Abstract
Ice sheets are currently ignored in global methane budgets1,2. Although ice sheets have been proposed to contain large reserves of methane that may contribute to a rise in atmospheric methane concentration if released during periods of rapid ice retreat3,4, no data exist on the current methane footprint of ice sheets. Here we find that subglacially produced methane is rapidly driven to the ice margin by the efficient drainage system of a subglacial catchment of the Greenland ice sheet. We report the continuous export of methane-supersaturated waters (CH4(aq)) from the ice-sheet bed during the melt season. Pulses of high CH4(aq) concentration coincide with supraglacially forced subglacial flushing events, confirming a subglacial source and highlighting the influence of melt on methane export. Sustained methane fluxes over the melt season are indicative of subglacial methane reserves that exceed methane export, with an estimated 6.3 tonnes (discharge-weighted mean; range from 2.4 to 11 tonnes) of CH4(aq) transported laterally from the ice-sheet bed. Stable-isotope analyses reveal a microbial origin for methane, probably from a mixture of inorganic and ancient organic carbon buried beneath the ice. We show that subglacial hydrology is crucial for controlling methane fluxes from the ice sheet, with efficient drainage limiting the extent of methane oxidation5 to about 17 per cent of methane exported. Atmospheric evasion is the main methane sink once runoff reaches the ice margin, with estimated diffusive fluxes (4.4 to 28 millimoles of CH4 per square metre per day) rivalling that of major world rivers6. Overall, our results indicate that ice sheets overlie extensive, biologically active methanogenic wetlands and that high rates of methane export to the atmosphere can occur via efficient subglacial drainage pathways. Our findings suggest that such environments have been previously underappreciated and should be considered in Earth’s methane budget.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data used in this article are available from the corresponding author on request. The 16S rRNA gene sequence data are available from the NCBI Sequence Read Archive (https://www.ncbi.nlm.nih.gov/sra) under BioProject PRJNA495593 (BioSamples SAMN10228172-SAMN10228185; SAMN10228190- SAMN10228206).
References
Kirschke, S. et al. Three decades of global methane sources and sinks. Nat. Geosci. 6, 813–823 (2013).
Schaefer, H. et al. A 21st century shift from fossil-fuel to biogenic methane emissions indicated by 13CH4. Science 352, 80–84 (2016).
Wadham, J. L., Tranter, M., Tulaczyk, S. & Sharp, M. Subglacial methanogenesis: a potential climatic amplifier? Global Biogeochem. Cy. 22, GB2021 (2008).
Wadham, J. L. et al. Potential methane reservoirs beneath Antarctica. Nature 488, 633–637 (2012).
Dieser, M. et al. Molecular and biogeochemical evidence for methane cycling beneath the western margin of the Greenland Ice Sheet. ISME J. 8, 2305–2316 (2014).
Stanley, E. H. et al. The ecology of methane in streams and rivers: patterns, controls, and global significance. Ecol. Monogr. 86, 146–171 (2016).
Weitemeyer, K. A. & Buffett, B. A. Accumulation and release of methane from clathrates below the Laurentide and Cordilleran ice sheets. Global Planet. Change 53, 176–187 (2006).
Michaud, A. B. et al. Microbial oxidation as a methane sink beneath the West Antarctic Ice Sheet. Nat. Geosci. 10, 582–586 (2017).
Petrenko, V. V. et al. Minimal geological methane emissions during the Younger Dryas–Preboreal abrupt warming event. Nature 548, 443–446 (2017).
Portnov, A., Vadakkepuliyambatta, S., Mienert, J. & Hubbard, A. Ice-sheet-driven methane storage and release in the Arctic. Nat. Commun. 7, (2016).
Souchez, R., Lemmens, M. & Chappellaz, J. Flow-induced mixing in the GRIP basal ice deduced from the CO2 and CH4 records. Geophys. Res. Lett. 22, 41–44 (1995).
Miteva, V., Teacher, C., Sowers, T. & Brenchley, J. Comparison of the microbial diversity at different depths of the GISP2 Greenland ice core in relationship to deposition climates. Environ. Microbiol. 11, 640–656 (2009).
Christner, B. C., Montross, G. G. & Priscu, J. C. Dissolved gases in frozen basal water from the NGRIP borehole: implications for biogeochemical processes beneath the Greenland Ice Sheet. Polar Biol. 35, 1735–1741 (2012).
Schmidt, M., Linke, P. & Esser, D. Recent development in IR sensor technology for monitoring subsea methane discharge. Mar. Technol. Soc. J. 47, 27–36 (2013).
Cowton, T., Nienow, P., Bartholomew, I., Sole, A. & Mair, D. Rapid erosion beneath the Greenland ice sheet. Geology 40, 343–346 (2012).
Kohler, T. J. et al. Carbon dating reveals a seasonal progression in the source of particulate organic carbon exported from the Greenland Ice Sheet. Geophys. Res. Lett. 44, 6209–6217 (2017).
Stibal, M. et al. Methanogenic potential of Arctic and Antarctic subglacial environments with contrasting organic carbon sources. Glob. Change Biol. 18, 3332–3345 (2012).
Bartholomew, I. et al. Supraglacial forcing of subglacial drainage in the ablation zone of the Greenland ice sheet. Geophys. Res. Lett. 38, L08502 (2011).
Raymond, P. A. et al. Global carbon dioxide emissions from inland waters. Nature 503, 355–359 (2013); erratum 507, 387 (2013).
Chandler, D. M. et al. Evolution of the subglacial drainage system beneath the Greenland Ice Sheet revealed by tracers. Nat. Geosci. 6, 195–198 (2013).
Hall, R. O., Kennedy, T. A. & Rosi-Marshall, E. J. Air–water oxygen exchange in a large whitewater river. Limnol. Oceanogr. Fluids Environ. 2, 1–11 (2012).
Maurice, L., Rawlins, B. G., Farr, G., Bell, R. & Gooddy, D. C. The influence of flow and bed slope on gas transfer in steep streams and their implications for evasion of CO2. J. Geophys. Res. Biogeosci. 122, 2862–2875 (2017).
Walter Anthony, K. M., Anthony, P., Grosse, G. & Chanton, J. Geologic methane seeps along boundaries of Arctic permafrost thaw and melting glaciers. Nat. Geosci. 5, 419–426 (2012).
Telling, J. et al. Rock comminution as a source of hydrogen for subglacial ecosystems. Nat. Geosci. 8, 851–855 (2015); erratum 8, 981 (2015).
Whiticar, M. J. Carbon and hydrogen isotope systematics of bacterial formation and oxidation of methane. Chem. Geol. 161, 291–314 (1999).
Etiope, G. & Sherwood Lollar, B. Abiotic methane on Earth. Rev. Geophys. 51, 276–299 (2013).
Walter, F., Chaput, J. & Lüthi, M. P. Thick sediments beneath Greenland’s ablation zone and their potential role in future ice sheet dynamics. Geology 42, 487–490 (2014).
Wingham, D. J., Siegert, M. J., Shepherd, A. & Muir, A. S. Rapid discharge connects Antarctic subglacial lakes. Nature 440, 1033–1036 (2006).
Beaton, A. D. et al. High-resolution in situ measurement of nitrate in runoff from the Greenland Ice Sheet. Environ. Sci. Technol. 51, 12518–12527 (2017).
Hawkings, J. R. et al. Ice sheets as a significant source of highly reactive nanoparticulate iron to the oceans. Nat. Commun. 5, 3929 (2014).
Ward, J. A. et al. Microbial hydrocarbon gases in the Witwatersrand Basin, South Africa: implications for the deep biosphere. Geochim. Cosmochim. Acta 68, 3239–3250 (2004).
Wiesenburg, D. A. & Guinasso, N. L., Jr. Equilibrium solubilities of methane, carbon monoxide, and hydrogen in water and sea water. J. Chem. Eng. Data 24, 356–360 (1979).
Raymond, P. A. et al. Scaling the gas transfer velocity and hydraulic geometry in streams and small rivers. Limnol. Oceanogr. Fluids Environ. 2, 41–53 (2012).
Wanninkhof, R. Relationship between wind speed and gas exchange over the ocean revisited. Limnol. Oceanogr. Methods 12, 351–362 (2014).
Sherwood Lollar, B., Hirschorn, S. K., Chartrand, M. M. G. & Lacrampe-Couloume, G. An approach for assessing total instrumental uncertainty in compound-specific carbon isotope analysis: implications for environmental remediation studies. Anal. Chem. 79, 3469–3475 (2007).
Davie, M. K. & Buffett, B. A. A numerical model for the formation of gas hydrate below the seafloor. J. Geophys. Res. 106, 497–514 (2001).
Tedesco, M. et al. Evidence and analysis of 2012 Greenland records from spaceborne observations, a regional climate model and reanalysis data. Cryosphere 7, 615–630 (2013).
Palmer, S., Shepherd, A., Nienow, P. & Joughin, I. Seasonal speedup of the Greenland Ice Sheet linked to routing of surface water. Earth Planet. Sci. Lett. 302, 423–428 (2011).
Acknowledgements
We thank all of those who assisted with fieldwork at LG, especially J. Hatton, as well as F. Sgouridis and J. Williams at the LOWTEX laboratory of the University of Bristol. This research is part of the UK NERC-funded DELVE programme (NERC grant NE/I008845/1 to J.L.W.). G.L.-G. was funded by the University of Bristol Scholarship Programme and a FRQNT Scholarship (number 185136). The work was also supported by a Leverhulme research fellowship to J.L.W., a UK NERC grant (NE/J02399X/1) to A.M.A. for DNA analyses, as well as Czech Science Foundation grants (GACR; 15-17346Y and 18-12630S) to M.S. Isotopic analyses were conducted by G. Lacrampe-Couloume at the University of Toronto with support provided by the Natural Sciences and Engineering Research Council of Canada (NSERC) to B.S.L. We also thank the Kangerlussuaq International Science Station, especially R. Møller, for support with field logistics, as well as M. A. Cooper, M. Macdonald and S. Hoffer for comments.
Reviewer information
Nature thanks J. Crawford and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.L.W. and G.L.-G. designed the study. B.S.L. supervised the stable-isotope analyses. S.A. performed the reaction-transport hydrate model calculations. P.F. assisted in the interpretation and analysis of the CONTROS HydroC CH4 raw results. G.L.-G., J.T., E.A.B., A.D.B., M.C.M. and J.R.H. conducted field logistical preparations. J.L.W., J.T. and M.S. led the 2015 Greenland field campaign. G.L.-G., A.D.B., A.J.T., J.T., E.A.B., J.R.H., T.J.K., J.D.Z. and M.S. collected the sensor field data. G.L.-G. and J.D.Z. collected manual water samples in the field. G.L.-G. and A.M.A. analysed molecular data. G.L.-G. performed the data analysis and wrote the manuscript with significant contribution from all co-authors.
Corresponding author
Ethics declarations
Competing interests
P.F. works for the sensor manufacturer Kongsberg Maritime Contros, but the sensor data discussed here were validated by independent measurements. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
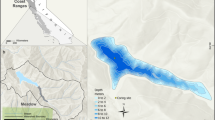
Extended Data Fig. 1 Leverett Glacier and proglacial stream.
a, Leverett Glacier (LG), with catchment boundaries38 outlined in grey; ‘SFJ’ denotes the Kangerlussuaq airport. b, Zoomed image of the LG, with the sampling site and portal marked by dots. c, Sensor deployment site during the early melt season, with the LG visible in the background; the image faces upstream. d, Sensor deployment site in late June; the image faces downstream. Also visible is the HydroC sensor inside a steel cage, during inspection and before redeployment. e, LG portal in late May, while still covered with both glacial and river ice. The photograph was taken one hour before the appearance of the glacial upwelling (see Supplementary Information 2b). The arrow marks the location of the chainsawed hole, shown in the inset (photograph taken on 10 May 2015). f, LG portal in mid-July 2015. Map images from USGS/NASA Landsat.
Extended Data Fig. 2 Comparison of CH4(aq) concentrations measured with the HydroC sensor and from manual samples.
a, CH4(aq) time series. Red points correspond to the HydroC pump power during operation. The continuous line depicts HydroC measurements, with the dashed section corresponding to times when the sensor experienced low pump power and thus a reduced water flow induced by the pump (19 June to 1 July). Open circles correspond to manual samples. The thin grey-shaded area around the CH4(aq) time series corresponds to the uncertainty of the HydroC measurements (about 3%). The uncertainty on manual measurements, indicated by the error bars, reflects the error on vial internal pressures and volumes (119 ± 0.76 ml, where the uncertainty represents standard deviation; internal pressures are derived from volumes—see Methods for details). b, Regression plot between the HydroC and manual sample measurements. Only manual samples taken during times when the HydroC pump power was above about 7 W were considered for the regression (black circles, black line); grey circles correspond to samples taken during times of lower pump power. Horizontal error bars reflect errors on manual measurements; vertical error bars are smaller than the size of the markers. The orange dashed line depicts a hypothetical 1:1 relationship between the sensor and manual measurements.
Extended Data Fig. 3 Combination plot of δ13C-CO2 and δ13C-CH4 of LG runoff.
Points denote δ13C CO2-CH4 values for LG manual samples. Methanogenesis and microbial oxidation classification zones are derived and adapted from ref. 25.
Extended Data Fig. 4 LG 16S rRNA gene sequences related to methanotrophic and methanogenic clades.
a, Relative abundance of the dominant operational taxonomical units related to bacterial methanotrophs (OTU00009) and archaeal methanogens. The box mid-lines represent medians; the IQR is represented by the lower and upper box boundaries and denote the 25th and 75th percentiles, respectively; whiskers indicate confidence intervals of 1.5 times the IQR, and points are outliers. b, c, Maximum-likelihood trees of 16S rRNA sequences related to methanotrophs rooted with the sequences of Clostridium frigoriphilum (b) and methanogens rooted with the sequences of Acidibilus sulfurireducens and Caldisphaera draconis (c).
Extended Data Fig. 5 Relationship between rates of subglacial methanogenesis, sediment thickness and observed annual CH4 flux at LG.
Each panel corresponds to the different yearly lateral CH4(aq) flux estimates measured in 2015 (see Fig. 2). Each line type corresponds to the sediment thickness required under different catchment area conditions: 100% (solid line), 50% (long-dashed line) or 10% (short-dashed line) of the subglacial catchment to contribute to the observed CH4 flux. Any point on a line corresponds to the methanogenesis rate and subglacial sediment thickness required to generate the observed lateral CH4 flux. The four points on each line correspond to known methanogenic rates recorded from different subglacial habitats17.
Extended Data Fig. 6 Summary of model conditions required for subglacial methane hydrate formation.
a–d, Model results for a fixed methanogenic depth (100 m) but varying methanogenic rates (R2 to R10; that is, 2 × 10−15 to 10 × 10−15 grams CH4 per gram of sediment per second). e–h, Outputs of model runs under a fixed methanogenic rate (5 × 10−15 grams CH4 per gram of sediment per second) but varying methanogenic depths (20–100 m). a, b, e, f, Vertical profiles of methane solubility, dissolved methane and methane hydrates; methane concentrations are normalized to equilibrium concentration. c, g, Time required for methane hydrate formation under the modelled conditions. d, h, Diffusive CH4 flux at the sediment–ice interface under methane hydrate conditions assuming three different catchment cover areas for methane hydrates (that is, 10%, 50% and 100% of the LG catchment), compared to the three lateral flux scenarios (a, b, d; Fig. 2); see Supplementary Information 2f.
Extended Data Fig. 7 Extended time series of geochemical measurements from the LG proglacial river.
a–d, The EC, pH and SSC time series include those depicted in Fig. 1 but extend to measurements before and after the methane record. It should be noted that the CH4(aq) data in c are the HydroC partial pressure (in microatmospheres) measurements. Left and right vertical axes correspond to black and orange datasets, respectively.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, a Supplementary Discussion and Supplementary References.
Rights and permissions
About this article
Cite this article
Lamarche-Gagnon, G., Wadham, J.L., Sherwood Lollar, B. et al. Greenland melt drives continuous export of methane from the ice-sheet bed. Nature 565, 73–77 (2019). https://doi.org/10.1038/s41586-018-0800-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0800-0
This article is cited by
-
Groundwater springs formed during glacial retreat are a large source of methane in the high Arctic
Nature Geoscience (2023)
-
Methylotrophic Communities Associated with a Greenland Ice Sheet Methane Release Hotspot
Microbial Ecology (2023)
-
Impact of interannual and multidecadal trends on methane-climate feedbacks and sensitivity
Nature Communications (2022)
-
Storage and export of microbial biomass across the western Greenland Ice Sheet
Nature Communications (2021)
-
Active methanogenesis during the melting of Marinoan snowball Earth
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.