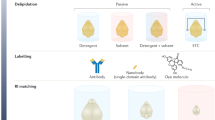
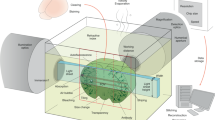
Abstract
State-of-the-art tissue-clearing methods provide subcellular-level optical access to intact tissues from individual organs and even to some entire mammals. When combined with light-sheet microscopy and automated approaches to image analysis, existing tissue-clearing methods can speed up and may reduce the cost of conventional histology by several orders of magnitude. In addition, tissue-clearing chemistry allows whole-organ antibody labelling, which can be applied even to thick human tissues. By combining the most powerful labelling, clearing, imaging and data-analysis tools, scientists are extracting structural and functional cellular and subcellular information on complex mammalian bodies and large human specimens at an accelerated pace. The rapid generation of terabyte-scale imaging data furthermore creates a high demand for efficient computational approaches that tackle challenges in large-scale data analysis and management. In this Review, we discuss how tissue-clearing methods could provide an unbiased, system-level view of mammalian bodies and human specimens and discuss future opportunities for the use of these methods in human neuroscience.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
09 March 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41583-020-0291-5
References
Spalteholz, W. Über das Durchsichtigmachen von Menschlichen und Tierischen Präparaten. (S. Hirzel, 1914).
Chung, K. et al. Structural and molecular interrogation of intact biological systems. Nature 497, 332–337 (2013). This article is the first to demonstrate in situ synthesis of a hydrogel and its fusion with tissue via covalent bonds to form a cleared tissue–hydrogel hybrid.
Yang, B. et al. Single-cell phenotyping within transparent intact tissue through whole-body clearing. Cell 158, 945–958 (2014). This article first reports clearing of adult rodents via the vasculature; RNA detection in cleared tissues with single-molecule resolution by single-molecule fluorescence in situ hybridization and that specific formulations of tissue–hydrogel hybrids can significantly expand.
Renier, N. et al. iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 159, 896–910 (2014).
Susaki, E. A. et al. Whole-brain imaging with single-cell resolution using chemical cocktails and computational analysis. Cell 157, 726–739 (2014). This article first reports on the delipidation activity of amino alcohols and the high-performance hydrophilic tissue-clearing method CUBIC and reports tissue clearing of the marmoset brain.
Tainaka, K. et al. Whole-body imaging with single-cell resolution by tissue decolorization. Cell 159, 911–924 (2014). This article first demonstrates that decolourization of haem is possible in mild chemical conditions by amino alcohols, and reports whole-body imaging of the adult mouse.
Ertürk, A. et al. Three-dimensional imaging of the unsectioned adult spinal cord to assess axon regeneration and glial responses after injury. Nat. Med. 18, 166–171 (2012). This work demonstrates the application of an organic solvent-clearing method (3DISCO) on the intact adult mouse CNS to study neurodegeneration and regeneration.
Belle, M. et al. A simple method for 3D analysis of immunolabeled axonal tracts in a transparent nervous system. Cell Rep. 9, 1191–1201 (2014). This work is the first to combine whole-mount immunostaining, 3DISCO clearing and light-sheet microscopy to analyse axon guidance defects in mutant mice.
Treweek, J. B. et al. Whole-body tissue stabilization and selective extractions via tissue-hydrogel hybrids for high-resolution intact circuit mapping and phenotyping. Nat. Protoc. 10, 1860–1896 (2015).
Costantini, I. et al. A versatile clearing agent for multi-modal brain imaging. Sci. Rep. 5, 9808 (2015).
Klingberg, A. et al. Fully automated evaluation of total glomerular number and capillary tuft size in nephritic kidneys using lightsheet microscopy. J. Am. Soc. Nephrol. 28, 452–459 (2017).
Hama, H. et al. Scale: a chemical approach for fluorescence imaging and reconstruction of transparent mouse brain. Nat. Neurosci. 14, 1481–1488 (2011).
Ke, M.-T., Fujimoto, S. & Imai, T. SeeDB: a simple and morphology-preserving optical clearing agent for neuronal circuit reconstruction. Nat. Neurosci. 16, 1154–1161 (2013).
Dodt, H. U. et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat. Methods 4, 331–336 (2007).
Susaki, E. A. & Ueda, H. R. Whole-body and whole-organ clearing and imaging techniques with single-cell resolution: toward organism-level systems biology in mammals. Cell Chem. Biol. 23, 137–157 (2016).
Tainaka, K., Kuno, A., Kubota, S. I., Murakami, T. & Ueda, H. R. Chemical principles in tissue clearing and staining protocols for whole-body cell profiling. Annu. Rev. Cell Dev. Biol. 32, 713–741 (2016).
Murakami, T. C. et al. A three-dimensional single-cell-resolution whole-brain atlas using CUBIC-X expansion microscopy and tissue clearing. Nat. Neurosci. 21, 625–637 (2018). This article first demonstrates the polymer-gel property of a fixed mouse brain in the absence of exogenous polymers, reports expansion microscopy using hydrophilic reagents and reports the generation of a 3D single-cell-resolution mouse brain atlas, the CUBIC-Atlas.
Amiya, T. & Tanaka, T. Phase transitions in crosslinked gels of natural polymers. Macromolecules 20, 1162–1164 (1987).
Shibayama, M. & Tanaka, T. in Responsive Gels: Volume Transitions I 1–62 (Springer, 1993).
Lorentz, H. A. Ueber die Beziehung zwischen der Fortpflanzungsgeschwindigkeit des Lichtes und der Körperdichte. Ann. Phys. 9, 641–665 (1880).
Lorenz, L. Ueber die Refractionsconstante. Ann. Phys. 11, 70–103 (1880).
Tainaka, K. et al. Chemical landscape for tissue clearing based on hydrophilic reagents. Cell Rep. 24, 2196–2210 (2018). This article first reports the chemical profiling of hydrophilic tissue-clearing reagents and proposes chemical principles of hydrophilic tissue-clearing methods.
Belle, M. et al. Tridimensional visualization and analysis of early human development. Cell 169, 161–173 e112 (2017). This study uses 3D imaging of cleared human embryos and fetuses to provide the first comprehensive description of the development of the human peripheral nervous system and the innervation pattern of many organs in intact specimens.
Kim, S. Y. et al. Stochastic electrotransport selectively enhances the transport of highly electromobile molecules. Proc. Natl Acad. Sci. USA 112, E6274–E6283 (2015).
Murray, E. et al. Simple, scalable proteomic imaging for high-dimensional profiling of intact systems. Cell 163, 1500–1514 (2015). This article is the first to demonstrate that controlling the chemical interaction time and kinetics achieves more uniform and scalable processing of large-scale tissues.
Renier, N. et al. Mapping of brain activity by automated volume analysis of immediate early genes. Cell 165, 1789–1802 (2016).
Kubota, S. I. et al. Whole-body profiling of cancer metastasis with single-cell resolution. Cell Rep. 20, 236–250 (2017).
Cai, R. et al. Panoptic imaging of transparent mice reveals whole-body neuronal projections and skull–meninges connections. Nat. Neurosci. 22, 317–327 (2019). This study shows that vDISCO boosted fluorescent protein signal in intact transparent mice, thereby allowing detection of widespread CNS trauma effects, and reveals short vascular connections between the skull marrow and brain meninges.
Deverman, B. E. et al. Cre-dependent selection yields AAV variants for widespread gene transfer to the adult brain. Nat. Biotechnol. 34, 204–209 (2016). This is the first article to report efficient crossing of the blood–brain barrier in adult rodents by an engineered adeno-associated virus vector able to package the cargo of choice. A Cre-enzyme-based viral-vector screening method is introduced and it is demonstrated how whole-body tissue clearing can facilitate transduction maps of systemically delivered genes.
Bedbrook, C. N., Deverman, B. E. & Gradinaru, V. Viral strategies for targeting the central and peripheral nervous systems. Annu. Rev. Neurosci. 41, 323–348 (2018).
Keller, P. J. & Ahrens, M. B. Visualizing whole-brain activity and development at the single-cell level using light-sheet microscopy. Neuron 85, 462–483 (2015).
Vigouroux, R. J., Belle, M. & Chédotal, A. Neuroscience in the third dimension: shedding new light on the brain with tissue clearing. Mol. Brain 10, 33 (2017).
Ertürk, A. et al. Three-dimensional imaging of solvent-cleared organs using 3DISCO. Nat. Protoc. 7, 1983–1995 (2012).
Liu, Z. et al. Immune homeostasis enforced by co-localized effector and regulatory T cells. Nature 528, 225–230 (2015).
Acar, M. et al. Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature 526, 126–130 (2015).
Espinosa-Medina, I. et al. Parasympathetic ganglia derive from Schwann cell precursors. Science 345, 87–90 (2014).
Oshimori, N., Oristian, D. & Fuchs, E. TGF-β promotes heterogeneity and drug resistance in squamous cell carcinoma. Cell 160, 963–976 (2015).
Garofalo, S. et al. Enriched environment reduces glioma growth through immune and non-immune mechanisms in mice. Nat. Commun. 6, 6623 (2015).
von Neubeck, B. et al. An inhibitory antibody targeting carbonic anhydrase XII abrogates chemoresistance and significantly reduces lung metastases in an orthotopic breast cancer model in vivo. Int. J. Cancer 143, 2065–2075 (2018).
Tanaka, N. et al. Whole-tissue biopsy phenotyping of three-dimensional tumours reveals patterns of cancer heterogeneity. Nat. Biomed. Eng. 1, 796–806 (2017).
Garvalov, B. K. & Ertürk, A. Seeing whole-tumour heterogeneity. Nat. Biomed. Eng. 1, 772–774 (2017).
Pan, C. et al. Shrinkage-mediated imaging of entire organs and organisms using uDISCO. Nat. Methods 13, 859–867 (2016).
Herisson, F. et al. Direct vascular channels connect skull bone marrow and the brain surface enabling myeloid cell migration. Nat. Neurosci. 21, 1209–1217 (2018).
Pan, C. et al. Deep learning reveals cancer metastasis and therapeutic antibody targeting in whole body. Cell 179, 1661–1676.e19 (2019). This works describes a novel method to detect and quantify cancer metastasis and antibody drug targeting at cellular resolution in the entire mouse using whole-body clearing and deep learning.
Chiang, A. S. et al. Three-dimensional mapping of brain neuropils in the cockroach, Diploptera punctata. J. Comp. Neurol. 440, 1–11 (2001).
Liu, Y.-C. & Chiang, A.-S. High-resolution confocal imaging and three-dimensional rendering. Methods 30, 86–93 (2003).
Richardson, D. S. & Lichtman, J. W. Clarifying Tissue Clearing. Cell 162, 246–257 (2015).
Hirshburg, J., Choi, B., Nelson, J. S. & Yeh, A. T. Correlation between collagen solubility and skin optical clearing using sugars. Lasers Surg. Med. 39, 140–144 (2007).
Hama, H. et al. ScaleS: an optical clearing palette for biological imaging. Nat. Neurosci. 18, 1518–1529 (2015).
Chance, B., Liu, H., Kitai, T. & Zhang, Y. Effects of solutes on optical properties of biological materials: models, cells, and tissues. Anal. Biochem. 227, 351–362 (1995).
Ke, M.-T. et al. Super-resolution mapping of neuronal circuitry with an index-optimized clearing agent. Cell Rep. 14, 2718–2732 (2016).
Susaki, E. A. et al. Advanced CUBIC protocols for whole-brain and whole-body clearing and imaging. Nat. Protoc. 10, 1709–1727 (2015).
Tatsuki, F. et al. Involvement of Ca2+-dependent hyperpolarization in sleep duration in mammals. Neuron 90, 70–85 (2016).
Economo, M. N. et al. A platform for brain-wide imaging and reconstruction of individual neurons. eLife 5, e10566 (2016).
Wang, L. et al. The coding of valence and identity in the mammalian taste system. Nature 558, 127–131 (2018).
Justus, D. et al. Glutamatergic synaptic integration of locomotion speed via septoentorhinal projections. Nat. Neurosci. 20, 16–19 (2017).
Romanov, R. A. et al. Molecular interrogation of hypothalamic organization reveals distinct dopamine neuronal subtypes. Nat. Neurosci. 20, 176–188 (2017).
Lanjakornsiripan, D. et al. Layer-specific morphological and molecular differences in neocortical astrocytes and their dependence on neuronal layers. Nat. Commun. 9, 1623 (2018).
Rousso, D. L. et al. Two pairs of on and off retinal ganglion cells are defined by intersectional patterns of transcription factor expression. Cell Rep. 15, 1930–1944 (2016).
Chen, J. Y. et al. Hoxb5 marks long-term haematopoietic stem cells and reveals a homogenous perivascular niche. Nature 530, 223–227 (2016).
Cuccarese, M. F. et al. Heterogeneity of macrophage infiltration and therapeutic response in lung carcinoma revealed by 3D organ imaging. Nat. Commun. 8, 14293 (2017).
Davis, F. M. et al. Single-cell lineage tracing in the mammary gland reveals stochastic clonal dispersion of stem/progenitor cell progeny. Nat. Commun. 7, 13053 (2016).
Li, J. et al. Single-cell lineage tracing reveals that oriented cell division contributes to trabecular morphogenesis and regional specification. Cell Rep. 15, 158–170 (2016).
Yamamoto, J. et al. Neuronal signals regulate obesity induced beta-cell proliferation by FoxM1 dependent mechanism. Nat. Commun. 8, 1930 (2017).
Chen, F., Tillberg, P. W. & Boyden, E. S. Expansion microscopy. Science 347, 543–548 (2015). This article first reports the concept of expansion microscopy based on an exogenous hydrogel polymer.
Matsumoto, K. et al. Advanced CUBIC tissue clearing for whole-organ cell profiling. Nat. Protoc. 14, 3506–3537 (2019).
Tomer, R., Ye, L., Hsueh, B. & Deisseroth, K. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat. Protoc. 9, 1682–1697 (2014).
Gradinaru, V., Treweek, J., Overton, K. & Deisseroth, K. Hydrogel-tissue chemistry: principles and applications. Annu. Rev. Biophys. 47, 355–376 (2018).
Ku, T. et al. Multiplexed and scalable super-resolution imaging of three-dimensional protein localization in size-adjustable tissues. Nat. Biotechnol. 34, 973–981 (2016).
Sylwestrak, E. L., Rajasethupathy, P., Wright, M. A., Jaffe, A. & Deisseroth, K. Multiplexed intact-tissue transcriptional analysis at cellular resolution. Cell 164, 792–804 (2016).
Park, Y.-G. et al. Protection of tissue physicochemical properties using polyfunctional crosslinkers. Nat. Biotechnol. 37, 73–83 (2019). This article is the first to demonstrate protection of tissue and biomolecular properties against harsh stress using polyfunctional crosslinkers.
Renner, M. et al. Self-organized developmental patterning and differentiation in cerebral organoids. EMBO J. 36, 1316–1329 (2017).
Canter, R. G. et al. 3D mapping reveals network-specific amyloid progression and subcortical susceptibility in mice. Commun. Biol. 2, 360 (2019).
Greenbaum, A. et al. Bone CLARITY: clearing, imaging, and computational analysis of osteoprogenitors within intact bone marrow. Sci. Transl Med. 9, eaah6518 (2017). This article reports clearing of whole bone with preserved marrow and imaging endogenous fluorescence with single-cell resolution throughout the marrow by light-sheet microscopy.
Greenbaum, A., Jang, M. J., Challis, C. & Gradinaru, V. Q&A: how can advances in tissue clearing and optogenetics contribute to our understanding of normal and diseased biology? BMC Biol. 15, 87 (2017).
Shah, S. et al. Single-molecule RNA detection at depth via hybridization chain reaction and tissue hydrogel embedding and clearing. Development 143, 2862–2867 (2016).
DePas, W. H. et al. Exposing the three-dimensional biogeography and metabolic states of pathogens in cystic fibrosis sputum via hydrogel embedding, clearing, and rRNA labeling. mBio 7, e00796–e00816 (2016).
Treweek, J. B. & Gradinaru, V. Extracting structural and functional features of widely distributed biological circuits with single cell resolution via tissue clearing and delivery vectors. Curr. Opin. Biotechnol. 40, 193–207 (2016).
Menegas, W. et al. Dopamine neurons projecting to the posterior striatum form an anatomically distinct subclass. eLife 4, e10032 (2015).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 20, 1172–1179 (2017). This article reports novel viral vectors to deliver cargo to neurons throughout the brain and body via the bloodstream, and a complementary vector toolbox for sparse stochastic labelling for morphological assessment. These delivery methods for Golgi-like genetic labelling can complement tissue clearing and microscopy methods to yield refined maps of the nervous system across the brain and body.
Robinson, J. E. & Gradinaru, V. Dopaminergic dysfunction in neurodevelopmental disorders: recent advances and synergistic technologies to aid basic research. Curr. Opin. Neurobiol. 48, 17–29 (2018).
Liebmann, T. et al. Three-dimensional study of Alzheimer’s disease hallmarks using the iDISCO clearing method. Cell Rep. 16, 1138–1152 (2016).
Welniarz, Q. et al. Non cell-autonomous role of DCC in the guidance of the corticospinal tract at the midline. Sci. Rep. 7, 410 (2017).
Hruska, M., Henderson, N., Le Marchand, S. J., Jafri, H. & Dalva, M. B. Synaptic nanomodules underlie the organization and plasticity of spine synapses. Nat. Neurosci. 21, 671–682 (2018).
Ando, K. et al. Inside Alzheimer brain with CLARITY: senile plaques, neurofibrillary tangles and axons in 3-D. Acta Neuropathol. 128, 457–459 (2014).
Morawski, M. et al. Developing 3D microscopy with CLARITY on human brain tissue: towards a tool for informing and validating MRI-based histology. Neuroimage 182, 417–428 (2018).
Phillips, J. et al. Development of passive CLARITY and immunofluorescent labelling of multiple proteins in human cerebellum: understanding mechanisms of neurodegeneration in mitochondrial disease. Sci. Rep. 6, 26013 (2016).
Liu, A. K. L. et al. Bringing CLARITY to the human brain: visualization of Lewy pathology in three dimensions. Neuropathol. Appl. Neurobiol. 42, 573–587 (2016).
Lee, E. et al. ACT-PRESTO: rapid and consistent tissue clearing and labeling method for 3-dimensional (3D) imaging. Sci. Rep. 6, 18631 (2016).
Lai, H. M. et al. Next generation histology methods for three-dimensional imaging of fresh and archival human brain tissues. Nat. Commun. 9, 1066 (2018).
Allen, J. S., Damasio, H. & Grabowski, T. J. Normal neuroanatomical variation in the human brain: an MRI-volumetric study. Am. J. Phys. Anthropol. 118, 341–358 (2002).
Birey, F. et al. Assembly of functionally integrated human forebrain spheroids. Nature 545, 54–59 (2017).
Casoni, F. et al. Development of the neurons controlling fertility in humans: new insights from 3D imaging and transparent fetal brains. Development 143, 3969–3981 (2016).
Hsueh, B. et al. Pathways to clinical CLARITY: volumetric analysis of irregular, soft, and heterogeneous tissues in development and disease. Sci. Rep. 7, 5899 (2017).
Behjati, S., Lindsay, S., Teichmann, S. A. & Haniffa, M. Mapping human development at single-cell resolution. Development 145, dev152561 (2018).
Kieffer, C., Ladinsky, M. S., Ninh, A., Galimidi, R. P. & Bjorkman, P. J. Longitudinal imaging of HIV-1 spread in humanized mice with parallel 3D immunofluorescence and electron tomography. eLife 6, e23282 (2017).
Dantzer, R., O’Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci. 9, 46–56 (2008).
Glaser, A. K. et al. Light-sheet microscopy for slide-free non-destructive pathology of large clinical specimens. Nat. Biomed. Eng. 1, 0084 (2017).
Nojima, S. et al. CUBIC pathology: three-dimensional imaging for pathological diagnosis. Sci. Rep. 7, 9269 (2017).
Royen, M. E. et al. Three-dimensional microscopic analysis of clinical prostate specimens. Histopathology 69, 985–992 (2016).
Huisken, J., Swoger, J., Del Bene, F., Wittbrodt, J. & Stelzer, E. H. Optical sectioning deep inside live embryos by selective plane illumination microscopy. Science 305, 1007–1009 (2004).
Keller, P. J., Schmidt, A. D., Wittbrodt, J. & Stelzer, E. H. K. Reconstruction of zebrafish early embryonic development by scanned light sheet microscopy. Science 322, 1065–1069 (2008).
Voie, A. H., Burns, D. H. & Spelman, F. A. Orthogonal-plane fluorescence optical sectioning: three-dimensional imaging of macroscopic biological specimens. J. Microsc. 170, 229–236 (1993).
Ryan, D. P. et al. Automatic and adaptive heterogeneous refractive index compensation for light-sheet microscopy. Nat. Commun. 8, 612 (2017).
Tomer, R. et al. SPED light sheet microscopy: fast mapping of biological system structure and function. Cell 163, 1796–1806 (2015).
Gómez-Gaviro, M. V. et al. Optimized CUBIC protocol for 3D imaging of chicken embryos at single-cell resolution. Development 44, 2092–2097 (2017).
Stefaniuk, M. et al. Light-sheet microscopy imaging of a whole cleared rat brain with Thy1-GFP transgene. Sci. Rep. 6, 28209 (2016).
Niedworok, C. J. et al. Charting monosynaptic connectivity maps by two-color light-sheet fluorescence microscopy. Cell Rep. 2, 1375–1386 (2012).
Planchon, T. A. et al. Rapid three-dimensional isotropic imaging of living cells using Bessel beam plane illumination. Nat. Methods 8, 417–423 (2011).
Chen, B. C. et al. Lattice light-sheet microscopy: imaging molecules to embryos at high spatiotemporal resolution. Science 346, 1257998 (2014). This article describes lattice light-sheet microscopy, which uses very thin laser light sheets for fluorescence generation and introduces a light-efficient concept for high-resolution imaging of a field of view of approximately 100 µm.
Wu, Y. C. et al. Spatially isotropic four-dimensional imaging with dual-view plane illumination microscopy. Nat. Biotechnol. 31, 1032–1038 (2013). This article describes diSPIM light-sheet microscopy, which rapidly acquires two orthogonal views of a specimen with a field of view of several hundred micrometres and hence combines high imaging speeds with a high, spatially isotropic resolution of 330 nm.
Chhetri, R. K. et al. Whole-animal functional and developmental imaging with isotropic spatial resolution. Nat. Methods 12, 1171–1178 (2015). By allowing simultaneous orthogonal four-view imaging with a field of view approaching a 1 mm, IsoView light-sheet microscopy achieves a volume throughput of more than 108 µm3 per second and a high, spatially isotropic resolution of 400 nm.
Swoger, J., Verveer, P., Greger, K., Huisken, J. & Stelzer, E. H. Multi-view image fusion improves resolution in three-dimensional microscopy. Opt. Express 15, 8029–8042 (2007).
Royer, L. A. et al. Adaptive light-sheet microscopy for long-term, high-resolution imaging in living organisms. Nat. Biotechnol. 34, 1267–1278 (2016).
Royer, L. A., Lemon, W. C., Chhetri, R. K. & Keller, P. J. A practical guide to adaptive light-sheet microscopy. Nat. Protoc. 13, 2462–2500 (2018).
Silvestri, L. et al. RAPID: real-time image-based autofocus for all wide-field optical microscopy systems. Preprint at bioRxiv https://doi.org/10.1101/170555 (2017).
Hörl, D. et al. BigStitcher: reconstructing high-resolution image datasets of cleared and expanded samples. Nat. Methods 16, 870–874 (2019). This artcle describes BigStitcher, which offers highly customizable stitching of arbitrarily large image mosaics and multiview selective plane illumination microscopy data building on the BigDataViewer infrastructure in Fiji.
Gao, R. et al. Cortical column and whole-brain imaging with molecular contrast and nanoscale resolution. Science 363, eaau8302 (2019).
Dean, K. M., Roudot, P., Welf, E. S., Danuser, G. & Fiolka, R. Deconvolution-free subcellular imaging with axially swept light sheet microscopy. Biophys. J. 108, 2807–2815 (2015).
Pende, M. et al. High-resolution ultramicroscopy of the developing and adult nervous system in optically cleared Drosophila melanogaster. Nat. Commun. 9, 4731 (2018).
Amat, F. et al. Efficient processing and analysis of large-scale light-sheet microscopy data. Nat. Protoc. 10, 1679–1696 (2015).
Pietzsch, T., Saalfeld, S., Preibisch, S. & Tomancak, P. BigDataViewer: visualization and processing for large image data sets. Nat. Methods 12, 481–483 (2015). This article describes BigDataViewer, which provides a well-engineered software framework to navigate and compute on arbitrarily large image data sets that is integrated into the popular Fiji ecosystem.
Schindelin, J., Rueden, C. T., Hiner, M. C. & Eliceiri, K. W. The ImageJ ecosystem: an open platform for biomedical image analysis. Mol. Reprod. Dev. 82, 518–529 (2015).
Pietzsch, T., Preibisch, S., Tomančák, P. & Saalfeld, S. ImgLib2—generic image processing in Java. Bioinformatics 28, 3009–3011 (2012).
Preibisch, S., Saalfeld, S., Schindelin, J. & Tomancak, P. Software for bead-based registration of selective plane illumination microscopy data. Nat. Methods 7, 418–419 (2010).
Preibisch, S. et al. Efficient Bayesian-based multiview deconvolution. Nat. Methods 11, 645–648 (2014).
Balazs, B., Deschamps, J., Albert, M., Ries, J. & Hufnagel, L. A real-time compression library for microscopy images. Preprint at bioRxiv https://doi.org/10.1101/164624 (2017).
Cheeseman, B. L., Günther, U., Susik, M., Gonciarz, K. & Sbalzarini, I. F. Forget pixels: adaptive particle representation of fluorescence microscopy images. Nat. Commun. 9, 5160 (2018).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Tomer, R., Denes, A. S., Tessmar-Raible, K. & Arendt, D. Profiling by image registration reveals common origin of annelid mushroom bodies and vertebrate pallium. Cell 142, 800–809 (2010).
Heckscher, E. S. et al. Atlas-builder software and the eNeuro atlas: resources for developmental biology and neuroscience. Development 141, 2524–2532 (2014).
Ronneberger, O. et al. ViBE-Z: a framework for 3D virtual colocalization analysis in zebrafish larval brains. Nat. Methods 9, 735–742 (2012).
Bogovic, J. A., Hanslovsky, P., Wong, A. & Saalfeld, S. in 2016 IEEE 13th International Symposium on Biomedical Imaging (ISBI) 1123–1126 (IEEE, 2016).
Zheng, Z. et al. A complete electron microscopy volume of the brain of adult Drosophila melanogaster. Cell 174, 730–743.e22 (2017).
Sommer, C. S., Kothe C., Hamprecht U. & Ilastik F. A. in 2011 IEEE International Symposium on Biomedical Imaging: From Nano to Macro 230–233 (IEEE, 2011).
Fürth, D. et al. An interactive framework for whole-brain maps at cellular resolution. Nat. Neurosci. 21, 139–149 (2018).
Oh, S. W. et al. A mesoscale connectome of the mouse brain. Nature 508, 207–214 (2014).
Lein, E. S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
Saalfeld, S., Cardona, A., Hartenstein, V. & Tomancak, P. CATMAID: collaborative annotation toolkit for massive amounts of image data. Bioinformatics 25, 1984–1986 (2009).
Schneider-Mizell, C. M. et al. Quantitative neuroanatomy for connectomics in Drosophila. eLife 5, e12059 (2016). This article describes the first applications of crowdsourcing of connectome tracing through the Web-based CATMAID framework.
Amat, F. et al. Fast, accurate reconstruction of cell lineages from large-scale fluorescence microscopy data. Nat. Methods 11, 951–958 (2014).
Bria, A. & Iannello, G. TeraStitcher - a tool for fast automatic 3D-stitching of teravoxel-sized microscopy images. BMC Bioinforma. 13, 316 (2012).
Wolff, C. et al. Multi-view light-sheet imaging and tracking with the MaMuT software reveals the cell lineage of a direct developing arthropod limb. eLife 7, e34410 (2018).
Moffitt, J. R. et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 362, eaau5324 (2018).
Bakutkin, V. V., Maksimova, I. L., Semyonova, T. N., Tuchin, V. V. & Kon, I. L. in Ophthalmic Technologies V 137–142 (SPIE, 1995).
Zimnyakov, D. A., Tuchin, V. V., Michin, A. A., Kon, I. L. & Serov, A. N. in Ophthalmic Technologies VI 233–243 (SPIE, 1996).
Tuchin, V. V. et al. in Photon Propagation in Tissues II 118–143 (SPIE, 1996).
Tuchin, V. V. et al. Light propagation in tissues with controlled optical properties. J. Biomed. Opt. 2, 401–417 (1997).
Bashkatov, A. N. et al. in Ophthalmic Technologies IX 311–320 (SPIE, 1999).
Tuchin, V. V. et al. Optics of living tissues with controlled scattering properties. Proc. SPIE 3863, 10–21 (1999).
Tuchin, V. V., Xu, X. & Wang, R. K. Dynamic optical coherence tomography in studies of optical clearing, sedimentation, and aggregation of immersed blood. Appl. Opt. 41, 258–271 (2002).
Xu, X., Wang, R. K., Elder, J. B. & Tuchin, V. V. Effect of dextran-induced changes in refractive index and aggregation on optical properties of whole blood. Phys. Med. Biol. 48, 1205–1221 (2003).
Liu, H., Beauvoit, B., Kimura, M. & Chance, B. Dependence of tissue optical properties on solute-induced changes in refractive index and osmolarity. J. Biomed. Opt. 1, 200–211 (1996).
Vargas, O., Chan, E. K., Barton, J. K., Rylander, H. G. & Welch, A. J. Use of an agent to reduce scattering in skin. Lasers Surg. Med. 24, 133–141 (1999).
Vargas, G., Chan, K. F., Thomsen, S. L. & Welch, A. J. Use of osmotically active agents to alter optical properties of tissue: effects on the detected fluorescence signal measured through skin. Lasers Surg. Med. 29, 213–220 (2001).
Wang, R. K., Xu, X., Tuchin, V. V. & Elder, J. B. Concurrent enhancement of imaging depth and contrast for optical coherence tomography by hyperosmotic agents. JOSA B 18, 948–953 (2001).
Xu, X. & Wang, R. K. The role of water desorption on optical clearing of biotissue: studied with near infrared reflectance spectroscopy. Med. Phys. 30, 1246–1253 (2003).
Jiang, J. & Wang, R. K. Comparing the synergistic effects of oleic acid and dimethyl sulfoxide as vehicles for optical clearing of skin tissue in vitro. Phys. Med. Biol. 49, 5283–5294 (2004).
Choi, B. et al. Determination of chemical agent optical clearing potential using in vitro human skin. Lasers Surg. Med. 36, 72–75 (2005).
Staudt, T., Lang, M. C., Medda, R., Engelhardt, J. & Hell, S. W. 2,2′-Thiodiethanol: a new water soluble mounting medium for high resolution optical microscopy. Microsc. Res. Tech. 70, 1–9 (2007).
Aoyagi, Y., Kawakami, R., Osanai, H., Hibi, T. & Nemoto, T. A rapid optical clearing protocol using 2,2′-thiodiethanol for microscopic observation of fixed mouse brain. PLOS ONE 10, e0116280 (2015).
Kuwajima, T. et al. ClearT: a detergent- and solvent-free clearing method for neuronal and non-neuronal tissue. Development 140, 1364–1368 (2013).
Tsai, P. S. et al. Correlations of neuronal and microvascular densities in murine cortex revealed by direct counting and colocalization of nuclei and vessels. J. Neurosci. 29, 14553–14570 (2009).
Hou, B. et al. Scalable and DiI-compatible optical clearance of the mammalian brain. Front Neuroanat. 9, 19 (2015).
Diogo, R., Siomava, N. & Gitton, Y. Development of human limb muscles based on whole-mount immunostaining and the links between ontogeny and evolution. Development 146, dev180349 (2019).
LaVision BioTec. The UltraMicroscope setup. LaVision BioTec https://www.lavisionbiotec.com/products/UltraMicroscope/specification.html (2019).
Zeiss. Lightsheet Z1. Zeiss https://www.zeiss.com/microscopy/us/products/imaging-systems/lightsheet-z-1.html#downloads (2019).
Engelbrecht, C. J. & Stelzer, E. H. Resolution enhancement in a light-sheet-based microscope (SPIM). Opt. Lett. 31, 1477–1479 (2006).
Schwarz, M. K. et al. Fluorescent-protein stabilization and high-resolution imaging of cleared, intact mouse brains. PLOS ONE 10, e0124650 (2015).
Jing, D. et al. Tissue clearing of both hard and soft tissue organs with the PEGASOS method. Cell Res. 28, 803–818 (2018).
Becker, K., Jährling, N., Saghafi, S., Weiler, R. & Dodt, H. U. Chemical clearing and dehydration of GFP expressing mouse brains. PLOS ONE 7, e33916 (2012).
Scott, G. D., Blum, E. D., Fryer, A. D. & Jacoby, D. B. Tissue optical clearing, three-dimensional imaging, and computer morphometry in whole mouse lungs and human airways. Am. J. Respir. Cell. Mol. Biol. 51, 43–55 (2014).
Ertürk, A., Lafkas, D. & Chalouni, C. Imaging cleared intact biological systems at a cellular level by 3DISCO. J. Vis. Exp. https://doi.org/10.3791/5138 (2014).
Epp, J. R. et al. Optimization of CLARITY for clearing whole-brain and other intact organs. eNeuro 2, ENEURO.0022-15.2015 (2015).
Abe, T. et al. Visualization of cell cycle in mouse embryos with Fucci2 reporter directed by Rosa26 promoter. Development 140, 237–246 (2013).
Breuss, M. et al. The expression of tubb2b undergoes a developmental transition in murine cortical neurons. J. Comp. Neurol. 523, 2161–2186 (2015).
Sekitani, T. et al. Ultraflexible organic amplifier with biocompatible gel electrodes. Nat. Commun. 7, 11425 (2016).
Mizutani, H. et al. Transparency-enhancing technology allows three-dimensional assessment of gastrointestinal mucosa: a porcine model. Pathol. Int. 68, 102–108 (2018).
Warner, C. A. et al. An optical clearing technique for plant tissues allowing deep imaging and compatible with fluorescence microscopy. Plant. Physiol. 166, 1684–1687 (2014).
Hasegawa, J. et al. Three-dimensional imaging of plant organs using a simple and rapid transparency technique. Plant Cell Physiol. 57, 462–472 (2016).
Chen, L. et al. UbasM: an effective balanced optical clearing method for intact biomedical imaging. Sci. Rep. 7, 12218 (2017).
Kurihara, D., Mizuta, Y., Sato, Y. & Higashiyama, T. ClearSee: a rapid optical clearing reagent for whole-plant fluorescence imaging. Development 142, 4168–4179 (2015).
Wang, Z. et al. Imaging transparent intact cardiac tissue with single-cell resolution. Biomed. Opt. Express 9, 423–436 (2018).
Yu, T. et al. RTF: a rapid and versatile tissue optical clearing method. Sci. Rep. 8, 1964 (2018).
Li, W., Germain, R. N. & Gerner, M. Y. Multiplex, quantitative cellular analysis in large tissue volumes with clearing-enhanced 3D microscopy (Ce3D). Proc. Natl Acad. Sci. USA 114, E7321–E7330 (2017).
Lai, H. M. et al. Rationalisation and validation of an acrylamide-free procedure in three-dimensional histological imaging. PLOS ONE 11, e0158628 (2016).
Xu, N. et al. Fast free-of-acrylamide clearing tissue (FACT)—an optimized new protocol for rapid, high-resolution imaging of three-dimensional brain tissue. Sci. Rep. 7, 9895 (2017).
Perbellini, F. et al. Free-of-Acrylamide SDS-based tissue clearing (FASTClear) for three dimensional visualization of myocardial tissue. Sci. Rep. 7, 5188 (2017).
Acknowledgements
The authors thank E. A. Susaki for help in compiling Table 1 and Supplementary Table 1 for current tissue-clearing protocols and reagents, K. Matsumoto and Y. Shinohara for drawing the chemical structures in the supplementary information, T. Mano for contributing to the CUBIC figure, R. Cai and C. Pan for contributing to the uDISCO figure, S. R. Kumar, G. M. Coughlin, R. Challis and C. Challis for contributing to the viral-assisted spectral tracing figure and Y.-G. Park, C. H. Sohn, T. Ku, V. Lilascharoen and B. K. Lim for contributing to the SHIELD figure. The authors also gratefully acknowledge grant support from Brain/MINDS, the Basic Science and Platform Technology Program for Innovative Biological Medicine (AMED/MEXT), the Japan Society for the Promotion of Science (Grant-in-Aid for Scientific Research (S)) and the Human Frontier Science Program Research Grant Program (HFSP RGP0019/2018) (H.R.U.), the Munich Cluster for Systems Neurology (SyNergy), the Fritz Thyssen Stiftung and the Deutsche Forschungsgemeinschaft (A.E.), the David and Lucile Packard Foundation (Packard Fellowship), the McKnight Foundation, the US National Institutes of Health (NIH) (1-DP2-ES027992; U01MH117072), the NCSOFT Cultural Foundation and the Koreaan Institute for Basic Science (IBS-R026-D1) (K.C.), the NIH BRAIN Initiative, the NIH Office of the Director and the US National Science Foundation (NeuroNex) (V.G.), LABEX LIFESENSES (reference ANR-10-LABX-65) managed by the French Agence National de la Recherche within the Investissements d’Avenir programme under reference ANR-11-IDEX-0004-02 (A.C.), the European Regional Development Fund in the framework of the Czech IT4Innovations National Supercomputing Center path to exascale project, project number CZ.02.1.01/0.0/0.0/16_013/0001791, within the Czech Research, Development and Education Operational Programme (P.T.) and the Howard Hughes Medical Institute (P.J.K.).
Author information
Authors and Affiliations
Contributions
H.R.U., A.E., K.C., V.G., A.C. and P.J.K. researched data for the article. All the authors contributed to substantial discussion of its content, wrote the article and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
H.R.U. is a co-inventor on a patent applications covering the CUBIC reagents (PCT/JP2014/070618 (pending), patent applicant is RIKEN, other co-inventors are E. A. Susaki and K. Tainaka; PCT/JP2017/016410 (pending), patent applicant is RIKEN, other co-inventors are K. Tainaka and T. Murakami) and a co-founder of CUBICStars Inc. A.E. is the applicant and the inventor on a patent application for technologies relating to vDISCO clearing (PCT/EP2018/063098 (pending)). K.C. is the inventor or a co-inventor on patents and patent applications for CLARITY (PCT/US2013/031066 (active), patent applicant is Stanford University, co-inventor is K. A. Deisseroth), stochastic electrotransport (PCT/US2015/024297 (active), patent applicant is MIT), SHIELD (PCT/US2016/064538 (pending), applicant is Massachusetts Institute of Technology (MIT), other co-inventors are E. Murray and J. H. Cho), SWITCH (PCT/US2016/064538 (pending), applicant is MIT, other co-inventors are E. Murray and J. H. Cho) and MAP (PCT/US2017/030285 (pending), applicant is MIT, other co-inventors are T. Ku, J. M. Swaney and J. Y. Park) and a co-founder of LifeCanvas Technologies. V.G. is a co-inventor on patent applications covering PACT and PARS (PCT/US2014/048985 (active), applicant is California Institute of Technology, other co-inventors are V. Gradinaru and B. Yang) and adeno-associated virus (US14/485,024 (active), applicant is California Institute of Technology, other co-inventors are B. E. Deverman, P. H. Patterson and V. Gradinaru) technologies. P.J.K. is an inventor or co-inventor on patents and patent applications covering multiview imaging (US14/049,470 (active), applicant is Howard Hughes Medical Institute) and adaptive light-sheet microscopy (PCT/US2017/038970 (pending), applicant is Howard Hughes Medical Institute, other co-inventors are R. K. Chhetri and L. A. Royer). P.T. and A.C. declare no competing interests.
Additional information
Peer review information
Nature Reviews Neuroscience thanks S. Gentleman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Glossary
- Tissue clearing
-
A method to make a biological specimen transparent by minimization of light scattering and light absorption by the biological specimen.
- Hydrophobic tissue clearing
-
One of three major tissue-clearing methods; it uses hydrophobic (water-immiscible) reagents. It is also referred to as ‘solvent tissue clearing’.
- Hydrophilic tissue clearing
-
One of three major tissue-clearing methods; it uses hydrophilic (water-miscible) reagents. It is also referred to as ‘aqueous tissue clearing’ but does not always involve the use of water.
- Refractive index
-
(RI). The ratio of the speed of light in a vacuum to its speed in a specified medium. The RI of a vacuum is 1 by definition, whereas the RI of water is ~1.33.
- Light-sheet microscopy
-
A technique that allows fast, high-resolution imaging of large biological specimens with low light exposure by rapidly acquiring images of thin optical sections illuminated by laser light sheets.
- Passive CLARITY technique
-
(PACT). A technique that allows flexible hydrogel formulation and clearing without the need to use electrophoresis.
- Refractive index-matching solution
-
A solution that is compatible with the passive CLARITY technique, perfusion-assisted agent release in situ and CLARITY. It provides high-resolution imaging at depth by further reducing light scattering in both cleared and uncleared samples.
- Perfusion-assisted agent release in situ
-
(PARS). A method that allows whole-rodent clearing and labelling. It uses the intrinsic circulatory system (the vasculature) to deliver clearing agents and labels instead of relying on passive diffusion, which can be prohibitively slow for large organs or whole organisms.
- Hydrogel-based tissue clearing
-
One of three major tissue-clearing methods; it crosslinks biological specimens to make a synthetic hydrogel.
- sCMOS detectors
-
Scientific-grade CMOS-based cameras that offer a large sensor area, high pixel count, low noise, high frame rate, high dynamic range and high quantum yield, all of which are highly desirable properties for detectors used in widefield fluorescence light microscopy.
- Hierarchical data format version 5
-
(HDF5). A hierarchical data format and versatile data model to manage and represent extremely large and complex data objects.
- Compute unified device architecture
-
(CUDA). A parallel computing platform and programming model developed by Nvidia for general computing on its own graphics processing units.
- Adaptive particle representation
-
A content-adaptive representation of fluorescence microscopy images that overcomes storage and processing bottlenecks in big microscopy image data.
Rights and permissions
About this article
Cite this article
Ueda, H.R., Ertürk, A., Chung, K. et al. Tissue clearing and its applications in neuroscience. Nat Rev Neurosci 21, 61–79 (2020). https://doi.org/10.1038/s41583-019-0250-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41583-019-0250-1
This article is cited by
-
Breaking boundaries in whole-body imaging and disease understanding with wildDISCO
Nature Biotechnology (2024)
-
Virtual reality-empowered deep-learning analysis of brain cells
Nature Methods (2024)
-
Whole-body cellular mapping in mouse using standard IgG antibodies
Nature Biotechnology (2024)
-
Current and future applications of light-sheet imaging for identifying molecular and developmental processes in autism spectrum disorders
Molecular Psychiatry (2024)
-
Mapping of individual sensory nerve axons from digits to spinal cord with the transparent embedding solvent system
Cell Research (2024)