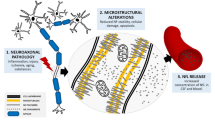
Abstract
Neuroaxonal damage is the pathological substrate of permanent disability in various neurological disorders. Reliable quantification and longitudinal follow-up of such damage are important for assessing disease activity, monitoring treatment responses, facilitating treatment development and determining prognosis. The neurofilament proteins have promise in this context because their levels rise upon neuroaxonal damage not only in the cerebrospinal fluid (CSF) but also in blood, and they indicate neuroaxonal injury independent of causal pathways. First-generation (immunoblot) and second-generation (enzyme-linked immunosorbent assay) neurofilament assays had limited sensitivity. Third-generation (electrochemiluminescence) and particularly fourth-generation (single-molecule array) assays enable the reliable measurement of neurofilaments throughout the range of concentrations found in blood samples. This technological advancement has paved the way to investigate neurofilaments in a range of neurological disorders. Here, we review what is known about the structure and function of neurofilaments, discuss analytical aspects and knowledge of age-dependent normal ranges of neurofilaments and provide a comprehensive overview of studies on neurofilament light chain as a marker of axonal injury in different neurological disorders, including multiple sclerosis, neurodegenerative dementia, stroke, traumatic brain injury, amyotrophic lateral sclerosis and Parkinson disease. We also consider work needed to explore the value of this axonal damage marker in managing neurological diseases in daily practice.
Key points
-
Neuronal damage and loss are the pathological substrates of permanent disability in various acute and chronic neurological disorders.
-
Levels of neurofilament proteins increase in cerebrospinal fluid (CSF) and in the blood upon neuroaxonal damage.
-
First-generation (immunoblot) and second-generation (enzyme-linked immunosorbent assay) neurofilament assays captured only the tip of the iceberg in disease.
-
Third-generation (electrochemiluminescence) and fourth-generation (single-molecule array) assays permit highly sensitive, longitudinal detection of blood neurofilament levels even in mild disease and in healthy controls.
-
Multicentre studies are underway to consolidate neurofilaments as biomarkers that reflect brain tissue damage, enabling longitudinal monitoring of disease activity and drug effects in clinical trials in neurological diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Deisenhammer, F. et al. EFNS guidelines on disease-specific CSF investigations. Eur. J. Neurol. 16, 760–770 (2009).
Petzold, A., Keir, G., Green, A. J., Giovannoni, G. & Thompson, E. J. A specific ELISA for measuring neurofilament heavy chain phosphoforms. J. Immunol. Methods 278, 179–190 (2003).
Petzold, A., Rejdak, K. & Plant, G. T. Axonal degeneration and inflammation in acute optic neuritis. J. Neurol. Neurosurg. Psychiatry 75, 1178–1180 (2004).
Gaiottino, J. et al. Increased neurofilament light chain blood levels in neurodegenerative neurological diseases. PLoS ONE 8, e75091 (2013).
Kuhle, J. et al. Serum neurofilament light chain is a biomarker of human spinal cord injury severity and outcome. J. Neurol. Neurosurg. Psychiatry 86, 273–279 (2015).
Gisslen, M. et al. Plasma concentration of the neurofilament light protein (NFL) is a biomarker of CNS injury in HIV infection: a cross-sectional study. EBioMedicine 3, 135–140 (2016). This study was the first time that NfL was measured with a single-molecule array assay and showed that CSF and plasma concentrations were highly correlated, indicating that plasma NfL concentration is a promising marker in HIV-associated brain damage.
Kuhle, J. et al. Comparison of three analytical platforms for quantification of the neurofilament light chain in blood samples: ELISA, electrochemiluminescence immunoassay and Simoa. Clin. Chem. Lab. Med. 54, 1655–1661 (2016).
Disanto, G. et al. Serum neurofilament light: a biomarker of neuronal damage in multiple sclerosis. Ann. Neurol. 81, 857–870 (2017). This study was the first in which serum NfL levels in MS were measured with a Simoa assay; higher CSF and serum levels and clinical and MRI disease activity were associated with higher risk of relapses and disability worsening.
Yuan, A., Rao, M. V., Veeranna & Nixon, R. A. Neurofilaments and neurofilament proteins in health and disease. Cold Spring Harb. Perspect. Biol. 9, a018309 (2017). This paper provides a comprehensive and detailed overview of the structure and biological roles of the neurofilament proteins.
Dyson, H. J. & Wright, P. E. Intrinsically unstructured proteins and their functions. Nat. Rev. Mol. Cell Biol. 6, 197–208 (2005).
Petzold, A. Neurofilament phosphoforms: surrogate markers for axonal injury, degeneration and loss. J. Neurol. Sci. 233, 183–198 (2005).
Herrmann, H. & Aebi, U. Intermediate filaments: structure and assembly. Cold Spring Harb. Perspect. Biol. 8, a018242 (2016).
Nixon, R. A. & Sihag, R. K. Neurofilament phosphorylation: a new look at regulation and function. Trends Neurosci. 14, 501–506 (1991).
Beck, R., Deek, J. & Safinya, C. R. Structures and interactions in ‘bottlebrush’ neurofilaments: the role of charged disordered proteins in forming hydrogel networks. Biochem. Soc. Trans. 40, 1027–1031 (2012).
Barry, D. M. et al. Expansion of neurofilament medium C terminus increases axonal diameter independent of increases in conduction velocity or myelin thickness. J. Neurosci. 32, 6209–6219 (2012).
Rao, M. V. et al. The neurofilament middle molecular mass subunit carboxyl-terminal tail domains is essential for the radial growth and cytoskeletal architecture of axons but not for regulating neurofilament transport rate. J. Cell Biol. 163, 1021–1031 (2003).
Brown, R. H. Jr & Al-Chalabi, A. Amyotrophic lateral sclerosis. N. Engl. J. Med. 377, 1602 (2017).
Rosengren, L. E., Karlsson, J. E., Karlsson, J. O., Persson, L. I. & Wikkelso, C. Patients with amyotrophic lateral sclerosis and other neurodegenerative diseases have increased levels of neurofilament protein in CSF. J. Neurochem. 67, 2013–2018 (1996).
Norgren, N., Karlsson, J. E., Rosengren, L. & Stigbrand, T. Monoclonal antibodies selective for low molecular weight neurofilaments. Hybrid. Hybridomics 21, 53–59 (2002). This paper provides a seminal description of NfL‑specific monoclonal antibodies that are now used in most studies.
Norgren, N., Rosengren, L. & Stigbrand, T. Elevated neurofilament levels in neurological diseases. Brain Res. 987, 25–31 (2003).
Petzold, A. et al. In vivo monitoring of neuronal loss in traumatic brain injury: a microdialysis study. Brain 134, 464–483 (2011).
Zucchi, E. et al. A motor neuron strategy to save time and energy in neurodegeneration: adaptive protein stoichiometry. J. Neurochem. https://doi.org/10.1111/jnc.14542 (2018).
Petzold, A., Keir, G., Warren, J., Fox, N. & Rossor, M. N. A systematic review and meta-analysis of CSF neurofilament protein levels as biomarkers in dementia. Neurodegener. Dis. 4, 185–194 (2007).
Petzold, A. et al. Neurofilament ELISA validation. J. Immunol. Methods 352, 23–31 (2010). This study was an evaluation of the most widely used NfL ELISA kit across 35 different laboratories, and identified that a lack of accurate and consistent protein standards is the main reason for a poor inter-laboratory coefficient of variation.
Kuhle, J. et al. A highly sensitive electrochemiluminescence immunoassay for the neurofilament heavy chain protein. J. Neuroimmunol. 220, 114–119 (2010).
Kuhle, J. et al. Neurofilament heavy chain in CSF correlates with relapses and disability in multiple sclerosis. Neurology 76, 1206–1213 (2011).
Kuhle, J. et al. Serum neurofilament is associated with progression of brain atrophy and disability in early MS. Neurology 88, 826–831 (2017).
Rissin, D. M. et al. Single-molecule enzyme-linked immunosorbent assay detects serum proteins at subfemtomolar concentrations. Nat. Biotechnol. 28, 595–599 (2010). This study provided the initial description of the high-sensitivity, fourth-generation Simoa assay technology that enables reliable quantification of NfL in serum and plasma samples.
Kuhle, J. et al. Serum neurofilament light chain in early relapsing remitting MS is increased and correlates with CSF levels and with MRI measures of disease severity. Mult. Scler. 22, 1550–1559 (2016).
Piehl, F. et al. Plasma neurofilament light chain levels in patients with MS switching from injectable therapies to fingolimod. Mult. Scler. 24, 1046–1054 (2018).
Bacioglu, M. et al. Neurofilament light chain in blood and CSF as marker of disease progression in mouse models and in neurodegenerative diseases. Neuron 91, 56–66 (2016).
Wilke, C. et al. Neurofilament light chain in FTD is elevated not only in cerebrospinal fluid, but also in serum. J. Neurol. Neurosurg. Psychiatry 87, 1270–1272 (2016).
Weydt, P. et al. Neurofilament levels as biomarkers in asymptomatic and symptomatic familial amyotrophic lateral sclerosis. Ann. Neurol. 79, 152–158 (2016). This study of asymptomatic and symptomatic ALS mutation carriers and family controls showed that blood NfL levels are normal in the asymptomatic stage and increase at the time of early symptom onset, thereby linking NfL to the symptomatic phase of ALS.
Lu, C. H. et al. Neurofilament light chain: a prognostic biomarker in amyotrophic lateral sclerosis. Neurology 84, 2247–2257 (2015).
Meeter, L. H. et al. Neurofilament light chain: a biomarker for genetic frontotemporal dementia. Ann. Clin. Transl Neurol. 3, 623–636 (2016).
Novakova, L. et al. Monitoring disease activity in multiple sclerosis using serum neurofilament light protein. Neurology 89, 2230–2237 (2017).
Martinez-Morillo, E. et al. Neurofilament medium polypeptide (NFM) protein concentration is increased in CSF and serum samples from patients with brain injury. Clin. Chem. Lab. Med. 53, 1575–1584 (2015).
Barro, C. et al. Serum neurofilament as a predictor of disease worsening and brain and spinal cord atrophy in multiple sclerosis. Brain 141, 2382–2391 (2018).
Yilmaz, A. et al. Neurofilament light chain protein as a marker of neuronal injury: review of its use in HIV-1 infection and reference values for HIV-negative controls. Expert. Rev. Mol. Diagn. 17, 761–770 (2017).
Reiber, H. Flow rate of cerebrospinal fluid (CSF) — a concept common to normal blood-CSF barrier function and to dysfunction in neurological diseases. J. Neurol. Sci. 122, 189–203 (1994).
Idland, A. V. et al. CSF neurofilament light levels predict hippocampal atrophy in cognitively healthy older adults. Neurobiol. Aging 49, 138–144 (2017).
Szaro, B. G. & Strong, M. J. Post-transcriptional control of neurofilaments: new roles in development, regeneration and neurodegenerative disease. Trends Neurosci. 33, 27–37 (2010).
Mattsson, N., Andreasson, U., Zetterberg, H. & Blennow, K. Alzheimer’s Disease Neuroimaging Initiative. Association of plasma neurofilament light with neurodegeneration in patients with Alzheimer disease. JAMA Neurol. 74, 557–566 (2017). In this paper, plasma NfL was associated with Alzheimer disease diagnosis and cognitive performance, suggesting that NfL is a noninvasive biomarker for this disease.
Bischof, A. et al. Serum neurofilament light chain: a biomarker of neuronal injury in vasculitic neuropathy. Ann. Rheum. Dis. 77, 1093–1094 (2017).
Sandelius, A. et al. Plasma neurofilament light chain concentration in the inherited peripheral neuropathies. Neurology 90, e518–e524 (2018).
Brownlee, W. J., Hardy, T. A., Fazekas, F. & Miller, D. H. Diagnosis of multiple sclerosis: progress and challenges. Lancet 389, 1336–1346 (2017).
Lucchinetti, C. F. et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 365, 2188–2197 (2011).
Huizinga, R., Gerritsen, W., Heijmans, N. & Amor, S. Axonal loss and gray matter pathology as a direct result of autoimmunity to neurofilaments. Neurobiol. Dis. 32, 461–470 (2008).
Trapp, B. D. et al. Axonal transection in the lesions of multiple sclerosis. N. Engl. J. Med. 338, 278–285 (1998).
Kuhlmann, T., Lingfeld, G., Bitsch, A., Schuchardt, J. & Bruck, W. Acute axonal damage in multiple sclerosis is most extensive in early disease stages and decreases over time. Brain 125, 2202–2212 (2002).
Tallantyre, E. C. et al. Clinico-pathological evidence that axonal loss underlies disability in progressive multiple sclerosis. Mult. Scler. 16, 406–411 (2010).
Rocca, M. A. et al. Brain MRI atrophy quantification in MS: From methods to clinical application. Neurology 88, 403–413 (2017).
Lycke, J. N., Karlsson, J. E., Andersen, O. & Rosengren, L. E. Neurofilament protein in cerebrospinal fluid: a potential marker of activity in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 64, 402–404 (1998). This work identified for the first time that neurofilament levels in CSF could be a biomarker in MS.
Norgren, N. et al. Neurofilament and glial fibrillary acidic protein in multiple sclerosis. Neurology 63, 1586–1590 (2004).
Teunissen, C. E. et al. Combination of CSF N-acetylaspartate and neurofilaments in multiple sclerosis. Neurology 72, 1322–1329 (2009).
Khalil, M. et al. CSF neurofilament and N-acetylaspartate related brain changes in clinically isolated syndrome. Mult. Scler. 19, 436–442 (2013).
Arrambide, G. et al. Neurofilament light chain level is a weak risk factor for the development of MS. Neurology 87, 1076–1084 (2016).
Petzold, A. et al. CSF neurofilament levels: a potential prognostic marker in Guillain-Barre syndrome. Neurology 67, 1071–1073 (2006).
Petzold, A., Steenwijk, M. D., Eikelenboom, J. M., Wattjes, M. P. & Uitdehaag, B. M. Elevated CSF neurofilament proteins predict brain atrophy: a 15-year follow-up study. Mult. Scler. 22, 1154–1162 (2016).
Gunnarsson, M. et al. Axonal damage in relapsing multiple sclerosis is markedly reduced by natalizumab. Ann. Neurol. 69, 83–89 (2011). This longitudinal study showed a treatment effect of natalizumab on CSF levels of NfL in relapsing–remitting MS, regardless of previous treatment and whether patients experienced a relapse in the 3 months before natalizumab treatment.
Kuhle, J. et al. Fingolimod and CSF neurofilament light chain levels in relapsing-remitting multiple sclerosis. Neurology 84, 1639–1643 (2015).
Novakova, L. et al. Cerebrospinal fluid biomarkers of inflammation and degeneration as measures of fingolimod efficacy in multiple sclerosis. Mult. Scler. 23, 62–71 (2017).
Novakova, L. et al. Cerebrospinal fluid biomarkers as a measure of disease activity and treatment efficacy in relapsing-remitting multiple sclerosis. J. Neurochem. 141, 296–304 (2017).
Axelsson, M. et al. Immunosuppressive therapy reduces axonal damage in progressive multiple sclerosis. Mult. Scler. 20, 43–50 (2014).
Romme Christensen, J. et al. Natalizumab in progressive MS: results of an open-label, phase 2A, proof-of-concept trial. Neurology 82, 1499–1507 (2014).
Malmestrom, C., Haghighi, S., Rosengren, L., Andersen, O. & Lycke, J. Neurofilament light protein and glial fibrillary acidic protein as biological markers in MS. Neurology 61, 1720–1725 (2003).
Rosengren, L. E., Karlsson, J. E., Sjogren, M., Blennow, K. & Wallin, A. Neurofilament protein levels in CSF are increased in dementia. Neurology 52, 1090–1093 (1999).
Sjogren, M. et al. Cytoskeleton proteins in CSF distinguish frontotemporal dementia from AD. Neurology 54, 1960–1964 (2000).
Olsson, B. et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: a systematic review and meta-analysis. Lancet Neurol. 15, 673–684 (2016).
Mattsson, N. et al. Cerebrospinal fluid tau, neurogranin, and neurofilament light in Alzheimer’s disease. EMBO Mol. Med. 8, 1184–1196 (2016).
Zetterberg, H. Neurofilament light: a dynamic cross-disease fluid biomarker for neurodegeneration. Neuron 91, 1–3 (2016).
Zetterberg, H. et al. Association of cerebrospinal fluid neurofilament light concentration with Alzheimer disease progression. JAMA Neurol. 73, 60–67 (2015).
Rohrer, J. D. et al. Serum neurofilament light chain protein is a measure of disease intensity in frontotemporal dementia. Neurology 87, 1329–1336 (2016).
Skillback, T. et al. CSF neurofilament light differs in neurodegenerative diseases and predicts severity and survival. Neurology 83, 1945–1953 (2014).
Pijnenburg, Y. A., Verwey, N. A., van der Flier, W. M., Scheltens, P. & Teunissen, C. E. Discriminative and prognostic potential of cerebrospinal fluid phosphoTau/tau ratio and neurofilaments for frontotemporal dementia subtypes. Alzheimers Dement. (Amst.) 1, 505–512 (2015).
Andreasson, U., Blennow, K. & Zetterberg, H. Update on ultrasensitive technologies to facilitate research on blood biomarkers for central nervous system disorders. Alzheimers Dement. (Amst.) 3, 98–102 (2016).
Weston, P. S. J. et al. Serum neurofilament light in familial Alzheimer disease: a marker of early neurodegeneration. Neurology 89, 2167–2175 (2017).
Hansson, O. et al. Blood-based NfL: a biomarker for differential diagnosis of parkinsonian disorder. Neurology 88, 930–937 (2017).
Steinacker, P. et al. Neurofilament as a blood marker for diagnosis and monitoring of primary progressive aphasias. Neurology 88, 961–969 (2017).
Steinacker, P. et al. Neurofilaments in blood and CSF for diagnosis and prediction of onset in Creutzfeldt-Jakob disease. Sci. Rep. 6, 38737 (2016).
Nylen, K. et al. CSF -neurofilament correlates with outcome after aneurysmal subarachnoid hemorrhage. Neurosci. Lett. 404, 132–136 (2006).
Lewis, S. B., Wolper, R. A., Miralia, L., Yang, C. & Shaw, G. Detection of phosphorylated NF-H in the cerebrospinal fluid and blood of aneurysmal subarachnoid hemorrhage patients. J. Cereb. Blood Flow Metab. 28, 1261–1271 (2008).
Zanier, E. R. et al. Neurofilament light chain levels in ventricular cerebrospinal fluid after acute aneurysmal subarachnoid haemorrhage. J. Neurol. Neurosurg. Psychiatry 82, 157–159 (2011).
Traenka, C. et al. Serum neurofilament light chain levels are associated with clinical characteristics and outcome in patients with cervical artery dissection. Cerebrovasc. Dis. 40, 222–227 (2015).
Gattringer, T. et al. Serum neurofilament light is sensitive to active cerebral small vessel disease. Neurology 89, 2108–2114 (2017). This study provided evidence of a possible role for serum NfL as a marker of ongoing cerebral small vessel disease and for the occurrence of new vascular MRI lesions, even when these lesions are clinically silent.
Bergman, J. et al. Neurofilament light in CSF and serum is a sensitive marker for axonal white matter injury in MS. Neurol. Neuroimmunol. Neuroinflamm. 3, e271 (2016).
Blennow, K. et al. Traumatic brain injuries. Nat. Rev. Dis. Primers 2, 16084 (2016).
McKee, A. C. et al. The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol. 131, 75–86 (2016).
Dekosky, S. T., Blennow, K., Ikonomovic, M. D. & Gandy, S. Acute and chronic traumatic encephalopathies: pathogenesis and biomarkers. Nat. Rev. Neurol. 9, 192–200 (2013).
Zetterberg, H. et al. Neurochemical aftermath of amateur boxing. Arch. Neurol. 63, 1277–1280 (2006).
Neselius, S. et al. CSF-biomarkers in Olympic boxing: diagnosis and effects of repetitive head trauma. PLoS ONE 7, e33606 (2012).
Shahim, P. et al. Serum neurofilament light protein predicts clinical outcome in traumatic brain injury. Sci. Rep. 6, 36791 (2016).
Al Nimer, F. et al. Comparative assessment of the prognostic value of biomarkers in traumatic brain injury reveals an independent role for serum levels of neurofilament light. PLoS ONE 10, e0132177 (2015).
Shahim, P., Zetterberg, H., Tegner, Y. & Blennow, K. Serum neurofilament light as a biomarker for mild traumatic brain injury in contact sports. Neurology 88, 1788–1794 (2017).
Oliver, J. M. et al. Serum neurofilament light in american football athletes over the course of a season. J. Neurotrauma 33, 1784–1789 (2016).
Al-Chalabi, A. et al. Amyotrophic lateral sclerosis: moving towards a new classification system. Lancet Neurol. 15, 1182–1194 (2016).
Brettschneider, J., Petzold, A., Sussmuth, S. D., Ludolph, A. C. & Tumani, H. Axonal damage markers in cerebrospinal fluid are increased in ALS. Neurology 66, 852–856 (2006).
Zetterberg, H., Jacobsson, J., Rosengren, L., Blennow, K. & Andersen, P. M. Cerebrospinal fluid neurofilament light levels in amyotrophic lateral sclerosis: impact of SOD1 genotype. Eur. J. Neurol. 14, 1329–1333 (2007).
Mendonca, D. M. et al. Neurofilament heavy subunit in cerebrospinal fluid: a biomarker of amyotrophic lateral sclerosis? Amyotroph. Lateral Scler. 12, 144–147 (2011).
McCombe, P. A. et al. Serial measurements of phosphorylated neurofilament-heavy in the serum of subjects with amyotrophic lateral sclerosis. J. Neurol. Sci. 353, 122–129 (2015).
Li, S. et al. Phosphorylated neurofilament heavy chain levels in paired plasma and CSF of amyotrophic lateral sclerosis. J. Neurol. Sci. 367, 269–274 (2016).
Steinacker, P. et al. Neurofilaments in the diagnosis of motoneuron diseases: a prospective study on 455 patients. J. Neurol. Neurosurg. Psychiatry 87, 12–20 (2016).
De, S. M. et al. Comparison of elevated phosphorylated neurofilament heavy chains in serum and cerebrospinal fluid of patients with amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 89, 367–373 (2017).
Poesen, K. et al. Neurofilament markers for ALS correlate with extent of upper and lower motor neuron disease. Neurology 88, 2302–2309 (2017).
Oeckl, P. P. et al. Multicenter validation of CSF neurofilaments as diagnostic biomarkers for ALS. Amyotroph. Lateral. Scler. Frontotemporal Degener. 17, 404–413 (2016).
Gendron, T. F. et al. Phosphorylated neurofilament heavy chain: a biomarker of survival for C9ORF72-associated amyotrophic lateral sclerosis. Ann. Neurol. 82, 139–146 (2017).
Lehmer, C. et al. Poly-GP in cerebrospinal fluid links C9orf72-associated dipeptide repeat expression to the asymptomatic phase of ALS/FTD. EMBO Mol. Med. 9, 859–868 (2017).
Feneberg, E. et al. Multicenter evaluation of neurofilaments in early symptom onset amyotrophic lateral sclerosis. Neurology 90, e22–e30 (2018).
Boylan, K. B. et al. Phosphorylated neurofilament heavy subunit (pNF-H) in peripheral blood and CSF as a potential prognostic biomarker in amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 84, 467–472 (2013).
Menke, R. A. et al. CSF neurofilament light chain reflects corticospinal tract degeneration in ALS. Ann. Clin. Transl Neurol. 2, 748–755 (2015).
Volkening, K., Leystra-Lantz, C., Yang, W., Jaffee, H. & Strong, M. J. Tar DNA binding protein of 43 kDa (TDP-43), 14-3-3 proteins and copper/zinc superoxide dismutase (SOD1) interact to modulate NFL mRNA stability. Implications for altered RNA processing in amyotrophic lateral sclerosis (ALS). Brain Res. 1305, 168–182 (2009).
Halbgebauer, S., Ockl, P., Wirth, K., Steinacker, P. & Otto, M. Protein biomarkers in Parkinson’s disease: focus on cerebrospinal fluid markers and synaptic proteins. Mov. Disord. 31, 848–860 (2016).
Eusebi, P. et al. Diagnostic utility of cerebrospinal fluid alpha-synuclein in Parkinson’s disease: a systematic review and meta-analysis. Mov. Disord. 32, 1389–1400 (2017).
Holmberg, B., Rosengren, L., Karlsson, J. E. & Johnels, B. Increased cerebrospinal fluid levels of neurofilament protein in progressive supranuclear palsy and multiple-system atrophy compared with Parkinson’s disease. Mov. Disord. 13, 70–77 (1998).
Brettschneider, J. et al. Neurofilament heavy-chain NfH(SMI35) in cerebrospinal fluid supports the differential diagnosis of Parkinsonian syndromes. Mov. Disord. 21, 2224–2227 (2006).
Hall, S. et al. Accuracy of a panel of 5 cerebrospinal fluid biomarkers in the differential diagnosis of patients with dementia and/or parkinsonian disorders. Arch. Neurol. 69, 1445–1452 (2012).
Magdalinou, N. K. et al. A panel of nine cerebrospinal fluid biomarkers may identify patients with atypical parkinsonian syndromes. J. Neurol. Neurosurg. Psychiatry 86, 1240–1247 (2015).
Rojas, J. C. et al. Plasma neurofilament light chain predicts progression in progressive supranuclear palsy. Ann. Clin. Transl Neurol. 3, 216–225 (2016).
Byrne, L. M. et al. Neurofilament light protein in blood as a potential biomarker of neurodegeneration in Huntington’s disease: a retrospective cohort analysis. Lancet Neurol. 16, 601–609 (2017).
Wild, E. J., Petzold, A., Keir, G. & Tabrizi, S. J. Plasma neurofilament heavy chain levels in Huntington’s disease. Neurosci. Lett. 417, 231–233 (2007).
Constantinescu, R., Romer, M., Oakes, D., Rosengren, L. & Kieburtz, K. Levels of the light subunit of neurofilament triplet protein in cerebrospinal fluid in Huntington’s disease. Parkinsonism Relat. Disord. 15, 245–248 (2009).
Niemela, V., Landtblom, A. M., Blennow, K. & Sundblom, J. Tau or neurofilament light -which is the more suitable biomarker for Huntington’s disease? PLoS ONE 12, e0172762 (2017).
Cousins, D. A. & Grunze, H. Interpreting magnetic resonance imaging findings in bipolar disorder. CNS Neurosci. Ther. 18, 201–207 (2012).
Jakobsson, J. et al. Elevated concentrations of neurofilament light chain in the cerebrospinal fluid of bipolar disorder patients. Neuropsychopharmacology 39, 2349–2356 (2014).
Isgren, A. et al. Markers of neuroinflammation and neuronal injury in bipolar disorder: relation to prospective clinical outcomes. Brain Behav. Immun. 65, 195–201 (2017).
Steinacker, P. et al. Diagnostic and prognostic significance of neurofilament light chain NF-L, but not progranulin and S100B, in the course of amyotrophic lateral sclerosis: Data from the German MND-net. Amyotroph. Lateral. Scler. Frontotemporal. Degener. 18, 112–119 (2017).
Acknowledgements
M.O. is supported by the German Federal Ministry for Education and Research (German FTLD consortium), the Thierry Latran Foundation and the ALS association. H.Z. is supported by grants from the Swedish Research Council, the European Research Council and the Knut and Alice Wallenberg Foundation. K.B. is supported by grants from the Swedish Research Council, the Swedish Alzheimer Association, the Swedish Brain Foundation and the Torsten Söderberg Foundation. J.K. is supported by grants from the Swiss National Science Foundation (320030_160221). F.P. is supported by grants from the Swedish Research Council.
Author information
Authors and Affiliations
Contributions
All authors contributed to all aspects of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khalil, M., Teunissen, C.E., Otto, M. et al. Neurofilaments as biomarkers in neurological disorders. Nat Rev Neurol 14, 577–589 (2018). https://doi.org/10.1038/s41582-018-0058-z
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41582-018-0058-z
This article is cited by
-
Cluster analysis dissecting cognitive deficits in older adults with major depressive disorder and the association with neurofilament light chain
BMC Geriatrics (2024)
-
Correlation analysis between smoke exposure and serum neurofilament light chain in adults: a cross-sectional study
BMC Public Health (2024)
-
Study protocol: fish oil supplement in prevention of oxaliplatin-induced peripheral neuropathy in adjuvant colorectal cancer patients – a randomized controlled trial. (OxaNeuro)
BMC Cancer (2024)
-
The impact of kidney function on plasma neurofilament light and phospho-tau 181 in a community-based cohort: the Shanghai Aging Study
Alzheimer's Research & Therapy (2024)
-
Serum neurofilament indicates accelerated neurodegeneration and predicts mortality in late-stage Parkinson’s disease
npj Parkinson's Disease (2024)