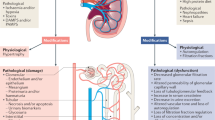
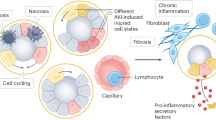
Abstract
The acute loss of kidney function has been diagnosed for many decades using the serum concentration of creatinine — a muscle metabolite that is an insensitive and non-specific marker of kidney function, but is now used for the very definition of acute kidney injury (AKI). Fortunately, myriad new tools have now been developed to better understand the relationship between acute tubular injury and elevation in serum creatinine (SCr). These tools include unbiased gene and protein expression analyses in kidney, urine and blood, the localization of specific gene transcripts in pathological biopsy samples by rapid in-situ RNA technology and single-cell RNA-sequencing analyses. However, this molecular approach to AKI has produced a series of unexpected problems, because the expression of specific kidney-derived molecules that are indicative of injury often do not correlate with SCr levels. This discrepancy between kidney injury markers and SCr level can be reconciled by the recognition that many separate subtypes of AKI exist, each with distinct patterning of molecular markers of tubular injury and SCr data. In this Review, we describe the weaknesses of isolated SCr-based diagnoses, the clinical and molecular subtyping of acute tubular injury, and the role of non-invasive biomarkers in clinical phenotyping. We propose a conceptual model that synthesizes molecular and physiological data along a time course spanning from acute cellular injury to organ failure.
Key points
-
Current definitions of acute kidney injury (AKI), based on serum creatinine (SCr) level, focus on loss of kidney function rather than kidney injury.
-
AKI definitions cannot provide an acute measurement of loss of function, however, because SCr is a quantitative functional marker only at the steady state.
-
Current AKI metrics can neither detect kidney injury in real time nor distinguish dramatically different types of kidney injury.
-
Molecular analyses of acutely damaged kidneys have detected cellular and segment-specific responses to injurious stimuli, prior to and distinct from the loss of function as measured by SCr.
-
As a result, molecular analyses have detected different types of acute tubular injury and have re-characterized the concept of the kidney response to noxious stimuli into biomarker-positive ʻinjury’ and biomarker-negative ʻno injury’.
-
A conceptual model places ʻtubular injury’ (biomarkers) upstream of ʻloss of function’ (AKI metrics), providing a unifying ʻinjury’ and ʻloss of function’ sequence consistent with biological pathways.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Heberden, W. Commentaries on the History and Cure of Diseases (Wells and Lilly, 1818).
Eknoyan, G. Emergence of the concept of acute renal failure. Am. J. Nephrol. 22, 225–230 (2002).
Cameron, J. S. Bright’s disease today: the pathogenesis and treatment of glomerulonephritis–I. Br. Med. J. 4, 87–90 (1972).
Smith, H. W. The Kidney: Structure and Function in Health and Disease (Oxford University Press, 1951).
Bellomo, R. et al. Acute renal failure — definition, outcome measures, animal models, fluid therapy and information technology needs: the second international consensus conference of the acute dialysis quality initiative (ADQI) group. Crit. Care 8, R204–212 (2004).
Mehta, R. L. et al. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit. Care 11, R31 (2007).
Khwaja, A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin. Pract. 120, c179–c184 (2012).
Xu, K. et al. Unique transcriptional programs identify subtypes of AKI. J. Am. Soc. Nephrol. 28, 1729–1740 (2017).
Uchino, S., Bellomo, R., Bagshaw, S. M. & Goldsmith, D. Transient azotaemia is associated with a high risk of death in hospitalized patients. Nephrol. Dial. Transpl. 25, 1833–1839 (2010).
Vanmassenhove, J., Van Biesen, W., Vanholder, R. & Lameire, N. Subclinical AKI: ready for primetime in clinical practice? J. Nephrol. 32, 9–16 (2018).
Haase, M., Kellum, J. A. & Ronco, C. Subclinical AKI — an emerging syndrome with important consequences. Nat. Rev. Nephrol. 8, 735–739 (2012).
Ronco, C., Kellum, J. A. & Haase, M. Subclinical AKI is still AKI. Crit. Care 16, 313 (2012).
Huen, S. C. & Parikh, C. R. Molecular phenotyping of clinical AKI with novel urinary biomarkers. Am. J. Physiol. Ren. Physiol. 309, F406–F413 (2015).
Fujii, T., Uchino, S., Takinami, M. & Bellomo, R. Subacute kidney injury in hospitalized patients. Clin. J. Am. Soc. Nephrol. 9, 457–461 (2014).
Nickolas, T. L. et al. Diagnostic and prognostic stratification in the emergency department using urinary biomarkers of nephron damage: a multicenter prospective cohort study. J. Am. Coll. Cardiol. 59, 246–255 (2012).
Mehta, S. et al. The prognostic importance of duration of AKI: a systematic review and meta-analysis. BMC Nephrol. 19, 91 (2018).
Au, V., Feit, J., Barasch, J., Sladen, R. N. & Wagener, G. Urinary neutrophil gelatinase-associated lipocalin (NGAL) distinguishes sustained from transient acute kidney injury after general surgery. Kidney Int. Rep. 1, 3–9 (2016).
Freda, B. J., Knee, A. B., Braden, G. L., Visintainer, P. F. & Thakar, C. V. Effect of transient and sustained acute kidney injury on readmissions in acute decompensated heart failure. Am. J. Cardiol. 119, 1809–1814 (2017).
Moriyama, N. et al. Early development of acute kidney injury is an independent predictor of in-hospital mortality in patients with acute myocardial infarction. J. Cardiol. 69, 79–83 (2017).
Basu, R. K., Kaddourah, A., Goldstein, S. L. & Investigators, A. S. Assessment of a renal angina index for prediction of severe acute kidney injury in critically ill children: a multicentre, multinational, prospective observational study. Lancet Child. Adolesc. Health 2, 112–120 (2018).
Yang, X. et al. Urinary matrix metalloproteinase-7 predicts severe AKI and poor outcomes after cardiac surgery. J. Am. Soc. Nephrol. 28, 3373–3382 (2017).
Efstratiadis, G. et al. Rhabdomyolysis updated. Hippokratia 11, 129–137 (2007).
Walid, M. S. Blood urea nitrogen/creatinine ratio in rhabdomyolysis. Indian J. Nephrol. 18, 173–174 (2008).
Doi, K. et al. Reduced production of creatinine limits its use as marker of kidney injury in sepsis. J. Am. Soc. Nephrol. 20, 1217–1221 (2009).
Vanholder, R., Sever, M. S., Erek, E. & Lameire, N. Rhabdomyolysis. J. Am. Soc. Nephrol. 11, 1553–1561 (2000).
Schetz, M., Gunst, J. & Van den Berghe, G. The impact of using estimated GFR versus creatinine clearance on the evaluation of recovery from acute kidney injury in the ICU. Intensive Care Med. 40, 1709–1717 (2014).
Ravn, B., Prowle, J. R., Martensson, J., Martling, C. R. & Bell, M. Superiority of serum cystatin C over creatinine in prediction of long-term prognosis at discharge from ICU. Crit. Care Med. 45, e932–e940 (2017).
Wilson, F. P., Sheehan, J. M., Mariani, L. H. & Berns, J. S. Creatinine generation is reduced in patients requiring continuous venovenous hemodialysis and independently predicts mortality. Nephrol. Dial. Transpl. 27, 4088–4094 (2012).
Sise, M. E. et al. Urine neutrophil gelatinase-associated lipocalin identifies unilateral and bilateral urinary tract obstruction. Nephrol. Dial. Transpl. 26, 4132–4135 (2011).
Decoste, R., Himmelman, J. G. & Grantmyre, J. Acute renal infarct without apparent cause: a case report and review of the literature. Can. Urol. Assoc. J. 9, E237–239 (2015).
Ramcharan, T. & Matas, A. J. Long-term (20–37 years) follow-up of living kidney donors. Am. J. Transpl. 2, 959–964 (2002).
Molitoris, B. A. Therapeutic translation in acute kidney injury: the epithelial/endothelial axis. J. Clin. Invest. 124, 2355–2363 (2014).
Sharma, A., Mucino, M. J. & Ronco, C. Renal functional reserve and renal recovery after acute kidney injury. Nephron Clin. Pract. 127, 94–100 (2014).
Moretti, C. et al. Androgens and body composition in the aging male. J. Endocrinol. Invest. 28, 56–64 (2005).
Kimmel, P. L., Lew, S. Q. & Bosch, J. P. Nutrition, ageing and GFR: is age-associated decline inevitable? Nephrol. Dial. Transpl. 11, 85–88 (1996).
Musso, C. G. et al. Creatinine reabsorption by the aged kidney. Int. Urol. Nephrol. 41, 727–731 (2009).
Sjostrom, P. A., Odlind, B. G. & Wolgast, M. Extensive tubular secretion and reabsorption of creatinine in humans. Scand. J. Urol. Nephrol. 22, 129–131 (1988).
Gault, M. H. & Cockcroft, D. W. Letter: creatinine clearance and age. Lancet 2, 612–613 (1975).
Cockcroft, D. W. & Gault, M. H. Prediction of creatinine clearance from serum creatinine. Nephron 16, 31–41 (1976).
Inker, L. A. et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N. Engl. J. Med. 367, 20–29 (2012).
Schaeffner, E. S. et al. Two novel equations to estimate kidney function in persons aged 70 years or older. Ann. Intern. Med. 157, 471–481 (2012).
Sutherland, S. M. et al. AKI in hospitalized children: comparing the pRIFLE, AKIN, and KDIGO definitions. Clin. J. Am. Soc. Nephrol. 10, 554–561 (2015).
Toto, R. D. Conventional measurement of renal function utilizing serum creatinine, creatinine clearance, inulin and para-aminohippuric acid clearance. Curr. Opin. Nephrol. Hypertens. 4, 505–509 (1995).
Coca, S. G. et al. First post-operative urinary kidney injury biomarkers and association with the duration of AKI in the TRIBE-AKI Cohort. PLOS ONE 11, e0161098 (2016).
Coca, S. G., King, J. T., Jr, Rosenthal, R. A., Perkal, M. F. & Parikh, C. R. The duration of postoperative acute kidney injury is an additional parameter predicting long-term survival in diabetic veterans. Kidney Int. 78, 926–933 (2010).
Brown, J. R., Kramer, R. S., Coca, S. G. & Parikh, C. R. Duration of acute kidney injury impacts long-term survival after cardiac surgery. Ann. Thorac. Surg. 90, 1142–1148 (2010).
Safirstein, R. L. Acute renal failure: from renal physiology to the renal transcriptome. Kidney Int. Suppl., S62–S66 (2004).
Johnson, A. C. M. & Zager, R. A. Mechanisms underlying increased timp2 and igfbp7 urinary excretion in experimental AKI. J. Am. Soc. Nephrol. 29, 2157–2167 (2018).
Johnson, A. C. & Zager, R. A. Plasma and urinary p21: potential biomarkers of AKI and renal aging. Am. J. Physiol. Ren. Physiol. 315, F1329–F1335 (2018).
Garner, A. E., Lewington, A. J. & Barth, J. H. Detection of patients with acute kidney injury by the clinical laboratory using rises in serum creatinine: comparison of proposed definitions and a laboratory delta check. Ann. Clin. Biochem. 49, 59–62 (2012).
Lin, J. et al. False-positive rate of AKI using consensus creatinine-based criteria. Clin. J. Am. Soc. Nephrol. 10, 1723–1731 (2015).
Labban, B. et al. The role of kidney biopsy in heart transplant candidates with kidney disease. Transplantion 89, 887–893 (2010).
Bergler-Klein, J. et al. The long-term effect of simultaneous heart and kidney transplantation on native renal function. Transplantion 71, 1597–1600 (2001).
Moledina, D. G. et al. Performance of serum creatinine and kidney injury biomarkers for diagnosing histologic acute tubular injury. Am. J. Kidney Dis. 70, 807–816 (2017).
Gay, L. et al. Mouse TU tagging: a chemical/genetic intersectional method for purifying cell type-specific nascent RNA. Genes Dev. 27, 98–115 (2013).
Yuen, P. S., Jo, S. K., Holly, M. K., Hu, X. & Star, R. A. Ischemic and nephrotoxic acute renal failure are distinguished by their broad transcriptomic responses. Physiol. Genomics 25, 375–386 (2006).
Zager, R. A. Alterations of intravascular volume: influence on renal susceptibility to ischemic injury. J. Lab. Clin. Med. 108, 60–69 (1986).
Nickolas, T. L. et al. Sensitivity and specificity of a single emergency department measurement of urinary neutrophil gelatinase-associated lipocalin for diagnosing acute kidney injury. Ann. Intern. Med. 148, 810–819 (2008).
Belcher, J. M. et al. Kidney biomarkers and differential diagnosis of patients with cirrhosis and acute kidney injury. Hepatology 60, 622–632 (2014).
Verna, E. C. et al. Urinary neutrophil gelatinase-associated lipocalin predicts mortality and identifies acute kidney injury in cirrhosis. Dig. Dis. Sci. 57, 2362–2370 (2012).
Singer, E. et al. Urinary neutrophil gelatinase-associated lipocalin distinguishes pre-renal from intrinsic renal failure and predicts outcomes. Kidney Int. 80, 405–414 (2011).
Ahmad, T. et al. Worsening renal function in patients with acute heart failure undergoing aggressive diuresis is not associated with tubular injury. Circulation 137, 2016–2028 (2018).
Heyman, S. N., Rosenberger, C. & Rosen, S. Experimental ischemia-reperfusion: biases and myths-the proximal vs. distal hypoxic tubular injury debate revisited. Kidney Int. 77, 9–16 (2010).
Liu, J. et al. Cell-specific translational profiling in acute kidney injury. J. Clin. Invest. 124, 1242–1254 (2014).
Lautrette, A. et al. Angiotensin II and EGF receptor cross-talk in chronic kidney diseases: a new therapeutic approach. Nat. Med. 11, 867–874 (2005).
Azroyan, A. et al. Renal intercalated cells sense and mediate inflammation via the P2Y14 receptor. PLOS ONE 10, e0121419 (2015).
Gburek, J. et al. Renal uptake of myoglobin is mediated by the endocytic receptors megalin and cubilin. Am. J. Physiol. Ren. Physiol. 285, F451–458 (2003).
Prozialeck, W. C. & Edwards, J. R. Mechanisms of cadmium-induced proximal tubule injury: new insights with implications for biomonitoring and therapeutic interventions. J Pharmacol. Exp. Ther. 343, 2–12 (2012).
Schmitz, C. et al. Megalin deficiency offers protection from renal aminoglycoside accumulation. J. Biol. Chem. 277, 618–622 (2002).
Servais, H. et al. Gentamicin-induced apoptosis in LLC-PK1 cells: involvement of lysosomes and mitochondria. Toxicol. Appl. Pharmacol. 206, 321–333 (2005).
Servais, H. et al. Renal cell apoptosis induced by nephrotoxic drugs: cellular and molecular mechanisms and potential approaches to modulation. Apoptosis 13, 11–32 (2008).
Prozialeck, W. C. et al. Expression of kidney injury molecule-1 (Kim-1) in relation to necrosis and apoptosis during the early stages of Cd-induced proximal tubule injury. Toxicol. Appl. Pharmacol. 238, 306–314 (2009).
McWilliam, S. J. et al. Mechanism-based urinary biomarkers to identify the potential for aminoglycoside-induced nephrotoxicity in premature neonates: a proof-of-concept study. PLOS ONE 7, e43809 (2012).
Blank, M., Thompson, A., Hausner, E. & Rouse, R. Biomarkers of drug-induced acute kidney injury: a regulatory perspective. Expert. Opin. Drug. Metab. Toxicol. 14, 929–936 (2018).
Matheis, K. A. et al. Cross-study and cross-omics comparisons of three nephrotoxic compounds reveal mechanistic insights and new candidate biomarkers. Toxicol. Appl. Pharmacol. 252, 112–122 (2011).
Woodcock, J. & Jenkins, J. Review submission of the qualification of seven biomarkers of drug-induced nephrotoxicity in rats. Department of Health and Human Services https://c-path.org//wp-content/uploads/2014/09/PSTC-NWG-20080414-FDA-FinalConclusion.pdf (2008).
European Medicines Agency. Final report on the pilot Joint EMEA/FDA VXDS experience on qualification of nephrotoxicity biomarkers. EMA https://c-path.org//wp-content/uploads/2014/09/PSTC-NWG-20080523-EMA-FinalConclusion.pdf (2008).
Rasi, G. Letter of support for PSTC translational drug-induced kidney injury (DIKI) biomarkers. European Medicines Agency https://c-path.org//wp-content/uploads/2014/11/letter-of-support-for-PSTC-translational-drug-induced-kidney-injury-DIKI-biomarkers.pdf (2014).
Woodcock, J. Biomarker letter of support. FDA https://c-path.org//wp-content/uploads/2014/09/PSTC-NWG-2014820-FDA-LetterOfSupport.pdf (2014).
Rasi, G. Letter of support for drug-induced renal tubular injury biomarker(s). European Medicines Agency https://www.ema.europa.eu/en/documents/other/letter-support-drug-induced-renal-tubular-injury-biomarkers_en.pdf (2016).
Woodcock, J. Letter of Support for Drug-Induced Renal Tubular Injury Biomarker(s). FDA https://www.fda.gov/media/102623/download (2016).
Leptak, C. & Stockbridge, N. Qualification determination letter. FDA https://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/DrugDevelopmentToolsQualificationProgram/BiomarkerQualificationProgram/UCM618888.pdf (2018).
El-Achkar, T. M. et al. Sepsis induces changes in the expression and distribution of Toll-like receptor 4 in the rat kidney. Am. J. Physiol. Ren. Physiol. 290, F1034–F1043 (2006).
Krivan, S. et al. Increased expression of Toll-like receptors 2, 3, 4 and 7 mRNA in the kidney and intestine of a septic mouse model. Sci. Rep. 9, 4010 (2019).
Lieberthal, W. & Nigam, S. K. Acute renal failure. I. relative importance of proximal vs. distal tubular injury. Am. J. Physiol. 275, F623–F631 (1998).
Epstein, F. H. Oxygen and renal metabolism. Kidney Int. 51, 381–385 (1997).
Bagnasco, S., Good, D., Balaban, R. & Burg, M. Lactate production in isolated segments of the rat nephron. Am. J. Physiol. 248, F522–F526 (1985).
Brezis, M., Rosen, S., Silva, P. & Epstein, F. H. Transport activity modifies thick ascending limb damage in the isolated perfused kidney. Kidney Int. 25, 65–72 (1984).
di Mari, J. F., Davis, R. & Safirstein, R. L. MAPK activation determines renal epithelial cell survival during oxidative injury. Am. J. Physiol. 277, F195–F203 (1999).
Schofield, C. J. & Ratcliffe, P. J. Oxygen sensing by HIF hydroxylases. Nat. Rev. Mol. Cell Biol. 5, 343–354 (2004).
Sharfuddin, A. A. & Molitoris, B. A. Pathophysiology of ischemic acute kidney injury. Nat. Rev. Nephrol. 7, 189–200 (2011).
Ratliff, B. B., Rabadi, M. M., Vasko, R., Yasuda, K. & Goligorsky, M. S. Messengers without borders: mediators of systemic inflammatory response in AKI. J. Am. Soc. Nephrol. 24, 529–536 (2013).
Kalogeris, T., Baines, C. P., Krenz, M. & Korthuis, R. J. Cell biology of ischemia/reperfusion injury. Int. Rev. Cell Mol. Biol. 298, 229–317 (2012).
Kanda, J. et al. An AKI biomarker lipocalin 2 in the blood derives from the kidney in renal injury but from neutrophils in normal and infected conditions. Clin. Exp. Nephrol. 19, 99–106 (2015).
Paragas, N. et al. The Ngal reporter mouse detects the response of the kidney to injury in real time. Nat. Med. 17, 216–222 (2011).
Lankadeva, Y. R. et al. Alterations in regional kidney oxygenation during expansion of extracellular fluid volume in conscious healthy sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 315, R1242–R1250 (2018).
Evans, R. G. et al. Urinary oxygen tension: a clinical window on the health of the renal medulla? Am. J. Physiol. Regul. Integr. Comp. Physiol. 306, R45–R50 (2014).
Lankadeva, Y. R., Kosaka, J., Evans, R. G., Bellomo, R. & May, C. N. Urinary oxygenation as a surrogate measure of medullary oxygenation during angiotensin ii therapy in septic acute kidney injury. Crit. Care Med. 46, e41–e48 (2018).
Sgouralis, I. et al. Bladder urine oxygen tension for assessing renal medullary oxygenation in rabbits: experimental and modeling studies. Am. J. Physiol. Regul. Integr. Comp. Physiol. 311, R532–R544 (2016).
Zhu, M. Z. L. et al. Urinary hypoxia: an intraoperative marker of risk of cardiac surgery-associated acute kidney injury. Nephrol. Dial. Transpl. 33, 2191–2201 (2018).
Kellum, J. A. & Prowle, J. R. Paradigms of acute kidney injury in the intensive care setting. Nat. Rev. Nephrol. 14, 217–230 (2018).
Angus, D. C. & van der Poll, T. Severe sepsis and septic shock. N. Engl. J. Med. 369, 840–851 (2013).
Brown, K. A. et al. Neutrophils in development of multiple organ failure in sepsis. Lancet 368, 157–169 (2006).
Gomez, H. et al. A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock. 41, 3–11 (2014).
Ince, C. et al. The endothelium in sepsis. Shock 45, 259–270 (2016).
Prowle, J. R. & Bellomo, R. Sepsis-associated acute kidney injury: macrohemodynamic and microhemodynamic alterations in the renal circulation. Semin. Nephrol. 35, 64–74 (2015).
Post, E. H. et al. Changes in kidney perfusion and renal cortex metabolism in septic shock: an experimental study. J. Surg. Res. 207, 145–154 (2017).
Takasu, O. et al. Mechanisms of cardiac and renal dysfunction in patients dying of sepsis. Am. J. Respir. Crit. Care Med. 187, 509–517 (2013).
Tran, M. et al. PGC-1alpha promotes recovery after acute kidney injury during systemic inflammation in mice. J. Clin. Invest. 121, 4003–4014 (2011).
Brealey, D. & Singer, M. Mitochondrial dysfunction in sepsis. Curr. Infect. Dis. Rep. 5, 365–371 (2003).
Parikh, S. M. et al. Mitochondrial function and disturbances in the septic kidney. Semin. Nephrol. 35, 108–119 (2015).
Bailly, V. et al. Shedding of kidney injury molecule-1, a putative adhesion protein involved in renal regeneration. J. Biol. Chem. 277, 39739–39748 (2002).
Nickolas, T. L. et al. NGAL (Lcn2) monomer is associated with tubulointerstitial damage in chronic kidney disease. Kidney Int. 82, 718–722 (2012).
Yan, L., Borregaard, N., Kjeldsen, L. & Moses, M. A. The high molecular weight urinary matrix metalloproteinase (MMP) activity is a complex of gelatinase B/MMP-9 and neutrophil gelatinase-associated lipocalin (NGAL). Modulation of MMP-9 activity by NGAL. J. Biol. Chem. 276, 37258–37265 (2001).
Ichimura, T. et al. Kidney injury molecule-1 (KIM-1), a putative epithelial cell adhesion molecule containing a novel immunoglobulin domain, is up-regulated in renal cells after injury. J. Biol. Chem. 273, 4135–4142 (1998).
Mishra, J. et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365, 1231–1238 (2005).
Dong, L., Ma, Q., Bennett, M. & Devarajan, P. Urinary biomarkers of cell cycle arrest are delayed predictors of acute kidney injury after pediatric cardiopulmonary bypass. Pediatr. Nephrol. 32, 2351–2360 (2017).
Krawczeski, C. D. et al. Temporal relationship and predictive value of urinary acute kidney injury biomarkers after pediatric cardiopulmonary bypass. J. Am. Coll. Cardiol. 58, 2301–2309 (2011).
Mishra, J. et al. Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J. Am. Soc. Nephrol. 14, 2534–2543 (2003).
Paragas, N. et al. Urinary NGAL marks cystic disease in HIV-associated nephropathy. J. Am. Soc. Nephrol. 20, 1687–1692 (2009).
Paragas, N. et al. alpha-Intercalated cells defend the urinary system from bacterial infection. J. Clin. Invest. 124, 2963–2976 (2014).
Wang, F. et al. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J. Mol. Diagn. 14, 22–29 (2012).
Emlet, D. R. et al. Insulin-like growth factor binding protein 7 and tissue inhibitor of metalloproteinases-2: differential expression and secretion in human kidney tubule cells. Am. J. Physiol. Ren. Physiol. 312, F284–F296 (2017).
Mar, D. et al. Heterogeneity of epigenetic changes at ischemia/reperfusion- and endotoxin-induced acute kidney injury genes. Kidney Int. 88, 734–744 (2015).
Basu, R. K. et al. Combining functional and tubular damage biomarkers improves diagnostic precision for acute kidney injury after cardiac surgery. J. Am. Coll. Cardiol. 64, 2753–2762 (2014).
Vaidya, V. S. et al. Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies. Nat. Biotechnol. 28, 478–485 (2010).
Mori, K. et al. Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury. J. Clin. Invest. 115, 610–621 (2005).
da Rocha, E. P. et al. Urinary neutrophil gelatinase-associated lipocalin is excellent predictor of acute kidney injury in septic elderly patients. Aging Dis. 9, 182–191 (2018).
Park, H. S. et al. Urinary neutrophil gelatinase-associated lipocalin as a biomarker of acute kidney injury in sepsis patients in the emergency department. Clin. Chim. Acta 495, 552–555 (2019).
Srisawat, N. et al. Neutrophil gelatinase associated lipocalin (NGAL) in leptospirosis acute kidney injury: a multicenter study in thailand. PLOS ONE 10, e0143367 (2015).
Urbschat, A. et al. Serum and urinary NGAL but not KIM-1 raises in human postrenal AKI. Eur. J. Clin. Invest. 44, 652–659 (2014).
Forster, C. S. & Devarajan, P. Neutrophil gelatinase-associated lipocalin: utility in urologic conditions. Pediatr. Nephrol. 32, 377–381 (2017).
Kostic, D. et al. The role of renal biomarkers to predict the need of surgery in congenital urinary tract obstruction in infants. J. Pediatr. Urol. 15, 242.e1–249.e9 (2019).
Goetz, D. H. et al. The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol. Cell 10, 1033–1043 (2002).
Arai, S. et al. Apoptosis inhibitor of macrophage protein enhances intraluminal debris clearance and ameliorates acute kidney injury in mice. Nat. Med. 22, 183–193 (2016).
Murray, P. T. et al. Potential use of biomarkers in acute kidney injury: report and summary of recommendations from the 10th acute dialysis quality initiative consensus conference. Kidney Int. 85, 513–521 (2014).
Haase, M. et al. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J. Am. Coll. Cardiol. 57, 1752–1761 (2011).
Basu, R. K. et al. Incorporation of biomarkers with the renal angina index for prediction of severe AKI in critically ill children. Clin. J. Am. Soc. Nephrol. 9, 654–662 (2014).
Molitoris, B. A. & Reilly, E. S. Quantifying glomerular filtration rates in acute kidney injury: a requirement for translational success. Semin. Nephrol. 36, 31–41 (2016).
Hollinger, A. et al. Proenkephalin A 119-159 (Penkid) is an early biomarker of septic acute kidney injury: the kidney in sepsis and septic shock (Kid-SSS) study. Kidney Int. Rep. 3, 1424–1433 (2018).
Denning, G. M. et al. Proenkephalin expression and enkephalin release are widely observed in non-neuronal tissues. Peptides 29, 83–92 (2008).
Rosen, S., Brezis, M. & Stillman, I. The pathology of nephrotoxic injury: a reappraisal. Min. Electrolyte Metab. 20, 174–180 (1994).
Heyman, S. N., Rosen, S., Fuchs, S., Epstein, F. H. & Brezis, M. Myoglobinuric acute renal failure in the rat: a role for medullary hypoperfusion, hypoxia, and tubular obstruction. J. Am. Soc. Nephrol. 7, 1066–1074 (1996).
Alexanian, R., Barlogie, B. & Dixon, D. Renal failure in multiple myeloma. Pathogenesis and prognostic implications. Arch. Intern. Med. 150, 1693–1695 (1990).
Cohen, D. J., Sherman, W. H., Osserman, E. F. & Appel, G. B. Acute renal failure in patients with multiple myeloma. Am. J. Med. 76, 247–256 (1984).
Perazella, M. A. Onco-nephrology: renal toxicities of chemotherapeutic agents. Clin. J. Am. Soc. Nephrol. 7, 1713–1721 (2012).
Ghane Shahrbaf, F. & Assadi, F. Drug-induced renal disorders. J. Ren. Inj. Prev. 4, 57–60 (2015).
Stacul, F. et al. Strategies to reduce the risk of contrast-induced nephropathy. Am. J. Cardiol. 98, 59K–77K (2006).
Goldfarb, S., McCullough, P. A., McDermott, J. & Gay, S. B. Contrast-induced acute kidney injury: specialty-specific protocols for interventional radiology, diagnostic computed tomography radiology, and interventional cardiology. Mayo Clin. Proc. 84, 170–179 (2009).
Schrier, R. W. Nephrology forum: acute renal failure. Kidney Int. 15, 205–216 (1979).
Gines, P. & Schrier, R. W. Renal failure in cirrhosis. N. Engl. J. Med. 361, 1279–1290 (2009).
Charlton, J. R. et al. Late onset neonatal acute kidney injury: results from the AWAKEN Study. Pediatr. Res. 85, 339–348 (2018).
Takaya, Y. et al. Impact of onset time of acute kidney injury on outcomes in patients with acute decompensated heart failure. Heart Vessel. 31, 60–65 (2016).
Acknowledgements
J.B. is supported by NIH 1U54DK104309, NIH 2R01DK073462, UG3 DK114926, T32-DK108741 and a Columbia Precision Medicine Pilot Award. P.D. is funded by NIH P50 DK096418, NIH R01HL133695, NIH 1R01HL132551.
Author information
Authors and Affiliations
Contributions
B.D.D.O., K.X., T.H.S., K.K., V.D.D., N.P.T., J.B. and P.D. researched data for the article. All authors contributed equally to discussion of the article’s content, writing the article and review/editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.B. and P.D. are co-inventors on patents (US57766204P; US8592170; US797710; EP1766395B1; EP1616184) for the use of NGAL in kidney disease.
Additional information
Peer review information
Nature Reviews Nephrology thanks M. Ostermann, J. Prowle and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Thiouracil tagging
-
Tagging of newly synthesized RNA. The Uprt gene is activated in the Rosa locus by segment-specific Cre drivers. 4-Thiouracil is then introduced at the time of choosing and 4 h later, thiouracil-labelled RNA is extracted from the whole organ without the need for cell dissociation.
- Warm IRI
-
Arterial ischaemia–reperfusion injury of the kidney in the setting of normal body temperature (37 oC).
- Sterile inflammation
-
The presence of inflammatory cells (neutrophils, macrophages and lymphocytes) in the absence of overt infection with bacteria or virus.
- RNA pulldown
-
A process of extraction and purification of labelled RNA from an organ.
- Partial pressure of oxygen
-
(pO2). The percentage of atmosphere occupied by oxygen gas, multiplied by the total atmospheric pressure.
- Fine mapping
-
The use of high-resolution microscopy, whereby single cells and cellular details are microscopically discernible.
- Siderophore
-
An organic chemical that binds with high affinity to iron. Bacteria create many different types of complex siderophores (catecholates, hydroxamates, carboxylates) to capture host iron from serum, urine and cells. Metabolic fragments, such as catecholates, can serve as siderophores in mammals. NGAL protein binds catechols and catecholate siderophores enterochelin with high affinity.
- Nutritional immunity
-
A process of sequestering critical nutrients needed for bacterial growth. This includes the capture of iron-bound siderophores by NGAL and more generally the capture of iron by transferrin and lactoferrin.
Rights and permissions
About this article
Cite this article
Desanti De Oliveira, B., Xu, K., Shen, T.H. et al. Molecular nephrology: types of acute tubular injury. Nat Rev Nephrol 15, 599–612 (2019). https://doi.org/10.1038/s41581-019-0184-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-019-0184-x
This article is cited by
-
Urinary non-albumin protein-creatinine ratio is an independent predictor of mortality in patients with type 2 diabetes: a retrospective cohort study
Scientific Reports (2024)
-
Advances in pediatric acute kidney injury pathobiology: a report from the 26th Acute Disease Quality Initiative (ADQI) conference
Pediatric Nephrology (2024)
-
A risk-stratified assessment of biomarker-based acute kidney injury phenotypes in children
Pediatric Research (2023)
-
Application of metabolomics in urolithiasis: the discovery and usage of succinate
Signal Transduction and Targeted Therapy (2023)
-
Acute kidney injury
Intensive Care Medicine (2023)