Abstract
Arrestin proteins bind to active, phosphorylated G-protein-coupled receptors (GPCRs), thereby preventing G-protein coupling, triggering receptor internalization and affecting various downstream signalling pathways1,2. Although there is a wealth of structural information detailing the interactions between GPCRs and G proteins, less is known about how arrestins engage GPCRs. Here we report a cryo-electron microscopy structure of full-length human neurotensin receptor 1 (NTSR1) in complex with truncated human β-arrestin 1 (βarr1(ΔCT)). We find that phosphorylation of NTSR1 is critical for the formation of a stable complex with βarr1(ΔCT), and identify phosphorylated sites in both the third intracellular loop and the C terminus that may promote this interaction. In addition, we observe a phosphatidylinositol-4,5-bisphosphate molecule forming a bridge between the membrane side of NTSR1 transmembrane segments 1 and 4 and the C-lobe of arrestin. Compared with a structure of a rhodopsin–arrestin-1 complex, in our structure arrestin is rotated by approximately 85° relative to the receptor. These findings highlight both conserved aspects and plasticity among arrestin–receptor interactions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The cryo-EM density map for the NTSR1–βarr1(ΔCT) complex has been deposited in the Electron Microscopy Data Bank under accession code EMD-20836. The coordinates for the model of NTSR1–βarr1(ΔCT) have been deposited in the PDB under accession number 6UP7. Proteomics data have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository and can be accessed at https://doi.org/10.6019/PXD016224. The data supporting the findings of this study are available as Supplementary Information (Supplementary Tables 1–39 and Supplementary Figs. 2–5). All other data are available upon request to the corresponding authors.
References
Scheerer, P. & Sommer, M. E. Structural mechanism of arrestin activation. Curr. Opin. Struct. Biol. 45, 160–169 (2017).
Hilger, D., Masureel, M. & Kobilka, B. K. Structure and dynamics of GPCR signaling complexes. Nat. Struct. Mol. Biol. 25, 4–12 (2018).
Rankovic, Z., Brust, T. F. & Bohn, L. M. Biased agonism: an emerging paradigm in GPCR drug discovery. Bioorg. Med. Chem. Lett. 26, 241–250 (2016).
Luttrell, L. M., Maudsley, S. & Bohn, L. M. Fulfilling the promise of “biased” G protein-coupled receptor agonism. Mol. Pharmacol. 88, 579–588 (2015).
Barak, L. S. et al. ML314: a biased neurotensin receptor ligand for methamphetamine abuse. ACS Chem. Biol. 11, 1880–1890 (2016).
Peterson, Y. K. & Luttrell, L. M. The diverse roles of arrestin scaffolds in G protein-coupled receptor signaling. Pharmacol. Rev. 69, 256–297 (2017).
Laporte, S. A. & Scott, M. G. H. β-Arrestins: multitask scaffolds orchestrating the where and when in cell signalling. Methods Mol. Biol. 1957, 9–55 (2019).
Krishna Kumar, K. et al. Structure of a signaling cannabinoid receptor 1-G protein complex. Cell 176, 448–458.e12 (2019).
Kato, H. E. et al. Conformational transitions of a neurotensin receptor 1–Gi1 complex. Nature 572, 80–85 (2019).
Maeda, S., Qu, Q., Robertson, M. J., Skiniotis, G. & Kobilka, B. K. Structures of the M1 and M2 muscarinic acetylcholine receptor/G-protein complexes. Science 364, 552–557 (2019).
Gao, Y. et al. Structures of the rhodopsin–transducin complex: insights into G-protein activation. Mol. Cell 75, 781–790 (2019).
García-Nafría, J. & Tate, C. G. Cryo-EM structures of GPCRs coupled to Gs, Gi and Go. Mol. Cell. Endocrinol. 488, 1–13 (2019).
Glukhova, A. et al. Rules of engagement: GPCRs and G proteins. ACS Pharmacol. Transl. Sci. 1, 73–83 (2018).
Zhou, X.E. et al. Identification of phosphorylation codes for arrestin recruitment by G protein-coupled receptors. Cell 170, 457–469.e13 (2017).
Yang, F. et al. Phospho-selective mechanisms of arrestin conformations and functions revealed by unnatural amino acid incorporation and 19F-NMR. Nat. Commun. 6, 8202 (2015).
Lohse, M. J., Benovic, J. L., Codina, J., Caron, M. G. & Lefkowitz, R. J. β-Arrestin: a protein that regulates β-adrenergic receptor function. Science 248, 1547–1550 (1990).
Rostène, W. H. & Alexander, M. J. Neurotensin and neuroendocrine regulation. Front. Neuroendocrinol. 18, 115–173 (1997).
Besserer-Offroy, É. et al. The signaling signature of the neurotensin type 1 receptor with endogenous ligands. Eur. J. Pharmacol. 805, 1–13 (2017).
White, J. F. et al. Structure of the agonist-bound neurotensin receptor. Nature 490, 508–513 (2012).
Egloff, P. et al. Structure of signaling-competent neurotensin receptor 1 obtained by directed evolution in Escherichia coli. Proc. Natl Acad. Sci. USA 111, E655–E662 (2014).
Krumm, B. E., White, J. F., Shah, P. & Grisshammer, R. Structural prerequisites for G-protein activation by the neurotensin receptor. Nat. Commun. 6, 7895 (2015).
Komolov, K. E. et al. Structural and functional analysis of a β2-adrenergic receptor complex with GRK5. Cell 169, 407–421.e16 (2017).
Komolov, K. E. & Benovic, J. L. G protein-coupled receptor kinases: Past, present and future. Cell. Signal. 41, 17–24 (2018).
Inagaki, S. et al. G protein-coupled receptor kinase 2 (GRK2) and 5 (GRK5) exhibit selective phosphorylation of the neurotensin receptor in vitro. Biochemistry 54, 4320–4329 (2015).
Vishnivetskiy, S. A. et al. An additional phosphate-binding element in arrestin molecule. Implications for the mechanism of arrestin activation. J. Biol. Chem. 275, 41049–41057 (2000).
Peisley, A. & Skiniotis, G. 2D projection analysis of GPCR complexes by negative stain electron microscopy. Methods Mol. Biol. 1335, 29–38 (2015).
Nakane, T., Kimanius, D., Lindahl, E. & Scheres, S. H. Characterisation of molecular motions in cryo-EM single-particle data by multi-body refinement in RELION. eLife 7, e36861 (2018).
Gurevich, V. V. & Gurevich, E. V. The structural basis of arrestin-mediated regulation of G-protein-coupled receptors. Pharmacol. Ther. 110, 465–502 (2006).
Gaidarov, I., Krupnick, J. G., Falck, J. R., Benovic, J. L. & Keen, J. H. Arrestin function in G protein-coupled receptor endocytosis requires phosphoinositide binding. EMBO J. 18, 871–881 (1999).
Chen, Q. et al. Structural basis of arrestin-3 activation and signaling. Nat. Commun. 8, 1427 (2017).
Milano, S. K., Kim, Y. M., Stefano, F. P., Benovic, J. L. & Brenner, C. Nonvisual arrestin oligomerization and cellular localization are regulated by inositol hexakisphosphate binding. J. Biol. Chem. 281, 9812–9823 (2006).
Lally, C. C., Bauer, B., Selent, J. & Sommer, M. E. C-edge loops of arrestin function as a membrane anchor. Nat. Commun. 8, 14258 (2017).
Kang, D. S. et al. Structure of an arrestin2–clathrin complex reveals a novel clathrin binding domain that modulates receptor trafficking. J. Biol. Chem. 284, 29860–29872 (2009).
Gurevich, V. V. et al. Arrestin interactions with G protein-coupled receptors. Direct binding studies of wild type and mutant arrestins with rhodopsin, β2-adrenergic, and m2 muscarinic cholinergic receptors. J. Biol. Chem. 270, 720–731 (1995).
Han, M., Gurevich, V. V., Vishnivetskiy, S. A., Sigler, P. B. & Schubert, C. Crystal structure of β-arrestin at 1.9 Å: possible mechanism of receptor binding and membrane translocation. Structure 9, 869–880 (2001).
Shukla, A. K. et al. Structure of active β-arrestin-1 bound to a G-protein-coupled receptor phosphopeptide. Nature 497, 137–141 (2013).
Szczepek, M. et al. Crystal structure of a common GPCR-binding interface for G protein and arrestin. Nat. Commun. 5, 4801 (2014).
Eichel, K. et al. Catalytic activation of β-arrestin by GPCRs. Nature 557, 381–386 (2018).
Yen, H. Y. et al. PtdIns(4,5)P2 stabilizes active states of GPCRs and enhances selectivity of G-protein coupling. Nature 559, 423–427 (2018).
Inoue, A. et al. Illuminating G-protein-coupling selectivity of GPCRs. Cell 177, 1933–1947.e25 (2019).
Staus, D. P. et al. Structure of the M2 muscarinic receptor–β-arrestin complex in a lipid nanodisc. Nature https://www.doi.org/10.1038/s41586-020-1954-0 (2020).
Eichel, K. & von Zastrow, M. Subcellular organization of GPCR signaling. Trends Pharmacol. Sci. 39, 200–208 (2018).
Eichel, K., Jullié, D. & von Zastrow, M. β-Arrestin drives MAP kinase signalling from clathrin-coated structures after GPCR dissociation. Nat. Cell Biol. 18, 303–310 (2016).
Lee, M. H. et al. The conformational signature of β-arrestin2 predicts its trafficking and signalling functions. Nature 531, 665–668 (2016).
Nuber, S. et al. β-Arrestin biosensors reveal a rapid, receptor-dependent activation/deactivation cycle. Nature 531, 661–664 (2016).
Goddard, T. D. et al. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Beyett, T. S. et al. Perturbation of the interactions of calmodulin with GRK5 using a natural product chemical probe. Proc. Natl Acad. Sci. USA 116, 15895–15900 (2019).
Schorb, M., Haberbosch, I., Hagen, W. J. H., Schwab, Y. & Mastronarde, D. N. Software tools for automated transmission electron microscopy. Nat. Methods 16, 471–477 (2019).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: Real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Heymann, J. B. & Belnap, D. M. Bsoft: image processing and molecular modeling for electron microscopy. J. Struct. Biol. 157, 3–18 (2007).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Adams, P. D. et al. The Phenix software for automated determination of macromolecular structures. Methods 55, 94–106 (2011).
Williams, C. J. et al. MolProbity: More and better reference data for improved all-atom structure validation. Protein Sci. 27, 293–315 (2018).
Dixon, A. S. et al. NanoLuc complementation reporter optimized for accurate measurement of protein interactions in cells. ACS Chem. Biol. 11, 400–408 (2016).
Acknowledgements
We thank M. Krawitzky and C. Adams for instrument and software access; R. Leib, F. Liu and M. Bern for discussions pertaining to mass spectrometry data analysis; G. Lam and B. Fitch for assistance with lipidomics analysis; K. Sato, Y. Sugamura and A. Inoue for plasmid construction and cell-based GPCR assays; and D. Mayer for suggestions on arrestin purification. This work was supported in part by National Institutes of Health grants R01NS028471 (B.K.K.), 1U19AI109662 (J.S.G.) and P30 CA124435 (for using the Stanford Cancer Institute Proteomics/Mass Spectrometry Shared Resource). Additional support to G.S. and B.K.K. was provided by the Mathers Foundation. B.K.K. is a Chan-Zuckerberg Biohub investigator. M.M. was supported by an American Heart Association postdoctoral fellowship (17POST33410958). J.J. is a Damon Runyon Fellow supported by the Damon Runyon Cancer Research Foundation (DRG-2318-18). A.I. was funded by the PRIME 18gm5910013 and the LEAP 18gm0010004 from the Japan Agency for Medical Research and Development (AMED) and KAKENHI 17K08264 from the Japan Society for the Promotion of Science (JSPS). H.E.K. was funded by KAKENHI 19H03163 from JSPS, The Naito Foundation, The Kurata Grants from The Hitachi Global Foundation, and Grant-in-Aid from the Tokyo Biochemical Research Foundation.
Author information
Authors and Affiliations
Contributions
W.H. initiated the project and performed GPCR screening to identify strong arrestin couplers with H.E.K. W.H. expressed and purified NTSR1, screened NTSR1 constructs and performed initial NTSR1–βarr1 complexing. M.M. established arrestin expression and purification, performed fluorescence polarization measurements, engineered pre-activated arrestin constructs and developed fluorescence and electron paramagnetic resonance reporters to guide the complex formation strategy. Q.Q. screened samples by negative-stain electron microscopy and cryo-EM, prepared grids, collected and processed cryo-EM data and generated the cryo-EM maps. J.J. expressed and purified GRK5, screened and optimized NTSR1 phosphorylation conditions, phosphorylated NTSR1 for cryo-EM studies, performed mass spectrometry experiments and analysed all mass spectrometry data. M.M. and J.J. established GRK5 expression and purification conditions and screened and optimized NTSR1–βarr1(ΔCT) complexing conditions. W.H. and J.J. chose crosslinkers to screen. W.H., M.M. and J.J. screened crosslinkers and optimized sample preparation for cryo-EM. W.H. and Q.Q. build the NTSR1–βarr1(ΔCT) model, with contributions from J.J. and M.J.R. K.C.N performed lipidomics measurements, which were overseen by J.J. and J.S.G. A.I. performed NanoBiT experiments. M.M., J.J., G.S. and B.K.K. wrote the paper with input from W.H. and Q.Q. B.K.K. and G.S. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
B.K.K. is a co-founder of and consultant for ConfometRx.
Additional information
Peer review information Nature thanks Oliver Clarke, Martin Lohse and John Tesmer for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Screening for GPCR–βarr1 complexes.
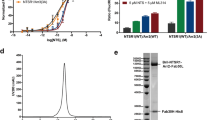
A fluorescence size-exclusion chromatography (FSEC) approach was used to identify GPCRs that display strong coupling to βarr1 in vitro. a, Overview of the screening strategy. Small-scale transfection and purification of candidate GFP-tagged GPCRs was followed by complexing with activated βarr1, denoted by βarr1*. βarr1 was activated in situ by mixing with Fab30 and V2Rpp (Methods) before being added (in excess) to the agonist-bound receptor. Complex formation was assessed by FSEC. b, Representative FSEC traces. The trace of NTSR1 + βarr1* (left) is notable in that it shows a shoulder peak, which indicates substantial complex formation, whereas the trace for β2 adrenergic receptor (β2AR, right) + βarr1* shows little or no complex formation. Experiments were performed independently twice, with similar results.
Extended Data Fig. 2 Phosphorylation of NTSR1 is crucial for the coupling of βarr1.
a, The phosphorylation state of NTSR1 was assessed using ion-exchange chromatography. Chromatograms of aliquots taken at various time points (T, shown in minutes) are overlaid, and the relative amounts of unphosphorylated and phosphorylated species were measured using an optimized stepped elution profile. This experiment was performed once. b, Graphical representation of normalized relative ratio of phosphorylated and unphosphorylated receptor from data shown in a. c, d, FSEC chromatogram for screening βarr1 constructs for forming a stable complex with either unphosphorylated (c) or GRK5-phosphorylated (d) NTSR1. Elution volumes for free receptor, free βarr1 and the receptor–βarr1 complex are shown. The unphosphorylated receptor does not form a complex with any of the βarr1 constructs tested, whereas the phosphorylated receptor couples moderately to full-length βarr1 and strongly to the pre-activated βarr1(ΔCT) construct. Experiments were performed independently twice, with similar results.
Extended Data Fig. 3 Overview of sample preparation and representative cryo-EM preparation.
a, Workflow for the assembly of the NTSR1–βarr1(ΔCT) complex. Receptor, GRK5 and βarr1(ΔCT) were purified separately. Phosphorylation of NTSR1 by GRK5 in the presence of diC8-PtdIns(4,5)P2 was monitored by analytical ion-exchange chromatography (shown in inset as gradient elution from a representative preparation; a stepped elution ion-exchange chromatography run is shown in Extended Data Fig. 2a), followed by M1 affinity purification. The phosphorylated receptor was combined with βarr1(ΔCT) and additional diC8-PtdIns(4,5)P2 and separated by size-exclusion chromatography (SEC) to isolate the NTSR1–βarr1(ΔCT) complex. The complex was labelled with sulfo-LC-SDA and irradiated with UV light. After a second round of SEC to re-isolate the complex, the sample was concentrated and used for cryo-EM. b, Representative sample preparation: phosphorylated NTSR1 was mixed with βarr1(ΔCT) and purified by SEC (blue curve, SEC 1). Complex fractions were combined and treated with crosslinker, quenched, then UV-irradiated. Lanes 2 and 3 of the SDS–PAGE gel show pooled samples before and after crosslinking and UV irradiation; about 25% of the sample is crosslinked (based on densitometry, top two bands relative to total). The crosslinked sample was re-run on SEC (red curve, SEC2) and SDS–PAGE of peak fractions is shown. Peak fractions were combined, concentrated and used for cryo-EM. Representative negative-stain electron microscopy images of the NTSR1–βarr1(ΔCT) complex show similar homogeneity, pre- and post-crosslinking. Enlarged views of rotated representative particles (denoted by yellow and blue circles) are shown. Four sample preparations, which all gave similar results by SEC, SDS–PAGE and negative-stain analysis, were used to generate the cryo-EM dataset.
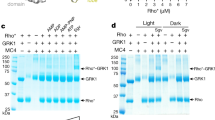
Extended Data Fig. 4 Mass spectrometry analysis of crosslinks in the NTSR1–βarr1(ΔCT) complex.
a, Lysine residues modified within the NTSR1–βarr1(ΔCT) complex after treatment with sulfo-LC-SDA were identified by mass spectrometry. Although mass spectrometry data were collected for each band observed by SDS–PAGE separately (Extended Data Fig. 3b), data were combined for analysis as the overall sample was imaged by cryo-EM. For each lysine residue in the complex the degree of modification was approximated as the percentage of times a lysine was found to be modified relative to the total number of times that lysine is observed across all peptides. The Cα for lysine residues that were observed to be modified less than 30% of the time are shown as grey spheres, whereas those found to be modified more than 30% of the time are shown as green spheres. A tabulated analysis is presented in Supplementary Tables 1–7. b, Of the modified lysine residues, several mainly formed dead-end crosslinks (reaction of the activated diazirene with water); those with more than 30% of the lysine residues resulting in dead-end crosslinks are shown as maroon spheres (Cα). c, Only one intermolecular crosslink is observed that is consistent with the structural model obtained from cryo-EM. This crosslink was localized to ICL1 (K91) and a β-strand (shown in green) in the arrestin C-lobe. d, Several intramolecular crosslinks within ICL2 and the adjacent helices are observed. Reactive lysine residues are shown as green spheres (for Cα), and crosslinks were localized to the region shown in green. e, Multiple intramolecular crosslinks within arrestin were observed. Each reactive lysine residue (shown as a Cα sphere) that forms an intrapeptide crosslink, or an intramolecular (within arrestin) crosslink, is coloured to match the region in which the observed crosslink could be localized. Each band was analysed once by mass spectrometry from a representative cryo-EM preparation.
Extended Data Fig. 5 Cryo-EM workflow.
Flow chart of cryo-EM data analysis. Global resolution was determined by FSC with a cut-off of 0.143. The mask used for sharpening was also used for global resolution determination.
Extended Data Fig. 6 Cryo-EM map resolution and model validation, with multi-body analysis revealing structural heterogeneity within the NTSR1–βarr1(ΔCT) complex.
a, Cross validation between the map and the model. The model was refined against one half map after displacement of atoms by 0.2 Å, and FSC curves were calculated between this model and the working half1 map (red), the free half2 map (green) and the final cryo-EM map (full dataset, black) by Mtriage implemented in PHENIX. b, Local resolution of the final 4.2 Å map was estimated by Bsoft. c, Euler angle distribution of the particle set used in the final map. d, Representative sections of the model with accompanying regions of density from the electron microscopy map. e, Masks used for multi-body refinement. f, The contributions of each of the twelve eigenvectors (numbered along the x-axis) to the variance of the overall final map. g, Maps corresponding to the second and ninth components of the first eigenvector (f) are aligned, showing swing-like motion of βarr1(ΔCT) with respect to NTSR1. h, Maps corresponding to first and tenth components of the second eigenvector (f) are superimposed, indicating the tilt-like motion of βarr1(ΔCT) with respect to NTSR1.
Extended Data Fig. 7 Arrestin orientation and interdomain twist.
a, Additional views of Fig. 1c, showing an overlay between NTSR1–βarr1(ΔCT) and Rho–Arr1: cytoplasmic (intracellular) view (left), side view (middle) and extracellular view (right). b, Compared with the inactive structure, βarr1(ΔCT) in complex with NTSR1 displays an additional interdomain twist between its N-lobe and its C-lobe, which is a hallmark of activation. Comparison of the interdomain twist between NTSR1–βarr1(ΔCT) (blue, present structure) and V2Rpp–βarr1 (orange, PDB: 4JQI) relative to inactive βarr1 (wheat, PDB: 1G4M). A smaller interdomain twist and displacement between respective C-lobes is observed for NTSR1-bound βarr1 (16°, 3.6 Å) compared with V2Rpp-bound βarr1 (22°, 4.6 Å), indicated by arrows drawn to relative scale. A similar comparison for rhodopsin-bound arrestin-1 (green, PDB: 5W0P) and pre-activated p44 arrestin-1 (purple, PDB: 4J2Q) relative to inactive arrestin-1 (pink, PDB: 1CF1) yields interdomain twist and displacement values of 20°, 5.4 Å and 23°, 6.1 Å, respectively.
Extended Data Fig. 8 Identification of GRK5 phosphorylation sites in human NTSR1 and comparison of phosphate-specific contacts in NTSR1–βarr1(ΔCT) to those in Rho–Arr1 and V2Rpp–βarr1.
a, Snake diagram of human NTSR1 showing identified phosphosites in ICL3 and the receptor C terminus. The trypsin/LysC digestion sites are shown in light blue. All four phosphosites for ICL3 are in a single peptide beginning with QAA at the end of TM5 and ending at PGR at the beginning of TM6. Phosphosites in the C terminus occur on two peptides, one containing S396, RPAFSR, as well as several missed cleavage variants; and one containing S401, S403, S404, T407 and T413. Tabulated fragment ions are presented in Supplementary Tables 8–39 and spectra are shown in Supplementary Figs. 2–5. Any post translational modifications outside of these two discussed regions are not shown for clarity. These experiments were performed independently twice, and analysed by two or three technical replicates each time, all with similar results. b, Overlay of arrestin structures, in which binding of phosphorylated receptor residues contributes to stabilizing the active state of arrestin. Left, equivalent phosphoresidues on different receptor C-tails (shown in stick representation and coloured accordingly: NTSR1, yellow; V2Rpp, red; rhodopsin, pink) contribute to arrestin activation by interacting with a lysine residue on the gate loop (K294, shown as Cα spheres), thereby breaking the polar core. Right, phosphoresidue pT347 in the V2Rpp interacts with residue K77 at the base of the finger loop. A similarly positioned phosphoresidue in the ICL3 of NTSR1 (pS287) probably interacts with arrestin residues R76/K77 in a similar manner.
Extended Data Fig. 9 Membrane context and curvature for NTSR1–βarr1(ΔCT).
a, Comparison of the structures of NTSR1–βarr1(ΔCT) and NTSR1–cGi, for which membrane-embedded regions were computed using the Orientations of Proteins in Membranes server (https://opm.phar.umich.edu/). The membrane is indicated by grey spheres to show the extent to which the C-edge would insert into the membrane. b, The extent of arrestin tilt observed in the NTSR1–βarr1(ΔCT) structure could also be relevant to the increased membrane curvature observed in endocytic vesicles. As an example, several membrane bilayers are drawn (in red) to scale, comparing the degree of curvature for vesicles ranging from 120 nm to 50 nm in diameter relative to a planar bilayer.
Supplementary information
Supplementary Information
This file contains Supplementary Figure 1, which shows that NTSR1-βarr1(ΔCT) complex samples contain diC8-PtdIns(4,5)P2, Supplementary Figures 2-5 show the spectra used for assignment of the GRK5 phosphosites in NTSR1 and Supplementary Figure 6, which shows the uncropped gel displayed in Extended Data Figure 3b. It also contains Supplementary Tables 1-7 which present information about crosslinks observed in the NTSR1-βarr1(ΔCT) complex. There is one table for each band excised from the SDS-PAGE gel, two for global analysis (one for each protein) and one for mapping crosslinks. Lastly it contains Supplementary Tables 8-39 that enumerate the observed fragmentation ions that led to the phosphosite assignments presented in the text.
Rights and permissions
About this article
Cite this article
Huang, W., Masureel, M., Qu, Q. et al. Structure of the neurotensin receptor 1 in complex with β-arrestin 1. Nature 579, 303–308 (2020). https://doi.org/10.1038/s41586-020-1953-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-1953-1
This article is cited by
-
Molecular mechanism of GPCR spatial organization at the plasma membrane
Nature Chemical Biology (2024)
-
Disordered-to-ordered transitions in assembly factors allow the complex II catalytic subunit to switch binding partners
Nature Communications (2024)
-
Structural insights into the activation and inhibition of CXC chemokine receptor 3
Nature Structural & Molecular Biology (2024)
-
Cryo-electron microscopy for GPCR research and drug discovery in endocrinology and metabolism
Nature Reviews Endocrinology (2024)
-
Ligand efficacy modulates conformational dynamics of the µ-opioid receptor
Nature (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.