Abstract
Prolonged exposure to microbial products such as lipopolysaccharide can induce a form of innate immune memory that blunts subsequent responses to unrelated pathogens, known as lipopolysaccharide tolerance. Sepsis is a dysregulated systemic immune response to disseminated infection that has a high mortality rate. In some patients, sepsis results in a period of immunosuppression (known as ‘immunoparalysis’)1 characterized by reduced inflammatory cytokine output2, increased secondary infection3 and an increased risk of organ failure and mortality4. Lipopolysaccharide tolerance recapitulates several key features of sepsis-associated immunosuppression5. Although various epigenetic changes have previously been observed in tolerized macrophages6,7,8, the molecular basis of tolerance, immunoparalysis and other forms of innate immune memory has remained unclear. Here we perform a screen for tolerance-associated microRNAs and identify miR-221 and miR-222 as regulators of the functional reprogramming of macrophages during lipopolysaccharide tolerization. Prolonged stimulation with lipopolysaccharide in mice leads to increased expression of miR-221 and mir-222, both of which regulate brahma-related gene 1 (Brg1, also known as Smarca4). This increased expression causes the transcriptional silencing of a subset of inflammatory genes that depend on chromatin remodelling mediated by SWI/SNF (switch/sucrose non-fermentable) and STAT (signal transducer and activator of transcription), which in turn promotes tolerance. In patients with sepsis, increased expression of miR-221 and miR-222 correlates with immunoparalysis and increased organ damage. Our results show that specific microRNAs can regulate macrophage tolerization and may serve as biomarkers of immunoparalysis and poor prognosis in patients with sepsis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hotchkiss, R. S., Monneret, G. & Payen, D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat. Rev. Immunol. 13, 862–874 (2013).
Ertel, W. et al. Downregulation of proinflammatory cytokine release in whole blood from septic patients. Blood 85, 1341–1347 (1995).
Otto, G. P. et al. The late phase of sepsis is characterized by an increased microbiological burden and death rate. Crit. Care 15, R183 (2011).
Boomer, J. S. et al. Immunosuppression in patients who die of sepsis and multiple organ failure. J. Am. Med. Assoc. 306, 2594–2605 (2011).
Cavaillon, J. M. & Adib-Conquy, M. Bench-to-bedside review: endotoxin tolerance as a model of leukocyte reprogramming in sepsis. Crit. Care 10, 233 (2006).
Saeed, S. et al. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science 345, 1251086 (2014).
Foster, S. L., Hargreaves, D. C. & Medzhitov, R. Gene-specific control of inflammation by TLR-induced chromatin modifications. Nature 447, 972–978 (2007).
Chen, J. & Ivashkiv, L. B. IFN-γ abrogates endotoxin tolerance by facilitating Toll-like receptor-induced chromatin remodeling. Proc. Natl Acad. Sci. USA 107, 19438–19443 (2010).
Mages, J., Dietrich, H. & Lang, R. A genome-wide analysis of LPS tolerance in macrophages. Immunobiology 212, 723–737 (2008).
Fraker, D. L., Stovroff, M. C., Merino, M. J. & Norton, J. A. Tolerance to tumor necrosis factor in rats and the relationship to endotoxin tolerance and toxicity. J. Exp. Med. 168, 95–105 (1988).
Cavaillon, J.-M., Pitton, C. & Fitting, C. Endotoxin tolerance is not a LPS-specific phenomenon: partial mimicry with IL-1, IL-10 and TGFβ. J. Endotoxin Res. 1, 21–29 (1994).
Di Leva, G. et al. MicroRNA cluster 221–222 and estrogen receptor α interactions in breast cancer. J. Natl. Cancer Inst. 102, 706–721 (2010).
Stanton, B. Z. et al. Smarca4 ATPase mutations disrupt direct eviction of PRC1 from chromatin. Nat. Genet. 49, 282–288 (2017).
Ramirez-Carrozzi, V. R. et al. Selective and antagonistic functions of SWI/SNF and Mi-2β nucleosome remodeling complexes during an inflammatory response. Genes Dev. 20, 282–296 (2006).
Ramirez-Carrozzi, V. R. et al. A unifying model for the selective regulation of inducible transcription by CpG islands and nucleosome remodeling. Cell 138, 114–128 (2009).
Saccani, S., Pantano, S. & Natoli, G. Two waves of nuclear factor κB recruitment to target promoters. J. Exp. Med. 193, 1351–1359 (2001).
Agalioti, T., Chen, G. & Thanos, D. Deciphering the transcriptional histone acetylation code for a human gene. Cell 111, 381–392 (2002).
Park, C. Y. et al. A resource for the conditional ablation of microRNAs in the mouse. Cell Reports 1, 385–391 (2012).
Ourthiague, D. R. et al. Limited specificity of IRF3 and ISGF3 in the transcriptional innate-immune response to double-stranded RNA. J. Leukoc. Biol. 98, 119–128 (2015).
Mancino, A. et al. A dual cis-regulatory code links IRF8 to constitutive and inducible gene expression in macrophages. Genes Dev. 29, 394–408 (2015).
Tong, A. J. et al. A stringent systems approach uncovers gene-specific mechanisms regulating inflammation. Cell 165, 165–179 (2016).
Garber, M. et al. A high-throughput chromatin immunoprecipitation approach reveals principles of dynamic gene regulation in mammals. Mol. Cell 47, 810–822 (2012).
Cohen, M. et al. Chronic exposure to TGFβ1 regulates myeloid cell inflammatory response in an IRF7-dependent manner. EMBO J. 33, 2906–2921 (2014).
Langlais, D., Barreiro, L. B. & Gros, P. The macrophage IRF8/IRF1 regulome is required for protection against infections and is associated with chronic inflammation. J. Exp. Med. 213, 585–603 (2016).
Ostuni, R. et al. Latent enhancers activated by stimulation in differentiated cells. Cell 152, 157–171 (2013).
Huang, M. et al. Chromatin-remodelling factor BRG1 selectively activates a subset of interferon-α-inducible genes. Nat. Cell Biol. 4, 774–781 (2002).
Ni, Z. et al. Apical role for BRG1 in cytokine-induced promoter assembly. Proc. Natl Acad. Sci. USA 102, 14611–14616 (2005).
Levy, M. M. et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Intensive Care Med. 29, 530–538 (2003).
Kadoch, C. et al. Proteomic and bioinformatic analysis of mammalian SWI/SNF complexes identifies extensive roles in human malignancy. Nat. Genet. 45, 592–601 (2013).
Bultman, S. et al. A Brg1 null mutation in the mouse reveals functional differences among mammalian SWI/SNF complexes. Mol. Cell 6, 1287–1295 (2000).
Cheng, S. C. et al. Broad defects in the energy metabolism of leukocytes underlie immunoparalysis in sepsis. Nat. Immunol. 17, 406–413 (2016).
Leentjens, J. et al. Reversal of immunoparalysis in humans in vivo: a double-blind, placebo-controlled, randomized pilot study. Am. J. Respir. Crit. Care Med. 186, 838–845 (2012).
Döcke, W. D. et al. Monocyte deactivation in septic patients: restoration by IFN-γ treatment. Nat. Med. 3, 678–681 (1997).
Zambelli, F., Pesole, G. & Pavesi, G. Pscan: finding over-represented transcription factor binding site motifs in sequences from co-regulated or co-expressed genes. Nucleic Acids Res. 37, W247–W252 (2009).
Roberson, S. M. & Walker, W. S. Immortalization of cloned mouse splenic macrophages with a retrovirus containing the v-raf/mil and v-myc oncogenes. Cell. Immunol. 116, 341–351 (1988).
Kertesz, M., Iovino, N., Unnerstall, U., Gaul, U. & Segal, E. The role of site accessibility in microRNA target recognition. Nat. Genet. 39, 1278–1284 (2007).
Griffiths-Jones, S., Grocock, R. J., van Dongen, S., Bateman, A. & Enright, A. J. miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res. 34, D140–D144 (2006).
Ran, F. A. et al. Genome engineering using the CRISPR–Cas9 system. Nat. Protocols 8, 2281–2308 (2013).
Afgan, E. et al. The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2016 update. Nucleic Acids Res. 44, W3–W10 (2016).
Bone, R. C. et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Chest 101, 1644–1655 (1992).
Moreau, R. et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology 144, 1426–1437.e9 (2013).
Rao, P. et al. IκBβ acts to inhibit and activate gene expression during the inflammatory response. Nature 466, 1115–1119 (2010).
Acknowledgements
We thank M. McManus (UCSF) for generously providing us with a targeting construct used in the generation of mir-221 mir-222 knockout mice. This work was supported by grants R21-AI116082 and R37-AI33443 to S.G. from the National Institutes of Health.
Reviewer information
Nature thanks G. Crabtree, R.Hotchkiss, M. Netea and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.J.S. performed the majority of the experiments and writing of the manuscript. R.G.B. performed the microRNA microarray experiment. G.M. performed experiments on patient samples. T.B., S.D.D. and D.E.F. assisted with experimental design and collected patient samples for the human portions of this study. M.S.H. assisted with experimental design and the writing of the manuscript. S.G. conceived the study, and provided guidance with experimental design and the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 In vitro modelling of tolerance and miR-222 induction upon prolonged LPS stimulation.
a, Schematic of experiments performed in b. b, Expression of LPS-response genes in control BMDMs that have undergone the given treatments. Four major expression patterns of LPS-response genes in response to tolerization were noted (n = 5 biologically independent samples). c, Schematic of experiments performed in d. d, Cytokine production—measured by ELISA—by BMDMs re-stimulated with LPS overnight after pre-treatment with LPS for the given periods of time. Time points chosen for miRNA microarray analysis have bars shaded in grey (n = 3 biologically independent samples). e, Schematic of strategy for experiments performed in Fig. 1. f, Comparison of microarray (x axis) and qPCR (y axis) measurements of LPS-induced upregulation of miRNAs. A linear regression showing the correlation between the two methods is plotted (n = 16 miRNAs tested). g, qPCR verification of LPS-induced change in expression of nine miRNAs (n = 3 biologically independent samples). h, Expression of miR-222 after stimulation of BMDMs by anti-inflammatory and tolerance-inducing factors for the given lengths of time (n = 5 biologically independent samples; Dex, dexamethasone). i, Expression of miR-222 in response to LPS alone, or LPS after pre-treatment of BMDMs with IFNγ (n = 4 biologically independent samples). For all bar and line graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for paired values.
Extended Data Fig. 2 Differential regulation of miR-222 and miR-221 and association of miR-222 with in vitro tolerance.
a–c, Expression of miR-221 and miR-222 in response to LPS stimulation of BMDMs (a, n = 4 biologically independent samples), peritoneal macrophages (b, n = 3 biologically independent samples for miR-222 and n = 4 biologically independent samples for miR-221) or monocytes isolated from the bone marrow (c, n = 3 biologically independent samples), as determined by qPCR. d, LPS-induced miR-221 and miR-222 expression in BMDMs with or without IFNγ pre-treatment, as determined by qPCR (n = 2 biologically independent samples). e, Schematic of experiments performed in f, g and Fig. 1c. f, g, LPS-induced gene expression at the mRNA (f) or primary transcript (g) level after miR-222 mimic transfection (n = 5 biologically independent samples). For all bar and line graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for paired values.
Extended Data Fig. 3 Tnf is a direct target of miR-222, but suppression of Tnf does not account for miR-222-mediated transcriptional silencing of late LPS-response genes.
a, Sequence and prediction scores of an miR-222-binding site in the Tnf UTR. b, Activity of a luciferase reporter construct in which the luciferase coding sequence is followed by either the complete Tnf UTR, or a UTR in which the predicted miR-222-binding site has been mutated to the sequence shown in a (n = 6 independent experiments). c, CRISPR–Cas9 targeting strategy to delete predicted binding sites. CDS, coding sequence. d, Clones of RAW cells were screened for successful deletion of the miR-222-binding site by PCR across the targeted region of the UTR, using genomic DNA from the given clonal line as a template. Screening for Tnf UTR deletion is shown. Experiment was repeated twice with similar results. e, Successful deletion of the miR-222-binding site in RAW cell clones was confirmed by sequencing genomic DNA of the given cell line. miR-222-binding site in the Tnf UTR is highlighted in yellow. f, LPS-induced Tnf expression in control and CRISPR–Cas9-targeted RAW cells (n = 4 independent experiments). g, Average effect of miR-222 mimic transfection on LPS-induced Tnf mRNA levels in either control mouse embryonic fibroblasts or mouse embryonic fibroblasts that have undergone CRISPR targeting and clonal selection for deletion of the miR-222-binding site. Average of the effects from the three clonal lines (n = 3 independent experiments) is shown. h, Wild-type BMDMs were transfected with a control or miR-222 mimic oligonucleotide. Twenty-four hours later, cells were pre-treated with an isotype control (IgG) or TNF-neutralizing (anti-TNF) antibody for two hours, and stimulated with 10 ng ml−1 LPS. Expression of the given genes was measured by qPCR (n = 4 biologically independent samples). i, Efficacy of TNF neutralization was confirmed by treating cells with IgG or anti-TNF as above, followed by stimulation with 100 ng ml−1 recombinant mouse TNF (n = 3 biologically independent samples). Gene upregulation was not detected (ND) in two out of three samples treated with anti-TNF. For all bar graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for paired values.
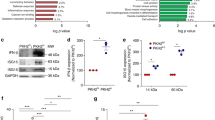
Extended Data Fig. 4 Evidence of miR-222 targeting of Brg1.
a, Example of gating used to exclude dead cells from flow cytometry analyses in c, g and Extended Data Fig. 6i. b, Example of gating used to distinguish cells with high versus low levels of IκBα, as analysed in c. c, Effect of miRNA overexpression (by viral transduction) on LPS-induced IκBα degradation in immortalized BMDMs, measured by flow cytometry (n = 4 independent experiments). d, Sequence and prediction scores of an miR-222-binding site in the Brg1 UTR. e, miR-222 and Brg1 mRNA levels in LPS-stimulated BMDMs (n = 3 biologically independent samples). f, Brg1 mRNA levels in resting BMDMs 24 h after transfection (n = 4 biologically independent samples). g, Effect of miRNA overexpression or antagonization (by viral transduction) on BRG1 levels in immortalized BMDMs, observed by flow cytometry. Representative of four independent experiments with similar results, quantified in h. h, Flow cytometry analysis of BRG1 protein levels in transduced immortalized BMDMs (n = 4 independent experiments). i, Activity of a luciferase reporter construct in which the luciferase coding sequence is followed by either the complete Brg1 UTR, or a UTR in which the predicted miR-222-binding site has been mutated to the sequence shown in d (n = 3 independent experiments). j, Quantification of the average effect of miR-222 mimic transfection on Brg1-dependent and Brg1-independent LPS-response genes (n = 3 biologically independent samples). Two-sided Student’s t-test for heteroscedastic values used to compare ratios (ratio of miR-222 overexpression to control) at peak LPS-induced expression times for Brg1-dependent versus Brg1-independent genes. k, l, ChIP for histone H3 acetylation (k) or histone H4 acetylation (l) after LPS stimulation of immortalized BMDMs transduced with overexpression constructs (k and l tested in same n = 3 independent experiments). m, Successful deletion of the miR-222-binding site in the Brg1 UTR in RAW cell clones was confirmed by sequencing genomic DNA of the given cell line. miR-222-binding site is highlighted in yellow. n, Effect of miR-222 overexpression (by oligonucleotide transfection) on LPS-induced gene expression in either a RAW cell line in which the Brg1–miR-222 binding site was deleted by CRISPR targeting (as shown in Extended Data Fig. 3c) or a cell line in which the binding site was not targeted for deletion (n = 5 independent experiments). For all bar graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for paired values.
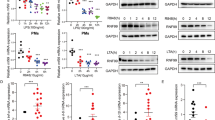
Extended Data Fig. 5 Comparison of miR-221 and miR-222, and effects of miR-221 and miR-222 deletion on the transcriptional response to LPS.
a, Alignment of the mature miR-221 and miR-222 sequences. The miRNA seed sequence is highlighted in yellow. b, Venn diagram displaying overlap between MicroCosm target predictions for mmu-miR-221 and mmu-miR-222. c, Alignment and computational scores of miR-221 sequence with predicted Brg1 UTR target site. Alignment of miR-222 sequence with the site is also shown. d, Brg1 expression in BMDMs transfected with the given oligonucleotide (n = 3 biologically independent samples). e, LPS-induced cytokine production in BMDMs transfected with given miRNA mimics, as measured by ELISA (n = 5 biologically independent samples). f, Schematic of the miR-221 and miR-222 locus after targeting with a construct designed to generate both complete and conditional mir-221 mir-222 knockout mice. g, Schematic of the miR-221 and miR-222 locus after breeding targeted mice (f) with EIIa-Cre mice, which results in complete deletion of miR-221 and miR-222. h, miRNA expression in BMDMs from littermates with a wild-type or mir-221 mir-222 knockout allele (n = 5 biologically independent samples). i, LPS-induced gene expression in naive or tolerized peritoneal macrophages isolated from wild-type or mir-221 mir-222 knockout littermates (n = 7 biologically independent samples). j, Heat map comparing the effect of Brg1 and Brm knockdown15 and mir-222 knockout on gene expression. Colours represent values of the given ratios; red indicates increased expression, white indicates no change and blue indicates decreased expression. k, Heat map of LPS-induced gene expression in wild-type and mir-221 mir-222 knockout macrophages. For all bar graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for paired (d, e) or heteroscedastic (i) values.
Extended Data Fig. 6 Gene Ontology and ChIP–seq analysis shows that genes affected by mir-221 mir-222 knockout have differential gene functions and transcription-factor binding at promoters.
a–f, Enriched GO terms (a–c) and transcription-factor binding at promoters (d–f) of genes that are expressed at higher (twofold or higher) or lower (0.5-fold or lower) levels in mir-221 mir-222 knockout macrophages after no stimulation (a, d, n = 647 genes higher, 565 genes lower), LPS stimulation (b, e, n = 143 genes higher, 121 genes lower) or LPS tolerization followed by re-stimulation (c, f, n = 123 genes higher,48 genes lower). PANTHER was used to identify GO terms. The top four terms for each category are shown; GO terms that are unique to either higher- or lower-expression gene subsets are highlighted. g, h, IRF and NF-κB subunit occupancy at gene promoters; gene subsets analysed are described in Fig. 2h. For transcription factor analyses, previously published ChIP–seq data were used20,21,22,23,24,25. i, RNA levels of genes in wild-type or mir-221 mir-222 knockout peritoneal macrophages, quantified by a single RNA sequencing experiment. j, qPCR for gene expression in wild-type BMDMs after Amaxa-based nucleofection of given overexpression construct (n = 3 biologically independent samples). For all bar graphs, centre value represents the mean and errors bars (if applicable) represent s.e.m.
Extended Data Fig. 7 mir-221 mir-222 knockout mice have an altered LPS response and knockout macrophages exhibit enhanced Salmonella uptake and clearance in vitro.
a, Schematic of experiments performed in b, c. b, Survival of naive or tolerized mice injected with high doses of LPS. c, Wild-type or mir-222 knockout littermates were tolerized to LPS before lethal LPS injection. The change in body temperature after final LPS injection was monitored for 24 h. d, Schematic of experiments performed in e. e, Survival of naive or tolerized mice injected with LPS and d-galactosamine. f, BMDMs from wild-type or mir-221 mir-222 knockout mice were spin-infected with a GFP-expressing strain of S. Typhimurium. Fluorescence was analysed by microscopy 60 min after infection. Representative of two independent experiments with similar results. g, BMDMs from wild-type or mir-221 mir-222 knockout mice were spin-infected with a GFP-expressing strain of S. Typhimurium. Fluorescence was analysed by flow cytometry 30 min post-infection. Representative of three independent experiments with similar results. h, Average fluorescence of infected BMDMs after early (left) or late (right) time points after infection (n = 4 biologically independent wild-type samples, 3 biologically independent knockout samples). i, Survival of S. Typhimurium after in vitro infection of BMDMs, determined by comparing CFUs after lysis of BMDMs at early and late time points of infection (n = 5 biologically independent wild-type samples, 4 biologically independent knockout samples). For all bar and line graphs, mean ± s.e.m. is plotted. *P < 0.05, **P < 0.01, determined by two-sided Student’s t-test for heteroscedastic values.
Extended Data Fig. 8 miR-221 and miR-222 are upregulated in human cells and patients with sepsis.
a, b, LPS-induced miRNA expression in undifferentiated (a) or phorbol 12-myristate 13-acetate-differentiated (b) human U937 cells (n = 3 independent experiments). c, Patient characteristics for data shown in Fig. 4a. APACHE, acute physiology and chronic health evaluation; SAPS, simplified acute physiology score. Categorical variables are given as n (percentage in parentheses) and continuous variables as median (interquartile range in parentheses). d, e, Baseline characteristics of patients with decompensated liver disease in the absence or presence of multiple organ failure syndrome (according to the EASL CLIF-C criteria for acute-on-chronic liver failure). Data in d correspond to peripheral blood mononuclear cell analyses (Fig. 4b–d). Median with interquartiles or frequencies and percentages are shown. P values from Mann–Whitney U test or Fisher’s exact test as appropriate (two-sided). *, comparing any infection to no infection; **, 4 out of 30 (13%) and 1 out of 10 (10%) patients were lost to follow-up within 30 days. Data in e correspond to monocyte analyses (Fig. 4f). Median with interquartiles or frequencies and percentages are shown. P values from Mann–Whitney U test or Fisher’s exact test as appropriate (two-sided). *, comparing any infection to no infection; **, 1 out of 10 (10%) patients was lost to follow-up within 30 days. f, Correlation between miR-221 and miR-222 levels in patients characterized in d (n = 30 patients). Bivariate nonparametric correlation analysis (Spearman’s rho) was used to identify correlations between variables and P values. g–j, Linear correlation of miR-222 expression and C-reactive protein (g), white blood cell count (h), creatinine levels (i) or model for end-stage liver disease score (j) in samples from the patient cohort described in d (n = 30 patients). Bivariate nonparametric correlation analysis (Spearman’s rho) was used to identify correlations between variables and P values. For line graphs, mean ± s.e.m. is plotted.
Extended Data Fig. 9 Model of the effect of miR-222 on LPS-induced macrophage tolerance.
a, Before an acute LPS stimulation, chromatin at BRG1-dependent gene promoters prevents binding of remodelling-dependent transcription factors and RNA polymerase. b, After an acute LPS stimulation, transcription factors such as STAT1 and STAT2 are recruited to gene promoters and stabilize BRG1 binding. c, d, BRG1 activity leads to chromatin remodelling (c), which enables recruitment of additional transcription factors, such as NF-κB, to the unwound DNA (d). This enables polymerase recruitment and licensing, which leads to gene transcription. e, After an initial LPS response, chromatin is ‘reset’ to an inhibitory state by negative regulators of chromatin accessibility. f, Upon LPS re-stimulation, transcription factors must again be recruited to gene promoters. However, miR-222 limits the level of BRG1. g, Lack of available BRG1 prevents chromatin remodelling at many gene promoters, and prevents downstream transcription factor recruitment. This prevents gene transcription from occurring in most cells.
Supplementary information
Rights and permissions
About this article
Cite this article
Seeley, J.J., Baker, R.G., Mohamed, G. et al. Induction of innate immune memory via microRNA targeting of chromatin remodelling factors. Nature 559, 114–119 (2018). https://doi.org/10.1038/s41586-018-0253-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0253-5
This article is cited by
-
MicroRNA-21a-5p inhibition alleviates systemic sclerosis by targeting STAT3 signaling
Journal of Translational Medicine (2024)
-
Cellular forgetting, desensitisation, stress and ageing in signalling networks. When do cells refuse to learn more?
Cellular and Molecular Life Sciences (2024)
-
A signature of immune-related genes correlating with clinical prognosis and immune microenvironment in sepsis
BMC Bioinformatics (2023)
-
Lean adipose tissue macrophage derived exosome confers immunoregulation to improve wound healing in diabetes
Journal of Nanobiotechnology (2023)
-
Apoptotic caspase-7 activation inhibits non-canonical pyroptosis by GSDMB cleavage
Cell Death & Differentiation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.