Abstract
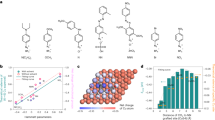
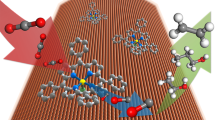
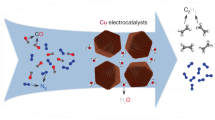
The electrocatalytic reduction of carbon dioxide, powered by renewable electricity, to produce valuable fuels and feedstocks provides a sustainable and carbon-neutral approach to the storage of energy produced by intermittent renewable sources1. However, the highly selective generation of economically desirable products such as ethylene from the carbon dioxide reduction reaction (CO2RR) remains a challenge2. Tuning the stabilities of intermediates to favour a desired reaction pathway can improve selectivity3,4,5, and this has recently been explored for the reaction on copper by controlling morphology6, grain boundaries7, facets8, oxidation state9 and dopants10. Unfortunately, the Faradaic efficiency for ethylene is still low in neutral media (60 per cent at a partial current density of 7 milliamperes per square centimetre in the best catalyst reported so far9), resulting in a low energy efficiency. Here we present a molecular tuning strategy—the functionalization of the surface of electrocatalysts with organic molecules—that stabilizes intermediates for more selective CO2RR to ethylene. Using electrochemical, operando/in situ spectroscopic and computational studies, we investigate the influence of a library of molecules, derived by electro-dimerization of arylpyridiniums11, adsorbed on copper. We find that the adhered molecules improve the stabilization of an ‘atop-bound’ CO intermediate (that is, an intermediate bound to a single copper atom), thereby favouring further reduction to ethylene. As a result of this strategy, we report the CO2RR to ethylene with a Faradaic efficiency of 72 per cent at a partial current density of 230 milliamperes per square centimetre in a liquid-electrolyte flow cell in a neutral medium. We report stable ethylene electrosynthesis for 190 hours in a system based on a membrane-electrode assembly that provides a full-cell energy efficiency of 20 per cent. We anticipate that this may be generalized to enable molecular strategies to complement heterogeneous catalysts by stabilizing intermediates through local molecular tuning.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: insights into materials design. Science 355, eaad4998 (2017).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Li, Y. & Sun, Q. Recent advances in breaking scaling relations for effective electrochemical conversion of CO2. Adv. Energy Mater. 6, 1600463 (2016).
Calle-Vallejo, F. & Koper, M. T. Theoretical considerations on the electroreduction of CO to C2 species on Cu(100) electrodes. Angew. Chem. Int. Ed. 52, 7282–7285 (2013).
Montoya, J. H., Shi, C., Chan, K. & Nørskov, J. K. Theoretical insights into a CO dimerization mechanism in CO2 electroreduction. J. Phys. Chem. Lett. 6, 2032–2037 (2015).
Yang, K. D. et al. Morphology-directed selective production of ethylene or ethane from CO2 on a Cu mesopore electrode. Angew. Chem. Int. Ed. 56, 796–800 (2017).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
Jiang, K. et al. Metal ion cycling of Cu foil for selective C–C coupling in electrochemical CO2 reduction. Nat. Catal. 1, 111–119 (2018).
Mistry, H. et al. Highly selective plasma-activated copper catalysts for carbon dioxide reduction to ethylene. Nat. Commun. 7, 12123 (2016).
Zhou, Y. et al. Dopant-induced electron localization drives CO2 reduction to C2 hydrocarbons. Nat. Chem. 10, 974–980 (2018).
Han, Z., Kortlever, R., Chen, H. Y., Peters, J. C. & Agapie, T. CO2 reduction selective for C≥2 products on polycrystalline copper with N-substituted pyridinium additives. ACS Cent. Sci. 3, 853–859 (2017).
Rosen, B. A. et al. Ionic liquid-mediated selective conversion of CO2 to CO at low overpotentials. Science 334, 643–644 (2011).
Masel, R. I. & Rosen, B. A. Catalyst mixtures. US patent 8,956,990 (2015).
Masel, R. I. & Rosen, B. A. Electrochemical devices comprising novel catalyst mixtures. US patent 9,464,359 (2016).
Masel, R. I. & Rosen, B. A. Catalyst mixtures. US patent 9,566,574 (2017).
Barton Cole, E. et al. Using a one-electron shuttle for the multielectron reduction of CO2 to methanol: kinetic, mechanistic, and structural insights. J. Am. Chem. Soc. 132, 11539–11551 (2010).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Li, J. et al. Efficient electrocatalytic CO2 reduction on a three-phase interface. Nat. Catal. 1, 592–600 (2018).
Ma, S. et al. One-step electrosynthesis of ethylene and ethanol from CO2 in an alkaline electrolyzer. J. Power Sources 301, 219–228 (2016).
Jouny, M., Luc, W. W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Sheppard, N. & Nguyen, T. T. in Advances in Infrared and Raman Spectroscopy Vol. 5 (eds Hawes Clark, R. J. & Hester, R. E.) 67 (Heyden, 1978).
Gunathunge, C. M. et al. Spectroscopic observation of reversible surface reconstruction of copper electrodes under CO2 reduction. J. Phys. Chem. C 121, 12337–12344 (2017).
Heyes, J., Dunwell, M. & Xu, B. CO2 reduction on Cu at low overpotentials with surface-enhanced in situ spectroscopy. J. Phys. Chem. C 120, 17334–17341 (2016).
Akemann, W. & Otto, A. Vibrational modes of CO adsorbed on disordered copper films. J. Raman Spectrosc. 22, 797–803 (1991).
Xiao, H., Goddard, W. A., Cheng, T. & Liu, Y. Cu metal embedded in oxidized matrix catalyst to promote CO2 activation and CO dimerization for electrochemical reduction of CO2. Proc. Natl Acad. Sci. USA 114, 6685–6688 (2017).
Cole, E. B., Sivasankar, N., Parajuli, R. & Keets, K. A. Reducing carbon dioxide to products. US patent 8,845,878 (2014).
Acknowledgements
This work was financially supported by the Ontario Research Fund: Research Excellence Program, the Natural Sciences and Engineering Research Council (NSERC) of Canada, the CIFAR Bio-Inspired Solar Energy program and the Joint Centre of Artificial Synthesis, a DOE Energy Innovation Hub, supported through the Office of Science of the US Department of Energy under award no. DE-SC0004993. All DFT computations were performed on the IBM BlueGene/Q supercomputer with support from the Southern Ontario Smart Computing Innovation Platform (SOSCIP). SOSCIP is funded by the Federal Economic Development Agency of Southern Ontario, the Province of Ontario, IBM Canada Ltd, Ontario Centres of Excellence, Mitacs and 15 Ontario academic member institutions. This research was enabled in part by support provided by Compute Ontario (www.computeontario.ca) and Compute Canada (www.computecanada.ca). This research used synchrotron resources of the Advanced Photon Source (APS), an Office of Science User Facility operated for the US Department of Energy (DOE) Office of Science by Argonne National Laboratory, and was supported by the US DOE under contract no. DE-AC02-06CH11357, and the Canadian Light Source and its funding partners. We thank T. Wu and L. Ma for technical support at 9BM beamline of APS. D.S. acknowledges the NSERC E.W.R. Steacie Memorial Fellowship. A.T. acknowledges Marie Skłodowska-Curie Fellowship H2020-MSCA-IF-2017 (793471). J.L. acknowledges the Banting postdoctoral fellowship. C.M.G. acknowledges NSERC for funding in the form of a postdoctoral fellowship from the government of Canada. J.P.E. thanks NSERC, Hatch and the Government of Ontario for their support through graduate scholarships.
Author information
Authors and Affiliations
Contributions
E.H.S., T.A. and J.C.P. supervised this project. F.L. and Y.L. carried out electrochemical experiments. A.T. and A.R.-H. carried out molecule synthesis and characterizations. Z.W. carried out DFT calculations. C.M.G. and F.L. conducted in situ Raman measurement. F.L. and A.O. carried out the membrane–electrode-assembly experiments. J.L. and F.L. performed XAS measurements. Y.W. carried out scanning electron microscopy and electrochemical impedance spectroscopy measurements. J.P.E. measured the contact angle. C.M. carried out the Comsol modelling. L.T. carried out EPR measurement under the supervision of R.D.B. M.L. performed part of the electrochemical experiments. Z.-Q.L., X.W. and H.L. provided help in NMR analysis. C.M.G., C.P.O. and Y.X. provided help in membrane-electrode assembly measurements. C.-S.T. carried out AFM measurement. D.-H.N. conducted X-ray diffraction measurement. R.Q.-B. carried out X-ray photoelectron spectroscopy measurement. C.T.D., T.T.Z., Y.C.L. and Z.H. provided help in materials synthesis and characterizations. F.L. and E.H.S. wrote the manuscript. All authors discussed the results and assisted during manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Muhyun Baik, Richard I. Masel and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file includes Methods, Supplementary Figures 1–34, Supplementary Notes 1–4 and Supplementary Tables 1–5
Rights and permissions
About this article
Cite this article
Li, F., Thevenon, A., Rosas-Hernández, A. et al. Molecular tuning of CO2-to-ethylene conversion. Nature 577, 509–513 (2020). https://doi.org/10.1038/s41586-019-1782-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1782-2
This article is cited by
-
Selective and energy-efficient electrosynthesis of ethylene from CO2 by tuning the valence of Cu catalysts through aryl diazonium functionalization
Nature Energy (2024)
-
Impact of palladium/palladium hydride conversion on electrochemical CO2 reduction via in-situ transmission electron microscopy and diffraction
Nature Communications (2024)
-
Analysis of metal–organic framework-based photosynthetic CO2 reduction
Nature Synthesis (2024)
-
Site-selective protonation enables efficient carbon monoxide electroreduction to acetate
Nature Communications (2024)
-
Acidic media enables oxygen-tolerant electrosynthesis of multicarbon products from simulated flue gas
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.