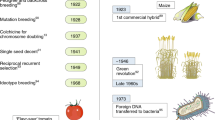
Abstract
The current trajectory for crop yields is insufficient to nourish the world’s population by 20501. Greater and more consistent crop production must be achieved against a backdrop of climatic stress that limits yields, owing to shifts in pests and pathogens, precipitation, heat-waves and other weather extremes. Here we consider the potential of plant sciences to address post-Green Revolution challenges in agriculture and explore emerging strategies for enhancing sustainable crop production and resilience in a changing climate. Accelerated crop improvement must leverage naturally evolved traits and transformative engineering driven by mechanistic understanding, to yield the resilient production systems that are needed to ensure future harvests.
Similar content being viewed by others
Main
The Green Revolution of the 1960s enabled a steep increase in the yields of major staple grain crops (wheat, corn and rice) to address the caloric needs of an increasing global population. This was accomplished through elite variety breeding, hybrid crop development, fertilizer application and advances in management through substantial public investment2. The consequent rise in food security benefitted many regions of the world and improved agricultural development (particularly in India and southeast Asia), reducing poverty and malnourishment.
By the 1980s, molecular and transformation technologies propelled the delivery of the first bioengineered genes into plant genomes. Currently, the most widely adopted genetically modified traits are resistance to herbicides and insects in crops with large markets (maize, soybean, cotton and Brassica napus (canola)). Although herbicide- and insect-resistance traits greatly lessened soil tillage and insecticide use, respectively, they require careful management to avoid natural selection of resistance in weeds or pests3,4. Despite engineered traits with clear benefits to farmers and end-users (including virus-resistant papaya5, drought-tolerant corn6, rice7 and bananas8 fortified with provitamin A, non-browning apples9 and low-acrylamide potatoes10), the acceptance of genetically modified traits is equivocal in some countries, and the cultivation of genetically modified crops is largely banned in the European Union.
Future food security will require reducing crop losses due to environmental factors, including climate change, as well as transformative advances that provide major gains in yields. More recent genomic technologies have expedited breeding and trait development for increased environmental resilience and productivity. Genetic diversity is now readily explored at nucleotide-scale precision, using genome-wide association studies and other gene-mapping methods paired with advanced phenotyping systems. The identification of loci that contribute to traits, coupled with molecular-marker-assisted breeding, enables the rapid selection of new genetic combinations in elite varieties. Complementary to breeding approaches, advances in the spatial and temporal regulation of engineered genes and pathways are increasingly accelerated by the targeted editing of genomes using CRISPR–Cas technology. A greater understanding of plant mechanisms that increase yields in variable environments is essential to drive the necessary gains in crop improvement, which can be fuelled by genetic diversity and implemented by genome-scale breeding, finely-tuned gene engineering and more-precise agronomic management practices.
Post-Green Revolution challenges
Despite the marked effect of the Green Revolution on food security, there were uneven consequences for human nutrition, the resilience of crops to stress, and the environment2. Asian populations benefitted from the increased production of staple grains, and the adoption of irrigation across vast areas2,11. The limited rise in food security in sub-Saharan Africa and other impoverished areas can be traced to geographically skewed support and a lack of investment in orphan crops2. An unintended consequence has been that fruits and vegetables rich in macronutrients have been displaced by calorie-rich and higher-value grain crops in some areas2. Moving forward, an increased production of nutrient-rich vegetable, pulse, tuber and cereal crops, and a broadening of the global reach of agricultural advances, is necessary to achieve food and nutritional security12.
Climatic stress and disease management
The increasing frequency of debilitating heat-waves, droughts, torrential rains and other weather extremes experienced across the globe negatively affects agricultural productivity, and is projected to do so1,13 (Fig. 1). Climatic constraints can occur independently or together (as with heat and aridity), and in either case reduce the level of productivity that is predicted for a well-managed environment (the yield potential). An unanticipated consequence of the development of high-yield varieties for locations with advanced cultivation practices has been a loss of genetic variation that is associated with resilience to suboptimal environments. It is imperative to breed crops that carry a diversity of resistance genes and/or to plant a diversity of varieties, as this approach minimizes the ability of pathogens to overcome resistance14. An increasing occurrence of extreme weather events, together with dire projections of climate change, makes the improvement of crop resilience to environmental (abiotic) and pathogen (biotic) stress of paramount importance for feeding a growing global population.
a–c, Maps indicate the yield losses caused by aridity stress averaged from 1950–2000 (a), heat stress averaged from 1994–2010 (b) and nutrient stress in 2009 (c). National data for each crop were previously compiled13, and are here averaged and re-plotted using the maps package in R153. d, Number of large flood events from 1985 to 2010154 by country.
Fertilizer use
The combination of high-yield crop varieties and the widespread use of inorganic fertilizers markedly improved crop production, with clear benefits in terms of food security15. This has translated to excessive anthropogenic release of reactive nitrogen16 and phosphate17 into the environment. Inorganic fertilizers have pushed the global nitrogen and phosphorus cycles well beyond their estimated safe operating space18, with considerable negative effects on biodiversity, human health and the atmosphere19. Their use presents a paradox, as the optimization of plant nutrition stabilizes yields and has helped to reduce expansions in crop area in light of population growth, yet nitrogenous fertilizers contribute substantially to the greenhouse gas emissions that promote climate change19.
Paths forward
The agriculture of the next decades must satisfy demands for nutritious food, fibre and animal feed in a highly variable climate, and also mitigate the effects of agriculture on the environment. This is a tall order. Key to addressing the challenge is a deeper understanding of genetic variation and the molecular, cellular and developmental pathways by which plants dynamically respond to and interact with their environment and pathogens, while maintaining growth, efficiency of nutrient use and fitness. New crop varieties ideally will have genetic combinations that alleviate losses from the multiple environmental and pest constraints that are encountered during the crop lifecycle in a farmer’s field. An important emerging and non-trivial goal is to optimize the efficiency of photosynthesis, water and nutrient use, including the fostering of beneficial interactions between plants and microorganisms that can promote nutrient acquisition. The integration of mechanistic understanding, genetic variation and genome-scale breeding towards technological solutions will be essential. Here we review advances and emerging directions within the plant sciences that may bolster yield-defining traits and resilience (Fig. 2, Box 1).
Overview of traits that provide increased resilience and yield in variable environments. a, Pathogen recognition by cell-surface and intracellular receptors (resistance proteins). Manipulation of host cells by pathogen-secreted effectors to promote infection can be recognized by resistance proteins and converted to disease resistance. b, Flooding survival via opposing regulation of gibberellin (GA). Semidwarf 1 (SD1), Snorkel 1 and Snorkel 2 (SK1/2) confer escape by accelerated elongation growth. Submergence 1A (SUB1A) confers tolerance by quiescence of growth. c, Root growth towards moisture involves transcriptional regulators (indol-3-acetic acid inhibitor protein 3 (IAA3) and auxin response factor (ARF7)), and is regulated by the hormones ABA and auxin. d, HKT1 (high-affinity K+ transporter sub-family 1) mediates sodium (Na+) exclusion from leaves. e, In developing seed tissues, catabolism of T6P aids the movement of photo-assimilate carbohydrate (CHO) from leaves to sinks in developing florets. f, Optimizing photosynthetic light harvesting and CO2 fixation by altering photosynthetic protein abundance and minimizing photorespiration. PS, photosystem. g, Dynamic control of stomatal aperture by pairs of epidermal guard cells lessens desiccation. h, Symbiotic plant–microorganism interactions facilitate the uptake of essential nutrients. NH4+, ammonium; PO4−, phosphate; NO3−, nitrate.
Protection from new and re-emerging diseases
The reliance on major pathogen-resistance genes bred into crop monocultures provides short-term protection against diseases, as seen in the boom-and-bust cycles of resistance over the past century and in the spread of new diseases across continents. A more complete molecular-genetic framework now exists for general and specific resistance to microbial pathogens mediated by a two-layered receptor signalling system (Fig. 2a). At the plant cell surface, families of pattern-recognition receptors register the presence of microorganisms20. Inside host cells, large panels of nucleotide-binding domain leucine-rich-repeat receptors (NLRs) detect activities of invasive pathogenic strains21. Advances in elucidating receptor–pathogen recognition and activation mechanisms at a protein-structural level provide strategies towards the rational design of receptor proteins that are tailored to intercept broader or alternative disease agents21,22. Newly engineered resistance traits can then be transferred to elite varieties of crops to confer resistance against modern diseases. There have been notable successes in the inter-family transgenic transfer of a pattern-recognition-receptor gene to potato, tomato, Medicago, wheat and rice20,23, indicating that surface receptors that are restricted to particular plant lineages can confer immunity in unrelated species. Transfer of the wheat Pm3e resistance gene against powdery mildew to a susceptible wheat variety has produced effective mildew resistance in field trials24. The engineering of pathogen-induced translational control of a key Arabidopsis immunity component in rice25 has provided promising disease-resistance benefits in initial crop field trials, apparently without a yield penalty. The incorporation of new surface- and intracellular-receptor recognition and signal transduction modules into crops is also on the horizon, building on knowledge of receptor functional partnerships and resistance network architectures21,26. Success in this area—especially as climates change—will require tight immune-receptor control, which can require co-evolved factors27,28, because modified receptors without requisite constraints are often mis-activated and cause necrosis and reduced plant health. Knowledge in this arena may also lead to strategies that lessen food spoilage by pathogens after harvesting.
Harnessing resistance from diverse germplasm
With increased access to diverse genetic variation found in crops and natural populations of wild relatives14, the door is open to recovering disease-resistance traits; many of these are encoded by genes for pattern recognition and NLRs that were lost during domestication, or that have evolved independently in different plant lineages14,29. Advances in genome sequencing and assembly technologies, coupled with new methods for capturing near-complete immune-receptor gene panels from complex genomes, hold promise for attaining sustainable disease resistance30,31,32. Merging these approaches with genome-wide association studies taps into immune-receptor gene variants that have adapted to local environments and pathogen populations to help to increase the resilience of future crops. The stacking (or ‘pyramiding’) of several resistance genes with different recognition spectra and environmental optima into a single background is now a credible strategy for achieving more durable disease resistance. Nevertheless, assembling appropriate gene combinations in elite varieties of crops remains a challenge.
Investigations of plant genomes within and across species provide insight into the evolutionary forces that have shaped the architecture and function of genes related to immunity. This will aid the design of new resistance traits33. The isolation and characterization of genes associated with disease susceptibility in the host has also gained prominence34. The proactive deployment of modified susceptibility genes in crops will become possible as geographical sampling of pathogen genomes and populations increases35,36. A recessive barley mildew resistance locus o (mlo) mutant that breeders have successfully used for 75 years against powdery mildew disease has been engineered into hexaploid wheat using mutagenesis by transcription-activator-like effector nuclease, or by combining mutations selected in three wheat MLO loci37,38. Thus, the characterization and manipulation of host–pathogen infection processes for the establishment of disease can generate novel resistance mechanisms in crops that are not necessarily found in natural populations. As a cautionary note, some newly engineered crop lines have displayed unexpected phenotypes and vulnerabilities to disease39, which underscores the need for rigorous performance testing of new material in field settings over multiple seasons.
Pathogen resistance in a shifting climate
Given the current trends in climate, attaining durable resistance in high-yield crops will require a greater knowledge of pathogen population dynamics and plant host responses to temperature. The alarming spread of devastating disease agents—such as the bacterium Xylella fastidiosa that attacks olives and woody crops in southern Europe, or the ug99 strain of stem rust fungus Puccinia graminis f. sp. tritici that affects wheat across parts of Africa and Asia—is attributed in part to warmer climates, and presents a complex biogeographical and epidemiological problem40,41. Moreover, surface- and intracellular-immunity systems appear to respond differently to changes in temperature, owing to effects on the microorganisms and hosts that are not completely understood42,43. Although immunity mediated by intracellular receptors tends to be less efficient as temperatures increase, some resistance genes confer protection at higher temperatures44,45. Although these findings are reassuring, they highlight the need for more genotype × environment studies in crops to stabilize resistance in increasingly precarious climates.
Resilience to abiotic stress
Abiotic stresses associated with climate change that destabilize yields include flooding, drought, soil salinity and extreme temperatures (Fig. 1). Resilience mechanisms have been mobilized for crop improvement through the identification of genes that are associated with key traits and signal transduction pathways, followed by breeding or engineering46,47 (Fig. 2b–f). Attaining resilience without affecting overall yield is a considerable challenge.
Flooding
Floods regularly limit yields48. Rice is exceptionally resilient against flooding, yet over 30% of the acreage cultivated with rice suffers yield loss owing to plant submergence49. SUBMERGENCE 1 (SUB1), identified in a flooding-tolerant landrace of rice, encodes a cluster of genes for ethylene-responsive transcription factors, including the submergence-activated SUB1A-150. The SUB1A transcription factor curbs the activation of genes that promote the breakdown of leaf sugars and starch (photo-assimilate) that would otherwise fuel growth to escape an inundation51 (Fig. 2b). The introduction of SUB1A-1, through marker-assisted breeding, into high-yield varieties now provides an additional week or more of submergence tolerance—without compromising yields under non-submergence conditions52.
The submergence tolerance by growth quiescence provided by SUB1A-1 contrasts with the accelerated underwater elongation of the shoots of varieties of crops that are adapted to progressive seasonal floods in delta regions. Deepwater varieties invest photo-assimilate into the extension of submerged stem internodes (Fig. 2b). This requires the SNORKEL 1 (SK1) and SNORKEL 2 (SK2) genes that encode transcription factors that are similar to SUB1A53, as well as biosynthesis of the hormone gibberellin. Gibberellin biosynthesis involves a functional allele of SEMIDWARF1 (SD1)54, the gene that—when mutated—determines the short stature of Green Revolution rice55. This knowledge can improve yields in low-lying areas that affected by climate change.
The alleles of SUB1A, SK1, SK2 and SD1 that are key to flooding survival are found in wild Oryza species56, which indicates that they arose in ancestral populations in flooded ecosystems. Evolution has modified the same growth-response network involving the hormones ethylene and gibberellin to achieve submergence tolerance or escape in diverse species of wetland plants48. Pathways to improved flooding tolerance include manipulation of root traits associated with waterlogging tolerance that involve a conserved mechanism48 and the oxygen-dependent turnover of SUB1A-1-like transcription factors, accomplished in several species57,58,59,60. There are other opportunities to protect yields in wet climates. Torrential rain and hail can cause yield losses of 50% or more, owing to premature pod shattering in oil crops. The identification of genes that control pod shattering in Arabidopsis61 enabled the gene-targeted molecular breeding of optimized pod-shattering properties in canola that is now increasingly planted by farmers.
Drought
Drought and other dehydration or osmotic stresses (salinity and cold) stimulate the production of the hormone abscisic acid (ABA) in plants. Although the mechanisms of the initial sensing of osmotic stress and signalling in response to osmotic stress remain poorly understood, the elucidation of the ABA receptor and signal transduction mechanisms62,63 has exposed new avenues for the enhancement of dehydration tolerance. This includes ABA receptor overexpression64,65 and engineering to respond to exogenously sprayed small molecules66,67, the overexpression of signal transduction components68,69,70 or the drought-driven repression of negative regulators of ABA signal transduction69,71.
ABA closes the adjustable stomatal pores on the leaf surface that allow gas exchange and thus reduces the water lost from plants during drought, but this response can be weak in crop varieties69,72. ABA also helps to regulate root growth in response to water availability, including inhibition of lateral root growth and enhancement of primary and secondary root growth. This developmental reprogramming allows roots to seek water. The DEEPER ROOTING (DRO1) gene of rice provides a deep root architecture in paddy fields, which bolsters yields under water-limited conditions73,74,75 (Fig. 2c). The identification of the major loci that control root traits associated with drought resilience has proven challenging owing to their quantitative nature and low heritability, which requires sophisticated belowground phenotyping and analytical methods76. Yet roots grow laterally towards moisture in soil77. New roots that access moisture emerge only on the side of a root that is moist, as a modification of a key auxin-response transcription factor on the dry side of a root impedes the developmental program78 (Fig. 2c). Knowledge such as this can inform strategies for the advanced breeding and engineering of improved resilience to drought, which continues to limit yields79,80.
Salinity
Irrigation substantially expands growing seasons and increases crop yields in many regions. Salt (sodium chloride) gradually accumulates in irrigated soils and is toxic to most crops; sodium accumulation is particularly detrimental in leaves. Approximately 40% of irrigated lands worldwide are affected by increased salt levels, and expansion of soil salinization is a major threat to crop performance47.
Plants encode a sodium-transporter gene sub-family named HKT181 (high-affinity K+ transporter) that provides protection from the over-accumulation of sodium in leaves82 (Fig. 2d). HKT1 mediates the removal of sodium, mainly in roots, from the xylem83,84, the vascular conduits that transport water and nutrients from roots to leaves. Major quantitative trait loci that enhance salt tolerance in wheat, wheat relatives and rice possess distinct HKT1 alleles47,83,85. This knowledge has enabled the marker-assisted breeding of wheat with a higher salt tolerance, resulting in a yield improvement of 25% under salinity stress in field trials85. Beneficial alleles of HKT1 may enhance salinity tolerance in other species, as has been shown in rice83. It will be necessary to combine HKTs with other strategies to further boost salinity resistance as land salinization continues to rise. The effects of salinity on root development also need to be factored into intervention strategies86,87. Natural variation in transporter genes and their regulation has also provided field-tested solutions for other toxic elements, including aluminium88,89 and boron90.
Extreme temperatures
Higher atmospheric levels of CO2 and other greenhouse gases are predicted to increase the frequency and duration of heat-waves91, which will lead to losses in crop yield—especially in arid regions92. Sensitivity to extreme temperatures varies during the plant lifecycle and across species. Low temperatures influence the germination, establishment, growth and viability of crops, except for those with temporal chilling or freezing resilience (such as winter wheat). Genetic variation in key transcriptional regulators of cold resilience is leveraged in breeding of several grain crops46. By contrast, warm temperatures promote growth until a threshold is reached above which yields precipitously diminish, especially when soil moisture is low or humidity is high93,94. Sensitivity to temperature extremes is heightened during reproduction, when it reduces male fertility and seed quality95. This presents a daunting challenge as protective responses are typically accompanied by reduced yield. Heat stress is an expanding threat in tropical regions, because, at high humidity, plants are less able to cool their leaves by transpiration via stomatal pores that control the trade-off between CO2 intake and water loss96. There is an urgent need for research and for ensuing genetic and engineered solutions that preserve crop productivity at increased temperatures (Box 1).
Metabolic control of resilience and yield
Breeding or engineering plants for a high yield potential in varied and variable environments is a potential solution for capturing effective resilience. Plants typically dampen growth and accelerate reproductive development as a consequence of stress. Yield maintenance under moderate drought was significantly improved in AQUAmax corn hybrids produced by selective and marker-assisted breeding97. The underlying genetic variants and mechanisms that enable these lines to conserve soil moisture and delay the accumulation of biomass until grain filling remain to be characterized. Higher yields under well-watered conditions as well as under moderate drought at the time of flowering was achieved in corn that expresses a metabolic enzyme that converts the low-abundance metabolite trehalose-6-phosphate (T6P) to trehalose in the phloem companion cells at the base of the ear and developing florets98 (Fig. 2e). This cell-specific modulation of T6P augments the mobilization of photosynthate to the unfertilized floret, and prolongs the photosynthetic activity of leaves during grain filling. The spatial modulation of T6P levels also regulates the draw of seed reserves into young elongating shoots of rice, particularly when dry seeds are sown directly into a flooded paddy99. The application of plant-permeable T6P analogues to wheat leaves increased seed filling and improved recovery from drought100. These examples illustrate the critical integration of metabolism and stress resilience to improve crops that can be provided by genetic variation and engineering.
Optimization of photosynthesis for yield
Modern crops are highly efficient at rapidly spreading their leaf canopies to maximize light interception, and at partitioning carbon and nutrients into seeds. However, crops are not as efficient at converting absorbed light energy into sugars through the process of photosynthesis101. This may be because the proteins and enzymes that mediate photosynthesis evolved in a low-light marine environment, which was very different from modern agronomic and atmospheric conditions102. However, the conservation of chloroplast transmembrane proteins that collect light energy and participate in electron transfer reactions within the chloroplast, along with the conservation of enzymes involved in carbon fixation, reduction and regeneration across plant species103 (Fig. 2f), has aided the modelling of photosynthesis104 and identification of numerous targets for increasing its efficiency101 (Fig. 3). Theoretical targets include expanding and optimizing light capture by the leaf canopy105,106,107, inducing a more rapid relaxation of non-photochemical quenching at photosystem II108, increasing the carboxylation capacity of the Rubisco enzyme as well as minimizing oxygenation and photorespiration109, enhancing the regenerative capacity of the carbon reduction cycle101, optimizing the electron transport chain110, converting crops from C3 to C4 metabolism111, and adding components of cyanobacterial or algal systems to pump CO2 or compartmentalize Rubisco101. Improving photosynthetic efficiency is neither a new nor a universally accepted idea. Some have argued that the selection pressures endured by photosynthesis render it unamenable to improvement112. Despite decades of research, the challenge of engineering Rubisco for improved specificity and carboxylation rate remains unmet113. However, some recent successes in engineering photosynthetic enzymes and introducing novel pathways into chloroplasts may lead to substantial gains in crop performance, as outlined below.
Transgenic manipulations of photosynthetic metabolism that lead to improved photosynthetic efficiency include (1) improving photosynthesis in a dynamic light environment by accelerating recovery from a photoprotected state, by overexpressing enzymes (such as photosystem II subunit S (PSBS) and VDE) that are involved in non-photochemical quenching (NPQ) (the dissipation of excess excitation energy as heat)116; (2) altering the CO2 response of stomata or the density of stomata on the leaf surface to increase the efficiency of water use120,122,123; (3) increasing the capacity for mesophyll conductance of CO2105; (4) improving the energy efficiency of carbon metabolism by altering mitochondrial enzymes155; (5) optimizing investment in light collection105; (6) increasing electron flow through the photosynthetic electron transport chain110; (7) altering Rubisco properties and activation to increase CO2 assimilation113,156; (8) bypassing photorespiration117; and (9) increasing the efficiency of ribulose 1,5-bisphosphate (RuBP) regeneration115.
Maize photosynthesis and fresh weight were enhanced by overexpressing the small and large subunits of Rubisco, together with an assembly chaperone protein114. In wheat, the overexpression of sedoheptulose-1,7-biphosphatase showed increased photosynthesis, and resulted in increased plant and grain biomass115. These genetic modifications to key crops are promising; their ultimate potential can be tested by incorporating the changes into elite varieties, and evaluation in the field. Photosynthetic manipulations also show promise in the field in the model plant tobacco. Re-engineering the expression of enzymes that control the induction, relaxation and amplitude of non-photochemical quenching successfully enhanced photosynthesis during natural light transitions, which resulted in 14–20% greater vegetative biomass in the field116. Even greater gains were observed by inserting enzymes involved in glycolate metabolism into chloroplasts to reduce photorespiration117. Coupling this with reduced expression of a glycolate and glycerate transporter, to minimize glycolate flux out of the chloroplast, raised vegetative biomass by 40% under field conditions117. These studies represent fundamental breakthroughs in understanding and engineering photosynthesis, which can now move from the proof-of-concept stage to expanded field testing.
Rising atmospheric CO2 and plant water loss
Crops lose between 100 and >400 water molecules through stomatal pores in leaves for every carbon atom that is fixed by photosynthesis, highlighting a fundamental trade-off between carbohydrate production and water use. Increases in CO2 concentrations inside leaves cause a reduction in the size of stomatal-pore apertures118. The continuing rise in atmospheric CO2 is increasingly narrowing stomatal pores, which can enhance the efficiency of water use by crops. However, many crops have weak or non-optimal stomatal CO2 responses. Advances have been made in understanding the signal transduction pathways that regulate water loss in response to CO2118, including that the stomatal CO2 response requires amplification by—but also includes unique components upstream of and parallel to—the ABA response pathway in guard cells119 (Fig. 2g). The upregulation of the stomatal CO2 response by guard-cell-targeted overexpression of carbonic anhydrases increased instantaneous water-use efficiency by about 44% in Arabidopsis, without a reduction in photosynthetic assimilation rates at ambient CO2120. On the other hand, C3 crops growing in nutrient-rich and water-sufficient humid regions could benefit from a weaker CO2-induced stomatal closing response, which may enhance growth owing to CO2 ‘fertilization’ in an atmosphere with an increased concentration ofCO2121. A complete understanding of the CO2 response pathway is needed to optimize and test water-use efficiency and gas-exchange strategies in the field.
Successful transgenic modifications in barley and rice have shown that reducing the density of stomata improves plant performance under water-restricted conditions122,123. Overexpression of the chloroplast photosystem II subunit S protein in tobacco was reported to lower stomatal conductance, and increased the efficiency of water use by field-grown plants124. The effective manipulation of stomatal function will require the discovery of the primary CO2 and/or bicarbonate sensors that control the stomatal CO2 response, as well as harnessing natural genetic variation in stomatal properties that could improve trade-offs between carbon gain and water loss in a world with high levels of atmospheric CO2125.
Technologies to reduce fertilizer use
Yields of crops are heavily dependent on sufficient nutrition (in particular, nitrogen and phosphorus) that is currently provided primarily through the application of inorganic fertilizers. In smallholder farming systems, crop productivity is limited by the availability of these nutrients15. Substantial advances have been made in understanding the mechanisms of nutrient uptake, transport and use in plants, with the aim of improving sufficiency126 (Fig. 2h). Balancing the use of photo-assimilate with nutrient uptake is critical for optimizing yields. The mutations that confer stem shortening in cereals, which facilitated the Green Revolution, brought with them unintended inefficiencies in nitrogen use that can be compensated for by changing the balance of transcription factors that control growth and nutrient use127. Breeding can also contribute to reducing nutrient imbalances through the optimization of rooting systems, nutrient transport activity and partitioning128.
In natural ecosystems, plants frequently engage with beneficial microorganisms that facilitate the uptake of limiting nutrients such as nitrogen and phosphate129. In agriculture, these beneficial associations are often dampened by supplied fertilizers, because plants suppress their interaction with symbionts when they perceive ample nutrients. Most plant species associate with arbuscular mycorrhizal fungi that greatly expand the root-surface area for nutrient uptake, and which actively mine immobilized phosphates from the soil130,131. Bringing associations with arbuscular mycorrhizal fungi more effectively into annual cropping systems with moderate fertilizer use could improve nutrient capture, and increase sustainability—particularly if the phosphate suppression of mycorrhization could be overridden. However, applications of strigolactones (the plant-derived low-phosphate signals to microorganisms) have so far been insufficient to override the suppression of mycorrhization132 and more research is therefore needed to obtain benefits from mycorrhizal associations in agriculture.
Engineering the nitrogen symbiosis
Some plants are colonized intracellularly by nitrogen-fixing bacteria that can deliver the complete nitrogen needs of the host plant. Associations such as this are limited to a select group of species, which presents an opportunity to radically improve nitrogen availability for cereal crops if the symbiosis trait can be transferred. Multiple avenues are being explored to achieve this—from equipping plants to associate with nitrogen-fixing bacteria to the transfer of the enzyme nitrogenase, which is responsible for nitrogen fixation. Studies of these processes in their native context have provided an understanding that was absent 30 years ago, when such approaches were first broached. Evolutionary, genomic and mechanistic studies suggest that relatively few genetic components might be needed to confer nitrogen-fixation capabilities. In the case of transferring nitrogenase to plants, the restriction of genetic components required was achieved by concatermerizing bacterial genetic units to create a minimal set of three genes that are necessary for the transfer of nitrogen fixation133. Moreover, some components of nitrogenase can be stably expressed in yeast and plants134.
The evolution of the nitrogen-fixing symbiosis in legumes used many components that function in associations with arbuscular mycorrhizal fungi129, which means that cereals possess some of the necessary building blocks and have the potential to streamline engineering efforts to transfer the nitrogen-fixing symbiosis. Recent phylogenomic approaches indicate that very minimal gene reduction (between two and seven genes) is associated with the loss of nitrogen fixation135,136, suggesting that a small set of genes could convert a species that associates with arbuscular mycorrhizal fungi into one that can also form nitrogen-fixing symbiosis. This considerable engineering challenge will require precise transcriptional and post-translational regulation of multiple heterologous genes in cereals. Additional challenges are the few well-characterized promoters for gene regulation in cereal roots, and bottlenecks associated with transformation of cereals that limit the scale of throughput required to test the engineering iterations that will be necessary to achieve nitrogen fixation.
Benefits of plant-associated microorganisms
The environment around and within plant roots includes microbial communities. These can be relatively restricted137 or dynamic138, and responsive to nutrient status139. Such communities or community members have the potential to protect plants against pathogen infections140,141 and, to some extent, drought142,143. A greater understanding of the mechanisms and the environmental conditions, including climate effects144, that control plant–microorganism assembly and activities may enable the engineering of microbial communities to optimize crop performance, particularly with microorganisms that are engineered using synthetic biology approaches. Current research indicates that some fungal species benefit host plants by enhancing phosphate uptake145, and within the diversity of cereal crops are lines that can host active communities of nitrogen-fixers146. The manipulation of microbial associations to improve crop resilience to environmental stresses is an area of intense research.
Prospectus on resilient crops
Research advances have provided innovative opportunities and technologies across the plant sciences, which can furnish solutions for addressing future food security (Box 1). The strategies described here for enhancing the resilience and sustainability of crops will only be realistic if they are part of an integrated approach to agriculture that is developed collaboratively with agronomists, engineers and farmers147. A critical challenge is the time from research discovery to true and widespread implementation in agriculture. Some high-impact breeding and genetically modified traits (for example, pest resistance mediated by individual Bacillus thuringiensis Cry proteins) have spread relatively rapidly. However, even in cases that involve breeding into diverse varieties, the time from initial discovery and development to broad use has often exceeded ten years79. Regulatory processes and intellectual property hurdles associated with technology can lead to additional delays in implementation. The robust assessment of varieties in variable field environments is essential to timely adoption. In the case of submergence-tolerant SUB1 rice varieties50, cooperation between scientists, breeders and farm advisers helped to achieve farmer acceptance and governmental certification within three years of gene characterization. The visible yield advantage of SUB1 varieties after flooding, and their lack of differences with the varieties they replaced, was key to their adoption. This success contrasts with the failure to provide many farmers in climate-vulnerable areas with the services of plant breeders to mobilize genetic variation for crop improvement148 and with selected or engineered genotypes that did not translate to the field46. Complementary approaches and technologies may provide viable opportunities (such as high-protein, salt-tolerant algae that require limited freshwater)—although new infrastructure, energy inputs and engineering solutions will be needed149.
Valuable genetic diversity for increasing crop resilience resides in cultivated landraces, heirloom varieties and the wild relatives of crops. Seed banks curate and distribute crop germplasm; the Crop Trust (https://www.croptrust.org) is one of the leading efforts to collect, conserve and use the approximately 50,000 species of wild relatives of crops150. These seed banks distribute germplasm that can be tapped for adaptations to abiotic and biotic stresses, but greater investment in high-throughput genotyping and phenotyping is needed to accelerate mapping, the identification of genes and mechanisms, and downstream breeding151.
Addressing yield loss due to climate change, salinity and (re)emerging diseases, weeds, parasitic plants and pests requires innovative technologies and proactive responses, not unlike the development of vaccines and innovations in modern medicine. The integration of genetic resources and transformative technologies, from genome editing to synthetic biology, are necessary to capture traits that increase global food security and reduce the effects of agriculture on the environment. An early failure of plant biotechnologists was in the lack of effective engagement with environmentalists, farmers and consumers on questions of health and safety, despite strict governmental procedures for the validation, release and monitoring of genetically modified crops. It is critical that the specific method used for crop improvement does not stymie the implementation of safe and effective solutions. Non-politicized regulatory systems are essential for scientific advances to scale to farmers within the timeframe needed.
The current timeline for increasing the resilience and sustainability of crops is too long. Crop varieties with new combinations or variants of disease-resistance genes are in preparation for use against newly emerged virulent pathogens. Advances in sequencing and the early detection of invasive pathogenic strains should enable better monitoring of disease, and therefore knowledge regarding where to deploy particular crop genotypes. The horizon for tailored panels of appropriately controlled genes that impart functional immunity in a commercial crop is years away. The most rapid translation to the field will be for small suites of genes from existing crop germplasm. For challenges that are difficult to overcome (such as resilience to heat and aridity during plant sexual reproduction98), disruptive advances such as the asexual propagation of seeds152 could lessen yield loss due to male infertility. Success in the engineering of improved photosynthesis, nutrient use and beneficial plant–microorganism interactions requires intensive investment, but could result in the gains needed.
The plant sciences have a critical role in meeting the food and fibre challenges of the future. Timely investments and research at many levels and collaborative efforts are paramount to deploying resilience mechanisms and improving the sustainability, yields and nutritional value of our crops.
References
Ray, D. K., Mueller, N. D., West, P. C. & Foley, J. A. Yield trends are insufficient to double global crop production by 2050. PLoS ONE 8, e66428 (2013).
Pingali, P. L. Green revolution: impacts, limits, and the path ahead. Proc. Natl Acad. Sci. USA 109, 12302–12308 (2012).
Duke, S. O. Perspectives on transgenic, herbicide-resistant crops in the United States almost 20 years after introduction. Pest Manag. Sci. 71, 652–657 (2015).
Tabashnik, B. E., Brévault, T. & Carrière, Y. Insect resistance to Bt crops: lessons from the first billion acres. Nat. Biotechnol. 31, 510–521 (2013).
Fitch, M. M. M., Manshardt, R. M., Gonsalves, D., Slightom, J. L. & Sanford, J. C. Virus resistant papaya plants derived from tissues bombarded with the coat protein gene of papaya ringspot virus. Bio/Technology 10, 1466–1472 (1992).
Castiglioni, P. et al. Bacterial RNA chaperones confer abiotic stress tolerance in plants and improved grain yield in maize under water-limited conditions. Plant Physiol. 147, 446–455 (2008).
Potrykus, I. From the concept of totipotency to biofortified cereals. Annu. Rev. Plant Biol. 66, 1–22 (2015).
Paul, J.-Y. et al. Golden bananas in the field: elevated fruit pro-vitamin A from the expression of a single banana transgene. Plant Biotechnol. J. 15, 520–532 (2017).
Murata, M. et al. A transgenic apple callus showing reduced polyphenol oxidase activity and lower browning potential. Biosci. Biotechnol. Biochem. 65, 383–388 (2001).
Rommens, C. M., Yan, H., Swords, K., Richael, C. & Ye, J. Low-acrylamide French fries and potato chips. Plant Biotechnol. J. 6, 843–853 (2008).
FAO. How to Feed the World in 2050, http://www.fao.org/fileadmin/templates/wsfs/docs/expert_paper/How_to_Feed_the_World_in_2050.pdf (FAO, 2009).
Herrero, M. et al. Farming and the geography of nutrient production for human use: a transdisciplinary analysis. Lancet Planet. Health 1, e33–e42 (2017).
Mills, G. et al. Closing the global ozone yield gap: quantification and cobenefits for multistress tolerance. Glob. Chang. Biol. 24, 4869–4893 (2018).
Nelson, R., Wiesner-Hanks, T., Wisser, R. & Balint-Kurti, P. Navigating complexity to breed disease-resistant crops. Nat. Rev. Genet. 19, 21–33 (2018).
Mueller, N. D. et al. Closing yield gaps through nutrient and water management. Nature 490, 254–257 (2012).
Fowler, D. et al. The global nitrogen cycle in the twenty-first century. Phil. Trans. R. Soc. Lond. B 368, 20130164 (2013).
Qiu, J. Phosphate fertilizer warning for China. Nature News https://doi.org/10.1038/news.2010.498 (2010).
Steffen, W. et al. Planetary boundaries: guiding human development on a changing planet. Science 347, 1259855 (2015).
Fowler, D. et al. Effects of global change during the 21st century on the nitrogen cycle. Atmos. Chem. Phys. 15, 13849–13893 (2015).
Boutrot, F. & Zipfel, C. Function, discovery, and exploitation of plant pattern recognition receptors for broad-spectrum disease resistance. Annu. Rev. Phytopathol. 55, 257–286 (2017).
Monteiro, F. & Nishimura, M. T. Structural, functional, and genomic diversity of plant NLR proteins: an evolved resource for rational engineering of plant immunity. Annu. Rev. Phytopathol. 56, 243–267 (2018).
De la Concepcion, J. C. et al. Polymorphic residues in rice NLRs expand binding and response to effectors of the blast pathogen. Nat. Plants 4, 576–585 (2018).
Pfeilmeier, S. et al. Expression of the Arabidopsis thaliana immune receptor EFR in Medicago truncatula reduces infection by a root pathogenic bacterium, but not nitrogen-fixing rhizobial symbiosis. Plant Biotechnol. J. 17, 569–579 (2019).
Koller, T. et al. Field grown transgenic Pm3e wheat lines show powdery mildew resistance and no fitness costs associated with high transgene expression. Transgenic Res. 28, 9–20 (2019).
Xu, G. et al. uORF-mediated translation allows engineered plant disease resistance without fitness costs. Nature 545, 491–494 (2017). The engineering of a pathogen-responsive upstream open reading frame cassette fused to a central Arabidopsis disease-resistance component (NPR1) in transgenic rice creates a crop line that exhibits broad-spectrum disease resistance without a yield penalty.
Wu, C.-H., Derevnina, L. & Kamoun, S. Receptor networks underpin plant immunity. Science 360, 1300–1301 (2018).
Hörger, A. C. et al. Balancing selection at the tomato RCR3 Guardee gene family maintains variation in strength of pathogen defense. PLoS Genet. 8, e1002813 (2012).
Chen, C., E, Z. & Lin, H.-X. Evolution and molecular control of hybrid incompatibility in plants. Front. Plant Sci. 7, 1208 (2016).
Dangl, J. L., Horvath, D. M. & Staskawicz, B. J. Pivoting the plant immune system from dissection to deployment. Science 341, 746–751 (2013).
Witek, K. et al. Accelerated cloning of a potato late blight-resistance gene using RenSeq and SMRT sequencing. Nat. Biotechnol. 34, 656–660 (2016).
Bevan, M. W. et al. Genomic innovation for crop improvement. Nature 543, 346–354 (2017).
Bailey, P. C. et al. Dominant integration locus drives continuous diversification of plant immune receptors with exogenous domain fusions. Genome Biol. 19, 23 (2018).
Arora, S. et al. Resistance gene cloning from a wild crop relative by sequence capture and association genetics. Nat. Biotechnol. 37, 139–143 (2019).
van Schie, C. C. N. & Takken, F. L. W. Susceptibility genes 101: how to be a good host. Annu. Rev. Phytopathol. 52, 551–581 (2014).
Bezrutczyk, M. et al. Sugar flux and signaling in plant–microbe interactions. Plant J. 93, 675–685 (2018). The engineering of recently identified plant sugar transporters, which provide pathogens with carbohydrates, enables the restriction of disease through reducing the nutrients available to these pathogens.
Gomez, M. A. et al. Simultaneous CRISPR/Cas9-mediated editing of cassava eIF4E isoforms nCBP-1 and nCBP-2 reduces cassava brown streak disease symptom severity and incidence. Plant Biotechnol. J. 17, 421–434 (2019).
Wang, Y. et al. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotechnol. 32, 947–951 (2014).
Acevedo-Garcia, J. et al. mlo-based powdery mildew resistance in hexaploid bread wheat generated by a non-transgenic TILLING approach. Plant Biotechnol. J. 15, 367–378 (2017).
Mehta, D. et al. Linking CRISPR-Cas9 interference in cassava to the evolution of editing-resistant geminiviruses. Genome Biol. 20, 80 (2019).
Bebber, D. P., Ramotowski, M. A. T. & Gurr, S. J. Crop pests and pathogens move polewards in a warming world. Nat. Clim. Chang. 3, 985–988 (2013).
Almeida, R. P. P. et al. Addressing the new global threat of Xylella fastidiosa. Phytopathology 109, 172–174 (2019).
Cheng, C. et al. Plant immune response to pathogens differs with changing temperatures. Nat. Commun. 4, 2530 (2013).
Velásquez, A. C., Castroverde, C. D. M. & He, S. Y. Plant-pathogen warfare under changing climate conditions. Curr. Biol. 28, R619–R634 (2018).
Chen, S., Zhang, W., Bolus, S., Rouse, M. N. & Dubcovsky, J. Identification and characterization of wheat stem rust resistance gene Sr21 effective against the Ug99 race group at high temperature. PLoS Genet. 14, e1007287 (2018). Resistance (NLR) genes that function at high temperature are effective against a fungal strain (ug99) that causes wheat stem rust disease and is threatening global wheat production.
Zhang, W. et al. Identification and characterization of Sr13, a tetraploid wheat gene that confers resistance to the Ug99 stem rust race group. Proc. Natl Acad. Sci. USA 114, E9483–E9492 (2017).
Mickelbart, M. V., Hasegawa, P. M. & Bailey-Serres, J. Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat. Rev. Genet. 16, 237–251 (2015).
Ismail, A. M. & Horie, T. Genomics, physiology, and molecular breeding approaches for improving salt tolerance. Annu. Rev. Plant Biol. 68, 405–434 (2017).
Voesenek, L. A. C. J. & Bailey-Serres, J. Flood adaptive traits and processes: an overview. New Phytol. 206, 57–73 (2015).
Singh, S., Mackill, D. J. & Ismail, A. M. Physiological basis of tolerance to complete submergence in rice involves genetic factors in addition to the SUB1 gene. AoB Plants 6, plu060 (2014).
Xu, K. et al. Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature 442, 705–708 (2006).
Fukao, T., Xu, K., Ronald, P. C. & Bailey-Serres, J. A variable cluster of ethylene response factor-like genes regulates metabolic and developmental acclimation responses to submergence in rice. Plant Cell 18, 2021–2034 (2006).
Dar, M. H. et al. No yield penalty under favorable conditions paving the way for successful adoption of flood tolerant rice. Sci. Rep. 8, 9245 (2018).
Hattori, Y. et al. The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460, 1026–1030 (2009).
Kuroha, T. et al. Ethylene–gibberellin signaling underlies adaptation of rice to periodic flooding. Science 361, 181–186 (2018). A loss-of-function allele of SEMIDWARF 1 confers short stature in Green Revolution rice, whereas a functional ethylene-induced allele enables elongation of submerged internodes for grain production above floodwaters.
Ashikari, M. et al. Loss-of-function of a rice gibberellin biosynthetic gene, GA20 oxidase (GA20ox-2), led to the rice ‘green revolution’. Breed. Sci. 52, 143–150 (2002).
Stein, J. C. et al. Genomes of 13 domesticated and wild rice relatives highlight genetic conservation, turnover and innovation across the genus Oryza. Nat. Genet. 50, 285–296 (2018). This inventory of genetic variation in the Oryza genus, and other recent pan-genome studies of crops, empower the exploration of phylogenetic relationships and genetic variation at disease-resistance and stress-resilience loci.
Gibbs, D. J. et al. Homeostatic response to hypoxia is regulated by the N-end rule pathway in plants. Nature 479, 415–418 (2011).
Licausi, F. et al. Oxygen sensing in plants is mediated by an N-end rule pathway for protein destabilization. Nature 479, 419–422 (2011).
Mendiondo, G. M. et al. Enhanced waterlogging tolerance in barley by manipulation of expression of the N-end rule pathway E3 ligase PROTEOLYSIS6. Plant Biotechnol. J. 14, 40–50 (2016).
Lin, C.-C. et al. Regulatory cascade involving transcriptional and N-end rule pathways in rice under submergence. Proc. Natl Acad. Sci. USA 116, 3300–3309 (2019). The submergence tolerance regulator SUB1A is unusual among ethylene-responsive transcription factors—including two related transcription factors encoded by genes it activates—in its insensitivity to oxygen-dependent turnover.
Liljegren, S. J. et al. SHATTERPROOF MADS-box genes control seed dispersal in Arabidopsis. Nature 404, 766–770 (2000).
Park, S.-Y. et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324, 1068–1071 (2009).
Ma, Y. et al. Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324, 1064–1068 (2009).
Zhao, Y. et al. ABA receptor PYL9 promotes drought resistance and leaf senescence. Proc. Natl Acad. Sci. USA 113, 1949–1954 (2016).
Yang, Z. et al. Leveraging abscisic acid receptors for efficient water use in Arabidopsis. Proc. Natl Acad. Sci. USA 113, 6791–6796 (2016).
Park, S.-Y. et al. Agrochemical control of plant water use using engineered abscisic acid receptors. Nature 520, 545–548 (2015).
Mega, R. et al. Tuning water-use efficiency and drought tolerance in wheat using abscisic acid receptors. Nat. Plants 5, 153–159 (2019).
Kudo, M. et al. A gene-stacking approach to overcome the trade-off between drought stress tolerance and growth in Arabidopsis. Plant J. 97, 240–256 (2019).
Hu, H. & Xiong, L. Genetic engineering and breeding of drought-resistant crops. Annu. Rev. Plant Biol. 65, 715–741 (2014).
Décima Oneto, C. et al. Water deficit stress tolerance in maize conferred by expression of an isopentenyltransferase (IPT) gene driven by a stress- and maturation-induced promoter. J. Biotechnol. 220, 66–77 (2016).
Wang, Y. et al. Molecular tailoring of farnesylation for plant drought tolerance and yield protection. Plant J. 43, 413–424 (2005).
Saradadevi, R., Palta, J. A. & Siddique, K. H. M. ABA-mediated stomatal response in regulating water use during the development of terminal drought in wheat. Front. Plant Sci. 8, 1251 (2017).
Uga, Y. et al. Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat. Genet. 45, 1097–1102 (2013).
Arai-Sanoh, Y. et al. Deep rooting conferred by DEEPER ROOTING 1 enhances rice yield in paddy fields. Sci. Rep. 4, 5563 (2014).
Li, X. et al. Genetic control of the root system in rice under normal and drought stress conditions by genome-wide association study. PLoS Genet. 13, e1006889 (2017).
Bray, A. L. & Topp, C. N. The quantitative genetic control of root architecture in maize. Plant Cell Physiol. 59, 1919–1930 (2018).
Bao, Y. et al. Plant roots use a patterning mechanism to position lateral root branches toward available water. Proc. Natl Acad. Sci. USA 111, 9319–9324 (2014).
Orosa-Puente, B. et al. Root branching toward water involves posttranslational modification of transcription factor ARF7. Science 362, 1407–1410 (2018).
Hall, A. J. & Richards, R. A. Prognosis for genetic improvement of yield potential and water-limited yield of major grain crops. Field Crops Res. 143, 18–33 (2013).
Nuccio, M. L., Paul, M., Bate, N. J., Cohn, J. & Cutler, S. R. Where are the drought tolerant crops? An assessment of more than two decades of plant biotechnology effort in crop improvement. Plant Sci. 273, 110–119 (2018).
Rubio, F., Gassmann, W. & Schroeder, J. I. Sodium-driven potassium uptake by the plant potassium transporter HKT1 and mutations conferring salt tolerance. Science 270, 1660–1663 (1995).
Mäser, P. et al. Altered shoot/root Na+ distribution and bifurcating salt sensitivity in Arabidopsis by genetic disruption of the Na+ transporter AtHKT1. FEBS Lett. 531, 157–161 (2002).
Ren, Z.-H. et al. A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat. Genet. 37, 1141–1146 (2005).
Sunarpi, et al. Enhanced salt tolerance mediated by AtHKT1 transporter-induced Na+ unloading from xylem vessels to xylem parenchyma cells. Plant J. 44, 928–938 (2005).
Munns, R. et al. Wheat grain yield on saline soils is improved by an ancestral Na+ transporter gene. Nat. Biotechnol. 30, 360–364 (2012).An HKT1 allele that confers sodium tolerance is crossed into a commercial durum wheat variety, thus enhancing yield under salinity stress.
Barberon, M. et al. Adaptation of root function by nutrient-induced plasticity of endodermal differentiation. Cell 164, 447–459 (2016).
Duan, L. et al. Endodermal ABA signaling promotes lateral root quiescence during salt stress in Arabidopsis seedlings. Plant Cell 25, 324–341 (2013).Root growth dynamics under salinity stress are regulated by processes in a specific cell layer, and effective gene engineering of these dynamics may require use of promoters with defined spatial and temporal activity.
Huang, C. F. et al. A bacterial-type ABC transporter is involved in aluminum tolerance in rice. Plant Cell 21, 655–667 (2009).
Maron, L. G. et al. Aluminum tolerance in maize is associated with higher MATE1 gene copy number. Proc. Natl Acad. Sci. USA 110, 5241–5246 (2013).
Pallotta, M. et al. Molecular basis of adaptation to high soil boron in wheat landraces and elite cultivars. Nature 514, 88–91 (2014).The molecular characterization of loci associated with boron tolerance uncovers variation in gene copy number and expression patterns that translate to the field.
Horton, R. M., Mankin, J. S., Lesk, C., Coffel, E. & Raymond, C. A review of recent advances in research on extreme heat events. Curr. Clim. Change Rep. 2, 242–259 (2016).
Moore, F. C., Baldos, U., Hertel, T. & Diaz, D. New science of climate change impacts on agriculture implies higher social cost of carbon. Nat. Commun. 8, 1607 (2017).
Lobell, D. B. et al. Greater sensitivity to drought accompanies maize yield increase in the U.S. Midwest. Science 344, 516–519 (2014).
Zhao, C. et al. Temperature increase reduces global yields of major crops in four independent estimates. Proc. Natl Acad. Sci. USA 114, 9326–9331 (2017).
Bita, C. E. & Gerats, T. Plant tolerance to high temperature in a changing environment: scientific fundamentals and production of heat stress-tolerant crops. Front. Plant Sci. 4, 273 (2013).
Jackson, R. D., Idso, S. B., Reginato, R. J. & Pinter, P. J. Jr. Canopy temperature as a crop water stress indicator. Wat. Resour. Res. 17, 1133–1138 (1981).
Cooper, M., Gho, C., Leafgren, R., Tang, T. & Messina, C. Breeding drought-tolerant maize hybrids for the US corn-belt: discovery to product. J. Exp. Bot. 65, 6191–6204 (2014).
Nuccio, M. L. et al. Expression of trehalose-6-phosphate phosphatase in maize ears improves yield in well-watered and drought conditions. Nat. Biotechnol. 33, 862–869 (2015).Improving drought resilience of maize yield was achieved through the design of a promoter that targets maize-ear tissue in a stage-specific manner.
Kretzschmar, T. et al. A trehalose-6-phosphate phosphatase enhances anaerobic germination tolerance in rice. Nat. Plants 1, 15124 (2015).
Griffiths, C. A. et al. Chemical intervention in plant sugar signalling increases yield and resilience. Nature 540, 574–578 (2016).The effective delivery of membrane-permeable and activatable small molecules could provide a new management strategy for drought protection.
Zhu, X.-G., Long, S. P. & Ort, D. R. Improving photosynthetic efficiency for greater yield. Annu. Rev. Plant Biol. 61, 235–261 (2010).
Dann, M. & Leister, D. Enhancing (crop) plant photosynthesis by introducing novel genetic diversity. Phil. Trans. R. Soc. Lond. B 372, 20160380 (2017).
Blankenship, R. E. Early evolution of photosynthesis. Plant Physiol. 154, 434–438 (2010).
Farquhar, G. D., von Caemmerer, S. & Berry, J. A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149, 78–90 (1980).
Long, S. P., Marshall-Colon, A. & Zhu, X.-G. Meeting the global food demand of the future by engineering crop photosynthesis and yield potential. Cell 161, 56–66 (2015).
Ort, D. R. et al. Redesigning photosynthesis to sustainably meet global food and bioenergy demand. Proc. Natl Acad. Sci. USA 112, 8529–8536 (2015).
Evans, J. R. & Clarke, V. C. The nitrogen cost of photosynthesis. J. Exp. Bot. 70, 7–15 (2019).
Murchie, E. H. & Niyogi, K. K. Manipulation of photoprotection to improve plant photosynthesis. Plant Physiol. 155, 86–92 (2011).
Betti, M. et al. Manipulating photorespiration to increase plant productivity: recent advances and perspectives for crop improvement. J. Exp. Bot. 67, 2977–2988 (2016).
Simkin, A. J., McAusland, L., Lawson, T. & Raines, C. A. Overexpression of the RieskeFeS protein increases electron transport rates and biomass yield. Plant Physiol. 175, 134–145 (2017).
von Caemmerer, S., Quick, W. P. & Furbank, R. T. The development of C4 rice: current progress and future challenges. Science 336, 1671–1672 (2012).
Evans, L. T. Adapting and improving crops: the endless task. Phil. Trans. R. Soc. Lond. B 352, 901–906 (1997).
Sharwood, R. E. Engineering chloroplasts to improve Rubisco catalysis: prospects for translating improvements into food and fiber crops. New Phytol. 213, 494–510 (2017).
Salesse-Smith, C. E. et al. Overexpression of Rubisco subunits with RAF1 increases Rubisco content in maize. Nat. Plants 4, 802–810 (2018).
Driever, S. M. et al. Increased SBPase activity improves photosynthesis and grain yield in wheat grown in greenhouse conditions. Phil. Trans. R. Soc. Lond. B 372, 20160384 (2017).
Kromdijk, J. et al. Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354, 857–861 (2016).Transgenic tobacco with accelerated interconversion of violaxanthin and zeaxanthin in the xanthophyll cycle, and increased amounts of a photosystem II subunit, yielded 15% greater production of plant biomass in natural field conditions.
South, P. F., Cavanagh, A. P., Liu, H. W. & Ort, D. R. Synthetic glycolate metabolism pathways stimulate crop growth and productivity in the field. Science 363, eaat9077 (2019).Transgenic tobacco with synthetic glycolate metabolism in the chloroplast showed enhanced photosynthesis and increased biomass in the field.
Zhang, J. et al. Insights into the molecular mechanisms of CO2-mediated regulation of stomatal movements. Curr. Biol. 28, R1356–R1363 (2018).
Hsu, P.-K. et al. Abscisic acid-independent stomatal CO2 signal transduction pathway and convergence of CO2 and ABA signaling downstream of OST1 kinase. Proc. Natl Acad. Sci. USA 115, E9971–E9980 (2018).
Hu, H. et al. Carbonic anhydrases are upstream regulators of CO2-controlled stomatal movements in guard cells. Nat. Cell Biol. 12, 87–93 (2010).A strategy for improving water use efficiency via modulating the stomatal CO 2 response is shown.
Franks, P. J. et al. Sensitivity of plants to changing atmospheric CO2 concentration: from the geological past to the next century. New Phytol. 197, 1077–1094 (2013).
Hughes, J. et al. Reducing stomatal density in barley improves drought tolerance without impacting on yield. Plant Physiol. 174, 776–787 (2017).
Caine, R. S. et al. Rice with reduced stomatal density conserves water and has improved drought tolerance under future climate conditions. New Phytol. 221, 371–384 (2019).
Głowacka, K. et al. Photosystem II subunit S overexpression increases the efficiency of water use in a field-grown crop. Nat. Commun. 9, 868 (2018).
Rogers, A., Ainsworth, E. A. & Leakey, A. D. B. Will elevated carbon dioxide concentration amplify the benefits of nitrogen fixation in legumes? Plant Physiol. 151, 1009–1016 (2009).
Schroeder, J. I. et al. Using membrane transporters to improve crops for sustainable food production. Nature 497, 60–66 (2013).
Li, S. et al. Modulating plant growth-metabolism coordination for sustainable agriculture. Nature 560, 595–600 (2018).
Cormier, F. et al. Breeding for increased nitrogen-use efficiency: a review for wheat (T. aestivum L.). Plant Breed. 135, 255–278 (2016).
Oldroyd, G. E. D. Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nat. Rev. Microbiol. 11, 252–263 (2013).
Harrison, M. J. Signaling in the arbuscular mycorrhizal symbiosis. Annu. Rev. Microbiol. 59, 19–42 (2005).
Choi, J., Summers, W. & Paszkowski, U. Mechanisms underlying establishment of arbuscular mycorrhizal symbioses. Annu. Rev. Phytopathol. 56, 135–160 (2018).
Foo, E., Yoneyama, K., Hugill, C. J., Quittenden, L. J. & Reid, J. B. Strigolactones and the regulation of pea symbioses in response to nitrate and phosphate deficiency. Mol. Plant 6, 76–87 (2013).
Yang, J. et al. Polyprotein strategy for stoichiometric assembly of nitrogen fixation components for synthetic biology. Proc. Natl Acad. Sci. USA 115, E8509–E8517 (2018).
Burén, S. & Rubio, L. M. State of the art in eukaryotic nitrogenase engineering. FEMS Microbiol. Lett. 365, fnx274 (2018).
Griesmann, M. et al. Phylogenomics reveals multiple losses of nitrogen-fixing root nodule symbiosis. Science 361, eaat1743 (2018).
van Velzen, R. et al. Comparative genomics of the nonlegume Parasponia reveals insights into evolution of nitrogen-fixing rhizobium symbioses. Proc. Natl Acad. Sci. USA 115, E4700–E4709 (2018).
Busby, P. E. et al. Research priorities for harnessing plant microbiomes in sustainable agriculture. PLoS Biol. 15, e2001793 (2017).
Edwards, J. A. et al. Compositional shifts in root-associated bacterial and archaeal microbiota track the plant life cycle in field-grown rice. PLoS Biol. 16, e2003862 (2018).
Castrillo, G. et al. Root microbiota drive direct integration of phosphate stress and immunity. Nature 543, 513–518 (2017).
Pieterse, C. M. J. et al. Induced systemic resistance by beneficial microbes. Annu. Rev. Phytopathol. 52, 347–375 (2014).
Kwak, M.-J. et al. Rhizosphere microbiome structure alters to enable wilt resistance in tomato. Nat. Biotechnol. 36, 1100–1109 (2018).Comparative rhizosphere metagenomics enables the identification of protective microbial strains from the root-associated microbiota of disease-resistant tomato plants.
Subramanian, K. S., Charest, C., Dwyer, L. M. & Hamilton, R. I. Arbuscular mycorrhizas and water relations in maize under drought stress at tasselling. New Phytol. 129, 643–650 (1995).
Augé, R. M. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 11, 3–42 (2001).
Fitzpatrick, C. R. et al. Assembly and ecological function of the root microbiome across angiosperm plant species. Proc. Natl Acad. Sci. USA 115, E1157–E1165 (2018).
Hiruma, K. et al. Root endophyte Colletotrichum tofieldiae confers plant fitness benefits that are phosphate status dependent. Cell 165, 464–474 (2016).
Van Deynze, A. et al. Nitrogen fixation in a landrace of maize is supported by a mucilage-associated diazotrophic microbiota. PLoS Biol. 16, e2006352 (2018).In this study, a maize landrace is demonstrated to stimulate symbiosis with microbes that have nitrogenase activity, which contributes to nitrogen nutrition.
Challinor, A. J., Koehler, A.-K., Ramirez-Villegas, J., Whitfield, S. & Das, B. Current warming will reduce yields unless maize breeding and seed systems adapt immediately. Nat. Clim. Chang. 6, 954–958 (2016).
Atlin, G. N., Cairns, J. E. & Das, B. Rapid breeding and varietal replacement are critical to adaptation of cropping systems in the developing world to climate change. Glob. Food Sec. 12, 31–37 (2017).
Walsh, M. J. et al. Algal food and fuel coproduction can mitigate greenhouse gas emissions while improving land and water-use efficiency. Environ. Res. Lett. 11, 114006 (2016).
Dempewolf, H. et al. Adapting agriculture to climate change: a global initiative to collect, conserve, and use crop wild relatives. Agroecol. Sustain. Food Syst. 38, 369–377 (2014).
McCouch, S. et al. Agriculture: feeding the future. Nature 499, 23–24 (2013).
Khanday, I., Skinner, D., Yang, B., Mercier, R. & Sundaresan, V. A male-expressed rice embryogenic trigger redirected for asexual propagation through seeds. Nature 565, 91–95 (2019).
Becker, R. A. & Wilks, A. R. Maps: draw geographical maps, v.3.3.0, https://cran.r-project.org/package=maps/ (2018).
Brakenridge, G. R. Global Active Archive of Large Flood Events (Dartmouth Flood Observatory, University of Colorado), http://floodobservatory.colorado.edu/Archives/index.html (accessed 2019).
López-Calcagno, P. E. et al. Overexpressing the H-protein of the glycine cleavage system increases biomass yield in glasshouse and field-grown transgenic tobacco plants. Plant Biotechnol. J. 17, 141–151 (2019).
Lin, M. T., Occhialini, A., Andralojc, P. J., Parry, M. A. J. & Hanson, M. R. A faster Rubisco with potential to increase photosynthesis in crops. Nature 513, 547–550 (2014).
Acknowledgements
We apologize to those authors whose research could not be cited owing to space limits. We thank P. J. Franks and E. Buckler for discussions, and A. Digrado and C. Benjamin for assistance with figures. Research in the authors’ laboratories was supported by grants from the US National Science Foundation to J.I.S. (MCB-1900567) and J.B.-S. (IOS-1546879; IOS-1810468; IOS-1856749); the US National Institutes of Health to J.I.S. (GM060396-ES010337); the National Institute of Food and Agriculture to J.B.-S. (2017-67013-26194; 2019-67013-29313); the Max-Planck Society and the Deutsche Forschungsgemeinschaft (DFG; German Research Foundation) under Germany’s Excellence Strategy (EXC-2048/1, project 390686111 and Priority Program 2125 ‘DECRyPT’) to J.E.P.; a sub-award to E.A.A. from the University of Illinois as part of the Realizing Increased Photosynthetic Efficiency (RIPE) project (OPP1060461), UK AID and the Foundation for Food and Agricultural Research; and the Engineering the Nitrogen Symbiosis in Africa project (OPP1172165) sponsored by the Bill & Melinda Gates Foundation and Gatsby Foundation grants to G.E.D.O.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Martin van Ittersum, Pamela Ronald and Cyril Zipfel for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bailey-Serres, J., Parker, J.E., Ainsworth, E.A. et al. Genetic strategies for improving crop yields. Nature 575, 109–118 (2019). https://doi.org/10.1038/s41586-019-1679-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1679-0
This article is cited by
-
Transcriptomics of temperature-sensitive R gene-mediated resistance identifies a WAKL10 protein interaction network
Scientific Reports (2024)
-
Emergency remedial measures to salvage losses from Chinese wheat post-harvest sprouting
npj Sustainable Agriculture (2024)
-
Technology-enabled great leap in deciphering plant genomes
Nature Plants (2024)
-
Identification, Evolutionary Relationship Analysis of the Trehalose-6-Phosphate Synthase (TPS) Gene Family in Common Bean (Phaseolus vulgaris) and Their Expression in Response to Cold Stress
Journal of Plant Growth Regulation (2024)
-
Assessment of variability and genetic divergence of longan genotypes (Dimocarpus longan Lour.) based on qualitative and quantitative traits
Euphytica (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.