Abstract
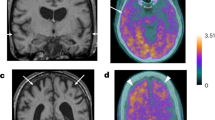
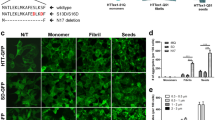
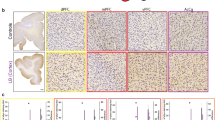
We previously reported1 the presence of amyloid-β protein (Aβ) deposits in individuals with Creutzfeldt–Jakob disease (CJD) who had been treated during childhood with human cadaveric pituitary-derived growth hormone (c-hGH) contaminated with prions. The marked deposition of parenchymal and vascular Aβ in these relatively young individuals with treatment-induced (iatrogenic) CJD (iCJD), in contrast to other prion-disease patients and population controls, allied with the ability of Alzheimer’s disease brain homogenates to seed Aβ deposition in laboratory animals, led us to argue that the implicated c-hGH batches might have been contaminated with Aβ seeds as well as with prions. However, this was necessarily an association, and not an experimental, study in humans and causality could not be concluded. Given the public health importance of our hypothesis, we proceeded to identify and biochemically analyse archived vials of c-hGH. Here we show that certain c-hGH batches to which patients with iCJD and Aβ pathology were exposed have substantial levels of Aβ40, Aβ42 and tau proteins, and that this material can seed the formation of Aβ plaques and cerebral Aβ−amyloid angiopathy in intracerebrally inoculated mice expressing a mutant, humanized amyloid precursor protein. These results confirm the presence of Aβ seeds in archived c-hGH vials and are consistent with the hypothesized iatrogenic human transmission of Aβ pathology. This experimental confirmation has implications for both the prevention and the treatment of Alzheimer’s disease, and should prompt a review of the risk of iatrogenic transmission of Aβ seeds by medical and surgical procedures long recognized to pose a risk of accidental prion transmission2,3.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jaunmuktane, Z. et al. Evidence for human transmission of amyloid-β pathology and cerebral amyloid angiopathy. Nature 525, 247–250 (2015); erratum 526, 595 (2015).
Collinge, J. Mammalian prions and their wider relevance in neurodegenerative diseases. Nature 539, 217–226 (2016).
Walsh, D. M. & Selkoe, D. J. A critical appraisal of the pathogenic protein spread hypothesis of neurodegeneration. Nat. Rev. Neurosci. 17, 251–260 (2016).
Jucker, M. & Walker, L. C. Self-propagation of pathogenic protein aggregates in neurodegenerative diseases. Nature 501, 45–51 (2013).
Brown, P. et al. Iatrogenic Creutzfeldt–Jakob disease at the millennium. Neurology 55, 1075–1081 (2000).
Collinge, J. et al. Kuru in the 21st century—an acquired human prion disease with very long incubation periods. Lancet 367, 2068–2074 (2006).
Swerdlow, A. J., Higgins, C. D., Adlard, P., Jones, M. E. & Preece, M. A. Creutzfeldt–Jakob disease in United Kingdom patients treated with human pituitary growth hormone. Neurology 61, 783–791 (2003).
Charidimou, A. et al. Emerging concepts in sporadic cerebral amyloid angiopathy. Brain 140, 1829–1850 (2017).
Biffi, A. & Greenberg, S. M. Cerebral amyloid angiopathy: a systematic review. J. Clin. Neurol. 7, 1–9 (2011).
Revesz, T. et al. Cerebral amyloid angiopathies: a pathologic, biochemical, and genetic view. J. Neuropathol. Exp. Neurol. 62, 885–898 (2003).
Levy, E. et al. Mutation of the Alzheimer’s disease amyloid gene in hereditary cerebral hemorrhage, Dutch type. Science 248, 1124–1126 (1990).
Hendriks, L. et al. Presenile dementia and cerebral haemorrhage linked to a mutation at codon 692 of the β-amyloid precursor protein gene. Nat. Genet. 1, 218–221 (1992).
Selkoe, D. J. & Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 8, 595–608 (2016).
Feeney, C. et al. Seeds of neuroendocrine doubt. Nature 535, E1–E2 (2016).
Adams, H. H. H., A Swanson, S., Hofman, A. & Ikram, M. A. Amyloid-β transmission or unexamined bias? Nature 537, E7–E9 (2016).
Collinge, J., Jaunmuktane, Z., Mead, S., Rudge, P. & Brandner, S. Collinge et al. reply. Nature 537, E7–E9 (2016).
Collinge, J., Jaunmuktane, Z., Mead, S., Rudge, P. & Brandner, S. Collinge et al. reply. Nature 535, E2–E3 (2016).
Rudge, P. et al. Iatrogenic CJD due to pituitary-derived growth hormone with genetically determined incubation times of up to 40 years. Brain 138, 3386–3399 (2015).
Milner, R. D. Human growth hormone (UK). Arch. Dis. Child. 54, 733–734 (1979).
Nilsson, P., Saito, T. & Saido, T. C. New mouse model of Alzheimer’s. ACS Chem. Neurosci. 5, 499–502 (2014).
Saito, T. et al. Single App knock-in mouse models of Alzheimer’s disease. Nat. Neurosci. 17, 661–663 (2014).
Eisele, Y. S. et al. Peripherally applied Aβ-containing inoculates induce cerebral β-amyloidosis. Science 330, 980–982 (2010).
Frontzek, K., Lutz, M. I., Aguzzi, A., Kovacs, G. G. & Budka, H. Amyloid-β pathology and cerebral amyloid angiopathy are frequent in iatrogenic Creutzfeldt–Jakob disease after dural grafting. Swiss Med. Wkly 146, w14287 (2016).
Kovacs, G. G. et al. Dura mater is a potential source of Aβ seeds. Acta Neuropathol. 131, 911–923 (2016).
Hamaguchi, T. et al. Significant association of cadaveric dura mater grafting with subpial Aβ deposition and meningeal amyloid angiopathy. Acta Neuropathol. 132, 313–315 (2016).
Ritchie, D. L. et al. Amyloid-β accumulation in the CNS in human growth hormone recipients in the UK. Acta Neuropathol. 134, 221–240 (2017).
Duyckaerts, C. et al. Neuropathology of iatrogenic Creutzfeldt–Jakob disease and immunoassay of French cadaver-sourced growth hormone batches suggest possible transmission of tauopathy and long incubation periods for the transmission of Abeta pathology. Acta Neuropathol. 135, 201–212 (2018).
Jaunmuktane, Z. et al. Evidence of amyloid-β cerebral amyloid angiopathy transmission through neurosurgery. Acta Neuropathol. 135, 671–679 (2018).
Daviglus, M. L. et al. Risk factors and preventive interventions for Alzheimer disease: state of the science. Arch. Neurol. 68, 1185–1190 (2011).
O’Meara, E. S. et al. Alzheimer’s disease and history of blood transfusion by apolipoprotein-E genotype. Neuroepidemiology 16, 86–93 (1997).
Mably, A. J. et al. Anti-Aβ antibodies incapable of reducing cerebral Aβ oligomers fail to attenuate spatial reference memory deficits in J20 mice. Neurobiol. Dis. 82, 372–384 (2015).
Kanmert, D. et al. C-terminally truncated forms of tau, but not full-length tau or its C-terminal fragments, are released from neurons independently of cell death. J. Neurosci. 35, 10851–10865 (2015).
van der Laak, J. A., Pahlplatz, M. M., Hanselaar, A. G. & de Wilde, P. C. Hue-saturation-density (HSD) model for stain recognition in digital images from transmitted light microscopy. Cytometry 39, 275–284 (2000).
Acknowledgements
This work was funded by the UK Medical Research Council (MRC); the National Institute of Health Research (NIHR) University College London Hospitals (UCLH)/University College London (UCL) Biomedical Research Centre; the Leonard Wolfson Experimental Neurology Centre; and a grant to D.M.W. from the National Institute on Aging (AG046275). We thank the Queen Square Brain Bank for Neurological Disorders (supported by the Reta Lila Weston Trust for Medical Research, the Progressive Supranuclear Palsy (Europe) Association and the MRC) at the UCL Institute of Neurology, University College London; and the Oxford Brain Bank (supported by the MRC, the NIHR Oxford Biomedical Research Centre and the Brains for Dementia Research programme, jointly funded by Alzheimer’s Research UK and Alzheimer’s Society) for providing the UK human brain tissue samples. We thank M. Ellis for image analysis; Z. Jaunmuktane for advice on CAA scoring; and G. Graham, C. Fitzhugh, R. Labesse-Garbal and other staff of the MRC Prion Unit Biological Services facility for animal inoculation, observation and care. We thank M. Farmer and E. Quarterman for technical assistance; O. Avwenagha and J. Wadsworth for assistance in selecting and processing tissue samples; and E. Noble for assistance with assay development. We thank P. Adlard for help in identifying growth-hormone batches for this study and M. Sutton for providing c-hGH vials from archived stores at Public Health England Porton Down. Antibodies m266, 2G3 and 21F12 were gifts from P. Seubert and D. Schenk, Elan Pharmaceuticals.
Author information
Authors and Affiliations
Contributions
S.A.P. and M.A.F. coordinated animal experiments and performed data analysis. J.L., T.N. and S.B. performed neuropathological analysis. D.X.T., Z.C., D.M. and D.M.W. performed and analysed biochemical assays. T.Saito and T.Saido provided NL-F mice. P.R. coordinated the identification and sourcing of relevant archival c-hGH batches. J.C. oversaw the study and drafted the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
J.C. is a shareholder and director of D-Gen Limited, an academic spin-out company working in the field of prion-disease diagnosis, decontamination and therapeutics.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Time course of CAA and Aβ deposition in control- and AD-brain-inoculated AppNL-F/NL-F mice.
Mice were inoculated with either control-brain homogenates (a–c, g–i, m–o, s–u) or AD-brain homogenates (d–f, j–l, p–r, v–x) and culled at the stated times. Aβ deposition was assessed on sagittal sections (a, d, g, j, m, p, s, v). CAA (b, e, h, k, n, q, t, w) and cerebellar deposition (c, f, i, l, o, r, u, x) were evident only in AD-brain-inoculated animals. Boxes denote areas magnified to the right. Scale bars represent 1.4 mm for whole sections (a, d, g, j, m, p, s, v), 25 µm for CAA (b, e, h, k, n, q, t, w), and 50 µm for the cerebellar region (c, f, i, l, o, r, u, x).
Extended Data Fig. 2 Aβ plaques and CAA in AppNL-F/NL-F mice following inoculation with c-hGH preparations.
AppNL-F/NL-F mice were inoculated with c-hGH batch HWP 42 (a, c–f, k–n) or HWP 51 (b, g–j, o–r) and culled after 240 days. Aβ deposition was assessed on sagittal sections (a, b). Black and red boxes denote areas magnified to better show cerebellar Aβ deposits (c–j) and CAA (k–r), respectively, in the middle and lower panels. Scale bars represent 1.1 mm for whole sections (a, b) and 50 µm for the cerebellar region and CAA (c–r).
Supplementary information
Source data
Rights and permissions
About this article
Cite this article
Purro, S.A., Farrow, M.A., Linehan, J. et al. Transmission of amyloid-β protein pathology from cadaveric pituitary growth hormone. Nature 564, 415–419 (2018). https://doi.org/10.1038/s41586-018-0790-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0790-y
This article is cited by
-
Signs of ‘transmissible’ Alzheimer’s seen in people who received growth hormone
Nature (2024)
-
Evidence for iatrogenic transmission of Alzheimer’s disease
Nature Medicine (2024)
-
Iatrogenic Alzheimer’s disease in recipients of cadaveric pituitary-derived growth hormone
Nature Medicine (2024)
-
Spontaneous intracerebral haemorrhage associated with early-onset cerebral amyloid angiopathy and Alzheimer’s disease neuropathological changes five decades after cadaveric dura mater graft
Acta Neuropathologica Communications (2023)
-
Opinion: more mouse models and more translation needed for ALS
Molecular Neurodegeneration (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.