Abstract
The mechanisms that extend lifespan in humans are poorly understood. Here we show that extended longevity in humans is associated with a distinct transcriptome signature in the cerebral cortex that is characterized by downregulation of genes related to neural excitation and synaptic function. In Caenorhabditis elegans, neural excitation increases with age and inhibition of excitation globally, or in glutamatergic or cholinergic neurons, increases longevity. Furthermore, longevity is dynamically regulated by the excitatory–inhibitory balance of neural circuits. The transcription factor REST is upregulated in humans with extended longevity and represses excitation-related genes. Notably, REST-deficient mice exhibit increased cortical activity and neuronal excitability during ageing. Similarly, loss-of-function mutations in the C. elegans REST orthologue genes spr-3 and spr-4 elevate neural excitation and reduce the lifespan of long-lived daf-2 mutants. In wild-type worms, overexpression of spr-4 suppresses excitation and extends lifespan. REST, SPR-3, SPR-4 and reduced excitation activate the longevity-associated transcription factors FOXO1 and DAF-16 in mammals and worms, respectively. These findings reveal a conserved mechanism of ageing that is mediated by neural circuit activity and regulated by REST.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data from the ROSMAP cohort are available under controlled use conditions set by human privacy regulations. To access the data, a data use agreement is needed. This registration is in place solely to ensure the anonymity of the ROSMAP study participants. Data can be requested on the Rush Alzheimer’s Disease Center Resource Sharing Hub at http://www.radc.rush.edu/. C. elegans RNA-seq data are available in the Gene Expression Omnibus (GEO) under accession number GSE123146.
Code availability
All code used in the analysis of data presented in this manuscript is available upon request.
References
Satoh, A., Imai, S. I. & Guarente, L. The brain, sirtuins, and ageing. Nat. Rev. Neurosci. 18, 362–374 (2017).
Bishop, N. A., Lu, T. & Yankner, B. A. Neural mechanisms of ageing and cognitive decline. Nature 464, 529–535 (2010).
Ailion, M., Inoue, T., Weaver, C. I., Holdcraft, R. W. & Thomas, J. H. Neurosecretory control of aging in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 96, 7394–7397 (1999).
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999).
Alcedo, J. & Kenyon, C. Regulation of C. elegans longevity by specific gustatory and olfactory neurons. Neuron 41, 45–55 (2004).
Bishop, N. A. & Guarente, L. Two neurons mediate diet-restriction-induced longevity in C. elegans. Nature 447, 545–549 (2007).
Wolkow, C. A., Kimura, K. D., Lee, M.-S. & Ruvkun, G. Regulation of C. elegans life-span by insulinlike signaling in the nervous system. Science 290, 147–150 (2000).
Lu, T. et al. Gene regulation and DNA damage in the ageing human brain. Nature 429, 883–891 (2004).
Mostafavi, S. et al. A molecular network of the aging human brain provides insights into the pathology and cognitive decline of Alzheimer’s disease. Nat. Neurosci. 21, 811–819 (2018).
Fromer, M. et al. Gene expression elucidates functional impact of polygenic risk for schizophrenia. Nat. Neurosci. 19, 1442–1453 (2016).
Gibbs, J. R. et al. Abundant quantitative trait loci exist for DNA methylation and gene expression in human brain. PLoS Genet. 6, e1000952 (2010).
Hilliard, M. A. et al. In vivo imaging of C. elegans ASH neurons: cellular response and adaptation to chemical repellents. EMBO J. 24, 63–72 (2005).
Pokala, N., Liu, Q., Gordus, A. & Bargmann, C. I. Inducible and titratable silencing of Caenorhabditis elegans neurons in vivo with histamine-gated chloride channels. Proc. Natl Acad. Sci. USA 111, 2770–2775 (2014).
Hamilton, B. et al. A systematic RNAi screen for longevity genes in C. elegans. Genes Dev. 19, 1544–1555 (2005).
Lu, T. et al. REST and stress resistance in ageing and Alzheimer’s disease. Nature 507, 448–454 (2014).
Hu, X. L. et al. Conditional deletion of NRSF in forebrain neurons accelerates epileptogenesis in the kindling model. Cereb. Cortex 21, 2158–2165 (2011).
Brennan, G. P. et al. Dual and opposing roles of microRNA-124 in epilepsy are mediated through inflammatory and NRSF-dependent gene networks. Cell Rep. 14, 2402–2412 (2016).
Gilbert, L. A. et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154, 442–451 (2013).
Dillin, A., Crawford, D. K. & Kenyon, C. Timing requirements for insulin/IGF-1 signallling in C. elegans. Science 298, 830–834 (2002).
Yang, P. et al. A C-terminal truncated mutation of spr-3 gene extends lifespan in Caenorhabditis elegans. Acta Biochim. Biophys. Sin. (Shanghai) 45, 540–548 (2013).
Calixto, A., Chelur, D., Topalidou, I., Chen, X. & Chalfie, M. Enhanced neuronal RNAi in C. elegans using SID-1. Nat. Methods 7, 554–559 (2010).
Lin, K., Dorman, J. B., Rodan, A. & Kenyon, C. daf-16: An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science 278, 1319–1322 (1997).
Evason, K., Huang, C., Yamben, I., Covey, D. F. & Kornfeld, K. Anticonvulsant medications extend worm life-span. Science 307, 258–262 (2005).
Evason, K., Collins, J. J., Huang, C., Hughes, S. & Kornfeld, K. Valproic acid extends Caenorhabditis elegans lifespan. Aging Cell 7, 305–317 (2008).
Chen, X. et al. Ethosuximide ameliorates neurodegenerative disease phenotypes by modulating DAF-16/FOXO target gene expression. Mol. Neurodegener. 10, 51 (2015).
Pozzi, D. et al. REST/NRSF-mediated intrinsic homeostasis protects neuronal networks from hyperexcitability. EMBO J. 32, 2994–3007 (2013).
Pecoraro-Bisogni, F. et al. REST-dependent presynaptic homeostasis induced by chronic neuronal hyperactivity. Mol. Neurobiol. 55, 4959–4972 (2018).
Li, Y. et al. Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum. Mol. Genet. 18, 4897–4904 (2009).
Bennett, D. A., Schneider, J. A., Arvanitakis, Z. & Wilson, R. S. Overview and findings from the religious orders study. Curr. Alzheimer Res. 9, 628–645 (2012).
Bennett, D. A. et al. Overview and findings from the Rush Memory and Aging Project. Curr. Alzheimer Res. 9, 646–663 (2012).
Mu, X. et al. A gene network downstream of transcription factor Math5 regulates retinal progenitor cell competence and ganglion cell fate. Dev. Biol. 280, 467–481 (2005).
Mao, C. A. et al. Neuronal transcriptional repressor REST suppresses an Atoh7-independent program for initiating retinal ganglion cell development. Dev. Biol. 349, 90–99 (2011).
Dhamne, S. C. et al. Replicable in vivo physiological and behavioral phenotypes of the Shank3B null mutant mouse model of autism. Mol. Autism 8, 26 (2017).
Kelly, E. et al. mGluR5 modulation of behavioral and epileptic phenotypes in a mouse model of tuberous sclerosis complex. Neuropsychopharmacology 43, 1457–1465 (2018).
Purtell, H. et al. Electrographic spikes are common in wildtype mice. Epilepsy Behav. 89, 94–98 (2018).
Yuskaitis, C. J. et al. A mouse model of DEPDC5-related epilepsy: neuronal loss of Depdc5 causes dysplastic and ectopic neurons, increased mTOR signaling, and seizure susceptibility. Neurobiol. Dis. 111, 91–101 (2018).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Lakowski, B. et al. Two suppressors of sel-12 encode C2H2 zinc-finger proteins that regulate presenilin transcription in Caenorhabditis elegans. Development 130, 2117–2128 (2003).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Ohnishi, N., Kuhara, A., Nakamura, F., Okochi, Y. & Mori, I. Bidirectional regulation of thermotaxis by glutamate transmissions in Caenorhabditis elegans. EMBO J. 30, 1376–1388 (2011).
Jarriault, S. & Greenwald, I. Suppressors of the egg-laying defective phenotype of sel-12 presenilin mutants implicate the CoREST corepressor complex in LIN-12/Notch signaling in C. elegans. Genes Dev. 16, 2713–2728 (2002).
Winston, W. M., Molodowitch, C. & Hunter, C. P. Systemic RNAi in C. elegans requires the putative transmembrane protein SID-1. Science 295, 2456–2459 (2002).
C. elegans Deletion Mutant Consortium. Large-scale screening for targeted knockouts in the Caenorhabditis elegans genome. G3 (Bethesda) 2, 1415–1425 (2012).
Nottke, A. C. et al. SPR-5 is a histone H3K4 demethylase with a role in meiotic double-strand break repair. Proc. Natl Acad. Sci. USA 108, 12805–12810 (2011).
Greer, E. L., Becker, B., Latza, C., Antebi, A. & Shi, Y. Mutation of C. elegans demethylase spr-5 extends transgenerational longevity. Cell Res. 26, 229–238 (2016).
Robert, V. J., Katic, I. & Bessereau, J. L. Mos1 transposition as a tool to engineer the Caenorhabditis elegans genome by homologous recombination. Methods 49, 263–269 (2009).
Kamath, R. S. & Ahringer, J. Genome-wide RNAi screening in Caenorhabditis elegans. Methods 30, 313–321 (2003).
Frøkjær-Jensen, C., Davis, M. W., Ailion, M. & Jorgensen, E. M. Improved Mos1-mediated transgenesis in C. elegans. Nat. Methods 9, 117–118 (2012).
Sarov, M. et al. A genome-scale resource for in vivo tag-based protein function exploration in C. elegans. Cell 150, 855–866 (2012).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
Sandelin, A., Alkema, W., Engström, P., Wasserman, W. W. & Lenhard, B. JASPAR: an open-access database for eukaryotic transcription factor binding profiles. Nucleic Acids Res. 32, D91–D94 (2004).
Grant, C. E., Bailey, T. L. & Noble, W. S. FIMO: scanning for occurrences of a given motif. Bioinformatics 27, 1017–1018 (2011).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinformatics 14, 128 (2013).
Zhang, Y. et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J. Neurosci. 34, 11929–11947 (2014).
Tepper, R. G. et al. PQM-1 complements DAF-16 as a key transcriptional regulator of DAF-2-mediated development and longevity. Cell 154, 676–690 (2013).
Alexa, A. & Rahnenfuhrer, J. topGO: enrichment analysis for gene ontology. R package version 2340 (2018).
Wang, M., Zhao, Y. & Zhang, B. Efficient test and visualization of multi-set intersections. Sci. Rep. 5, 16923 (2015).
Zaykin, D. V. Optimally weighted Z-test is a powerful method for combining probabilities in meta-analysis. J. Evol. Biol. 24, 1836–1841 (2011).
Dewey, M. Metap: meta analysis of significance values. R package Version 10 (2018).
Acknowledgements
We thank members of the Yankner laboratory for suggestions and discussion, and the Hsieh laboratory for providing the anti-mouse REST antibody. This work was supported by an NIH Director’s Pioneer Award (DP1OD006849) and NIH grants RO1AG046174 and RO1AG26651 to B.A.Y., RO1GM072551 to M.P.C., P30AG10161, R01AG15819, R01AG17917, R01AG36836 and U01AG46152 to D.A.B., EY024376 to C.-A.M., EY011930 to W.H.K. and K99AG050830 to J.M.Z., and grants from the Glenn Foundation for Medical Research and The Ludwig Family Foundation to B.A.Y. Data were generated as part of the CMC, supported by funding from Takeda Pharmaceuticals Company Limited, F. Hoffman-La Roche Ltd and NIH grants R01MH085542, R01MH093725, P50MH066392, P50MH080405, R01MH097276, RO1-MH-075916, P50M096891, P50MH084053S1, R37MH057881, R37MH057881S1, HHSN271201300031C, AG02219, AG05138 and MH06692.
Author information
Authors and Affiliations
Contributions
J.M.Z., P.O. and N.D. performed experiments in C. elegans; D.D. performed statistical analysis and informatics on human brain and C. elegans RNA-seq; L.A. and D.D. analysed Rest cKO mice and human brain sections; L.A., S.C.D. and A.R. performed PET–CT and electrophysiological analysis of mice; J.M.Z. performed cell culture; C.-A.M., W.H.K. and G.M.C. contributed reagents; D.A.B. contributed brain samples and data from the ROSMAP cohort; J.M.Z., D.D., M.P.C. and B.A.Y. contributed to the overall study design; B.A.Y. directed the study; and B.A.Y., J.M.Z. and D.D. wrote the manuscript, which was examined by all authors.
Corresponding author
Ethics declarations
Competing interests
G.M.C. is a cofounder and senior advisor for GC Therapeutics, Inc, which uses transcription factors for therapeutics. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Nektarios Tavernarakis and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Fig. 1 Partitioning of the ageing human population for analysis of gene expression in the brain.
a–c, Adjusted gene expression profiles of age-associated genes were compared between cognitively normal aged individuals to derive a matrix of Pearson correlation coefficients that indicate the degree of similarity between any two cases in the ROSMAP (a, dorsolateral prefrontal cortex, n = 150 individuals), CMC (b, dorsolateral prefrontal cortex, n = 174 individuals) and Gibbs (c, frontal cortex, n = 40 individuals) cohorts. d–f, Most significantly enriched GO terms for upregulated genes in the cortex of cognitively normal individuals who lived to be ≥85 years old relative to individuals who lived to be ≤80 years old in the ROSMAP (d, n = 117 individuals), CMC (e, n = 155 individuals), and Gibbs (f, n = 37 individuals) cohorts. P values were calculated using Fisher’s exact test (see Methods). g, Meta-analysis of GO term enrichment for downregulated genes. Shown are selected GO terms related to excitatory and inhibitory synaptic transmission. The individual cohort enrichment P values were combined using Stouffer’s method (see Methods). NS, not significant (FDR > 0.1).
Extended Data Fig. 2 Ivermectin and nemadipine extend lifespan without interfering with worm motility.
a, Worms were transferred at day 8 to either standard NGM plates or plates containing ivermectin (Ive, 1 pg ml–1) or nemadipine (2 μM). Shown is a representative curve of an experiment repeated twice. Nemadipine versus wild-type, P = 3.2 × 10–4; ivermectin versus wild-type, P = 2.2 × 10–7 by log-rank test. Nemadipine, n = 81; ivermectin, n = 82; wild-type, n = 76. b, Day 2 worms treated with nemadipine or ivermectin for 24 h were transferred to liquid culture and thrashing rate was assessed using the Nemametrix wMicrotracker (see Methods). Shown are mean motility scores for the first 60 min ± s.e.m. Untreated, n = 17 wells; ivermectin, n = 17 wells; nemadipine, n = 16 wells. Each well contained about 10 worms. **P = 1.7 × 10–4 versus untreated, Mann–Whitney U-test with multiple testing correction by Holm’s method. Results are representative of an experiment replicated twice.
Extended Data Fig. 3 Repression of multiple neurotransmitter systems extends lifespan in C. elegans.
a–h, C. elegans lines expressing the transgenic HisCl1 channel in the indicated neuronal populations were treated with 10 mM histamine (His+) starting at adult day 1 (a, c, e, g) or day 8 (b, d, f, h) and compared to untreated controls (His–). P values calculated by log-rank test. See Supplementary Table 22 for individual n values and statistics. i, Mean lifespan extension ± s.e.m. for worms treated with histamine at days 1 or 8 relative to untreated controls for at least three independent replicates. *P < 0.05, **P < 0.01 by Student’s t-test. HisCl1was driven using the GAL4SK:VP64 system for the GABAergic (GABA), glutamatergic (GLUT) and cholinergic systems, using unc-47, eat-4, and unc-17 drivers, respectively (see Supplementary Table 19). j, Reduced ASH neuron excitation following inhibition of GABA activity at day 1 but not day 8. Shown is normalized maximum GCaMP fluorescence in day 1 and 8 unc-47:HisCl1 worms that were treated with 10 mM histamine (His (+)) on the indicated day, or untreated controls (His (–)). Day 1 His(–), n = 18 worms; day 1 His(+), n = 19 worms; day 8 His(–), n = 23 worms; day 8 His(+), n = 20 worms. *P = 1.1 × 10–3 by Mann–Whitney U-test.
Extended Data Fig. 4 Neural excitation, neuropeptide signalling and lifespan in C. elegans.
a, Increased excitation of ASH neurons following RNAi against the GABA vesicular transporter unc-47. GCaMP imaging was performed on worms with enhanced neuronal RNAi (see Fig. 3 legend and Methods) for unc-47 (n = 37) or controls (n = 43) at day 2. **P = 6.8 × 10–3 by Mann–Whitney U-test. b, RNAi of unc-47 reduces lifespan. Worms with enhanced neuronal RNAi were treated with unc-47 (n = 31) or control RNAi (n = 84). Shown is a representative lifespan analysis replicated three times. P = 1.3 × 10–6 by log-rank test. c, Reduction in synaptic neurotransmission or neuropeptide signalling extends lifespan in C. elegans. Mutations in genes affecting glutamatergic neurotransmission (eat-4), presynaptic function (unc-13) and neuropeptide signalling (egl-3) produce comparable lifespan extensions. WT, n = 57; eat-4(nj2), n = 54, P ≤ 2.2 × 10–16; unc-13(e51), n = 92, P = 3.6 × 10–14; egl-3(gk238), n = 35, P = 8.3 × 10–11 by log-rank test. Curves are representative of two independent replicates. d, Extension of lifespan by egl-3 RNAi in worms with enhanced neuronal RNAi. Shown are lifespan curves representative of two independent replicates. egl-3 RNAi (n = 47 worms); empty vector (n = 84 worms). P = 3.5 × 10–11 by log-rank test.
Extended Data Fig. 5 Gene regulation and neural activity associated with REST and extended longevity.
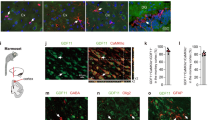
a, b, Expression of genes that are downregulated in individuals ≥85 years old versus ≤80 years old is inversely related to REST mRNA levels. Shown is linear regression analysis of normalized and adjusted REST mRNA levels and mean expression of all downregulated genes (a) and downregulated genes associated with the synaptic transmission GO term (b). Data are from the CMC cohort. Each point represents an individual case, n = 155 individuals. P values derived by a t-test for the slope of the regression line. Note similarity to the data for the ROSMAP cohort in Fig. 2a, b. c, d, Stratification by age group. Analysis of the ROSMAP cohort (c, n = 117 individuals) and the CMC cohort (d, n = 155 individuals) as in Fig. 2a, but stratified by age group. P values derived by t-test for the slope of the regression line. e, Loss of REST expression in conditional Rest knockout mice. Representative images of the cortex (top) and hippocampus (bottom) from Restlx/lx (control) and Nestin-Cre;Restlx/lx (Rest−/−) mice. Immunolabelling was performed with the anti-mouse REST-14 antibody directed against the REST C-terminal domain (Supplementary Table 20). Scale bar, 40 µm. Image is representative of an experiment replicated four times. f, Survival of Rest−/− and control mice following administration of the seizure-inducing agent PTZ (40 mg kg–1xr Rest−/− versus control by log-rank test. Control, n = 9; Rest−/−, n = 7.
Extended Data Fig. 6 Induction of spr-4 extends lifespan and suppresses neural excitation in C. elegans.
a, spr-4 mRNA levels in worms expressing a stably integrated dCas9::VP64 transgene in the presence (sgRNA(+)) or absence (sgRNA(–)) of four different sgRNAs targeting the spr-4 promoter. Transcript levels were determined by qRT–PCR and normalized to sgRNA(–) controls. Mean ± s.e.m., n = 3. Primer A, *P = 0.041; primer B, *P = 0.020 by one-sided Student’s t-test. b, dCas9::VP64-mediated elevation of SPR-4 protein levels. Left, representative images of the head region of heterozygous F1 progeny of the strains bearing a pspr-4::spr-4::gfp::spr-4utr transgene. Arrowheads indicate SPR-4::GFP-positive nuclei. Dashed red lines indicate the outline of the worm body. Scale bar, 40 μm. Middle, SPR-4::GFP protein levels are increased by dCAS9::VP64 mediated activation. Values represent mean ± s.e.m. sgRNA(–), n = 5 worms; sgRNA(+), n = 5 worms with 7–38 measurements per worm; *P = 0.022, one-sided Student’s t-test. Right, SPR-4::GFP expression appears in more cells following dCAS9::VP64-mediated activation. Values represent mean ± s.e.m. sgRNA(–), n = 4 worms; sgRNA(+), n = 4 worms. *P = 0.011, one-sided Student’s t-test. Shown is a representative experiment replicated three times. c, Extended lifespan in worms expressing an integrated dCas9::VP64 transgene and sgRNAs targeting the spr-4 promoter (sgRNA(+)) (n = 79 worms) relative to dCas9::VP64-expressing worms in the absence of sgRNAs (sgRNA(–)) (n = 57 worms). P = 5.5 × 10–9, log-rank test. Representative of an experiment replicated six times. d, Lifespans of worms expressing sgRNA targeting the spr-4 promoter in the presence (n = 87 worms) or absence (n = 58 worms) of dCas9::VP64. P = 3.7 × 10–7, log-rank test. Representative of an experiment replicated twice. e, Lifespans of dCas9::VP64-expressing worms in the presence (n = 51 worms) or absence (n = 58 worms) of sgRNAs on the spr-4(tm465) loss-of-function mutant background. P = 0.49, log-rank test. Representative of three independent replicates. f, Overexpression of spr-4 reduces neural excitation. GCaMP imaging was performed in ASH neurons in SPR-4-overexpressing (sgRNA(+)) and control (sgRNA(–)) worms in the lines described in c. Shown are maximum GCaMP fluorescence changes. sgRNA(–), n = 12 worms; sgRNA(+), n = 10 worms. *P = 0.025, Mann–Whitney U-test.
Extended Data Fig. 7 Lifespan extension by overexpression of spr-4 and inhibition of neural excitation depends on DAF-16.
a, Extension of lifespan by overexpression of spr-4 is dependent on DAF-16. Lifespans of worms overexpressing spr-4 (sgRNA(+); dCAS9::VP64) or not overexpressing spr-4 (sgRNA(–); dCAS9::VP64) following treatment with daf-16 RNAi or an empty vector control. sgRNA(+) EV (n = 29 worms) versus sgRNA(–) EV (n = 25 worms): P = 2.7 × 10–4; sgRNA(+) daf-16 (n = 18 worms) versus sgRNA(–) daf-16 (n = 29 worms): P = 0.20 by log-rank test. Representative of four independent replicates. b, c, Extension of lifespan by the inhibitors of neural excitation ivermectin and nemadipine is DAF-16-dependent. Shown are lifespan determinations for wild-type control and daf-16(mu86) mutant worms in the presence or absence of nemadipine (2 μM; b) or ivermectin (1 pg ml–1; c). b, WT, n = 69 worms; WT + Nema, n = 51; daf-16, n = 43; daf-16 + Nema, n = 67. WT + Nema versus WT, P = 9.9 × 10–8; daf-16 + Nema versus daf-16, P = 0.014; log-rank test. c, WT, n = 78 worms; WT + Ive, n = 77; daf-16, n = 27; daf-16 + Ive, n = 29. WT + Ive versus WT, P = 7.3 × 10–8; daf-16 + Ive versus daf-16, P = 0.22; log-rank test. Curves are representative of an experiment replicated two (nemadipine) or three (ivermectin) times. d, Inhibition of neural excitation with ivermectin elevates DAF-16 levels. Worms expressing a Daf-16::GFP transgene were treated for 10 days with 1 pg ml–1 ivermectin and assessed by confocal microscopy. Left, total DAF-16::GFP (mean ± s.e.m.). Untreated, n = 19 worms; ivermectin, n = 16 worms. **P = 2.5 × 10–7, Mann–Whitney U-test. Right, nuclear DAF-16::GFP. n = 5 worms per group, 50–61 nuclei per worm. *P = 0.013 by Student’s t-test. Results are representative of an experiment replicated twice. e, DAF-16 is not required for inhibition of neural excitation by nemadipine. Shown are maximum ASH GCaMP intensity changes for day 2 daf-16(mu86) mutant worms treated for 24 h with 2 μM nemadipine (untreated, n = 16 worms; nemadipine, n = 18 worms). P = 9.4 × 10–5, Mann–Whitney U-test. f, DAF-16 is not required for inhibition of neural excitation by ivermectin. Shown are data from day 2 worms treated for 24 h with 1 pg ml–1 ivermectin (control, n = 19 worms; ivermectin, n = 32 worms). P = 0.030, Mann–Whitney U-test.
Extended Data Fig. 8 SPR-3 and SPR-4 contribute to lifespan extension and gene regulation associated with reduced DAF-2 and insulin–IGF-like signalling.
a, Loss of function of SPR-3 and SPR-4 reduces the lifespan extension induced by daf-2 RNAi. Left, representative lifespan analysis of spr-4(by105);spr-3(ok2525) double mutant and wild-type worms following daf-2 or empty vector control RNAi. WT + EV, n = 54 worms; spr-4;3 + EV, n = 58 worms; WT + daf-2, n = 26 worms; spr-4;3 + daf-2, n = 54 worms. Right, values represent mean ± s.e.m. per cent lifespan extension (daf-2 RNAi versus EV control) in the indicated genotypes. WT, n = 6 independent experiments; spr-4(by105), n = 3, *P = 0.017 versus WT; spr-4(tm465), n = 4, **P = 0.0062 versus WT; spr-3(ok2525), n = 4, **P = 0.0018 versus WT; spr-4(by105);spr-3(ok2525), n = 4, **P = 0.0016 versus WT; Students t-test. See Supplementary Table 22 for individual lifespan data and statistics. b, Lifespan is unaffected by spr-4 and spr-3 mutations in a wild-type background. WT, n = 50 worms; spr-3(ok2525), n = 31; spr-4(by105);spr-3(ok2525), n = 32; spr-4(by105), n = 34; spr-4(tm465), n = 33.There were no reproducibly significant changes by the log-rank test in 3–6 independent experiments per genotype (see Supplementary Table 22). c, Quantification of lifespan extension in daf-2 mutant worms shown in Fig. 3b attributable to neuronal expression of spr-3 and spr-4. RNAi was targeted to neurons by neuronal expression of a sid-1 transgene in otherwise sid-1-null daf-2(1370) mutants (daf-2;p[neuron]:sid-1), and compared with untargeted RNAi in sid-1 wild-type daf-2(1370) mutants (daf-2). Values represent mean ± s.e.m. lifespan extension relative to the control sid-1(pk3321);p[neuron]:sid-1 worms treated with empty vector (n = 3 independent experiments). Significant lifespan effects were not observed for RNAi in the absence of the daf-2 mutation. *P < 0.05; **P < 0.01 by Student’s t-test. d, Gene expression determined by RNA-seq in day 2 adult worms. Differentially expressed genes (rows) and the indicated worm genotypes (columns) were clustered, and gene expression, transformed to a z-score per gene, is represented in a heat map. n = 3 independent replicates per genotype. e, Venn diagram illustrating the overlap in differentially expressed genes in daf-2 single mutant versus WT and spr-4;spr-3;daf-2 triple mutant versus daf-2 single mutant worms. P = 7 × 10–30, Fisher’s exact test with a one-sided alternative hypothesis. f, Long-lived daf-2 mutants do not show an age-related increase in neural excitation. Shown are maximum ASH GCaMP intensity changes in day 1–2 (n = 39) and day 14–16 (n = 20) daf-2(e1370) mutant worms. Note the absence of the age-related increase in excitation observed in wild-type ageing worms (Fig. 1e). P = 0.93, Mann–Whitney U-test. g, The spr-4;spr-3 double mutation in a wild-type background does not significantly affect neural excitation in ASH neurons. WT, n = 15 worms; spr-4;spr-3, n = 15 worms. P = 0.62, Mann–Whitney U-test. h, DAF-16 does not mediate suppression of neural excitation in the daf-2 mutant. RNAi against daf-16 was performed in daf-2(e1370) mutant worms on a sid-1(pk3321);p[neuron]:sid-1 background to augment RNAi in neurons (daf-16 RNAi, n = 20 worms, EV control, n = 12 worms). P = 0.33, Mann–Whitney U-test. i, Descriptions of the genes targeted by RNAi in Fig. 4d.
Extended Data Fig. 9 Regulation of DAF-16 by SPR-3 and SPR-4.
a, Reduced DAF-16 activation in spr-4;spr-3 mutants following daf-2 RNAi. Left confocal image, day 10 worms of the indicated genotypes expressing an integrated Daf-16::GFP transgene and treated with daf-2 RNAi or empty vector control since day 1 of adulthood. Images are maximum intensity z-projections. Scale bar, 40 μm. Left bar graph, mean ± s.e.m. GFP intensity in the peri-pharyngeal regions of spr-4;spr-3 double mutants relative to wild-type controls for a representative experiment replicated four times (see Methods for details of analysis). n = 8–12 worms per replicate. **P = 5.2 × 10–5, Welch’s t-test. Right confocal image, higher-magnification views of DAF-16::GFP and DAPI-labelled nuclei. Images are magnified confocal z-planes. Scale bar, 10 μm. Right bar graph, mean ± s.e.m. nuclear GFP intensity relative to the WT-EV control, n = 5 worms per genotype and 51–89 nuclei per worm. *P = 0.016, **P = 5.5 × 10–3 by ANOVA with post hoc Tukey test. Values and images are representative of an experiment replicated three times. b, Gene expression determined by RNA-seq in adult day 10 worms. Differentially expressed genes (rows) and replicates of the indicated worm genotypes (columns) were clustered, and gene expression, transformed into a z-score per gene, is represented in a heat map. n = 3 independent replicates per genotype. c, Venn diagram illustrating the overlap of differentially expressed genes in day 10 daf-2 versus wild-type and spr-4;spr-3;daf-2 versus daf-2 worms. P = 4 × 10–123, Fisher’s exact test with a one-sided alternative hypothesis. d, Overlap of class I daf-16 target genes (see Methods) with genes downregulated in day 10 spr-4;spr-3;daf-2 triple mutants relative to daf-2 single mutants. P values calculated using a hypergeometric distribution (see Methods). n.s., P = 0.99. e, Ivermectin increases DAF-16::GFP levels in spr-4;spr-3 worms following daf-2 RNAi. Left, confocal imaging of GFP fluorescence in ivermectin-treated (10 pg ml–1) and untreated worms. The red dashed lines indicate the worm body. Right, quantification of DAF-16::GFP (mean GFP intensity ± s.e.m., WT/Untreated, n = 12; WT/Ivermectin, n = 10; spr-4;spr-3/Untreated, n = 10; spr-4;spr-3/Ivermectin, n = 10. **P = 4.6 × 10–4 (spr-4;spr-3 versus WT/untreated), P = 2.6 × 10–4 (spr-4;spr-3/Ivermectin versus spr-4;spr-3/untreated) by Mann–Whitney U-test with multiple testing correction by Holm’s method. Shown is a representative experiment replicated three times.
Extended Data Fig. 10 Coregulation of FOXO1 and REST in the ageing brain and modulation by glutamatergic signalling.
a, Linear regression analysis of REST and FOXO mRNA levels in the prefrontal cortex of 174 cognitively intact individuals (age ≥60 years) from the CMC cohort determined by RNA-seq. P values derived from a t-test for the slope of the regression line and Bonferroni-corrected across all expressed genes. b, Colocalization of REST and FOXO1 in neurons of the aged human prefrontal cortex. Confocal immunofluorescence microscopy was performed in human prefrontal cortex using antibodies against REST (green, rabbit polyclonal; Bethyl), FOXO1 (red, goat polyclonal; LS-Bio) and the neuronal marker MAP2 (grey, chicken polyclonal; Abcam). Scale bar, 40 μm. The image shown is representative of immunofluorescence labelling performed in 30 individuals. See Supplementary Table 20 for additional information on antibodies. c, Inhibition of glutamatergic signalling in mouse cortical neuronal cultures elevates FOXO1 levels. Left, primary mouse cortical neuronal cultures treated with kynurenic acid (KYNA, 5 μM), AP5 (50 μM), NBQX (2 μM) or vehicle control were analysed by confocal immunofluorescence for FOXO1 or MAP2 and labelled with DAPI. Boxed areas are magnified in the lower three rows. Note that most FOXO1 in cultured neurons is cytoplasmic, but a detectable nuclear component overlaps with DAPI. Scale bar, 40 μm. Right, quantification (mean ± s.e.m.) of total and nuclear FOXO1 levels in MAP2-positive neurons. Control, n = 200; KYNA, n = 326; AP5, n = 148; NBQX, n = 197. FOXO1 total/KYNA, **P = 2.1 × 10–8; FOXO1 nuclear/KYNA, **P = 1.1 × 10–4; FOXO1 total/NBQX, **P = 8.8 × 10–13; FOXO1 nuclear/NBQX, **P = 5.2 × 10–6; Mann–Whitney U-test with multiple testing correction by Holm’s method. Shown is a representative experiment replicated three times.
Supplementary information
Supplementary Information
This file contains a Guide to Supplementary Tables 1-22 (see separate file for tables).
Supplementary Tables
This file contains Supplementary Tables 1-22 – see Supplementary Information document for guide.
Video 1
GCaMP recording of an ASH neuron in a day 2 wild-type worm.
Video 2
GCaMP recording of an ASH neuron in a day 2 spr-4;3;daf-2 worm.
Video 3
GCaMP recording of an ASH neuron in a day 2 daf-2 worm.
Video 4
Wild-type worms, day 2, untreated.
Video 5
Wild-type worms, day 2, treated with 2 μM nemadipine.
Video 6
Wild-type worms, day 2, treated with 1pg/mL ivermectin.
Rights and permissions
About this article
Cite this article
Zullo, J.M., Drake, D., Aron, L. et al. Regulation of lifespan by neural excitation and REST. Nature 574, 359–364 (2019). https://doi.org/10.1038/s41586-019-1647-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1647-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.