Abstract
Denisovans are members of a hominin group who are currently only known directly from fragmentary fossils, the genomes of which have been studied from a single site, Denisova Cave1,2,3 in Siberia. They are also known indirectly from their genetic legacy through gene flow into several low-altitude East Asian populations4,5 and high-altitude modern Tibetans6. The lack of morphologically informative Denisovan fossils hinders our ability to connect geographically and temporally dispersed fossil hominins from Asia and to understand in a coherent manner their relation to recent Asian populations. This includes understanding the genetic adaptation of humans to the high-altitude Tibetan Plateau7,8, which was inherited from the Denisovans. Here we report a Denisovan mandible, identified by ancient protein analysis9,10, found on the Tibetan Plateau in Baishiya Karst Cave, Xiahe, Gansu, China. We determine the mandible to be at least 160 thousand years old through U-series dating of an adhering carbonate matrix. The Xiahe specimen provides direct evidence of the Denisovans outside the Altai Mountains and its analysis unique insights into Denisovan mandibular and dental morphology. Our results indicate that archaic hominins occupied the Tibetan Plateau in the Middle Pleistocene epoch and successfully adapted to high-altitude hypoxic environments long before the regional arrival of modern Homo sapiens.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All the proteomic mass spectrometry data have been deposited in the ProteomeXchange Consortium repository (http://www.proteomexchange.org/) with the identifier PXD011377. Protein consensus sequences for the Xiahe hominin used for phylogenetic analysis are available in Supplementary Information 2. A surface scan model of the Xiahe mandible is publicly available at: https://www.eva.mpg.de/evolution/downloads/registration-form-xiahe.html.
Code availability
All R code used to generate protein deamidation and peptide cleavage patterns are available upon request from F.W. (frido.welker@palaeo.eu).
References
Krause, J. et al. The complete mitochondrial DNA genome of an unknown hominin from southern Siberia. Nature 464, 894–897 (2010).
Sawyer, S. et al. Nuclear and mitochondrial DNA sequences from two Denisovan individuals. Proc. Natl Acad. Sci. USA 112, 15696–15700 (2015).
Slon, V. et al. A fourth Denisovan individual. Sci. Adv. 3, e1700186 (2017).
Browning, S. R., Browning, B. L., Zhou, Y., Tucci, S. & Akey, J. M. Analysis of human sequence data reveals two pulses of archaic Denisovan admixture. Cell 173, 53–61 (2018).
Sankararaman, S., Mallick, S., Patterson, N. & Reich, D. The combined landscape of Denisovan and Neanderthal ancestry in present-day humans. Curr. Biol. 26, 1241–1247 (2016).
Huerta-Sánchez, E. et al. Altitude adaptation in Tibetans caused by introgression of Denisovan-like DNA. Nature 512, 194–197 (2014).
Chen, F. H. et al. Agriculture facilitated permanent human occupation of the Tibetan Plateau after 3600 B.P. Science 347, 248–250 (2015).
Meyer, M. C. et al. Permanent human occupation of the central Tibetan Plateau in the early Holocene. Science 355, 64–67 (2017).
Welker, F. Palaeoproteomics for human evolution studies. Quat. Sci. Rev. 190, 137–147 (2018).
Welker, F. Elucidation of cross-species proteomic effects in human and hominin bone proteome identification through a bioinformatics experiment. BMC Evol. Biol. 18, 23 (2018).
Reich, D. et al. Genetic history of an archaic hominin group from Denisova Cave in Siberia. Nature 468, 1053–1060 (2010).
Qin, P. & Stoneking, M. Denisovan ancestry in East Eurasian and Native American populations. Mol. Biol. Evol. 32, 2665–2674 (2015).
Skoglund, P. & Jakobsson, M. Archaic human ancestry in East Asia. Proc. Natl Acad. Sci. USA 108, 18301–18306 (2011).
Reich, D. et al. Denisova admixture and the first modern human dispersals into Southeast Asia and Oceania. Am. J. Hum. Genet. 89, 516–528 (2011).
Jeong, C. et al. Admixture facilitates genetic adaptations to high altitude in Tibet. Nat. Commun. 5, 3281 (2014).
Hackinger, S. et al. Wide distribution and altitude correlation of an archaic high-altitude-adaptive EPAS1 haplotype in the Himalayas. Hum. Genet. 135, 393–402 (2016).
Zhang, D. et al. History and possible mechanisms of prehistoric human migration to the Tibetan Plateau. Sci. China Earth Sci. 59, 1765–1778 (2016).
Yuan, B., Huang, W. & Zhang, D. New evidence for human occupation of the northern Tibetan Plateau, China during the Late Pleistocene. Chin. Sci. Bull. 52, 2675–2679 (2007).
Zhao, M. et al. Mitochondrial genome evidence reveals successful Late Paleolithic settlement on the Tibetan Plateau. Proc. Natl Acad. Sci. USA 106, 21230–21235 (2009).
Zhang, X. L. et al. The earliest human occupation of the high-altitude Tibetan Plateau 40 thousand to 30 thousand years ago. Science 362, 1049–1051 (2018).
Welker, F. et al. Palaeoproteomic evidence identifies archaic hominins associated with the Châtelperronian at the Grotte du Renne. Proc. Natl Acad. Sci. USA 113, 11162–11167 (2016).
Meyer, M. et al. A high-coverage genome sequence from an archaic Denisovan individual. Science 338, 222–226 (2012).
Bailey, S. E., Skinner, M. M. & Hublin, J.-J. What lies beneath? An evaluation of lower molar trigonid crest patterns based on both dentine and enamel expression. Am. J. Phys. Anthropol. 145, 505–518 (2011).
Chang, C.-H. et al. The first archaic Homo from Taiwan. Nat. Commun. 6, 6037 (2015).
Xing, S., Martinón-Torres, M., Bermúdez de Castro, J. M., Wu, X. & Liu, W. Hominin teeth from the early Late Pleistocene site of Xujiayao, Northern China. Am. J. Phys. Anthropol. 156, 224–240 (2015).
Douka, K. et al. Age estimates for hominin fossils and the onset of the Upper Palaeolithic at Denisova Cave. Nature 565, 640–644 (2019).
Prüfer, K. et al. The complete genome sequence of a Neanderthal from the Altai Mountains. Nature 505, 43–49 (2014).
Lisiecki, L. E. & Raymo, M. E. A Pliocene–Pleistocene stack of 57 globally distributed benthic δ18O records. Paleoceanography 20, PA1003 (2005).
Uemura, R. et al. Ranges of moisture-source temperature estimated from Antarctic ice cores stable isotope records over glacial–interglacial cycles. Clim. Past 8, 1109–1125 (2012).
North Greenland Ice Core Project members. High-resolution record of Northern Hemisphere climate extending into the last interglacial period. Nature 431, 147–151 (2004).
Beck, J. W. et al. A 550,000-year record of East Asian monsoon rainfall from 10Be in loess. Science 360, 877–881 (2018).
Madsen, D. B. et al. Early foraging settlement of the Tibetan Plateau highlands. Archaeol. Res. Asia 11, 15–26 (2017).
Brantingham, J. P. et al. in Developments in Quaternary Sciences, vol. 9 (eds Madsen, D. B. et al.) 129–150 (Elsevier, Amsterdam, 2007).
Shen, C.-C. et al. Variation of initial 230Th/232Th and limits of high precision U–Th dating of shallow-water corals. Geochim. Cosmochim. Acta 72, 4201–4223 (2008).
Shen, C.-C. et al. High-precision and high-resolution carbonate 230Th dating by MC-ICP-MS with SEM protocols. Geochim. Cosmochim. Acta 99, 71–86 (2012).
Wiley, D. F. et al. Evolutionary morphing. Proc. IEEE Visualization 431–438 (2005).
Gunz, P. & Mitteroecker, P. Semilandmarks: a method for quantifying curves and surfaces. Hystrix It. J. Mamm. 24, 103–109 (2013).
Mitteroecker, P. & Bookstein, F. Linear discrimination, ordination, and the visualization of selection gradients in modern morphometrics. Evol. Biol. 38, 100–114 (2011).
Copes, L. Comparative and Experimental Investigations of Cranial Robusticity in Mid-Pleistocene Hominins PhD thesis, Arizona State Univ. (2012).
Mardia, K. V., Bookstein, F. L. & Moreton, I. J. Statistical assessment of bilateral symmetry of shapes. Biometrika 92, 249–250 (2005).
Rohlf, F. J. & Slice, D. Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst. Biol. 39, 40–59 (1990).
Bookstein, F. L. Morphometric Tools for Landmark Data: Geometry and Biology (Cambridge Univ. Press, 1997).
Stelzer, S., Gunz, P., Neubauer, S. & Spoor, F. Using the covariation of extant hominoid upper and lower jaws to predict dental arcades of extinct hominins. J. Hum. Evol. 114, 154–175 (2018).
Stelzer, S., Neubauer, S., Hublin, J.-J., Spoor, F. & Gunz, P. Morphological trends in arcade shape and size in Middle Pleistocene Homo. Am. J. Phys. Anthropol. 168, 70–91 (2019).
Spoor, F. et al. Reconstructed Homo habilis type OH 7 suggests deep-rooted species diversity in early Homo. Nature 519, 83–86 (2015).
Mathematica v.5.1 (Wolfram Research, 2004).
Turner, C. G. II, Nichol, C. R. & Scott, G. R. in Advances in Dental Anthropology (eds Kelley, M. A. & Larsen, C. S.) 13–31 (Wiley-Liss, New York, 1991).
Skinner, M. M., Gunz, P., Wood, B. A. & Hublin, J.-J. Enamel–dentine junction (EDJ) morphology distinguishes the lower molars of Australopithecus africanus and Paranthropus robustus. J. Hum. Evol. 55, 979–988 (2008).
Skinner, M. M., Gunz, P., Wood, B. A., Boesch, C. & Hublin, J.-J. Discrimination of extant Pan species and subspecies using the enamel–dentine junction morphology of lower molars. Am. J. Phys. Anthropol. 140, 234–243 (2009).
Dabney, J. et al. Complete mitochondrial genome sequence of a Middle Pleistocene cave bear reconstructed from ultrashort DNA fragments. Proc. Natl Acad. Sci. USA 110, 15758–15763 (2013).
Gansauge, M.-T. & Meyer, M. Single-stranded DNA library preparation for the sequencing of ancient or damaged DNA. Nat. Protoc. 8, 737–748 (2013).
Fu, Q. et al. DNA analysis of an early modern human from Tianyuan Cave, China. Proc. Natl Acad. Sci. USA 110, 2223–2227 (2013).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Welker, F., Soressi, M., Rendu, W., Hublin, J.-J. & Collins, M. J. Using ZooMS to identify fragmentary bone from the late Middle/Early Upper Palaeolithic sequence of Les Cottés, France. J. Archaeol. Sci. 54, 279–286 (2015).
Welker, F. et al. Variations in glutamine deamidation for a Châtelperronian bone assemblage as measured by peptide mass fingerprinting of collagen. Sci. Technol. Archaeol. Res. 3, 15–27 (2017).
Chambers, M. C. et al. A cross-platform toolkit for mass spectrometry and proteomics. Nat. Biotechnol. 30, 918–920 (2012).
Zhang, J. et al. PEAKS DB: de novo sequencing assisted database search for sensitive and accurate peptide identification. Mol. Cell. Proteomics 11, M111.010587 (2012).
Hendy, J. et al. A guide to ancient protein studies. Nat. Ecol. Evol. 2, 791–799 (2018).
van Doorn, N. L., Wilson, J., Hollund, H., Soressi, M. & Collins, M. J. Site-specific deamidation of glutamine: a new marker of bone collagen deterioration. Rapid Commun. Mass Spectrom. 26, 2319–2327 (2012).
Castellano, S. et al. Patterns of coding variation in the complete exomes of three Neandertals. Proc. Natl Acad. Sci. USA 111, 6666–6671 (2014).
Meyer, M. et al. Nuclear DNA sequences from the Middle Pleistocene Sima de los Huesos hominins. Nature 531, 504–507 (2016).
Miller, M. A., Pfeiffer, W. & Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Gateway Computing Environments Workshop 1–8 (2010).
Hiess, J., Condon, D. J., McLean, N. & Noble, S. R. 238U/235U Systematics in terrestrial uranium-bearing minerals. Science 335, 1610–1614 (2012).
Cheng, H. et al. Improvements in 230Th dating, 230Th and 234U half-life values, and U–Th isotopic measurements by multi-collector inductively coupled plasma mass spectrometry. Earth Planet. Sci. Lett. 371–372, 82–91 (2013).
Jaffey, A. H., Flynn, K. F., Glendenin, L. E., Bentley, W. C. & Essling, A. M. Precision measurement of half-lives and specific activities of 235U and 238U. Phys. Rev. C 4, 1889–1906 (1971).
Welker, F. et al. Middle Pleistocene protein sequences from the rhinoceros genus Stephanorhinus and the phylogeny of extant and extinct Middle/Late Pleistocene Rhinocerotidae. PeerJ 5, e3033 (2017).
Acknowledgements
We thank the sixth Gung-Thang Living Buddha, the anonymous monk, L. L. Wang and her husband for providing us the opportunity to study the fossil; D. Madsen, J. Brantingham, D. Rhode, C. Perreault, J. S. Yang, T. Cheng, X. K. Shen, J. T. Yao, Z. X. Yang, J. Chen, X. Z. Huang, M. H. Qiu and C.-R. Huang for their assistance with the fieldwork and in the laboratory; members of the local government of Xiahe County and Ganjia town for help, the monks in the Baishiya temple and people from Bajiao Ancient City for their support of the fieldwork; O. Jöris, G. Smith, P. Ungar and R. Grün for discussions and comments; many curators and colleagues who, over the years, gave us access to recent and fossil hominin specimens for computed tomography scanning, photogrammetry or analysis; E. Trinkaus for providing comparative data; H. Temming, S. Tuepke, C. Molenaar and Diondo for their technical assistance; S. Pääbo, V. Slon and A. Ayinuer-Petri for ancient DNA analytical support. We received support from the Strategic Priority Research Program of Chinese Academy of Sciences, Pan-Third Pole Environment Study for a Green Silk Road (Pan-TPE) (XDA20040000) and National Natural Science Foundation of China (41620104007). D.J.Z. received support from National Natural Science Foundation of China (41771225). Fieldwork in 2018 was supported by the Second Tibetan Plateau Scientific Expedition (Project no. 4). U–Th dating was supported by the Science Vanguard Research Program of the Ministry of Science and Technology (107-2119-M-002-051) and the Higher Education Sprout Project of the Ministry of Education, Taiwan (107L901001). J.-J.H. and F.W. thank the Max Planck Society for providing financial support.
Reviewer information
Nature thanks Aida Gómez-Robles, Antonio Rosas and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
F.H.C., J.-J.H. and D.J.Z. designed the study. D.J.Z., G.R.D., G.H.D., H.W., J.W. and H.X. collected field data. D.J.Z., H.X. and J.W. carried out sampling and subsampling process. C.-C.S. and T.-L.Y. conducted U–Th dating of the crust carbonates. Q.M.F. performed the ancient DNA extractions. Mandibular metrical and non-metrical data were compiled and analysed by J.-J.H. and I.B. Geometric morphometric analyses of the mandible were performed by S.E.F., S.S., I.B., D.J.Z. and J.-J.H. Dental metrical and non-metrical data were compiled and analysed by S.E.B. and M.M.S. H.X., F.W., R.F. and S.D. performed proteomic analysis. F.H.C., F.W., J.-J.H., D.J.Z. and C.-C.S. wrote the paper with contributions of all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
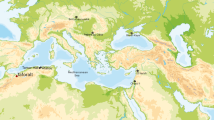
Extended Data Fig. 1 Location of Baishiya Karst Cave.
a, Location of Baishiya Karst Cave, archaic Homo sites, and selected Middle and Upper Palaeolithic sites in East and Central Asia. Numbers denoting the archaeological sites are: (1) Mezmaiskaya Cave; (2) Teshik-Tash Cave; (3) Okladnikov Cave; (4) Denisova Cave; (5) Kara-Bom; (6) Tongtian Cave; (7) Nyadem; (8) Quesang; (9) Chikhen Agui; (10) Shuidonggou; (11) Guanyin Cave, (12) Zhiren Cave; (13) Fuyan Cave; (14) Lingjing; (15) Xujiayao; (16) Tianyuan Cave; (17) Zhoukoudian Upper Cave; (18) Jinniushan. Denisovan ancestry is also detectable in other East and South Asian populations at low frequencies. b, Ganjia Basin with the Baishiya Karst Cave (red star) and two Palaeolithic sites (NML01, Nimalong01; WET01, Waerta01; blue triangles). Base maps generated by ArcGIS 10.3 using data provided by https://earthexplorer.usgs.gov/.
Extended Data Fig. 2 The Xiahe mandible with adhering carbonate crust.
a–f, Lateral (a), buccal (b), occlusal (c), inferior (d), anterior (e) and posterior (f) views. Sampling locations for ancient proteins and ancient DNA can be seen on the M2 and ascending ramus, and sampling location for U–Th dating on the inferior surface.
Extended Data Fig. 3 Surface model of the Xiahe mandible after digital removal of the adhering carbonate crust.
a–f, Lateral (a), buccal (b), occlusal (c), inferior (d), anterior (e) and posterior (f) views.
Extended Data Fig. 4 Preservation of the Xiahe dentine proteome.
a, Deamidation of five Xiahe proteins also identified in several proteomes from Middle and Late Pleistocene fossils. b, Glutamine deamidation of peptide P1105 observed in the zooarchaeology by mass spectrometry (ZooMS) analysis of the Xiahe dentine sample compared to reference data. n = number of specimens included. c, Correlation between deamidation observed in LC–MS/MS experiments for ammonium bicarbonate (AmBic) and acid-demineralization (Acid) extracts (R2 = 0.99). d, Length distribution of non-tryptic peptides in a Late Pleistocene Neanderthal and the Xiahe dentine proteome. AA, amino acids. Violins stretch from the minimum to the maximum value; box plots indicate the median (middle line), 25% and 75% (boxes) and stretch to 1.5× the interquartile range (whiskers). n = number of unique spectra included. Deamidation is based on quantitative MALDI–TOF-MS analysis (b) or semiquantitative LC–MS/MS spectral counting methods (a, c). ‘0%’ indicates an absence of deamidation and ‘100%’ indicates complete deamidation of asparagine and glutamine. a, d, Combined data for all six LC–MS/MS runs conducted on the Xiahe protein extracts. c, Combined data from the three replicates per extraction method. Samples are colour-coded according to geological age. Data were obtained from previous studies21,54,55,66.
Extended Data Fig. 5 Normalized frequencies of peptide cleavage patterns of selected proteins.
Spectral counts are based on the total number of ammonium bicarbonate and acid-demineralization PSMs (six runs) and include both N and C termini of each aligned PSM. Red colours indicate more PSMs than expected and green colours fewer PSMs than expected, compared to a random cleavage model for each protein separately. Note differences in colour scale. The title of each plot refers to the UniProt accession code for the relevant protein.
Extended Data Fig. 6 Geometric morphometrics of the mandible.
bgPCA of mandibular shape. Although some overlap exists, all groups show a distinct mandibular shape. Xiahe plots at the edge of the H. erectus distribution and within the range of Middle Pleistocene Homo. Surface models illustrate mandibular shape changes along bgPC1 (lateral view) and bgPC2 (lateral and superior view). Recent H. sapiens are shown in cyan, Upper Palaeolithic and Holocene H. sapiens in light blue, early H. sapiens in dark blue, Neanderthals in pink, H. erectus in green and other Middle Pleistocene fossil hominins in orange. SH, Sima de los Huesos. n = number of unique individuals contained in each hull. Further specimen names can be found in Supplementary Table 6.
Extended Data Fig. 7 Geometric morphometrics of the dental arcade.
a, bgPCA in form space. b, bgPCA in shape space. The wireframes illustrate form changes along bgPC1 and bgPC2. Colours are as in Extended Data Fig. 6. The wireframes show the form and shape changes along bgPC1 and bgPC2, respectively. Estimated wireframes used in the bgPCA are indicated by ‘est’. SH, Sima de los Huesos; D, Dmanisi. Further specimen names can be found in Supplementary Table 6. n = number of unique individuals contained in each hull.
Extended Data Fig. 8 Root morphology and EDJ shape.
a, The roots of the M1 are typical of lower molars with a mesial and distal plate-like root. There are mesial and distal plate-like roots on the M2; however, there is an additional accessory lingual root that splits off distally from the mesial root about 2/3 from the cervix. The P3 root is a Tomes’ form with a distinct lingual groove. b, A bgPCA of the EDJ ridge and cervix shape reveals a clear separation between early Homo and H. erectus on one side and Neanderthals and H. sapiens on the other side, with Middle Pleistocene hominins in between. Colours are as in Extended Data Fig. 6 for relevant groups. KRP, Krapina; LQ, La Quina. n = number of unique individuals contained in each hull. Note that the taxonomic status of Sangiran 5 is currently uncertain. Further specimen names can be found in Supplementary Table 6.
Supplementary information
Supplementary Information
This file contains Supplementary Information Sections 1-4, including an additional description of U-Th dating, comparative samples for morphological analysis, further notes on additional morphological results and reference data for palaeoproteomic phylogenetic analysis. It includes Supplementary Tables 1-5, Supplementary Figures 1-4 and additional references.
Supplementary Data
File in .fasta format containing consensus sequences derived from the Xiahe mandible proteomic analysis used for phylogenetic analysis.
Supplementary Table 6
This table contains specimen names and numbers used for the various morphological analysis conducted in comparison to the Xiahe mandible.
Rights and permissions
About this article
Cite this article
Chen, F., Welker, F., Shen, CC. et al. A late Middle Pleistocene Denisovan mandible from the Tibetan Plateau. Nature 569, 409–412 (2019). https://doi.org/10.1038/s41586-019-1139-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1139-x
This article is cited by
-
Preservation of proteins in the geosphere
Nature Ecology & Evolution (2024)
-
More than a decade of genetic research on the Denisovans
Nature Reviews Genetics (2024)
-
The Middle Pleistocene human metatarsal from Sedia del Diavolo (Rome, Italy)
Scientific Reports (2024)
-
Quaternary integrative stratigraphy, biotas, and paleogeographical evolution of the Qinghai-Tibetan Plateau and its surrounding areas
Science China Earth Sciences (2024)
-
New dating indicates intermittent human occupation of the Nwya Devu Paleolithic site on the high-altitude central Tibetan Plateau during the past 45,000 years
Science China Earth Sciences (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.