supported by a Singapore A*STAR National Science Scholarship (NSS) fellowship; M.H.d.M. was supported by Cystic Fibrosis Foundation Fellowship DEMORAA18F0; A.V.K. was supported by a National Science Centre (NCN) Poland mobility grant UMO-2019/32/T/NZ1/00459; and A.R. was supported by NIH T32 GM095450 and a National Science Foundation graduate research fellowship. V.K.M., J.D.M. and D.R.L. are supported by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
B.Y.M., M.H.d.M., S.B.P., V.K.M., J.D.M. and D.R.L. designed the study; M.H.d.M., J.Z. and D.E.B. designed, performed and analysed experiments to characterize DddA; B.Y.M. designed, performed and analysed nuclear and mitochondrial editing experiments; B.Y.M., M.H.d.M., A.R. and M.C.R. performed sequence analyses; F.H. performed microscopy; A.V.K. designed, performed and analysed mitochondrial biology experiments; B.Y.M., M.H.d.M., S.B.P., A.V.K., V.K.M., J.D.M. and D.R.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The Broad Institute and the University of Washington have filed provisional patent applications on base-editing systems described in this study, listing B.Y.M., M.H.d.M., S.B.P., J.D.M. and D.R.L. as inventors. D.R.L. is a consultant and co-founder of Prime Medicine, Beam Therapeutics, Pairwise Plants and Editas Medicine, companies that use genome editing. V.K.M. is a consultant to 5am Ventures and Janssen Pharmaceuticals.
Additional information
Peer review information Nature thanks Rahul Kohli, Fyodor Urnov and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Analysis of the bactericidal activity of DddA and its activity against dsDNA and RNA substrates.
a, Genomic context of dddA (purple) and dddIA (blue) in B. cenocepacia H111. b, Viability of B. cenocepacia ΔdddA ΔdddIA (recipient) over time during competition with B. cenocepacia donor strains carrying wild-type dddAtox or dddAtoxE1347A. Values and error bars represent the mean ± s.d. of three technical replicates. The experiment was repeated three times with similar results. c, α-VSV-g western blot analysis of total cell lysates of E. coli expressing the indicated deaminases tagged with VSV-G epitope. RNAP-β was used as a loading control. Results are representative of n = 2 independent biological replicates. d, In vitro DNA cytidine deamination assays using double-stranded 36-nt DNA substrates containing AC, TC, CC, and GC with a FAM fluorophore on the forward (A) or reverse (B) strand. Deamination activity results in a cleaved product (P). Images are representative of n = 2 independent biological replicates. e, f, Poisoned primer extension assay to detect deamination of cytidine in single-stranded (e) or double-stranded (f) RNA substrates. Images are representative of n = 2 independent biological replicates. A mix of RNA substrates containing the sequences GUCG or GUUG at the indicated ratios were incubated with purified DddAtox and reverse transcriptase. Primer extension was performed in reactions with ddGTP to terminate primer extension at cytidines. Cytidine deamination yields the 31-mer product.
Extended Data Fig. 2 DddAtox deaminates cytidines in bacteria with strong sequence context preference.
a, Number of SNPs from the indicated nucleotide classifications observed in E. coli Δudg following intoxication with DddAtox or DddAtox(E1347A). b, c, The position of SNPs on the chromosome of E. coli Δudg isolates intoxicated with DddAtox (b) or DddAtox(E1347A) (c). SNPs above the line indicate C-to-T transitions on the plus strand; SNPs below indicate C-to-T transitions on the minus strand. Other mutations are represented on the plus strand. Sequencing coverage was 203–265-fold. d, Deamination assay on DddAtox with double-stranded DNA substrates containing a single C with different nucleotides (A, T, C or G) at the position immediately 5′ of the C (red) (S, substrate; P, product). Images are representative of n = 3 independent biological replicates.
Extended Data Fig. 3 Base-editing efficiencies and indel frequencies of all DddAtox splits in HEK293T cells.
a–h, Each split was assayed in the aureus-N and aureus-C orientation (see Fig. 2b) across spacing region lengths of 12-bp (a), 17-bp (b), 23-bp (c), 28-bp (d), 33-bp (e), 39-bp (f), 44-bp (g) and 60-bp (h). Cells were collected 3 days post-transfection for DNA sequencing. Colours reflect the mean of n = 2 independent biological replicates.
Extended Data Fig. 4 TALE–split DddAtox proteins mediate efficient base editing in nuclear DNA of U2OS cells.
a, Left–G1333-DddAtox-N and Right–G1333-DddAtox-C bind DNA sequences within CCR5. Target cytosines are shown in purple and TALE binding sites are shown in blue. Two copies of UGI proteins (2×-UGI) were fused to the N- or C terminus through a 2- or 16-amino acid linker. Editing efficiencies and indel frequencies for the possible combinations of UGI positions and linker lengths are shown. In the absence of UGI protein, only C9-to-T9 edit was observed. b, Architecture of nuclear-targeting CCR5-DdCBE (see Fig. 3c for optimized DdCBE architecture targeting mtDNA). Target cytosines are shown in purple. c, Editing efficiencies and indel frequencies of cells treated with CCR5-DdCBE and ND6-DdCBE 3-days-post transfection are shown. Dead-DdCBEs containing the inactive DddAtox(E1347A) mutant were used as negative controls. d, Outcomes among edited alleles in which the specified target C is mutated are shown for the indicated base editor. Values and error bars in a, c and d reflect the mean ± s.d. of n = 3 independent biological replicates.
Extended Data Fig. 5 Unoptimized mitoTALE–split DddAtox fusions mediate modest editing of mitochondrial ND6 in HEK293T cells.
a, Architectures of non-UGI containing ND6-mitoTALE–DddAtox fusion pair. DddAtox was split at G1333 or G1397, with each half fused to either the left TALE or the right TALE. TALEs bind to mtDNA sequences (blue) that flank a 15-bp spacing region in mitochondrial ND6. Target cytosines are shown in purple. The last TALE repeat (*) did not match the reference genome9 (see Supplementary Table 4). b, mtDNA editing efficiencies of mitoTALE–DddAtox pairs in the listed split orientations. The dashed line is drawn at 0.1%. Values and error bars reflect the mean ± s.d. of n = 3 independent biological replicates.
Extended Data Fig. 6 DdCBE editing in HEK293T cells persist over multiple divisions while maintaining cell viability and mitochondrial DNA integrity.
a–e, Editing efficiencies for optimized ND6-DdCBE (a), MTND5P1-DdCBE (denoted ND5.1-DdCBE) (b), MTND5P2-DdCBE (denoted ND5.2-DdCBE) (c), ATP8-DdCBE (d) and BE2max and BE4max (e) are shown for each time point. C•G-to-T•A conversions at protein-coding genes that generate missense mutations (green) of the putative amino acid (red) are shown. f, Western blots of ND6-, ND5.1-, ND5.2- and ATP8-DdCBE at various time points. The right halves were Flag-tagged and the left halves were HA-tagged. Day 3 images are representative of n = 3 independent biological replicates; n = 1 for day 6 and day 12 images (see Supplementary Data 3 for uncropped images and fluorescent tagging of each half). Nuclear β-actin was used as a loading control. g, Cell viability was measured by recording the luminescence at the indicated time points. Luminescence values were normalized to the untreated control. h, DNA gel of PCR-amplified mtDNA captured as two amplicons (red). Images are representative of n = 3 independent biological replicates (see Supplementary Data 4 for uncropped images). i, mtDNA levels of DdCBE-edited cells were measured by qPCR relative to untreated cells. Values and error bars in a–e, g and i reflect the mean ± s.d. of n = 3 independent biological replicates. For a–e, asterisks indicate significant editing based on a comparison between indicated time points. *P < 0.05 and **P < 0.01 by Student’s two-tailed paired t-test. Individual P values are listed in Supplementary Table 7.
Extended Data Fig. 7 Stalling mtDNA replication impairs mitochondrial base editing in human cells.
a, Schematic of experimental design. Addition of doxycycline (Dox) induces the stable expression of a dominant-negative mutant of DNA polymerase-gamma containing a D1135A substitution (POLGdn) in a HEK293-derived cell line31. Total cell lysate was collected at indicated time points for western blotting of POLGdn in n = 3 independent biological replicates. b, mtDNA levels of uninduced (no Dox) and induced (+Dox) cells treated with indicated DdCBE 48 h post-transfection. mtDNA levels were measured by qPCR and normalized to uninduced cells without DdCBE treatment. c, Editing efficiencies of indicated DdCBE in uninduced and induced cells 48 h post-transfection. All values and error bars in b and c reflect the mean ± s.d. of n = 3 independent biological replicates.
Extended Data Fig. 8 Effect of DdCBE editing on mitochondrial function and mtDNA homeostasis.
a, mtDNA levels of ND4-edited cells measured by qPCR relative to cells treated with dead ND4-DdCBE. b, mtRNA levels of ND4-edited cells measured by reverse transcription-qPCR relative to cells treated with dead ND4-DdCBE. c–f, Confirmation of editing by Sanger sequencing and OCR of cells treated with ND5.1-DdCBE (c), ND5.2-DdCBE (d), MTND5P3-DdCBE (denoted ND5.3-DdCBE) (e) and ND1-DdCBE (f). Untreated cells were used as controls. All cells were collected 6 days post-transfection. For all Sanger sequencing plots, n = 3 independent biological replicates. All values and error bars shown in a, b and OCR plots in c–f reflect the mean ± s.e.m. of n = 3 independent biological replicates. For a and b, Student’s unpaired two-tailed t-test was applied. NS, not significant (P > 0.05).
Extended Data Fig. 9 Off-target editing activity of DdCBEs in nuclear DNA of HEK293T cells.
a–c, The on-target editing site in mtDNA and the corresponding nuclear DNA sequence with the greatest homology are shown for ND6-DdCBE (a), ND5.1-DdCBE (b) and ND4-DdCBE (c). TALE binding sites begin at N0 and are shown in blue. Target cytosines are in purple. Nucleotide mismatches between the mtDNA and nuclear pseudogene are in red. Editing efficiencies are measured by targeted amplicon sequencing 3 days post-transfection (a, b) or six days post-transfection (c) (see Methods for primer sequences). Each amplicon was sequenced at >44,000× coverage. All values and error bars reflect the mean ± s.d. of n = 3 independent biological replicates. Student’s unpaired two-tailed t-test was applied. *P < 0.05; NS, not significant (P > 0.05).
Extended Data Fig. 10 TALE arrays need to bind to mtDNA sequences positioned in close proximity to reassemble catalytically active DddAtox for off-target editing.
a, The identities and relative binding positions of each mismatched (MM) TALE–DddAtox half is shown. MM-1 and MM-2 contain a TALE-bound DddAtox half and a TALE-free DddAtox half. MM-3 and MM-4 contain DddAtox halves fused to TALE repeat arrays that bind to distant regions in mtDNA. ND6-Right TALE contains a permissive N-terminal domain (see Supplementary Table 4). b, The average percentage of genome-wide C•G-to-T•A off-target editing in mtDNA by indicated DdCBE and MM pairs are shown. The dashed line represents the percentage of endogenous C•G-to-T•A conversions in mtDNA as measured in the untreated control. Values and error bars reflect the mean ± s.e.m. of n = 3 independent biological replicates.
Extended Data Fig. 11 Predicted effects of off-target SNVs on mitochondrial DNA sequence and protein function.
a, Classification of off-target SNVs into noncoding or coding mutations. Mutations occurring in protein-coding regions of mtDNA were further categorized into synonymous, missense or nonsense mutations. b, For nonsynonymous SNVs, SIFT was used to predict the effect of these mutations on protein function. High- or low-confidence calls (indicated in parentheses) were made according to the standard parameters of the prediction software. c, Editing efficiencies of selected off-target TC bases in the indicated sequence contexts are shown. HEK293T cells were treated with the indicated DdCBE and collected 3 days post-transfection for DNA sequencing. Values and error bars reflect the mean ± s.d. of n = 3 independent biological replicates.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary Data Figures 1–4, Supplementary Tables 1, 4, 10-13 and Supplementary Sequences 1-4.
41586_2020_2477_MOESM3_ESM.xlsx
Supplementary Table 2 Schematic and sequences of guide RNAs for split-DddAtox–Cas9 screen. dSpCas9 guide RNAs (spG7 and spG6) are paired with with SaKKH guide RNAs (saG1 to saG4) to generate spacing regions with lengths between 12 and 60 bp.
41586_2020_2477_MOESM4_ESM.xlsx
Supplementary Table 3 Base percentages at each position of the EMX1 locus for DddAtox–Cas9 splits with no guide RNAs and monomers with their respective gRNAs. For G1333/G1397 DddAtox–dSpCas9 or G1333/G1397 DddAtox–SaKKH-Cas9(D10A) halves, these halves are directed to a site within EMX1 by a guide RNA spG4 (magenta) or saG4 (blue), respectively. The reciprocal DddAtox half of each fusion was absent. The 60-bp spacing region in highlighted in yellow. All shorter spacing regions are nested within the 60-bp spacing. Base percentages are obtained 3 days post-transfection and representative of n=2 independent biological replicates.
41586_2020_2477_MOESM5_ESM.xlsx
Supplementary Table 5 Indel frequencies of DdCBEs in their optimized orientations. Shown are the percent of indels in HEK293T cells for each DdCBE in its optimized split orientation (see Fig. 4a-g for on-target editing efficiencies of optimized DdCBEs). Cells treated with ND5.1-, ND5.2-, and ATP8-DdCBE were harvested 3 days post-transfection; cells treated with ND1-, ND2, ND4- and ND5.3-DdCBE were harvested 6 days post-transfection.
41586_2020_2477_MOESM6_ESM.xlsx
Supplementary Table 6 Base percentages at each position of the H-strand of mtDNA for indicated DdCBE in its optimized split orientation. TALE-binding sites are highlighted in blue. On-target C•G bases are highlighted in yellow. Base percentages are measured 3 days post-transfection of HEK293T cells.
41586_2020_2477_MOESM7_ESM.xlsx
Supplementary Table 7 P-values from comparison of editing efficiencies from time course experiments in HEK293T cells and U2OS cells. For a given DdCBE, the P-value for editing efficiencies of target cytidine across two cumulative timepoints is shown. P-values were calculated using the Student's two-tailed paired t-test. Entries are highlighted in red if the P-value indicated a significant difference (P<0.05).
41586_2020_2477_MOESM8_ESM.xlsx
Supplementary Table 8 Unique off-target SNVs mediated by DdCBEs. SNVs called by VarScan 2 were considered high-confidence if the percentage frequency of a given SNV is >0.1% in one or more replicates. For samples treated with DdCBEs, dead DdCBEs, and TALE-free G1397 DddAtox, the combined number of unique off-target SNVs from all three independent biological replicates that are absent in the untreated control are shown. For the untreated control, heteroplasmic mutations were excluded. SNV positions for each DdCBE treatment were from the NC_012920 reference genome. On-target SNVs are highlighted in red.
41586_2020_2477_MOESM9_ESM.xlsx
Supplementary Table 9 Overlapping off-target SNVs. Shown are the list of overlapping off-target SNVs between ND6- and ND5.1-DdCBE, ND6-, ND5.1- and ATP8-DdCBE and ND6-, ND5.1-, ND5.2- and ATP8-DdCBE. The average frequency of each SNV is shown.
Rights and permissions
About this article
Cite this article
Mok, B.Y., de Moraes, M.H., Zeng, J. et al. A bacterial cytidine deaminase toxin enables CRISPR-free mitochondrial base editing. Nature 583, 631–637 (2020). https://doi.org/10.1038/s41586-020-2477-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2477-4
This article is cited by
-
Targeted C•G-to-T•A base editing with TALE-cytosine deaminases in plants
BMC Biology (2024)
-
Focusing on mitochondria in the brain: from biology to therapeutics
Translational Neurodegeneration (2024)
-
Mitochondrial DNA editing in potato through mitoTALEN and mitoTALECD: molecular characterization and stability of editing events
Plant Methods (2024)
-
Single-cell analysis reveals context-dependent, cell-level selection of mtDNA
Nature (2024)
-
Discovering deaminases using AlphaFold2: a strategy to search for tool proteins for gene editing
Signal Transduction and Targeted Therapy (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.