Abstract
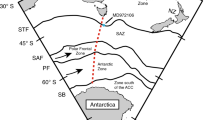
The cause of changes in atmospheric carbon dioxide (CO2) during the recent ice ages is yet to be fully explained. Most mechanisms for glacial–interglacial CO2 change have centred on carbon exchange with the deep ocean, owing to its large size and relatively rapid exchange with the atmosphere1. The Southern Ocean is thought to have a key role in this exchange, as much of the deep ocean is ventilated to the atmosphere in this region2. However, it is difficult to reconstruct changes in deep Southern Ocean carbon storage, so few direct tests of this hypothesis have been carried out. Here we present deep-sea coral boron isotope data that track the pH—and thus the CO2 chemistry—of the deep Southern Ocean over the past forty thousand years. At sites closest to the Antarctic continental margin, and most influenced by the deep southern waters that form the ocean’s lower overturning cell, we find a close relationship between ocean pH and atmospheric CO2: during intervals of low CO2, ocean pH is low, reflecting enhanced ocean carbon storage; and during intervals of rising CO2, ocean pH rises, reflecting loss of carbon from the ocean to the atmosphere. Correspondingly, at shallower sites we find rapid (millennial- to centennial-scale) decreases in pH during abrupt increases in CO2, reflecting the rapid transfer of carbon from the deep ocean to the upper ocean and atmosphere. Our findings confirm the importance of the deep Southern Ocean in ice-age CO2 change, and show that deep-ocean CO2 release can occur as a dynamic feedback to rapid climate change on centennial timescales.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data produced in this study are available in Extended Data Tables and will also be made available at the NOAA (https://www.ncdc.noaa.gov/paleo/study/25230) and Pangaea data repositories.
References
Broecker, W. S. Glacial to interglacial changes in ocean chemistry. Prog. Oceanogr. 11, 151–197 (1982).
Sarmiento, J. L. & Toggweiler, J. R. A new model for the role of the oceans in determining atmospheric pCO2. Nature 308, 621–624 (1984).
Sigman, D. M., Hain, M. P. & Haug, G. H. The polar ocean and glacial cycles in atmospheric CO2 concentration. Nature 466, 47–55 (2010).
Burke, A. & Robinson, L. F. The Southern Ocean’s role in carbon exchange during the last deglaciation. Science 335, 557–561 (2012).
Roberts, J. et al. Evolution of South Atlantic density and chemical stratification across the last deglaciation. Proc. Natl Acad. Sci. USA 113, 514–519 (2016).
Martinez-Garcia, A. et al. Iron fertilization of the Subantarctic Ocean during the last ice age. Science 343, 1347–1350 (2014).
Yu, J. et al. Deep South Atlantic carbonate chemistry and increased interocean deep water exchange during last deglaciation. Quat. Sci. Rev. 90, 80–89 (2014).
Rae, J. W. B. et al. Deep water formation in the North Pacific and deglacial CO2 rise. Paleoceanography 29, 645–667 (2014).
Yu, J. et al. Responses of the deep ocean carbonate system to carbon reorganization during the last glacial–interglacial cycle. Quat. Sci. Rev. 76, 39–52 (2013).
Rickaby, R. E. M., Elderfield, H., Roberts, N., Hillenbrand, C. D. & Mackensen, A. Evidence for elevated alkalinity in the glacial Southern Ocean. Paleoceanography 25, PA1209 (2010).
Ferrari, R. et al. Antarctic sea ice control on ocean circulation in present and glacial climates. Proc. Natl Acad. Sci. USA 111, 8753–8758 (2014).
Marcott, S. A. et al. Centennial-scale changes in the global carbon cycle during the last deglaciation. Nature 514, 616–619 (2014).
Adkins, J. F., McIntyre, K. & Schrag, D. P. The salinity, temperature, and δ18O of the glacial deep ocean. Science 298, 1769–1773 (2002).
Burke, A., Stewart, A. L., Adkins, J. F. & Ferrari, R. The glacial mid-depth radiocarbon bulge and its implications for the overturning circulation. Paleoceanography 30, 1021–1039 (2015).
Charles, C. D. et al. Millennial scale evolution of the Southern Ocean chemical divide. Quat. Sci. Rev. 29, 399–409 (2010).
Jaccard, S. L., Galbraith, E. D., Martinez-Garcia, A. & Anderson, R. F. Covariation of deep Southern Ocean oxygenation and atmospheric CO2 through the last ice age. Nature 530, 207–210 (2016).
Stephens, B. B. & Keeling, R. F. The influence of Antarctic sea ice on glacial-interglacial CO2 variations. Nature 404, 171–174 (2000).
Wolff, E. W. et al. Southern Ocean sea-ice extent, productivity and iron flux over the past eight glacial cycles. Nature 440, 491–496 (2006).
Abram, N. J., Wolff, E. W. & Curran, M. A. J. A review of sea ice proxy information from polar ice cores. Quat. Sci. Rev. 79, 168–183 (2013).
Galbraith, E. & de Lavergne, C. Response of a comprehensive climate model to a broad range of external forcings: relevance for deep ocean ventilation and the development of late Cenozoic ice ages. Clim. Dyn. https://doi.org/10.1007/s00382-018-4157-8 (2018).
Ahn, J. & Brook, E. J. Atmospheric CO2 and climate on millennial time scales during the last glacial period. Science 322, 83–85 (2008).
Stocker, T. F. The seesaw effect. Science 282, 61–62 (1998).
Anderson, R. F. et al. Wind-driven upwelling in the Southern Ocean and the deglacial rise in atmospheric CO2. Science 323, 1443–1448 (2009).
Abernathey, R. & Ferreira, D. Southern Ocean isopycnal mixing and ventilation changes driven by winds. Geophys. Res. Lett. 42, 10,357–10,365 (2015).
Chen, T. et al. Synchronous centennial abrupt events in the ocean and atmosphere during the last deglaciation. Science 349, 1537–1541 (2015).
Martínez-Botí, M. A. et al. Boron isotope evidence for oceanic carbon dioxide leakage during the last deglaciation. Nature 518, 219–222 (2015).
Broecker, W. S., Bond, G., Klas, M., Bonani, G. & Wolfli, W. A salt oscillator in the glacial Atlantic? 1. The concept. Paleoceanography 5, 469–477 (1990).
Members, W. D. P. et al. Precise interpolar phasing of abrupt climate change during the last ice age. Nature 520, 661–665 (2015).
Markle, B. R. et al. Global atmospheric teleconnections during Dansgaard-Oeschger events. Nat. Geosci. 10, 36–40 (2017).
Köhler, P., Knorr, G. & Bard, E. Permafrost thawing as a possible source of abrupt carbon release at the onset of the Bølling/Allerød. Nat. Commun. 5, 5520 (2014).
Galbraith, E. D. & Eggleston, S. A lower limit to atmospheric CO2 concentrations over the past 800,000 years. Nat. Geosci. 10, 295–298 (2017).
Bereiter, B., Shackleton, S., Baggenstos, D., Kawamura, K. & Severinghaus, J. Mean global ocean temperatures during the last glacial transition. Nature 553, 39–44 (2018).
Keeling, R. F. & Stephens, B. B. Antarctic sea ice and the control of Pleistocene climate instability. Paleoceanography 16, 112–131 (2001).
Key, R. M. et al. Global Ocean Data Analysis Project, Version 2 (GLODAPv2). http://doi.org/10.3334/CDIAC/OTG.NDP093_GLODAPv2 (2015).
Olsen, A. et al. The Global Ocean Data Analysis Project version 2 (GLODAPv2)—an internally consistent data product for the world ocean. Earth Syst. Sci. Data 8, 297–323 (2016).
Bereiter, B. et al. Revision of the EPICA Dome C CO2 record from 800 to 600 kyr before present. Geophys. Res. Lett. 42, 542–549 (2015).
Burke, A. et al. Reconnaissance dating: A new radiocarbon method applied to assessing the temporal distribution of Southern Ocean deep-sea corals. Deep Sea Res. Part I Oceanogr. Res. Pap. 57, 1510–1520 (2010).
Margolin, A. R. et al. Temporal and spatial distributions of cold-water corals in the Drake Passage: Insights from the last 35,000 years. Deep Sea Res. Part II Top. Stud. Oceanogr. 99, 237–248 (2014).
Spooner, P. T., Chen, T., Robinson, L. F. & Coath, C. Rapid uranium-series age screening of carbonates by laser ablation mass spectrometry. Quat. Geochronol. 31, 28–39 (2016).
Sinclair, D. J., Kinsley, L. P. & McCulloch, M. T. High resolution analysis of trace elements in corals by laser ablation ICP-MS. Geochim. Cosmochim. Acta 62, 1889–1901 (1998).
Robinson, L. F. et al. Primary U distribution in scleractinian corals and its implications for U series dating. Geochem. Geophys. Geosyst. 7, Q05022 (2006).
Gagnon, A. C., Adkins, J. F., Fernandez, D. P. & Robinson, L. F. Sr/Ca and Mg/Ca vital effects correlated with skeletal architecture in a scleractinian deep-sea coral and the role of Rayleigh fractionation. Earth Planet. Sci. Lett. 261, 280–295 (2007).
Rollion-Bard, C., Chaussidon, M. & France-Lanord, C. Biological control of internal pH in scleractinian corals: Implications on paleo-pH and paleo-temperature reconstructions. C. R. Geosci. 343, 397–405 (2011).
Stewart, J. A., Anagnostou, E. & Foster, G. L. An improved boron isotope pH proxy calibration for the deep-sea coral Desmophyllum dianthus through sub-sampling of fibrous aragonite. Chem. Geol. 447, 148–160 (2016).
Boyle, E. A. Cadmium, zinc, copper, and barium in foraminifera tests. Earth Planet. Sci. Lett. 53, 11–35 (1981).
Barker, S., Greaves, M. & Elderfield, H. A study of cleaning procedures used for foraminiferal Mg/Ca paleothermometry. Geochem. Geophys. Geosyst. 4, 8407 (2003).
Rae, J. W. B., Foster, G. L., Schmidt, D. N. & Elliott, T. Boron isotopes and B/Ca in benthic foraminifera: Proxies for the deep ocean carbonate system. Earth Planet. Sci. Lett. 302, 403–413 (2011).
Foster, G. L. et al. Interlaboratory comparison of boron isotope analyses of boric acid, seawater and marine CaCO3 by MC-ICPMS and NTIMS. Chem. Geol. 358, 1–14 (2013).
Kiss, E. Ion-exchange separation and spectrophotometric determination of boron in geological materials. Anal. Chim. Acta 211, 243–256 (1988).
Lemarchand, D., Gaillardet, J., Göpel, C. & Manhès, G. An optimized procedure for boron separation and mass spectrometry analysis for river samples. Chem. Geol. 182, 323–334 (2002).
Foster, G. L. Seawater pH, pCO2 and [CO3 =] variations in the Caribbean Sea over the last 130 kyr: A boron isotope and B/Ca study of planktic foraminifera. Earth Planet. Sci. Lett. 271, 254–266 (2008).
Al-Ammar, A. S., Gupta, R. K. & Barnes, R. M. Elimination of boron memory effect in inductively coupled plasma-mass spectrometry by ammonia gas injection into the spray chamber during analysis. Spectrochim. Acta B At. Spectrosc. 55, 629–635 (2000).
Misra, S., Owen, R., Kerr, J. & Greaves, M. Determination of δ11B by HR-ICP-MS from mass limited samples: application to natural carbonates and water samples. Geochim. Cosmochim. Acta 140, 531–552 (2014).
Rae, J. W. B. in Boron Isotopes 107–143 (Springer, 2018).
McCulloch, M. T. et al. in Boron Isotopes 145–162 (Springer, 2018).
Anagnostou, E., Huang, K. F., You, C. F., Sikes, E. L. & Sherrell, R. M. Evaluation of boron isotope ratio as a pH proxy in the deep sea coral Desmophyllum dianthus: evidence of physiological pH adjustment. Earth Planet. Sci. Lett. 349-350, 251–260 (2012).
Trotter, J. et al. Quantifying the pH ‘vital effect’ in the temperate zooxanthellate coral Cladocora caespitosa: validation of the boron seawater pH proxy. Earth Planet. Sci. Lett. 303, 163–173 (2011).
Venn, A. A. et al. Impact of seawater acidification on pH at the tissue-skeleton interface and calcification in reef corals. Proc. Natl Acad. Sci. USA 110, 1634–1639 (2013).
Allison, N., Cohen, I., Finch, A. A., Erez, J. & Tudhope, A. W. Corals concentrate dissolved inorganic carbon to facilitate calcification. Nat. Commun. 5, 5741 (2014).
McCulloch, M. et al. Resilience of cold-water scleractinian corals to ocean acidification: Boron isotopic systematics of pH and saturation state up-regulation. Geochim. Cosmochim. Acta 87, 21–34 (2012).
Gagnon, A. C., Adkins, J. F., Erez, J. & Eiler, J. M. Sr/Ca sensitivity to aragonite saturation state in cultured subsamples from a single colony of coral: mechanism of biomineralization during ocean acidification. Geochim. Cosmochim. Acta 105, 240–254 (2013).
Wang, X. T. et al. Deep-sea coral evidence for lower Southern Ocean surface nitrate concentrations during the last ice age. Proc. Natl Acad. Sci. USA 114, 3352–3357 (2017).
Acknowledgements
This work was supported by NERC Standard Grant NE/N003861/1 to J.W.B.R. and L.F.R., an NOAA Climate and Global Change VSP Fellowship to J.W.B.R, NERC Standard Grant NE/M004619/1 to A.B. and J.W.B.R., a NERC Strategic Environmental Science Capital Grant to A.B. and J.W.B.R., Marie Curie Career Integration Grant CIG14-631752 to A.B., an ERC consolidator grant to L.F.R., NSF grant OCE-1503129 to J.F.A., and NERC studentships to B.T. and E.L.
Reviewer information
Nature thanks C. Buizert and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.W.B.R., A.B., and L.F.R. designed the study. A.B., L.F.R., T.C., and T.L. collected and uranium–thorium dated the coral samples. J.W.B.R., B.T., E.L., C.C., R.G., J.A.S., and D.C.N. made boron isotope analyses. J.W.B.R., A.B., L.F.R., and J.F.A. developed the interpretation and all authors contributed to the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Deep Southern Ocean CO2 chemistry and atmospheric CO2 over the last 40,000 years.
Green triangles and blue diamonds show lower and upper cell deep-sea coral δ11B data, respectively. Individual subsamples are shown as small open symbols and mean values as larger filled symbols. Error bars on individual subsamples are equivalent to 2 s.d. analytical reproducibility and error bars on mean coral values represent 2 s.e. uncertainty on the mean of replicate subsamples (see Methods). Approximate pH values are given based on coral δ11B using the calibration in Extended Data Fig. 5, but uncertainty on this calibration is large (inset error bar), given the paucity of modern deep-sea coral data from low pH waters. Instead we focus on the δ11B values themselves, which provide a proxy of carbonate chemistry in their own right54. Synchronized ice core CO2 data36 are shown in grey symbols: circles from Dome C, dots from WAIS, stars from Taylor Dome, triangles from TALDICE, pluses from EDML, diamonds from Byrd, and squares from Siple Dome. Grey bands highlight intervals of CO2 rise.
Extended Data Fig. 2 Deep Southern Ocean CO2 chemistry and atmospheric CO2 over the last 40,000 years, highlighting the depths of upper cell corals.
Symbols and data are as plotted in Extended Data Fig. 1, but with the addition of the lower panel and annotations showing the depth in metres of each upper cell coral sample. No systematic offset is seen between samples from different depths. The only signal that occurs simultaneous with a change in depth is the decrease at ~11.5 kyr ago, but the jump back up to higher δ11B values following this event occurs without a change in depth, giving confidence that the excursion is not a depth-related signal. Furthermore the large excursion at ~14.7 kyr ago occurs without a significant change in depth. Note that all of the lower cell corals come from within 17 m water depth of each other.
Extended Data Fig. 3 Records of Southern Ocean biogeochemistry and CO2 over the last 40,000 years.
Data are plotted as in Fig. 2, but with opal flux23, a proxy for upwelling, deep sea coral δ15N62, a proxy for surface ocean nitrate consumption, and authigenic uranium concentrations16, a proxy for bottom water redox. The opal flux and authigenic uranium records combine two sediment cores: TN057-13-4PC in the younger part of the record (pluses) and TN057-14PC in the older part of the record (crosses). The opal flux records from each core are shown on separate scales. The coral δ15N data are grouped into samples from the Antarctic zone (AZ, blue) and Subantarctic zone (SAZ, red); smoothed fits to the data are shown, as provided in the original study62. Intervals of low CO2 during the last ice age are associated with low upwelling, an efficient biological pump, low oxygen water rich in respired carbon, and low-pH carbon-rich water in the deep Southern Ocean.
Extended Data Fig. 4 Deglacial records of Southern Ocean CO2 chemistry and opal fluxes, and climate over Antarctica and Greenland.
Data are plotted as in Figs. 2, 3, but with opal flux23, a proxy for upwelling, surface ocean-atmosphere CO2 difference, based on δ11B in planktic foraminifera26, and radiocarbon data4,25 from corals within these sample groupings, shown as 14C age offsets compared to the contemporaneous atmosphere. Intervals of rising CO2 in the atmosphere are associated with input of waters rich in CO2 and nutrients to the upper reaches of the Southern Ocean. Radiocarbon ages reflect the competing influences of upwelling of 14C-depleted waters and improved ventilation over the deglaciation.
Extended Data Fig. 5 Boron isotope calibration for modern D. dianthus.
Data are from open ocean sites44,56,60, with two additional recent (<1,650 years ago) samples from the Southern Ocean from this study. Water column δ11B of borate (B(OH)4−) values are as previously published or are calculated from carbonate chemistry data from nearby GLODAPv2 sites for the new samples, as described47,54. Note that the sensitivity of δ11B in carbonates to pH is based on the pH sensitivity of δ11B of borate. pH itself is not easily shown on a plot like this, as the relationship between δ11B of borate and pH is also somewhat influenced by water temperature, salinity, and depth54. A power law function was fitted to the data using Matlab’s curve fitting toolbox (solid line: δ11BCoral = −1.8214 × δ11BB(OH)4−−12.22 + 27.03; R2 = 0.57). Dashed lines show the 95% confidence intervals and give a measure of calibration uncertainty as shown in the error bar in Extended Data Fig. 1, although data from a given site may be able to record relative changes in pH more sensitively, as seen in many paleo-proxies.
Extended Data Fig. 6 Replicate subsamples from a D. dianthus septum.
To test for the potential influence of microstructural variability in composition, a coral septum was divided into four areas, which were then split into chunks of approximately 0.2, 0.8, 3.2 and 11 mg. These were then individually crushed, cleaned, and analysed. This sample treatment was designed to preserve heterogeneity between subsamples, although note that the clustering of subsamples of a given size from a certain area of the coral may lead to that group recording a slightly different signal (as seen in the 3.2 mg group). The lines in the middle panel show the mean and 2 s.d., excluding one outlier in the 11 mg group. δ11B is correlated with Mg/Ca and U/Ca, showing the influence of internal variability in coral composition.
Supplementary information
Supplementary Data
This file contains a zip folder which includes source data as used in Extended Data Tables 1-3 in txt format and an Excel workbook with all source data files combined.
Rights and permissions
About this article
Cite this article
Rae, J.W.B., Burke, A., Robinson, L.F. et al. CO2 storage and release in the deep Southern Ocean on millennial to centennial timescales. Nature 562, 569–573 (2018). https://doi.org/10.1038/s41586-018-0614-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0614-0
Keywords
This article is cited by
-
Southern Ocean glacial conditions and their influence on deglacial events
Nature Reviews Earth & Environment (2023)
-
Arctic and Antarctic forcing of ocean interior warming during the last deglaciation
Scientific Reports (2023)
-
Millennial atmospheric CO2 changes linked to ocean ventilation modes over past 150,000 years
Nature Geoscience (2023)
-
Variable ventilation ages in the equatorial Indian Ocean thermocline during the LGM
Scientific Reports (2023)
-
Regulation of CO2 by the sea in areas around Latin America in a context of climate change
Environmental Monitoring and Assessment (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.