Abstract
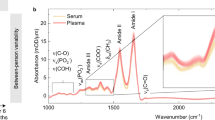
The proper functioning of living systems and physiological phenotypes depends on molecular composition. Yet simultaneous quantitative detection of a wide variety of molecules remains a challenge1,2,3,4,5,6,7,8. Here we show how broadband optical coherence opens up opportunities for fingerprinting complex molecular ensembles in their natural environment. Vibrationally excited molecules emit a coherent electric field following few-cycle infrared laser excitation9,10,11,12, and this field is specific to the sample’s molecular composition. Employing electro-optic sampling10,12,13,14,15, we directly measure this global molecular fingerprint down to field strengths 107 times weaker than that of the excitation. This enables transillumination of intact living systems with thicknesses of the order of 0.1 millimetres, permitting broadband infrared spectroscopic probing of human cells and plant leaves. In a proof-of-concept analysis of human blood serum, temporal isolation of the infrared electric-field fingerprint from its excitation along with its sampling with attosecond timing precision results in detection sensitivity of submicrograms per millilitre of blood serum and a detectable dynamic range of molecular concentration exceeding 105. This technique promises improved molecular sensitivity and molecular coverage for probing complex, real-world biological and medical settings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Geyer, P. E., Holdt, L. M., Teupser, D. & Mann, M. Revisiting biomarker discovery by plasma proteomics. Mol. Syst. Biol. 13, 942 (2017).
Barth, A. & Haris, P. I. Biological and Biomedical Infrared Spectroscopy (IOS Press, 2009).
Lasch, P. & Kneipp, J. Biomedical Vibrational Spectroscopy (Wiley, 2010).
Baker, M. J. et al. Developing and understanding biofluid vibrational spectroscopy: a critical review. Chem. Soc. Rev. 45, 1803–1818 (2016).
Hasin, Y., Seldin, M. & Lusis, A. Multi-omics approaches to disease. Genome Biol. 18, 83 (2017).
Chen, R. et al. Personal omics profiling reveals dynamic molecular and medical phenotypes. Cell 148, 1293–1307 (2012).
Türker-Kaya, S. & Huck, C. A review of mid-infrared and near-infrared imaging: principles, concepts and applications in plant tissue analysis. Molecules 22, 168 (2017).
Doherty, J., Cinque, G. & Gardner, P. Single-cell analysis using Fourier transform infrared microspectroscopy. Appl. Spectrosc. Rev. 52, 560–587 (2017).
Laubereau, A. & Kaiser, W. Vibrational dynamics of liquids and solids investigated by picosecond light pulses. Rev. Mod. Phys. 50, 607–665 (1978).
Sell, A., Scheu, R., Leitenstorfer, A. & Huber, R. Field-resolved detection of phase-locked infrared transients from a compact Er:fiber system tunable between 55 and 107 THz. Appl. Phys. Lett. 93, 251107 (2008).
Coddington, I., Swann, W. C. & Newbury, N. R. Time-domain spectroscopy of molecular free-induction decay in the infrared. Opt. Lett. 35, 1395–1397 (2010).
Kowligy, A. S. et al. Infrared electric field sampled frequency comb spectroscopy. Sci. Adv. 5, eaaw8794 (2019).
Wu, Q. & Zhang, X.-C. Free-space electro-optic sampling of terahertz beams. Appl. Phys. Lett. 67, 3523–3525 (1995).
Nahata, A., Weling, A. S. & Heinz, T. F. A wideband coherent terahertz spectroscopy system using optical rectification and electro-optic sampling. Appl. Phys. Lett. 69, 2321–2323 (1996).
Pupeza, I. et al. High-power sub-two-cycle mid-infrared pulses at 100 MHz repetition rate. Nat. Photon. 9, 721–724 (2015).
Gianazza, E., Miller, I., Palazzolo, L., Parravicini, C. & Eberini, I. With or without you—proteomics with or without major plasma/serum proteins. J. Proteomics 140, 62–80 (2016).
Dębska, B. & Guzowska-Świder, B. Fuzzy definition of molecular fragments in chemical structures. J. Chem. Inf. Comput. Sci. 40, 325–329 (2000).
Demtröder, W. Molecular Physics (Wiley, 2005).
Movasaghi, Z., Rehman, S. & ur Rehman, Dr. I. Fourier transform infrared (FTIR) spectroscopy of biological tissues. Appl. Spectrosc. Rev. 43, 134–179 (2008).
Griffiths, P. R. & De Haseth, J. A. Fourier Transform Infrared Spectrometry (Wiley, 2007).
Keilmann, F., Gohle, C. & Holzwarth, R. Time-domain mid-infrared frequency-comb spectrometer. Opt. Lett. 29, 1542–1544 (2004).
Newbury, N. R., Coddington, I. & Swann, W. Sensitivity of coherent dual-comb spectroscopy. Opt. Express 18, 7929–7945 (2010).
Villares, G., Hugi, A., Blaser, S. & Faist, J. Dual-comb spectroscopy based on quantum-cascade-laser frequency combs. Nat. Commun. 5, 5192 (2014).
Schwaighofer, A. et al. Beyond Fourier transform infrared spectroscopy: external cavity quantum cascade laser-based mid-infrared transmission spectroscopy of proteins in the amide I and amide II region. Anal. Chem. 90, 7072–7079 (2018).
Haas, J., Catalán, E. V., Piron, P., Karlsson, M. & Mizaikoff, B. Infrared spectroscopy based on broadly tunable quantum cascade lasers and polycrystalline diamond waveguides. Analyst 143, 5112–5119 (2018).
Ollesch, J. et al. An infrared spectroscopic blood test for non-small cell lung carcinoma and subtyping into pulmonary squamous cell carcinoma or adenocarcinoma. Biomed. Spectrosc. Imaging 5, 129–144 (2016).
Brandstetter, M., Volgger, L., Genner, A., Jungbauer, C. & Lendl, B. Direct determination of glucose, lactate and triglycerides in blood serum by a tunable quantum cascade laser-based mid-IR sensor. Appl. Phys. B 110, 233–239 (2013).
Baker, M. J. et al. Using Fourier transform IR spectroscopy to analyze biological materials. Nat. Protocols 9, 1771–1791 (2014).
Martin, M. C. et al. 3D spectral imaging with synchrotron Fourier transform infrared spectro-microtomography. Nat. Methods 10, 861–864 (2013).
Rohleder, D. et al. Comparison of mid-infrared and Raman spectroscopy in the quantitative analysis of serum. J. Biomed. Opt. 10, 031108 (2005).
Bhargava, R. Infrared spectroscopic imaging: the next generation. Appl. Spectrosc. 66, 1091–1120 (2012).
Quaroni, L., Zlateva, T., Wehbe, K. & Cinque, G. Infrared imaging of small molecules in living cells: from in vitro metabolic analysis to cytopathology. Faraday Discuss. 187, 259–271 (2016).
Bonnier, F. et al. Ultra-filtration of human serum for improved quantitative analysis of low molecular weight biomarkers using ATR-IR spectroscopy. Analyst 142, 1285–1298 (2017).
Haas, J. & Mizaikoff, B. Advances in mid-infrared spectroscopy for chemical analysis. Annu. Rev. Anal. Chem. 9, 45–68 (2016).
Lu, R. et al. High-sensitivity infrared attenuated total reflectance sensors for in situ multicomponent detection of volatile organic compounds in water. Nat. Protocols 11, 377–386 (2016).
Haase, K., Kröger-Lui, N., Pucci, A., Schönhals, A. & Petrich, W. Advancements in quantum cascade laser-based infrared microscopy of aqueous media. Faraday Discuss. 187, 119–134 (2016).
Haase, K., Kröger-Lui, N., Pucci, A., Schönhals, A. & Petrich, W. Real-time mid-infrared imaging of living microorganisms. J. Biophoton. 9, 61–66 (2016).
Gaida, C. et al. Watt-scale super-octave mid-infrared intrapulse difference frequency generation. Light Sci. Appl. 7, 94 (2018).
Seidel, M. et al. Multi-watt, multi-octave, mid-infrared femtosecond source. Science Advances 4, eaaq1526 (2018).
Butler, T. P. et al. Watt-scale 50-MHz source of single-cycle waveform-stable pulses in the molecular fingerprint region. Opt. Lett. 44, 1730–1733 (2019).
Pupeza, I. et al. Field-resolved spectroscopy in the molecular fingerprint region. In Lasers and Electro-Optics Europe & European Quantum Electronics Conf. (CLEO/Europe-EQEC) https://doi.org/10.1109/CLEOE-EQEC.2017.8086859 (IEEE, 2017).
Huber, M. et al. Detection sensitivity of field-resolved spectroscopy in the molecular fingerprint region. In Lasers and Electro-Optics Europe & European Quantum Electronics Conference (CLEO/Europe-EQEC) https://doi.org/10.1109/CLEOE-EQEC.2017.8086921 (IEEE, 2017).
Timmers, H. et al. Molecular fingerprinting with bright, broadband infrared frequency combs. Optica 5, 727–732 (2018).
Muraviev, A. V., Smolski, V. O., Loparo, Z. E. & Vodopyanov, K. L. Massively parallel sensing of trace molecules and their isotopologues with broadband subharmonic mid-infrared frequency combs. Nat. Photon. 12, 209–214 (2018).
Udem, T., Holzwarth, R. & Hänsch, T. W. Optical frequency metrology. Nature 416, 233–237 (2002).
Ye, J. & Cundiff, S. T. Femtosecond Optical Frequency Comb: Principle, Operation, And Applications (Springer, 2005).
Schweinberger, W. et al. Interferometric delay tracking for low-noise Mach-Zehnder-type scanning measurements. Opt. Express 27, 4789–4798 (2019).
Schubert, O. et al. Rapid-scan acousto-optical delay line with 34 kHz scan rate and 15 as precision. Opt. Lett. 38, 2907–2910 (2013).
Birarda, G. et al. IR-Live: fabrication of a low-cost plastic microfluidic device for infrared spectromicroscopy of living cells. Lab Chip 16, 1644–1651 (2016).
Max, J.-J. & Chapados, C. Glucose and fructose hydrates in aqueous solution by IR spectroscopy. J. Phys. Chem. A 111, 2679–2689 (2007).
Tsurumachi, N., Fuji, T., Kawato, S., Hattori, T. & Nakatsuka, H. Interferometric observation of femtosecond free induction decay. Opt. Lett. 19, 1867–1869 (1994).
Gallot, G. & Grischkowsky, D. Electro-optic detection of terahertz radiation. J. Opt. Soc. Am. B 16, 1204–1212 (1999).
Hobbs, P. C. D. Ultrasensitive laser measurements without tears. Appl. Opt. 36, 903–920 (1997).
Foltynowicz, A., Ban, T., Masłowski, P., Adler, F. & Ye, J. Quantum-noise-limited optical frequency comb spectroscopy. Phys. Rev. Lett. 107, 233002 (2011).
Buberl, T. Broadband interferometric subtraction of optical fields. Opt. Express 27, 2432–2443 (2019).
Tomberg, T., Muraviev, A., Ru, Q. & Vodopyanov, K. L. Background-free broadband absorption spectroscopy based on interferometric suppression with a sign-inverted waveform. Optica 6, 147–151 (2019).
Fritsch, K., Poetzlberger, M., Pervak, V., Brons, J. & Pronin, O. All-solid-state multipass spectral broadening to sub-20 fs. Opt. Lett. 43, 4643–4646 (2018).
Schulte, J., Sartorius, T., Weitenberg, J., Vernaleken, A. & Russbueldt, P. Nonlinear pulse compression in a multi-pass cell. Opt. Lett. 41, 4511–4514 (2016).
Huber, M. et al. Active intensity noise suppression for a broadband mid-infrared laser source. Opt. Express 25, 22499–22509 (2017).
Lanin, A. A., Voronin, A. A., Fedotov, A. B. & Zheltikov, A. M. Time-domain spectroscopy in the mid-infrared. Sci. Rep. 4, 1–8 (2014).
Acknowledgements
We thank D. Gerz, A. Zigman Kohlmaier, L. Fuerst and I. Kosse for their contributions and help with the measurements. We acknowledge the support of the Max Planck Society, the Center for Advanced Laser Applications of the Ludwig-Maximilians University and the King Saud University via the Researchers Supporting Project (NSRSP-2019/1).
Author information
Authors and Affiliations
Contributions
I.P., M.H., M.T., W.S., S.A.H., C.H., E.F., A. Apolonski, A. Azzeer, M.Z. and F.K. conceived the experiments. I.P., M.H., M.T., W.S., S.A.H., C.H., K.F., M.P., L.V., T.A, K.V.K., N.K., V.P., O.P., M.Z. and F.K. designed the experiments and analysed the experimental data. M.H., F.F. and M.Z. prepared the living systems. S.A.H., K.F., M.P. and O.P. designed and built the few-cycle near-infrared femtosecond laser source. I.P., M.H., W.S., S.A.H., C.H., L.V., V. P. and N.K. developed the optical system for the generation and electro-optic detection of waveform-stable MIR radiation. All authors contributed to evaluating the results and writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Comparison of FTS with FRS.
a, Schematic of FTS. b, Schematic of FRS. c, Portions of the background signal contributed by the sample response to the FTS (blue, first right-hand-side term of equation (1a)) and to the FRS (red, first right-hand-side term of equation (1b)) signals at a fixed delay (τ = 1,500 fs). For illustration purposes, the nonlinear upconversion efficiency was set to 1 and the ‘carved out’ effective window time length to 50 fs (without loss of generality). Example parameters: 190-fs Gaussian excitation pulse and 1,139-cm−1 DMSO2 absorption (see Extended Data Table 1).
Extended Data Fig. 2 Detection dynamic range and linearity of the instrument response.
a, Infrared excitation pulse (normalized to maximum), recorded with attenuating optical density (OD) filters instead of the cuvette in the beam path, for increasing attenuation and measurement time T. A 1,200-fs scan range and T = 16 s and T = 1,600 s were considered. Small variations of the pulse shape for different attenuations are attributed to slight dispersion variations among the OD filters. The attenuation-independent pulse shape confirms the instrument linearity over the entire parameter range considered. b, c, Spectral intensity (normalized to the maximum of the attenuation-free measurement) and phase of the signals in a, respectively. The detection noise floors in b were obtained by blocking the MIR signal and evaluating the mean of the (white) noise in the considered spectral range, and confirm the linear decrease of the noise floor with T. For the data in c, for all time-domain waveforms a super-Gaussian filter (width 700 fs, order 20) was applied.
Extended Data Fig. 3 Experimental setup.
See subsection ‘High-power femtosecond oscillator and generation of waveform-stable MIR Pulses’ in Supplementary Information section I for details. a, HWP, half-wave plate; QWP, quarter-wave plate; IDFG, intra-pulse difference frequency generation; IDT, interferometric delay tracking; Ge, germanium beam combiner. All steering mirrors for the MIR beam were gold-coated. In the bandwidth-optimized instrument setting, four custom dispersive mirrors were added to the MIR beam path (see text). The pulse was temporally compressed with customized dispersive optics. Pulse compression. EOS traces of the excitation pulse transmitted through water in the bandwidth-optimized instrument setting, with (blue) and without (red) four dispersive mirrors in the MIR beam path. c, As in b but on a logarithmic scale, visualizing the improved roll-off of the signal achieved with the dispersive optics.
Extended Data Fig. 4 Contributions to quantum efficiency in electro-optic sampling.
a, Frequency-resolved measurement of the noise of the balanced detection (black), and calculated shot noise (red). The dashed line indicates the lock-in frequency, and its peak stems from the chopper. b, Comparison of MIR power depletion after EOS crystal for the two different crystal thicknesses. The oscillations originate from interferences of the MIR pulse incident to the EOS crystal and MIR radiation generated therein (these oscillations do not affect the performance of EOS detection).
Extended Data Fig. 5 Measurement of noise contributions for the estimation of the performance of FTS with our femtosecond-laser-based source, our mechanical scan, and state-of-the-art infrared detection.
a, The setup mimics a FTS setup in the Mach–Zehnder configuration, with balanced lock-in detection. For lock-in frequency modulation, a mechanical chopper is placed in the ‘sample arm’. The two arms are recombined with a 50:50 beam splitter. The two outputs are detected with two independent MIR detectors (see text for details). The power impinging on each detector was limited to 450 mW, corresponding to a detector output voltage of 20 V. The relative intensity noise (RIN) spectrum of the source is recorded with an FFT-Analyzer in the range 0.1–100 kHz (before balanced detection). Balanced lock-in detection is performed with a lock-in amplifier with differential input. The beam block was used in the measurements shown in c. b, RIN spectrum of the free-running (red curve) and intensity-stabilized (blue curve) MIR beam (before the interferometer). The integrated RIN of the stabilized source from 1 Hz and 100 kHz is as low as 0.04%. c, Demodulated (after lock-in detection with a time constant of 1.6 ms and 4th-order filter) time-domain trace of detector noise (grey), local-oscillator signal with sample arm blocked (turquoise) and of the combination of both interferometer arms impinging on the balanced detection (blue). The inset shows a 1-second section of the signals, for a detailed comparison of the local-oscillator noise and the detector noise.
Extended Data Fig. 6 Simulations of time-domain decay of a molecular Lorentzian oscillator.
a, Fit of a Lorentzian oscillator to the 1,139 cm−1 absorption of (low-concentration) DMSO2. Black line, intensity transmission through pure, molecular DMSO2, determined by referencing the transmission spectrum of a 1 mg ml−1 solution to that of water, measured via FTIR, and normalizing to a 1-µm path. Green line, least-squares fit (1,080–1,190 cm−1) of a Lorentzian oscillator to the 1,139 cm−1 absorption, yielding a full width at half depth of 13.47 cm−1 and an absorption coefficient α = 11.96 cm−1. The numerical example shows the instantaneous and resonant parts of the electric field as described by equations (1) to (4) in Supplementary Information section II. The initial pulse is a Gaussian pulse with an intensity envelope (full width at half maximum) of 190 fs. The Lorentzian absorption band has a peak of α2z with α2 = 0.0024 cm−1, corresponding to a 200 ng ml−1 solution of DMSO2 in water, and a width δυ = 13.47 cm−1. These values were obtained from fitting a Lorentzian absorber to the 1,139 cm−1 band of the transmission spectrum of a 1 mg ml−1 solution obtained with FTIR and linear extrapolation to a concentration 5,000 times lower. b, Time-domain representation of the normalized envelope functions of the electric fields described (see key). A value of tB = 1.5 ps is chosen. The green vertical bars indicate the boundaries of the band-pass-filtered resonant response shown in green: 1.5 ps and 4 ps. c, Magnitudes of the Fourier transforms of the envelopes shown in a, normalized to C. At the absorption maximum, the discrepancy between the resonant response as in Supplementary Information section 2 and its approximation as in Supplementary Information section 3 is 1%, justifying this convenient approximation. The error introduced by band-pass filtering the resonant response between 1.5 ps and 4 ps compared to the high-pass time-filtered signal is 4%.
Extended Data Fig. 7 Spectral intensity of the Fourier-transformed temporal fingerprints of DMSO2.
Spectral intensity is shown for different concentrations, after high-pass-time-filtering at tB = 1,500 fs and subtraction of pure water reference, normalized to the spectral intensity of the reference pulse. Green dashed lines, modelled Lorentzian oscillator with the parameters derived from the fit in Extended Data Fig. 6. This model agrees excellently with the measured fingerprints, and confirms the minimum detectable absorbance predicted by equation (2) as well as the linear response of the instrument.
Extended Data Fig. 8 Principal component analysis.
a–d, Comparison of the loading vectors for the first principal component for the FTIR data (a) and the FRS data (b) from the serum spiking experiment, with the pre-processed GMF data (see text) of the FTIR (c) and FRS (d) measurements of a 1 mg ml−1 DMSO2 solution. We note that the FRS spectra are complex, so the real and imaginary parts were considered separately (and stitched to single vectors). e, Figure of merit (FOM) (colour scale in arbitrary units; see Supplementary Information section VI) quantifying the separation of classes according to the first principal component (the lower the FOM, the better the separation), evaluated for a large range of the beginning time tB and time window length Δt. The cross indicates parameters yielding optimum separation. f–i, Comparison of the loading vectors for the first principal component for the FTIR data (f) and the FRS data (g) from the sugar mixture experiment, with the pre-processed GMF data of the FTIR (h) and FRS (i). For the latter, the difference of the spectra of the 50/50 mixture and the pure maltose solution is shown. The real and imaginary parts were considered separately. j, FOM quantifying the separation of classes according to the first principal component, in analogy to e.
Extended Data Fig. 9 Absorption spectra of 10 mg ml−1 aqueous solutions of maltose and melibiose, measured by FRS and FTIR.
The difference in total absorption is due to the differing cuvette thickness. a, FRS; b, FTIR. OD, optical density.
Supplementary information
Supplementary Information
This file contains Supplementary Information sections 1–7 and Supplementary References.
Source data
Rights and permissions
About this article
Cite this article
Pupeza, I., Huber, M., Trubetskov, M. et al. Field-resolved infrared spectroscopy of biological systems. Nature 577, 52–59 (2020). https://doi.org/10.1038/s41586-019-1850-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1850-7
This article is cited by
-
Maximizing the efficiency of intrapulse difference frequency generation by pulse shaping and recycling
Applied Physics B (2024)
-
Mid-infrared cross-comb spectroscopy
Nature Communications (2023)
-
Few-femtosecond phase-sensitive detection of infrared electric fields with a third-order nonlinearity
Communications Physics (2023)
-
Upconversion time-stretch infrared spectroscopy
Light: Science & Applications (2023)
-
Effect of Bi2O3 on the Structural, Mechanical, and Radiation Gamma Shielding Properties of the Boro-Silica-Phosphate Glasses
Silicon (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.