Abstract
Metabotropic glutamate receptors are family C G-protein-coupled receptors. They form obligate dimers and possess extracellular ligand-binding Venus flytrap domains, which are linked by cysteine-rich domains to their 7-transmembrane domains. Spectroscopic studies show that signalling is a dynamic process, in which large-scale conformational changes underlie the transmission of signals from the extracellular Venus flytraps to the G protein-coupling domains—the 7-transmembrane domains—in the membrane. Here, using a combination of X-ray crystallography, cryo-electron microscopy and signalling studies, we present a structural framework for the activation mechanism of metabotropic glutamate receptor subtype 5. Our results show that agonist binding at the Venus flytraps leads to a compaction of the intersubunit dimer interface, thereby bringing the cysteine-rich domains into close proximity. Interactions between the cysteine-rich domains and the second extracellular loops of the receptor enable the rigid-body repositioning of the 7-transmembrane domains, which come into contact with each other to initiate signalling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its Supplementary Information. The X-ray crystal structures and structure factors of the apo mGlu5 ECD, of the apo mGlu5 ECD in complex with Nb43 and of the mGlu5 ECD in complex with Nb43 and l-quisqualate have been deposited in the Protein Data Bank under accession codes 6N4X, 6N4Y and 6N50, respectively. Cryo-EM maps of apo mGlu5 in nanodiscs, apo mGlu5 in complex with Nb43 and active mGlu5 in complex with Nb43 and l-quisqualate in GDN have been deposited in the Electron Microscopy Data Bank under accession codes EMD-0346, EMD-0347 and EMD-0345, respectively. The atomic coordinates of apo mGlu5 and active mGlu5 in complex with Nb43 and l-quisqualate have been deposited in the Protein Data Bank under accession codes 6N52 and 6N51, respectively.
Change history
27 February 2019
The surname of author Toon Laeremans was misspelled ‘Laermans’. This error has been corrected online.
References
Niswender, C. M. & Conn, P. J. Metabotropic glutamate receptors: physiology, pharmacology, and disease. Annu. Rev. Pharmacol. Toxicol. 50, 295–322 (2010).
Pin, J.-P. & Bettler, B. Organization and functions of mGlu and GABAB receptor complexes. Nature 540, 60–68 (2016).
Kunishima, N. et al. Structural basis of glutamate recognition by a dimeric metabotropic glutamate receptor. Nature 407, 971–977 (2000).
Kniazeff, J. et al. Closed state of both binding domains of homodimeric mGlu receptors is required for full activity. Nat. Struct. Mol. Biol. 11, 706–713 (2004).
Hlavackova, V. et al. Sequential inter- and intrasubunit rearrangements during activation of dimeric metabotropic glutamate receptor 1. Sci. Signal. 5, ra59 (2012).
Wu, H. et al. Structure of a class C GPCR metabotropic glutamate receptor 1 bound to an allosteric modulator. Science 344, 58–64 (2014).
Doré, A. S. et al. Structure of class C GPCR metabotropic glutamate receptor 5 transmembrane domain. Nature 511, 557–562 (2014).
Christopher, J. A. et al. Fragment and structure-based drug discovery for a class C GPCR: discovery of the mGlu5 negative allosteric modulator HTL14242 (3-chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J. Med. Chem. 58, 6653–6664 (2015).
Christopher, J. A. et al. Structure-based optimization strategies for G protein-coupled receptor (GPCR) allosteric modulators: a case study from analyses of new metabotropic glutamate receptor 5 (mGlu5) X-ray structures. J. Med. Chem. https://doi.org/10.1021/acs.jmedchem.7b01722 (2018).
Thal, D. M., Glukhova, A., Sexton, P. M. & Christopoulos, A. Structural insights into G-protein-coupled receptor allostery. Nature 559, 45–53 (2018).
Goudet, C. et al. Heptahelical domain of metabotropic glutamate receptor 5 behaves like rhodopsin-like receptors. Proc. Natl Acad. Sci. USA 101, 378–383 (2004).
El Moustaine, D. et al. Distinct roles of metabotropic glutamate receptor dimerization in agonist activation and G-protein coupling. Proc. Natl Acad. Sci. 109, 16342–16347 (2012).
Manglik, A., Kobilka, B. K. & Steyaert, J. Nanobodies to study G protein-coupled receptor structure and function. Annu. Rev. Pharmacol. Toxicol. 57, 19–37 (2017).
Kinney, G. G. et al. A novel selective positive allosteric modulator of metabotropic glutamate receptor subtype 5 has in vivo activity and antipsychotic-like effects in rat behavioral models. J. Pharmacol. Exp. Ther. 313, 199–206 (2005).
Gregory, K. J. et al. Investigating metabotropic glutamate receptor 5 allosteric modulator cooperativity, affinity, and agonism: enriching structure-function studies and structure-activity relationships. Mol. Pharmacol. 82, 860–875 (2012).
Bradley, S. J., Langmead, C. J., Watson, J. M. & Challiss, R. A. J. Quantitative analysis reveals multiple mechanisms of allosteric modulation of the mGlu5 receptor in rat astroglia. Mol. Pharmacol. 79, 874–885 (2011).
Noetzel, M. J. et al. Functional impact of allosteric agonist activity of selective positive allosteric modulators of metabotropic glutamate receptor subtype 5 in regulating central nervous system function. Mol. Pharmacol. 81, 120–133 (2012).
Bradley, S. J., Watson, J. M. & Challiss, R. A. J. Effects of positive allosteric modulators on single-cell oscillatory Ca2+ signaling initiated by the type 5 metabotropic glutamate receptor. Mol. Pharmacol. 76, 1302–1313 (2009).
Doumazane, E. et al. Illuminating the activation mechanisms and allosteric properties of metabotropic glutamate receptors. Proc. Natl Acad. Sci. USA 110, E1416–E1425 (2013).
Chappell, M. D. et al. Discovery of (1S,2R,3S,4S,5R,6R)-2-amino-3-[(3,4-difluorophenyl)sulfanylmethyl]-4-hydroxy-bicyclo[3.1.0]hexane-2,6-dicarboxylic acid hydrochloride (LY3020371·HCl): A potent, metabotropic glutamate 2/3 receptor antagonist with antidepressant-like activity. J. Med. Chem. 59, 10974–10993 (2016).
Huang, S. et al. Interdomain movements in metabotropic glutamate receptor activation. Proc. Natl Acad. Sci. USA 108, 15480–15485 (2011).
Siméon, F. G. et al. Synthesis and simple 18F-labeling of 3-fluoro-5-(2-(2-(fluoromethyl)thiazol-4-yl)ethynyl)benzonitrile as a high affinity radioligand for imaging monkey brain metabotropic glutamate subtype-5 receptors with positron emission tomography. J. Med. Chem. 50, 3256–3266 (2007).
Muto, T., Tsuchiya, D., Morikawa, K. & Jingami, H. Structures of the extracellular regions of the group II/III metabotropic glutamate receptors. Proc. Natl Acad. Sci. USA 104, 3759–3764 (2007).
Ray, K. & Hauschild, B. C. Cys-140 is critical for metabotropic glutamate receptor-1 dimerization. J. Biol. Chem. 275, 34245–34251 (2000).
Manglik, A. et al. Structural insights into the dynamic process of β2-adrenergic receptor signaling. Cell 161, 1101–1111 (2015).
Rosenbaum, D. M. et al. Structure and function of an irreversible agonist-β2 adrenoceptor complex. Nature 469, 236–240 (2011).
Levitz, J. et al. Mechanism of assembly and cooperativity of homomeric and heteromeric metabotropic glutamate receptors. Neuron 92, 143–159 (2016).
Stenkamp, R. E. Identifying G protein-coupled receptor dimers from crystal packings. Acta Crystallogr. D 74, 655–670 (2018).
Milligan, G. G protein-coupled receptor hetero-dimerization: contribution to pharmacology and function. Br. J. Pharmacol. 158, 5–14 (2009).
James, J. R., Oliveira, M. I., Carmo, A. M., Iaboni, A. & Davis, S. J. A rigorous experimental framework for detecting protein oligomerization using bioluminescence resonance energy transfer. Nat. Methods 3, 1001–1006 (2006).
Xue, L. et al. Major ligand-induced rearrangement of the heptahelical domain interface in a GPCR dimer. Nat. Chem. Biol. 11, 134–140 (2015).
Isberg, V. et al. Generic GPCR residue numbers – aligning topology maps while minding the gaps. Trends Pharmacol. Sci. 36, 22–31 (2015).
Rondard, P., Rovira, X., Goudet, C. & Pin, J.-P. in mGLU Receptors (ed. Ngomba, R.T. et al.) 129–147 (Springer, Cham, 2017).
Geng, Y., Bush, M., Mosyak, L., Wang, F. & Fan, Q. R. Structural mechanism of ligand activation in human GABAB receptor. Nature 504, 254–259 (2013).
Geng, Y. et al. Structural mechanism of ligand activation in human calcium-sensing receptor. eLife 5, e13662 (2016).
Martín, M., Sanz, J. M. & Cubero, A. Characterization of metabotropic glutamate receptors coupled to a pertussis toxin sensitive G-protein in bovine brain coated vesicles. FEBS Lett. 316, 191–196 (1993).
Doornbos, M. L. J. et al. Molecular mechanism of positive allosteric modulation of the metabotropic glutamate receptor 2 by JNJ-46281222. Br. J. Pharmacol. 173, 588–600 (2016).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Evans, P. Scaling and assessment of data quality. Acta Crystallogr. D 62, 72–82 (2006).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D 67, 235–242 (2011).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Adams, P. D. et al. The Phenix software for automated determination of macromolecular structures. Methods 55, 94–106 (2011).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Bricogne, G. et al. BUSTER v.2.10.3 (Global Phasing, Cambridge, 2017).
Smart, O. S. et al. Exploiting structure similarity in refinement: automated NCS and target-structure restraints in BUSTER. Acta Crystallogr. D 68, 368–380 (2012).
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997).
Williams, C. J. et al. MolProbity: More and better reference data for improved all-atom structure validation. Protein Sci. 27, 293–315 (2018).
Pardon, E. et al. A general protocol for the generation of nanobodies for structural biology. Nat. Protoc. 9, 674–693 (2014).
Cosford, N. D. P. et al. 3-[(2-Methyl-1,3-thiazol-4-yl)ethynyl]-pyridine: a potent and highly selective metabotropic glutamate subtype 5 receptor antagonist with anxiolytic activity. J. Med. Chem. 46, 204–206 (2003).
Vélez-Ruiz, G. A. & Sunahara, R. K. Reconstitution of G protein-coupled receptors into a model bilayer system: reconstituted high-density lipoprotein particles. Methods Mol. Biol. 756, 167–182 (2011).
Lindsley, C. W. et al. Discovery of positive allosteric modulators for the metabotropic glutamate receptor subtype 5 from a series of N-(1,3-diphenyl-1H- pyrazol-5-yl)benzamides that potentiate receptor function in vivo. J. Med. Chem. 47, 5825–5828 (2004).
Zheng, S. Q., Palovcak, E., Armache, J.-P., Verba, K. A., Cheng, Y. & Agard, D. A. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: Real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Scheres, S. H. W. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Heymann, J. B. & Belnap, D. M. Bsoft: image processing and molecular modeling for electron microscopy. J. Struct. Biol. 157, 3–18 (2007).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38, 27–28 (1996).
Phillips, J. C. et al. Scalable molecular dynamics with NAMD. J. Comput. Chem. 26, 1781–1802 (2005).
Robertson, M. J., Tirado-Rives, J. & Jorgensen, W. L. Improved peptide and protein torsional energetics with the OPLSAA force field. J. Chem. Theory Comput. 11, 3499–3509 (2015).
Acknowledgements
The work is supported by National Institutes of Health (NIH) grant R01 NS092695 (G.S., B.K.K. and J.M.M.) and NIH grant R01 NS028471 (B.K.K.). Research reported in this publication was supported by the National Institute of General Medical Sciences (NIGMS) of the NIH under award number T32GM007276 (A.K.). R.F. is funded by grant NNF15OC0015268 from the Novo Nordisk Foundation and the Stanford Bio-X Program. We thank H. Lundbeck for the synthesis of FFMTEB. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. Use of the Stanford Synchrotron Radiation Lightsource (SSRL), SLAC National Accelerator Laboratory, is supported by the DOE under contract no. DE-AC02-76SF00515. The SSRL Structural Molecular Biology Program is supported by the DOE Office of Biological and Environmental Research, and by the NIGMS (including P41GM103393). The content is solely the responsibility of the authors and does not necessarily represent the official view of the National Institutes of Health. B.K.K. is a Chan Zuckerberg Biohub Investigator.
Reviewer information
Nature thanks K. Gregory, J.-P. Pin and J. Rubinstein for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
A.K. developed purification schemes and purified all proteins for cryo-EM studies as well as mGlu5 ECD constructs for crystallography, obtained the ECD crystals with Nb43 and solved their structures, built and refined models of full-length mGlu5 from cryo-EM data, and wrote the manuscript. H.H. prepared cryo-EM grids, collected and processed cryo-EM data, and assisted with manuscript preparation. D.F. developed the purification protocol for crystallization of full-length mGlu5, obtained crystals of full-length mGlu5 and prepared mGlu5 samples for immunization. B.S. collected crystals of full-length mGlu5, collected and processed diffraction data and refined the model with assistance from M.C. and A.K. T.S.K. supervised D.F., B.S. and M.C. Y.Z. screened conditions for freezing cryo-EM grids of mGlu5. M.J.R. performed molecular dynamics simulations of mGlu5. T.L. and J.S. performed llama immunization, cDNA production, and selections by phage display. S.D. and J.T. performed early characterizations of mGlu5 by electron microscopy and helped to develop purification schemes. R.F. assisted with model building. W.I.W. assisted with model building, refinement and interpretation of all structures. J.M.M. conceived and performed all in vitro characterizations of ligands, mGlu5 constructs and mutants, and co-wrote the manuscript. G.S. and B.K.K. supervised the project and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
B.K.K. and T.S.K. are founders of and consultants for ConfometRx, Inc. D.F. and B.S. are employees of ConfometRx, Inc., and M.C. is a former employee of ConfometRx, Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Structural basis of Nb43 binding to mGlu5.
a, Cartoon view of the apo mGlu5 in complex with Nb43, coloured by b-factors. Notably, the CRDs are diametrically opposed and do not form a stable interface with each other, as reflected by increased b-factors relative to the VFT. b, The Nb43-binding interface, with a 2Fo − Fc map of residues comprising CDR3 at 1σ shown as blue mesh. c, d, Residues involved in Nb43 binding are located on helix L and the L–M loop, and are not conserved. e, The 2Fo − Fc map of the mGlu5 ECD bound to l-quisqualate and Nb43 is shown in grey mesh at 1σ around a cartoon representation of the refined model. The inset shows the quality of density in the l-quisqualate-binding pocket.
Extended Data Fig. 2 Overview of mGlu5 cryo-EM studies.
a, Two-dimensional negative-stain class averages of apo mGlu5 in LMNG shows several classes with split detergent micelles. b, c, Representative cryo-EM images of apo mGlu5 in nanodiscs (b) and active mGlu5 bound to l-quisqualate and Nb43 in GDN (c). d, e, Cryo-EM class averages of inactive (d) and active mGlu5 bound to l-quisqualate and Nb43 (e). f, Particle angular distribution of the final cryo-EM reconstructions of apo (left) and active (right) mGlu5.
Extended Data Fig. 3 Cryo-EM 3D reconstruction workflow.
a, b, Processing workflow of apo mGlu5 in MSP1D1 nanodiscs (a) and apo mGlu5 bound to Nb43 in GDN (b).
Extended Data Fig. 4 Cryo-EM active-state processing and map resolution calculations.
a, Flow chart indicating the processing of cryo-EM data for mGlu5 in complex with Nb43 and l-quisqualate. b, Three-dimensional density maps of mGlu5 (left) and mGlu5 in complex with Nb43 and l-quisqualate (right), coloured by local resolution. c, ‘Gold standard’ FSC curves from RELION indicate that the maps for both mGlu5 and mGlu5 in complex with Nb43 and l-quisqualate reach nominal resolutions of 4.0 Å at FSC = 0.143.
Extended Data Fig. 5 Agreement between the cryo-EM map and the model.
a–h, Representative cryo-EM densities and fitted atomic models for apo (a, b) and active (c–h) mGlu5. The B and C helices of the VFT and the 7TM domain are highlighted in a for the apo mGlu5 structure, and density and model fits are shown in b. Various helices of the VFT, as well as the ligand-binding pocket, CRD and TM6 are highlighted in c, and their cryo-EM density and fitted models are shown in d–h for active mGlu5 bound to Nb43 and l-quisqualate.
Extended Data Fig. 6 Comparison of the intersubunit interfaces in apo and active mGlu5.
Comparison of intersubunit interfaces in apo and active mGlu5 are shown for apo (top) and active (bottom) mGlu5. Contact regions (purple) show residues within 4 Å of the opposite subunit. Notably, apo mGlu5 lacks any interactions beyond the VFT.
Extended Data Fig. 7 Comparison of cryo-EM structures of full-length mGlu5 with crystal structures.
a, The conformation of the VFT and CRD portions of full-length apo mGlu5 obtained using cryo-EM (coloured cartoon) is almost identical to the conformation revealed by the crystal structure of the full-length apo mGlu5 (7TM domain not resolved; grey cartoon). b, Similarly to the apo-state structures, the conformation of the VFT and CRD portions of full-length Nb43-bound, active-state mGlu5 obtained by cryo-EM (coloured cartoon) is almost identical to the conformation revealed by the crystal structure of the ECD bound to l-quisqualate and Nb43. c, d, Despite being bound to a NAM (green) and a PAM (blue), the 7TM domains of in our full-length structures both align almost identically to a crystal structure of a NAM-bound 7TM domain in isolation (c) as well as to each other (d).
Extended Data Fig. 8 Molecular dynamics simulations of mGlu5 ECD-Nb43.
Simulation of the apo form of mGlu5 ECD both with and without Nb43. In the absence of Nb43, the ECD relaxes to an open conformation (right) with considerable separation between the bottom lobes of the VFT. The presence of Nb43 restricts opening of the ECD to an intermediate degree (middle) but does not stabilize the compact resting state (left).
Extended Data Fig. 9 Structural transitions in the 7TM domain upon activation.
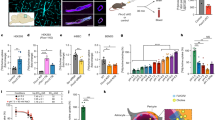
a, b, Activation involves a 20 Å translation of the 7TM domains relative to each other (a) followed by a 20° rotation around TM4 (b). The 7TM domains of the apo state are shown as shades of purple, whereas the active state 7TM domains are shown as shades of blue and teal. c, Model of the I7916.56C mutation on the active-state structure shows its ideal positioning for disulfide formation. d, Western blot analysis of disulfide formation involving I7916.56C in both wild type and the C129A background in the presence and absence of the NAM MPEP. Bars in d (bottom) represent mean ± s.e.m. from 8 independent experiments. Statistics were performed using repeated measures one-way ANOVA followed by Tukey’s multiple comparisons test and indicated P values were adjusted to account for multiple comparisons.
Supplementary information
Supplementary Data
This file contains Figures 1-3 and Tables 1-5. Figure 1 – In-vitro characterization of Nb43, Figure 2 - Constitutive activity of ELC2 mutants by IP1 accumulation, Figure 3 – Surface and total expression levels of receptor mutants compared to wild-type. Table 1 - The effect of L-glu as a competitive ligand and the allosteric ligands Nb43 and CDPPB as enhancers of [3H]-quisqualic acid binding, Table 2 - Saturation binding of [3H]-quisqualic acid in presence of vehicle, or a fixed concentration of Nb43 or CDPPB, Table 3 - Potency and Emax estimates of mGlu5 ligands alone and in combination with low receptor expression in presence of EAAT3,Table 4 - Effect of ECL2 deletion on orthosteric and allosteric receptor activation, Table 5 - Estimation of low mGlu5 expression levels by [3H]-MPEP binding
Video 1
: Activation triggered conformational changes in mGlu5. Map/model fits and morph between apo- and active- mGlu5 upon L-quisqualate binding. Agonist action leads to a compaction of the mGlu5 dimer
Rights and permissions
About this article
Cite this article
Koehl, A., Hu, H., Feng, D. et al. Structural insights into the activation of metabotropic glutamate receptors. Nature 566, 79–84 (2019). https://doi.org/10.1038/s41586-019-0881-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-0881-4
This article is cited by
-
Constitutive activation mechanism of a class C GPCR
Nature Structural & Molecular Biology (2024)
-
Absence of calcium-sensing receptor basal activity due to inter-subunit disulfide bridges
Communications Biology (2024)
-
Stepwise activation of a metabotropic glutamate receptor
Nature (2024)
-
Structure, function and drug discovery of GPCR signaling
Molecular Biomedicine (2023)
-
Steering G protein activation by mGlu heterodimer
Cell Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.