Abstract
Parenting is essential for the survival and wellbeing of mammalian offspring. However, we lack a circuit-level understanding of how distinct components of this behaviour are coordinated. Here we investigate how galanin-expressing neurons in the medial preoptic area (MPOAGal) of the hypothalamus coordinate motor, motivational, hormonal and social aspects of parenting in mice. These neurons integrate inputs from a large number of brain areas and the activation of these inputs depends on the animal’s sex and reproductive state. Subsets of MPOAGal neurons form discrete pools that are defined by their projection sites. While the MPOAGal population is active during all episodes of parental behaviour, individual pools are tuned to characteristic aspects of parenting. Optogenetic manipulation of MPOAGal projections mirrors this specificity, affecting discrete parenting components. This functional organization, reminiscent of the control of motor sequences by pools of spinal cord neurons, provides a new model for how discrete elements of a social behaviour are generated at the circuit level.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Dulac, C., O’Connell, L. A. & Wu, Z. Neural control of maternal and paternal behaviors. Science 345, 765–770 (2014).
Numan, M. & Insel, T. R. The Neurobiology of Parental Behavior (Springer, New York, 2011).
Wu, Z., Autry, A. E., Bergan, J. F., Watabe-Uchida, M. & Dulac, C. G. Galanin neurons in the medial preoptic area govern parental behaviour. Nature 509, 325–330 (2014).
Wickersham, I. R. et al. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647 (2007).
Marlin, B. J., Mitre, M., D’Amour, J. A., Chao, M. V. & Froemke, R. C. Oxytocin enables maternal behaviour by balancing cortical inhibition. Nature 520, 499–504 (2015).
Johnson, Z. V. & Young, L. J. Oxytocin and vasopressin neural networks: Implications for social behavioral diversity and translational neuroscience. Neurosci. Biobehav. Rev. 76, 87–98 (2017).
Bendesky, A. et al. The genetic basis of parental care evolution in monogamous mice. Nature 544, 434–439 (2017).
Tachikawa, K. S., Yoshihara, Y. & Kuroda, K. O. Behavioral transition from attack to parenting in male mice: a crucial role of the vomeronasal system. J. Neurosci. 33, 5120–5126 (2013).
Vom Saal, F. S. Time-contingent change in infanticide and parental behavior induced by ejaculation in male mice. Physiol. Behav. 34, 7–15 (1985).
Kohl, J., Autry, A. E. & Dulac, C. The neurobiology of parenting: a neural circuit perspective. BioEssays 39, 1–11 (2017).
Scott, N., Prigge, M., Yizhar, O. & Kimchi, T. A sexually dimorphic hypothalamic circuit controls maternal care and oxytocin secretion. Nature 525, 519–522 (2015).
Schwarz, L. A. et al. Viral-genetic tracing of the input–output organization of a central noradrenaline circuit. Nature 524, 88–92 (2015).
Gunaydin, L. A. et al. Natural neural projection dynamics underlying social behavior. Cell 157, 1535–1551 (2014).
Kudo, Y. et al. A single optical fiber fluorometric device for measurement of intracellular Ca2+ concentration: its application to hippocampal neurons in vitro and in vivo. Neuroscience 50, 619–625 (1992).
Elyada, Y. M. & Mizrahi, A. Becoming a mother—circuit plasticity underlying maternal behavior. Curr. Opin. Neurobiol. 35, 49–56 (2015).
Andermann, M. L. & Lowell, B. B. Toward a wiring diagram understanding of appetite control. Neuron 95, 757–778 (2017).
Li, Y. et al. Neuronal representation of social information in the medial amygdala of awake behaving mice. Cell 171, 1176–1190 (2017).
Remedios, R. et al. Social behaviour shapes hypothalamic neural ensemble representations of conspecific sex. Nature 550, 388–392 (2017).
Lee, G. & Gammie, S. C. GABAA receptor signaling in caudal periaqueductal gray regulates maternal aggression and maternal care in mice. Behav. Brain Res. 213, 230–237 (2010).
Salamone, J. D. & Correa, M. The mysterious motivational functions of mesolimbic dopamine. Neuron 76, 470–485 (2012).
McHenry, J. A. et al. Hormonal gain control of a medial preoptic area social reward circuit. Nat. Neurosci. 20, 449–458 (2017).
Seip, K. M. & Morrell, J. I. Transient inactivation of the ventral tegmental area selectively disrupts the expression of conditioned place preference for pup- but not cocaine-paired contexts. Behav. Neurosci. 123, 1325–1338 (2009).
Qualls-Creekmore, E. et al. Galanin-expressing GABA neurons in the lateral hypothalamus modulate food reward and noncompulsive locomotion. J. Neurosci. 37, 6053–6065 (2017).
Isogai, Y. et al. Molecular organization of vomeronasal chemoreception. Nature 478, 241–245 (2011).
Bergan, J. F., Ben-Shaul, Y. & Dulac, C. Sex-specific processing of social cues in the medial amygdala. eLife 3, e02743 (2014).
Yao, S., Bergan, J., Lanjuin, A. & Dulac, C. Oxytocin signaling in the medial amygdala is required for sex discrimination of social cues. eLife 6, e31373 (2017).
Hong, W., Kim, D. W. & Anderson, D. J. Antagonistic control of social versus repetitive self-grooming behaviors by separable amygdala neuronal subsets. Cell 158, 1348–1361 (2014).
Anderson, D. J. Circuit modules linking internal states and social behaviour in flies and mice. Nat. Rev. Neurosci. 17, 692–704 (2016).
Yang, T. & Shah, N. M. Molecular and neural control of sexually dimorphic social behaviors. Curr. Opin. Neurobiol. 38, 89–95 (2016).
Zimmerman, C. A., Leib, D. E. & Knight, Z. A. Neural circuits underlying thirst and fluid homeostasis. Nat. Rev. Neurosci. 18, 459–469 (2017).
Weber, F. & Dan, Y. Circuit-based interrogation of sleep control. Nature 538, 51–59 (2016).
Arber, S. Motor circuits in action: specification, connectivity, and function. Neuron 74, 975–989 (2012).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Miyamichi, K. et al. Dissecting local circuits: parvalbumin interneurons underlie broad feedback control of olfactory bulb output. Neuron 80, 1232–1245 (2013).
Beier, K. T. et al. Circuit architecture of VTA dopamine neurons revealed by systematic input–output mapping. Cell 162, 622–634 (2015).
Esposito, M. S., Capelli, P. & Arber, S. Brainstem nucleus MdV mediates skilled forelimb motor tasks. Nature 508, 351–356 (2014).
Franklin, K. B. J. & Paxinos, G. The Mouse Brain in Stereotaxic Coordinates 3rd edn (Academic, New York, 2007).
Eliava, M. et al. A new population of parvocellular oxytocin neurons controlling magnocellular neuron activity and inflammatory pain processing. Neuron 89, 1291–1304 (2016).
Xiao, L., Priest, M. F., Nasenbeny, J., Lu, T. & Kozorovitskiy, Y. Biased oxytocinergic modulation of midbrain dopamine systems. Neuron 95, 368–384 (2017).
Swanson, L. W. & Sawchenko, P. E. Hypothalamic integration: organization of the paraventricular and supraoptic nuclei. Annu. Rev. Neurosci 6, 269–324 (1983).
Wickersham, I. R., Finke, S., Conzelmann, K. K. & Callaway, E. M. Retrograde neuronal tracing with a deletion-mutant rabies virus. Nat. Methods 4, 47–49 (2007).
Osakada, F. et al. New rabies virus variants for monitoring and manipulating activity and gene expression in defined neural circuits. Neuron 71, 617–631 (2011).
Weible, A. P. et al. Transgenic targeting of recombinant rabies virus reveals monosynaptic connectivity of specific neurons. J. Neurosci. 30, 16509–16513 (2010).
Menegas, W., Babayan, B. M., Uchida, N. & Watabe-Uchida, M. Opposite initialization to novel cues in dopamine signaling in ventral and posterior striatum in mice. eLife 6, e21886 (2017).
Sukikara, M. H., Mota-Ortiz, S. R., Baldo, M. V., Felicio, L. F. & Canteras, N. S. The periaqueductal gray and its potential role in maternal behavior inhibition in response to predatory threats. Behav. Brain Res. 209, 226–233 (2010).
Fleming, A. S., Vaccarino, F. & Luebke, C. Amygdaloid inhibition of maternal behavior in the nulliparous female rat. Physiol. Behav. 25, 731–743 (1980).
Numan, M., Numan, M. J. & English, J. B. Excitotoxic amino acid injections into the medial amygdala facilitate maternal behavior in virgin female rats. Horm. Behav. 27, 56–81 (1993).
Sheehan, T., Paul, M., Amaral, E., Numan, M. J. & Numan, M. Evidence that the medial amygdala projects to the anterior/ventromedial hypothalamic nuclei to inhibit maternal behavior in rats. Neuroscience 106, 341–356 (2001).
Insel, T. R. & Harbaugh, C. R. Lesions of the hypothalamic paraventricular nucleus disrupt the initiation of maternal behavior. Physiol. Behav 45, 1033–1041 (1989).
Numan, M. & Corodimas, K. P. The effects of paraventricular hypothalamic lesions on maternal behavior in rats. Physiol. Behav. 35, 417–425 (1985).
Lee, G. & Gammie, S. C. GABAA receptor signaling in the lateral septum regulates maternal aggression in mice. Behav. Neurosci 123, 1169–1177 (2009).
D’Anna, K. L. & Gammie, S. C. Activation of corticotropin-releasing factor receptor 2 in lateral septum negatively regulates maternal defense. Behav. Neurosci 123, 356–368 (2009).
Hansen, S., Harthon, C., Wallin, E., Löfberg, L. & Svensson, K. Mesotelencephalic dopamine system and reproductive behavior in the female rat: effects of ventral tegmental 6-hydroxydopamine lesions on maternal and sexual responsiveness. Behav. Neurosci. 105, 588–598 (1991).
Hansen, S. Maternal behavior of female rats with 6-OHDA lesions in the ventral striatum: characterization of the pup retrieval deficit. Physiol. Behav. 55, 615–620 (1994).
Li, M. & Fleming, A. S. The nucleus accumbens shell is critical for normal expression of pup-retrieval in postpartum female rats. Behav. Brain Res. 145, 99–111 (2003).
Keer, S. E. & Stern, J. M. Dopamine receptor blockade in the nucleus accumbens inhibits maternal retrieval and licking, but enhances nursing behavior in lactating rats. Physiol. Behav. 67, 659–669 (1999).
Numan, M. et al. The effects of D1 or D2 dopamine receptor antagonism in the medial preoptic area, ventral pallidum, or nucleus accumbens on the maternal retrieval response and other aspects of maternal behavior in rats. Behav. Neurosci. 119, 1588–1604 (2005).
Numan, M. & Nagle, D. S. Preoptic area and substantia nigra interact in the control of maternal behavior in the rat. Behav. Neurosci. 97, 120–139 (1983).
Bridges, R. S., Mann, P. E. & Coppeta, J. S. Hypothalamic involvement in the regulation of maternal behaviour in the rat: inhibitory roles for the ventromedial hypothalamus and the dorsal/anterior hypothalamic areas. J. Neuroendocrinol. 11, 259–266 (1999).
Numan, M. & Numan, M. A lesion and neuroanatomical tract-tracing analysis of the role of the bed nucleus of the stria terminalis in retrieval behavior and other aspects of maternal responsiveness in rats. Dev. Psychobiol. 29, 23–51 (1996).
Numan, M., Rosenblatt, J. S. & Komisaruk, B. R. Medial preoptic area and onset of maternal behavior in the rat. J. Comp. Physiol. Psychol. 91, 146–164 (1977).
Bridges, R. S., Numan, M., Ronsheim, P. M., Mann, P. E. & Lupini, C. E. Central prolactin infusions stimulate maternal behavior in steroid-treated, nulliparous female rats. Proc. Natl Acad. Sci. USA 87, 8003–8007 (1990).
Numan, M. & Numan, M. J. Projection sites of medial preoptic area and ventral bed nucleus of the stria terminalis neurons that express Fos during maternal behavior in female rats. J. Neuroendocrinol. 9, 369–384 (1997).
Lonstein, J. S., Simmons, D. A., Swann, J. M. & Stern, J. M. Forebrain expression of c-fos due to active maternal behaviour in lactating rats. Neuroscience 82, 267–281 (1997).
Acknowledgements
We thank S. Sullivan for help with behaviour and mouse husbandry. E. Kremer (Montpellier) and R. Neve (MIT) provided viral vectors. E. Soucy and J. Greenwood helped design motivation assay. R. Hellmiss and K. Wilbur helped with illustrations. H. S. Knobloch-Bollmann provided advice on PVN cell types. We thank the members of the Dulac laboratory for comments on the manuscript. This work was supported by a Human Frontier Long-Term Fellowship, an EMBO Long-Term Fellowship and a Sir Henry Wellcome Fellowship to J.K., Fondation pour la Recherche Médicale grant SPE20150331860 to B.M.B., a NIH K99 Award and a NARSAD Young Investigator Award to A.E.A., a Howard Hughes Gilliam Fellowship to B.M.-R., a Harvard Mind Brain and Behavior faculty grant to N.U. and NIH grant 1R01HD082131-01A1 to C.D. C.D. and L.L. are investigators of the Howard Hughes Medical Institute.
Reviewer information
Nature thanks V. Grinevich and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.K. and C.D. conceived and designed the study. J.K performed and analysed tracing and optogenetics experiments. J.K. and B.M.B. performed and analysed photometry recordings. A.E.A. helped with in situ hybridization experiments, B.M.-R. helped with CTB tracing experiments, N.D.R. helped to analyse tracing data, and V.K. helped with optogenetics experiments. L.S.Z., K.M. and L.L. shared unpublished viral reagents. N.U. provided fibre photometry setup. J.K., B.M.B., N.D.R., A.E.A. and C.D. analysed and interpreted the results. J.K. and C.D. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
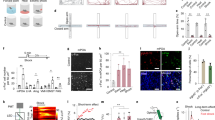
Extended Data Fig. 1 Putative functional roles of brain areas providing monosynaptic inputs into MPOAGal neurons.
a, Comparison between MPOAGal input fractions in virgin males (n = 3) and virgin females (n = 3) after rabies tracing (see Fig. 1a). Sexually dimorphic inputs are highlighted. Two-tailed t-tests, supraoptic nucleus (SON), **P = 0.0041; posteriomedial amygdalo-hippocampal area (AHPM), ***P = 0.0007; medial septum (MS), *P = 0.0133. b, Comparison between MPOAGal input fractions after rabies tracing was initiated from the right (n = 3) or left (n = 3) hemisphere in virgin females. No significant differences were found (P > 0.05; two-tailed paired t-test). c, Comparison between rabies-injected (ipsilateral (ipsi)) and non-injected (contralateral (contra)) MPOA of a mother after parental behaviour. Activated (Fos+) rabies+ neurons are shown (top, arrowheads). Fos+ neuron numbers are not significantly different between hemispheres (bottom, P = 0.43, 95% confidence interval −4.176–1.843; two-tailed paired t-test; n = 6). d, MPOAGal neurons receive monosynaptic inputs from magnocellular SONAVP neurons (mothers, 72.7 ± 9.3% overlap, n = 3; virgin females, 77.4 ± 4.3%, n = 3; fathers, 83.3 ± 3.3%, n = 3) but rarely from SONOXT neurons (mothers, 4.6 ± 4.2% overlap, n = 2; virgin females, 4.5 ± 1.0%, n = 2; fathers, 2.8 ± 1.8%, n = 2). Data are mean ± s.e.m. Scale bars, 100 μm (c) and 50 μm (d).
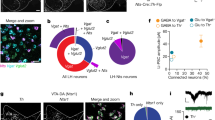
Extended Data Fig. 2 MPOAGal projections in males and downstream connectivity.
a, Synaptophysin–GFP (Syn–GFP) labelling of presynaptic sites in MPOAGal projections. b, Representative MPOAGal projections from a virgin male, identified by tdTomato fluorescence. c, Representative MPOAGal projections, identified by tdTomato fluorescence, after viral injection into the left MPOA. d, Fos+ fractions of virally labelled MPOAGal projections in fathers (n = 6, 3, 4, 3, 3, respectively, from top to bottom). Red line depicts the population average3. Data are mean ± s.e.m. e, Trans-synaptic retrograde rabies tracing from AVPeTH neurons. f, MPOAGal neurons presynaptic to AVPeTH neurons in females (left, indicated by arrowheads, 21.4% Gal+ neurons, 47 out of 220 neurons, n = 3) and males (right, 16.7% Gal+, 4 out of 24 neurons, n = 2). g, Direct and indirect MPOAGal to PVNOXT connectivity. Asterisk, AVPeTH neurons form excitatory synapses with PVNOXT neurons in females11. h, Conditional monosynaptic retrograde tracing initiated from PAG. i, j, Injection sites with mCherry+ starter neurons in PAG of Vgat-IRES-Cre (i, left) or Vglut2-IRES-Cre (j, left) mice. Presynaptic, rabies+Gal+ neurons are detected in MPOA when tracing is initiated from PAGVgat (i, right, indicated by arrowheads), but not PAGVglut2 (j, right), neurons. Scale bars, 50 μm (a, f and i, inset), 200 μm (i and j, left) 250 μm (b, c, inset and i and j, right) and 500 μm (c, left).
Extended Data Fig. 3 MPOAGal projections correspond to mostly non-overlapping neuronal subpopulations.
a, Control injection of a 1:1 mixture of CTB-488 and CTB-647 into PAG results in highly overlapping neuron populations in the MPOA (quantification in c). b, Strategy to determine collaterals between pairwise injected MPOAGal projections in Gal::cre+/−loxP-Stop-loxP-tdTomato+/− mice. An example with two double-labelled MPOAGal neurons is shown after injection of CTB-488 into PAG and CTB-647 into VTA (right, indicated by arrowheads). c, Quantification of data in a, b. Data are mean ± s.e.m. (n = 6, 6, 3, 3, 3, 3, 3, respectively, from top to bottom). d, Representative image from MPOA of Gal::cre+/−loxP-Stop-loxP-tdTomato+/− mouse after injection of CTB-647 into PAG. Note high overlap between Gal+ and CTB+ neurons. e, Frequency of Gal+ neurons in individual, CTB-labelled MPOA projections (n = 4, 6, 4, 3, 3, 3, respectively, from top to bottom). Red line depicts expected labelling frequency, based on proportion of Gal+ MPOA neurons3 (around 20%). c, e, Data are mean ± s.e.m. f, Distribution of cell bodies corresponding to specific MPOAGal projections. Individual MPOAGal projection areas in Gal::Cre virgin females were injected with Cre-dependent CAV2-FLEx-ZsGreen (see Fig. 2h). Only labelling patterns on the ipsilateral, injected side are shown and only two projection-specific subpopulations per side are displayed for clarity. Mouse brain images in this figure have been reproduced with permission from Elsevier37. g, Zones occupied by MPOAGal cell bodies projecting to MeA, PAG, VTA and PVN in anterior (left), central (middle) and posterior (right) MPOA. f, g, Distance from bregma is shown in mm. Scale bars, 50 μm (a, b and d, inset) and 250 μm (d).
Extended Data Fig. 4 MPOAGal projections barely collateralize.
a, Strategy to detect brain-wide axon collaterals of specific MPOAGal projections. b, Dense labelling of MPOAGal neurons after injection of retrograde tracer CAV into PAG and reporter AAV into MPOA. c, Absence of MPOAGal labelling in negative control without injection of CAV. d–f, Only minor axon collaterals are detectable from MPOAGal neurons projecting to PAG (d; n = 2 virgin males), VTA (e; n = 3 virgin males) or MeA (f; n = 2 virgin males). Note the MPOA to MeA fibre tract in BNST in f. Signal was enhanced using anti-GFP immunostaining (Methods). Scale bars, b, c, 400 μm (b, c), 100 μm (insets) and 150 μm (d–f).
Extended Data Fig. 5 Negative controls for monosynaptic retrograde tracing.
a, Absence of rabies+ background labelling in the MPOA of AAV- and rabies-injected C57BL/6 control mice (n = 2). b, Labelling of MPOAGal neurons after injection of CAV into PAG and starter AAVs into MPOA of Gal::cre mice (261 ± 19 neurons, n = 4). c, Near-absence of labelling in AAV-only negative control (11 ± 2 neurons, n = 2). d, Background rabies+ neurons were present in the following brain areas of CAV-, AAV- and rabies-injected C57BL/6 control mice (n = 3): MPOA, BNST, anterior hypothalamus (AH), PVN and SON. These areas were therefore excluded from analysis (see Fig. 2k, l and Methods). Scale bars, 400 μm (main images) and 150 μm (insets).
Extended Data Fig. 6 Histology of photometry recording experiments and tuning of MPOAGal neurons in other behavioural contexts.
a, Specific GCaMP6m expression in MPOAGal neurons (90.9 ± 4.3% overlap, n = 3, mothers). b–d, Implantation sites of optical fibres in the MPOA of Gal::cre+/−loxP-Stop-loxP-tdTomato+/− mother (b), virgin female (c) and father (d). e, Quantification of GCaMP+ neuron numbers in MPOA after AAV injection (‘Total’, n = 4) and after injection of HSV into individual projections (n = 5 each). Data for mothers are shown. Data are mean ± s.e.m. Two-tailed t-tests; Total versus PAG, VTA, MeA, ***P < 0.001, PAG versus MeA, **P = 0.0033. f–h, Expression of GCaMP6m in MPOAGal neurons after bilateral infection of axon terminals in PAG (f), VTA (g) or MeA (h) with Cre-dependent, GCaMP6m-expressing HSV. Insets show fibre implantation sites. i, j, Averaged recording traces from MPOAGal neuron activity during sniffing of accessible pups (i) or inaccessible pups enclosed in a wire mesh tea ball (j) in mothers (n = 4), virgin females (n = 3) and fathers (n = 5). k, l, Averaged recording traces from MPOAGal neuron activity during sniffing of female (k) or male (l) intruder in mothers (n = 4), virgin females (n = 3) and fathers (n = 5). Two-tailed t-tests; i, ***P < 0.0001, ***P < 0.0001, ***P = 0.0001 (left to right); j, *P = 0.0380; k, *P = 0.0219; l, *P = 0.0272. m–q, Averaged recording traces from MPOAGal neurons projecting to PAG (left, n = 10), VTA (middle, n = 12) or MeA (right, n = 8) during episodes of maternal behaviour. All traces and bar graphs are mean ± s.e.m. Scale bars, 50 μm (a), 400 μm (b–d), 1 mm (f–h) and 500 μm (f–h, insets).
Extended Data Fig. 7 Distribution of parental behaviours in mothers and virgin females.
Distribution of parental behaviours during 10-min pup interaction assays in mothers (a; n = 23) and virgin females (b; n = 20). In a, individuals exhibiting high pup sniffing are indicated in blue across plots, and individuals exhibiting high pup grooming are indicated in orange. In b, individuals exhibiting high pup sniffing are indicated in green. Note that y axis ranges are identical between a and b. Lines depict mean.
Extended Data Fig. 8 Behavioural specificity of MPOAGal projection stimulation.
a, Channelrhodopsin-2 (ChR2) expression in MPOAGal neurons (97.7 ± 0.2% overlap, virgin females, n = 2). Scale bar, 50 μm. b–g, Effect of activating PAG (b, c), VTA (d, e) or MeA (f, g) projections on time spent in nest in virgin females and virgin males (b, n = 13 females and n = 10 males; d, n = 9 females and n = 10 males; f, n = 10 females and n = 10 males) and number of pup-directed sniffing bouts (c, n = 13 females and n = 10 males; e, n = 9 females and n = 10 males; g, n = 10 females and n = 10 males). h–m, Effect of activating PAG (h, i), VTA (j, k) or MeA (l, m) projections on locomotion velocity (h, n = 13 females and n = 10 males; j, n = 8 females and n = 10 males; l, n = 10 females and n = 10 males) and moved distance (i, k, m). n, q, s, Effect of inhibiting PAG (n, n = 10 females), VTA (q, n = 10 females) or MeA (s, n = 11 females) projections on pup interactions. o, t, Effect of inhibiting PAG (o, n = 10 females) or MeA (t, n = 11 females) projections on number of barrier crosses. p, r, Effect of inhibiting PAG (p, n = 10 females) or MeA (r, n = 11 females) projections on chemoinvestigation of a male intruder. u–w, Effect of inhibiting PAG (u), VTA (v) or MeA (w) projections on locomotion velocity and moved distance (n = 10, 10, 11, respectively). Two-tailed paired t-tests; c, *P = 0.0135; f, *P = 0.03; n, *P = 0.0413, q: *P = 0.0264.
Supplementary information
Video 1
MPOAGal population activity during parenting
Pan-MPOAGal fibre photometry recording in a mother. While (1) no change in signal is visible during interactions with a food object (sniffing, retrieving of cracker), (2) sniffing pups in a wire mesh cage results in modest, and (3) sniffing and grooming of pups in the nest in strong increase in MPOAGal population activity
Video 2
Barrier crossing evoked by MPOAGal➝VTA activation
Optogenetic activation of MPOAGal➝VTA projections in a virgin Gal::Cre female evokes barrier crossing in a task assessing motivation to interact with pups. Photostimulation period indicated by white dot
Rights and permissions
About this article
Cite this article
Kohl, J., Babayan, B.M., Rubinstein, N.D. et al. Functional circuit architecture underlying parental behaviour. Nature 556, 326–331 (2018). https://doi.org/10.1038/s41586-018-0027-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0027-0
This article is cited by
-
Synchronized LFP rhythmicity in the social brain reflects the context of social encounters
Communications Biology (2024)
-
The dynamic state of a prefrontal–hypothalamic–midbrain circuit commands behavioral transitions
Nature Neuroscience (2024)
-
Activation of lateral preoptic neurons is associated with nest-building in male mice
Scientific Reports (2024)
-
Galaninergic and hypercapnia-activated neuronal projections to the ventral respiratory column
Brain Structure and Function (2024)
-
The medial preoptic area mediates depressive-like behaviors induced by ovarian hormone withdrawal through distinct GABAergic projections
Nature Neuroscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.