Abstract
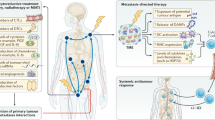
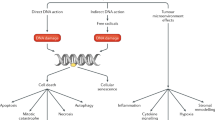
The majority of patients with prostate cancer who later develop lethal metastatic disease have high-risk localized disease at presentation, emphasizing the importance of effective treatment strategies at this stage. Multimodal treatment approaches that combine systemic and local therapies offer a promising strategy for improving the clinical outcomes of patients with high-risk localized prostate cancer. Combinations of neoadjuvant and adjuvant chemotherapy, hormonal therapy, or chemohormonal therapy are considered to be the standard of care in most solid tumours and should be investigated in the future for the treatment of prostate cancer to improve patient outcomes. However, although the combination of androgen deprivation therapy and radiotherapy is a standard of care in high-risk localized or locally advanced prostate cancer, the benefit of chemotherapy or chemohormonal therapy has yet to be demonstrated outside of the metastatic setting. Moreover, the benefit of neoadjuvant and/or adjuvant systemic therapies in combination with radical prostatectomy has not been proved. The development of next-generation hormonal agents, which have been approved for the treatment of castration-resistant prostate cancer, offers further therapeutic possibilities that are being assessed in early-phase clinical trials.
Key points
-
Patients with high-risk localized prostate cancer have a poor prognosis and require an appropriate treatment strategy that considers a multimodal approach and includes both local and systemic therapies.
-
No benefit has been proved for any neoadjuvant or adjuvant hormonal therapy in the case of radical prostatectomy.
-
No benefit has been proved for chemotherapy administered before or after radical prostatectomy.
-
Long-term androgen deprivation therapy in combination with radiotherapy was shown to markedly improve survival outcomes and should therefore be proposed for patients with high-risk localized prostate cancer.
-
Additional evidence of the benefit of chemohormonal therapy in combination with radiotherapy is required in the neoadjuvant and adjuvant settings.
-
Next-generation hormone therapies are still under investigation in the neoadjuvant or adjuvant settings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Winter, A. et al. Comparison of prostate cancer survival in Germany and the United States: can differences be attributed to differences in stage distributions? BJU Int. 119, 550–559 (2016).
D’Amico, A. V. et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280, 969–974 (1998).
Cooperberg, M. R., Broering, J. M. & Carroll, P. R. Time trends and local variation in primary treatment of localized prostate cancer. J. Clin. Oncol. 28, 1117–1123 (2010).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 66, 7–30 (2016).
Patrikidou, A. et al. Who dies from prostate cancer? Prostate Cancer Prostatic Dis. 17, 348–352 (2014).
Joniau, S. et al. Stratification of high-risk prostate cancer into prognostic categories: a European multi-institutional study. Eur. Urol. 67, 157–164 (2015).
Bach, C. et al. The status of surgery in the management of high-risk prostate cancer. Nat. Rev. Urol. 11, 342–351 (2014).
Mottet, N. et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur. Urol. 71, 618–629 (2017).
Gillessen, S. et al. Management of patients with advanced prostate cancer: recommendations of the St Gallen Advanced Prostate Cancer Consensus Conference (APCCC) 2015. Ann. Oncol. 26, 1589–1604 (2015).
Budaus, L. et al. Inverse stage migration in patients undergoing radical prostatectomy: results of 8916 European patients treated within the last decade. BJU Int. 108, 1256–1261 (2011).
Silberstein, J. L. et al. Reverse stage shift at a tertiary care center: escalating risk in men undergoing radical prostatectomy. Cancer 117, 4855–4860 (2011).
Tsai, H. K., D’Amico, A. V., Sadetsky, N., Chen, M. H. & Carroll, P. R. Androgen deprivation therapy for localized prostate cancer and the risk of cardiovascular mortality. J. Natl Cancer Inst. 99, 1516–1524 (2007).
Bosco, C. et al. Quantifying observational evidence for risk of fatal and nonfatal cardiovascular disease following androgen deprivation therapy for prostate cancer: a meta-analysis. Eur. Urol. 68, 386–396 (2015).
D’Amico, A. V. et al. Cancer-specific mortality after surgery or radiation for patients with clinically localized prostate cancer managed during the prostate-specific antigen era. J. Clin. Oncol. 21, 2163–2172 (2003).
Cooperberg, M. R., Vickers, A. J., Broering, J. M. & Carroll, P. R. Comparative risk-adjusted mortality outcomes after primary surgery, radiotherapy, or androgen-deprivation therapy for localized prostate cancer. Cancer 116, 5226–5234 (2010).
Zelefsky, M. J. et al. Metastasis after radical prostatectomy or external beam radiotherapy for patients with clinically localized prostate cancer: a comparison of clinical cohorts adjusted for case mix. J. Clin. Oncol. 28, 1508–1513 (2010).
Sooriakumaran, P. et al. Comparative effectiveness of radical prostatectomy and radiotherapy in prostate cancer: observational study of mortality outcomes. BMJ 348, g1502 (2014).
Petrelli, F. et al. Radical prostatectomy or radiotherapy in high-risk prostate cancer: a systematic review and metaanalysis. Clin. Genitourin. Cancer 12, 215–224 (2014).
Sweeney, C. J. et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N. Engl. J. Med. 373, 737–746 (2015).
Vale, C. L., Burdett, S. & Rydzewska, L. H. Correction to Lancet oncology 17: 248, 252. Addition of docetaxel or bisphosphonates to standard of care in men with localised or metastatic, hormone-sensitive prostate cancer: a systematic review and meta-analyses of aggregate data. Lancet Oncol. 17, 243–256 (2016).
James, N. D. et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 387, 1163–1177 (2016).
Tannock, I. F. et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 351, 1502–1512 (2004).
Fizazi, K. et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 13, 983–992 (2012).
Ryan, C. J. et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 16, 152–160 (2015).
Beer, T. M. et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N. Engl. J. Med. 371, 424–433 (2014).
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012).
de Bono, J. S. et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet 376, 1147–1154 (2010).
Bolla, M. et al. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 11, 1066–1073 (2010).
Roach, M. 3rd et al. Short-term neoadjuvant androgen deprivation therapy and external-beam radiotherapy for locally advanced prostate cancer: long-term results of RTOG 8610. J. Clin. Oncol. 26, 585–591 (2008).
Pilepich, M. V. et al. Androgen suppression adjuvant to definitive radiotherapy in prostate carcinoma —long-term results of phase III RTOG 85–31. Int. J. Radiat. Oncol. Biol. Phys. 61, 1285–1290 (2005).
Horwitz, E. M. et al. Ten-year follow-up of radiation therapy oncology group protocol 92-02: a phase III trial of the duration of elective androgen deprivation in locally advanced prostate cancer. J. Clin. Oncol. 26, 2497–2504 (2008).
D’Amico, A. V. et al. 6-Month androgen suppression plus radiation therapy versus radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA 292, 821–827 (2004).
Denham, J. W. et al. Short-term neoadjuvant androgen deprivation and radiotherapy for locally advanced prostate cancer: 10-year data from the TROG 96.01 randomised trial. Lancet Oncol. 12, 451–459 (2011).
Bolla, M. et al. Duration of androgen suppression in the treatment of prostate cancer. N. Engl. J. Med. 360, 2516–2527 (2009).
Widmark, A. et al. Endocrine treatment, with or without radiotherapy, in locally advanced prostate cancer (SPCG-7/SFUO-3): an open randomised phase III trial. Lancet 373, 301–308 (2009).
McKay, R. R., Choueiri, T. K. & Taplin, M. E. Rationale for and review of neoadjuvant therapy prior to radical prostatectomy for patients with high-risk prostate cancer. Drugs 73, 1417–1430 (2013).
Cha, E. K. & Eastham, J. A. Chemotherapy and novel therapeutics before radical prostatectomy for high-risk clinically localized prostate cancer. Urol. Oncol. 33, 217–225 (2015).
Pietzak, E. J. & Eastham, J. A. Neoadjuvant treatment of high-risk, clinically localized prostate cancer prior to radical prostatectomy. Curr. Urol. Rep. 17, 37 (2016).
Lou, D. Y. & Fong, L. Neoadjuvant therapy for localized prostate cancer: examining mechanism of action and efficacy within the tumor. Urol. Oncol. 34, 182–192 (2016).
Dreicer, R. et al. Phase II trial of neoadjuvant docetaxel before radical prostatectomy for locally advanced prostate cancer. Urology 63, 1138–1142 (2004).
Febbo, P. G. et al. Neoadjuvant docetaxel before radical prostatectomy in patients with high-risk localized prostate cancer. Clin. Cancer Res. 11, 5233–5240 (2005).
Magi-Galluzzi, C., Zhou, M., Reuther, A. M., Dreicer, R. & Klein, E. A. Neoadjuvant docetaxel treatment for locally advanced prostate cancer: a clinicopathologic study. Cancer 110, 1248–1254 (2007).
Chi, K. N. et al. Multicenter phase II study of combined neoadjuvant docetaxel and hormone therapy before radical prostatectomy for patients with high risk localized prostate cancer. J. Urol. 180, 565–570; discussion 570 (2008).
Prayer-Galetti, T. et al. Long-term follow-up of a neoadjuvant chemohormonal taxane-based phase II trial before radical prostatectomy in patients with non-metastatic high-risk prostate cancer. BJU Int. 100, 274–280 (2007).
Narita, S. et al. Short-term clinicopathological outcome of neoadjuvant chemohormonal therapy comprising complete androgen blockade, followed by treatment with docetaxel and estramustine phosphate before radical prostatectomy in Japanese patients with high-risk localized prostate cancer. World J. Surg. Oncol. 10, 1 (2012).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02543255 (2015).
Eastham, J. A. et al. Cancer and Leukemia Group B (CALGB) 90203: a randomized phase 3 study of radical prostatectomy alone versus estramustine and docetaxel before radical prostatectomy for patients with high-risk localized disease. Urology 62 (Suppl. 1), 55–62 (2003).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00430183 (2017).
Fujii, T. et al. Effectiveness of an adjuvant chemotherapy regimen for early-stage breast cancer: a systematic review and network meta-analysis. JAMA Oncol. 1, 1311–1318 (2015).
Wu, X. et al. Postoperative adjuvant chemotherapy for stage II colorectal cancer: a systematic review of 12 randomized controlled trials. J. Gastrointest. Surg. 16, 646–655 (2012).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00376792 (2018).
Ahlgren, G. et al. A randomized phase III trial between adjuvant docetaxel and surveillance after radical prostatectomy for high risk prostate cancer: results of SPCG12 [abstract 5001]. J. Clin. Oncol. https://doi.org/10.1200/JCO.2016.34.15_suppl.5001 (2016).
Schweizer, M. T. et al. Adjuvant leuprolide with or without docetaxel in patients with high-risk prostate cancer after radical prostatectomy (TAX-3501): important lessons for future trials. Cancer 119, 3610–3618 (2013).
Flaig, T. W. et al. Randomization reveals unexpected acute leukemias in Southwest Oncology Group prostate cancer trial. J. Clin. Oncol. 26, 1532–1536 (2008).
Dorff, T. B. et al. Adjuvant androgen deprivation for high-risk prostate cancer after radical prostatectomy: SWOG S9921 study. J. Clin. Oncol. 29, 2040–2045 (2011).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00132301 (2016).
Dalkin, B. L., Ahmann, F. R., Nagle, R. & Johnson, C. S. Randomized study of neoadjuvant testicular androgen ablation therapy before radical prostatectomy in men with clinically localized prostate cancer. J. Urol. 155, 1357–1360 (1996).
Labrie, F. et al. Neoadjuvant hormonal therapy: the Canadian experience. Urology 49, 56–64 (1997).
Fair, W. R., Rabbani, F., Bastar, A. & Betancourt, J. Neoadjuvant hormone therapy before radical prostatectomy: update on the Memorial Sloan-Kettering Cancer Center trials. Mol. Urol. 3, 253–260 (1999).
Schulman, C. C. et al. 4-Year follow-up results of a European prospective randomized study on neoadjuvant hormonal therapy prior to radical prostatectomy in T2-3N0M0 prostate cancer. European study group on neoadjuvant treatment of prostate cancer. Eur. Urol. 38, 706–713 (2000).
Soloway, M. S. et al. Neoadjuvant androgen ablation before radical prostatectomy in cT2bNxMo prostate cancer: 5-year results. J. Urol. 167, 112–116 (2002).
Selli, C. et al. Effects of complete androgen blockade for 12 and 24 weeks on the pathological stage and resection margin status of prostate cancer. J. Clin. Pathol. 55, 508–513 (2002).
Aus, G. et al. Three-month neoadjuvant hormonal therapy before radical prostatectomy: a 7-year follow-up of a randomized controlled trial. BJU Int. 90, 561–566 (2002).
Klotz, L. H. et al. Long-term followup of a randomized trial of 0 versus 3 months of neoadjuvant androgen ablation before radical prostatectomy. J. Urol. 170, 791–794 (2003).
Prezioso, D., Lotti, T., Polito, M. & Montironi, R. Neoadjuvant hormone treatment with leuprolide acetate depot 3.75 mg and cyproterone acetate, before radical prostatectomy: a randomized study. Urol. Int. 72, 189–195 (2004).
Yee, D. S. et al. Long-term follow-up of 3-month neoadjuvant hormone therapy before radical prostatectomy in a randomized trial. BJU Int. 105, 185–190 (2010).
Gravina, G. L. et al. Surgical and biologic outcomes after neoadjuvant bicalutamide treatment in prostate cancer. Urology 70, 728–733 (2007).
van der Kwast, T. H. et al. Prolonged neoadjuvant combined androgen blockade leads to a further reduction of prostatic tumor volume: three versus six months of endocrine therapy. Urology 53, 523–529 (1999).
Gleave, M. E. et al. Randomized comparative study of 3 versus 8-month neoadjuvant hormonal therapy before radical prostatectomy: biochemical and pathological effects. J. Urol. 166, 500–506; discussion 506–507 (2001).
Shelley, M. D. et al. A systematic review and meta-analysis of randomised trials of neo-adjuvant hormone therapy for localised and locally advanced prostate carcinoma. Cancer Treat. Rev. 35, 9–17 (2009).
Hu, J. et al. Neo-adjuvant hormone therapy for non-metastatic prostate cancer: a systematic review and meta-analysis of 5,194 patients. World J. Surg. Oncol. 13, 73 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01542021 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01674270 (2015).
Saad, F. et al. Efficacy outcomes by baseline prostate-specific antigen quartile in the AFFIRM trial. Eur. Urol. 67, 223–230 (2015).
Taplin, M. E. et al. Intense androgen-deprivation therapy with abiraterone acetate plus leuprolide acetate in patients with localized high-risk prostate cancer: results of a randomized phase II neoadjuvant study. J. Clin. Oncol. 32, 3705–3715 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01946165 (2017).
Efstathiou, E. et al. Neoadjuvant enzalutamide (ENZA) and abiraterone acetate (AA) plus leuprolide acetate (LHRHa) versus AA+ LHRHa in localized high-risk prostate cancer (LHRPC) [abstract 5002]. J. Clin. Oncol. https://doi.org/10.1200/JCO.2016.34.15_suppl.5002 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01547299 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02268175 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02903368 (2018).
Hsu, C.-Y., Joniau, S. & Van Poppel, H. Radical prostatectomy for locally advanced prostate cancer: technical aspects of radical prostatectomy. EAU Update Series 3, 90–97 (2005).
Soulie, M. et al. [Surgery of locally advanced prostate cancer]. Prog. Urol. 18, 1031–1037 (2008).
Donohue, J. F. et al. Poorly differentiated prostate cancer treated with radical prostatectomy: long-term outcome and incidence of pathological downgrading. J. Urol. 176, 991–995 (2006).
Roehl, K. A., Han, M., Ramos, C. G., Antenor, J. A. & Catalona, W. J. Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3,478 consecutive patients: long-term results. J. Urol. 172, 910–914 (2004).
Mason, M. D. et al. Final report of the intergroup randomized study of combined androgen-deprivation therapy plus radiotherapy versus androgen-deprivation therapy alone in locally advanced prostate cancer. J. Clin. Oncol. 33, 2143–2150 (2015).
Messing, E. M. et al. Immediate versus deferred androgen deprivation treatment in patients with node-positive prostate cancer after radical prostatectomy and pelvic lymphadenectomy. Lancet Oncol. 7, 472–479 (2006).
Wirth, M. P. et al. Prospective randomized trial comparing flutamide as adjuvant treatment versus observation after radical prostatectomy for locally advanced, lymph node-negative prostate cancer. Eur. Urol. 45, 267–270; discussion 270 (2004).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01442246 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01927627 (2017).
Fizazi, K. et al. Androgen deprivation therapy plus docetaxel and estramustine versus androgen deprivation therapy alone for high-risk localised prostate cancer (GETUG 12): a phase 3 randomised controlled trial. Lancet Oncol. 16, 787–794 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01952223 (2016).
Rosenthal, S. A. et al. A phase 3 trial of 2 years of androgen suppression and radiation therapy with or without adjuvant chemotherapy for high-risk prostate cancer: final results of radiation therapy oncology group phase 3 randomized trial NRG oncology RTOG 9902. Int. J. Radiat. Oncol. Biol. Phys. 93, 294–302 (2015).
Sandler, H. M. et al. A phase III protocol of androgen suppression (AS) and 3DCRT/IMRT versus AS and 3DCRT/IMRT followed by chemotherapy (CT) with doxetaxel and prednisone for localized, high-risk prostate cancer (RTOG 0521) [abstract LBA5022]. J. Clin. Oncol. https://doi.org/10.1200/jco.2015.33.18_suppl.lba5002 (2015).
Vale, C. L. et al. Addition of docetaxel or bisphosphonates to standard of care in men with localised or metastatic, hormone-sensitive prostate cancer: a systematic review and meta-analyses of aggregate data. Lancet Oncol. 17, 243–256 (2016).
Zapatero, A. et al. High-dose radiotherapy with short-term or long-term androgen deprivation in localised prostate cancer (DART01/05 GICOR): a randomised, controlled, phase 3 trial. Lancet Oncol. 16, 320–327 (2015).
D’Amico, A. V., Chen, M. H., Renshaw, A. A., Loffredo, M. & Kantoff, P. W. Androgen suppression and radiation versus radiation alone for prostate cancer: a randomized trial. JAMA 299, 289–295 (2008).
D’Amico, A. V., Chen, M. H., Renshaw, A., Loffredo, M. & Kantoff, P. W. Long-term follow-up of a randomized trial of radiation with or without androgen deprivation therapy for localized prostate cancer. JAMA 314, 1291–1293 (2015).
Konaka, H. et al. Tri-Modality therapy with I-125 brachytherapy, external beam radiation therapy, and short- or long-term hormone therapy for high-risk localized prostate cancer (TRIP): study protocol for a phase III, multicenter, randomized, controlled trial. BMC Cancer 12, 110 (2012).
Cho, E. et al. External beam radiation therapy and abiraterone in men with localized prostate cancer: safety and effect on tissue androgens. Int. J. Radiat. Oncol. Biol. Phys. 92, 236–243 (2015).
James, N. D. et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N. Engl. J. Med. 377, 338–351 (2017).
James, N. et al. Adding abiraterone for patients (pts) with high-risk prostate cancer (PCa) starting long-term androgen deprivation therapy (ADT): Outcomes in non-metastatic (M0) patients from STAMPEDE (NCT00268476) [abstract LBA34]. Ann. Oncol. https://doi.org/10.1093/annonc/mdx440.027 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02446444 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02064582 (2016).
Albertsen, P. C., Hanley, J. A. & Fine, J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA 293, 2095–2101 (2005).
Xie, W. et al. Metastasis-free survival is a strong surrogate of overall survival in localized prostate cancer. J. Clin. Oncol. 35, 3097–3104 (2017).
Hussain, M. et al. Neoadjuvant docetaxel and estramustine chemotherapy in high-risk/locallyadvanced prostate cancer. Urology 61, 774–780 (2003).
Friedman, J. et al. Neoadjuvant docetaxel and capecitabine in patients with high risk prostate cancer. J. Urol. 179, 911–915;discussion 915–916 (2008).
Sella, A. et al. Neoadjuvant chemohormonal therapy in poor-prognosis localized prostate cancer. Urology 71, 323–327 (2008).
Mellado, B. et al. Phase II trial of short-term neoadjuvant docetaxel and complete androgen blockade in high-risk prostate cancer. Br. J. Cancer 101, 1248–1252 (2009).
Vuky, J. et al. Phase II trial of neoadjuvant docetaxel and gefitinib followed by radical prostatectomy in patients with high-risk, locally advanced prostate cancer. Cancer 115, 784–791 (2009).
Garzotto, M. et al. Phase 1/2 study of preoperative docetaxel and mitoxantrone for high-risk prostate cancer. Cancer 116, 1699–1708 (2010).
Womble, P. R. et al. A phase II clinical trial of neoadjuvant ketoconazole and docetaxel chemotherapy before radical prostatectomy in high risk patients. J. Urol. 186, 882–887 (2011).
Ross, R. W. et al. Phase 2 study of neoadjuvant docetaxel plus bevacizumab in patients with high-risk localized prostate cancer: a prostate cancer clinical trials consortium trial. Cancer 118, 4777–4784 (2012).
Thalgott, M. et al. Long-term results of a phase II study with neoadjuvant docetaxel chemotherapy and complete androgen blockade in locally advanced and high-risk prostate cancer. J. Hematol. Oncol. 7, 20 (2014).
Zhao, B., Yerram, N. K., Gao, T., Dreicer, R. & Klein, E. A. Long-term survival of patients with locally advanced prostate cancer managed with neoadjuvant docetaxel and radical prostatectomy. Urol. Oncol. 33, 164 e119–164 e123 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02160353 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02028988 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01717053 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02023463 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02508636 (2016).
Acknowledgements
The authors thank the KPL medical team for help with the preparation of this manuscript. The authors acknowledge the institutional support from Janssen. The authors are fully responsible for the content of and editorial decisions for this manuscript.
Review criteria
An analytical review of the literature was conducted in September 2016 using the PubMed database, with no time limit, using the following keywords arranged in various combinations: “localized prostate cancer”, “non-metastatic castration-resistant prostate cancer”, “multimodal treatment”, “chemotherapy”, “prostatectomy”, “adjuvant”, “neoadjuvant”, “abiraterone acetate”, and “enzalutamide”. From this research, 239 articles were identified, restricted to full-text English-language, covering retrospective and prospective studies, as well as randomized controlled trials and meta-analyses on hormonal therapies and/or chemotherapy in the adjuvant or neoadjuvant setting. The abstracts of these articles were reviewed. Relevant articles with pathological (pathological complete response) or oncological (progression-free survival and overall survival) results were included in the analysis. Five abstracts from international congresses were also considered contributory and corresponding to the research carried out. Finally, 40 articles were selected and organized according to the local treatment performed, prostatectomy, or radiotherapy. Relevant references cited in the selected full-text articles were also added to the analysis. Thus, the final number of references was 62. Ongoing trials on hormonal and/or chemotherapy in the adjuvant or neoadjuvant setting were searched for on the clinicaltrials.gov website and are also presented.
Author information
Authors and Affiliations
Contributions
G. Pignot, D.M., E.G., and D.B. researched data for the article and made substantial contributions to discussion of the content. G. Pignot, D.M., and D.B. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
G. Pignot is a consultant for Sanofi and Janssen. D.M. is a consultant for Janssen. E.G. is a consultant for Janssen and Sanofi. J.-B.B. is a consultant for Astellas, Ferring, Ipsen, and Janssen. F.C.-S. is a consultant for Astellas, Janssen, and Sanofi. Y.L. is a consultant for Astellas, Janssen, Sanofi, MSD, Roche, and AstraZeneca and has received research funding from Sanofi. G. Ploussard is an advisory board member and a consultant for Astellas, Ferring, IPSEN, Janssen, and Pierre Fabre. P.S. is a consultant and expert for Astellas, AstraZeneca, Ferring, Ipsen, Janssen, Nanobiotix, Roche, Sanofi, and Takeda. M.-O.T. was an invited speaker in symposia organized by Janssen, Astellas, and Pfizer. S.V. is a consultant for Astellas, Janssen, and Sanofi. G. Pasticier is a consultant for Astellas, Janssen, and Sanofi. D.B. is a consultant and advisory board member for Janssen, Sanofi, and Ipsen.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Kattans’ nomogram: https://www.mskcc.org/nomograms/prostate
Rights and permissions
About this article
Cite this article
Pignot, G., Maillet, D., Gross, E. et al. Systemic treatments for high-risk localized prostate cancer. Nat Rev Urol 15, 498–510 (2018). https://doi.org/10.1038/s41585-018-0017-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-018-0017-x
This article is cited by
-
The nerve growth factor-delivered signals in prostate cancer and its associated microenvironment: when the dialogue replaces the monologue
Cell & Bioscience (2023)
-
Suboptimal use of hormonal therapy among German men with localized high-risk prostate Cancer during 2005 to 2015: analysis of registry data
BMC Cancer (2022)
-
Health-related quality of life and self-reported cognitive function in patients with delayed neurocognitive recovery after radical prostatectomy: a prospective follow-up study
Health and Quality of Life Outcomes (2021)
-
The androgen receptor/filamin A complex as a target in prostate cancer microenvironment
Cell Death & Disease (2021)
-
Considering the role of radical prostatectomy in 21st century prostate cancer care
Nature Reviews Urology (2020)